Abstract
Traumatic brain injury (TBI) is highly prevalent and there is currently no adequate treatment. Understanding the underlying mechanisms governing TBI and recovery remains an elusive goal. The heterogeneous nature of injury and individual’s response to injury have made understanding risk and susceptibility to TBI of great importance. Epidemiologic studies have provided evidence of sex-dependent differences following TBI. However, preclinical models of injury have largely focused on adult male animals. Here, we review 50 studies that have investigated TBI in both sexes using animal models. Results from these studies are highly variable and model dependent, but largely show females to have a protective advantage in behavioral outcomes and pathology following TBI. Further research of both sexes using newer models that better recapitulate mild and repetitive TBI is needed to characterize the nature of sex-dependent injury and recovery, and ultimately identifies targets for enhanced recovery.
Introduction
Traumatic brain injury (TBI) is a major public health burden. The number of TBIs per year is not known due to the many that go undiagnosed, but it has increased steadily over the past decade and is estimated that anywhere from 1.4 to 42 million occur every year.1–4
Most head trauma, up to 80% of all cases, fall into the mild subgroup of traumatic brain injury (mTBI), also known as concussion.5,6 Concussion occurs from a direct blow to or rapid acceleration-deceleration of the head, with or without loss of consciousness, that causes rapid onset of altered neurological function. Typical symptoms include headache, nausea, sensitivity to light, and impaired concentration and memory. 7 Although concussion produces symptoms that are understood to arise from brain pathology, clinical imaging abnormalities such as hemorrhage or skull fracture are not typically present.8,9 The diverse mechanisms by which concussion occurs and its varied clinical symptoms implicate heterogeneity of the underlying neuropathology. 10 Although most patients recover from concussion in a matter of hours to weeks, some remain symptomatic for months or even years.11–13
Understanding why some patients recover relatively quickly whereas others remain symptomatic is of the utmost importance. Preclinical studies over the past 30 years have yielded valuable information about various aspects of injury and recovery, such as the elaboration of a prolonged innate neuroinflammatory process and acute changes in phosphorylated tau deposition following concussion.14–16 Currently, the only clinical treatment for concussion is rest and supportive care. Because strict rest may actually confer worse and prolonged symptoms, active recovery methods have started to be implemented.17–19 In the search for direct biological interventions to treat concussion, animal models are critical for defining viable therapeutic targets for ameliorating symptoms and enhancing recovery. Given the heterogeneity with which concussion manifests in humans, it is important for animal models to address potential sources of inter-individual variation.
One area that has received increased attention as a possible modifier of outcome after concussion is biological sex. Multiple studies have shown that females are at greater risk than men for poor outcomes following concussion.20–23 Despite the fact that men are at greater risk for concussion due to greater participation in high-risk activities, women tend to report more symptoms and more persistent sequelae following concussion.22–25 These findings have been debated due to the subjective self-reporting involved to collect these measures, with some attributing the differences to societal pressure causing men to underreport symptoms. 26 Various mechanisms have been postulated as to why men and women have different outcomes following concussion and subconcussive injury, such as force of injury, number of injuries, skull and brain shape, neck strength, and hormonal influences.27,28 Further investigation, including in animal models, will be necessary to isolate individual aspects of injury and recovery to develop new modes and methods for treatment.
With a growing body of clinical evidence that sex may be an important factor conferring risk from TBI, defining the underlying mechanisms using animal models remains an important goal. This review seeks to summarize the sex-dependent findings from animal models of TBI to better understand what role sex differences play, and where opportunity may lie for future research to identify actionable targets for intervention.
Methods
This review used a structured search of PubMed to include all relevant articles through 2018. Search terms included “Sex Characteristics” or “Sex Factors”; “Disease Models. Animal”; and “Brain Injuries,” “Concussion,” “TBI,” and “mTBI.” Searches were limited to articles in English. Papers were screened to retain original research articles reporting sex effects related to TBI in animal models. Studies of stroke or other non-TBI injury models such as direct lesion, as well as reviews, editorials, letters and case reports, were excluded. References of included papers were reviewed to identify additional papers. A total of 50 articles were identified and are reviewed here (Table 1).
All papers examining sex differences after TBI.
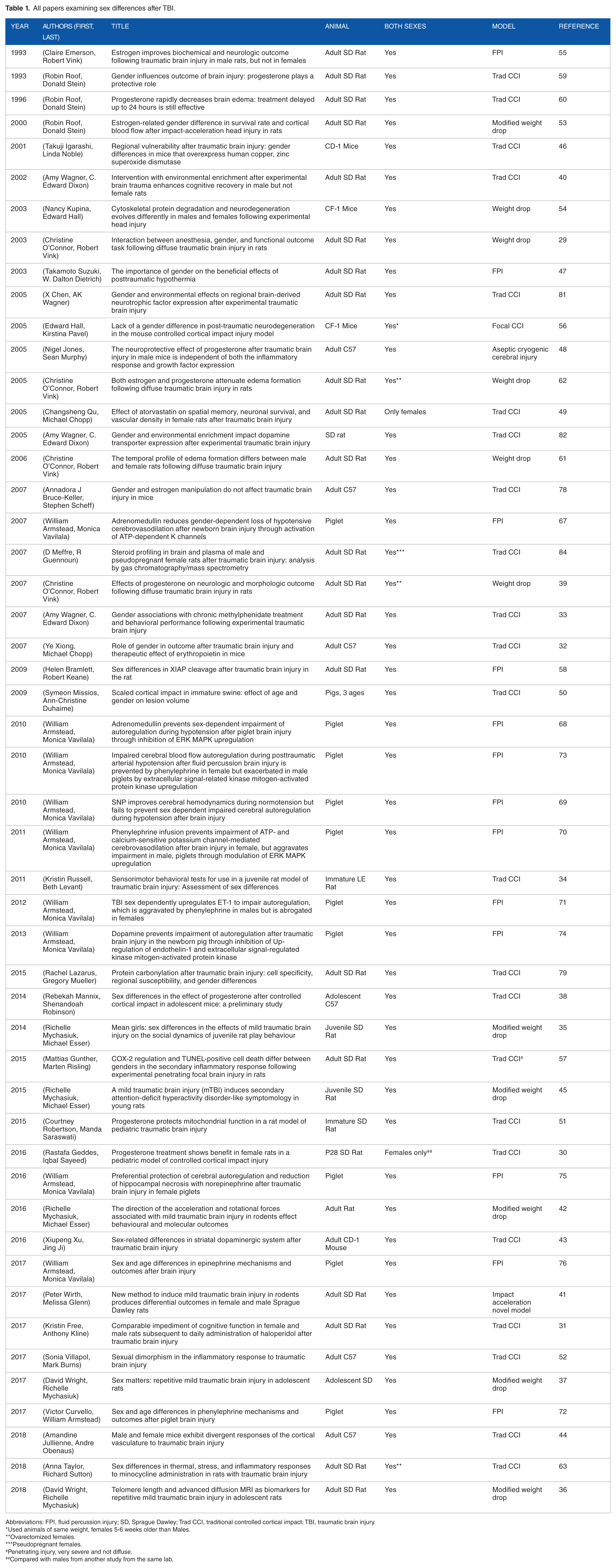
Abbreviations: FPI, fluid percussion injury; SD, Sprague Dawley; Trad CCI, traditional controlled cortical impact; TBI, traumatic brain injury.
*Used animals of same weight, females 5-6 weeks older than Males.
**ovarectomized females.
***pseudopregnant females.
#penetrating injury, very severe and not diffuse.
##compared with males from another study from the same lab.
Sex-Dependent Changes to Cognition and Behavior Following TBI
In studying the effects of TBI, 3 areas are most commonly assessed for behavioral changes: sensorimotor function, cognitive function, and anxiety/depression-like behavior. Most studies have reported that TBI is associated with reductions in motor performance across multiple tests. Studies that have examined sex have largely reported females to be more resilient than their male counterparts. O’Connor et al investigated the effects of different anesthetics on motor performance after TBI in male and female adult Sprague Dawley (SD) rats. They reported baseline sex differences in rotarod performance, with females outperforming males. 29 This finding was extended to post-injury performance. Both sexes were impaired following TBI in comparison with pre-injury, but females performed better than males, and their performance recovered at a faster rate. Other studies have also shown that females perform better than males on rotarod and other sensorimotor evaluations such as beam balance and wire grid foot fault.30–33 However, in a model of juvenile TBI, no sex differences were reported for sensorimotor tasks.34–36 In one of the few studies that reported on a repetitive mTBI model, adolescent female rats were more impaired than males in the beam balance task. 37 However, a subsequent study from the same group showed no sex difference in the beam balance using the same repetitive model.36,37 It is possible that these prepubescent animals showed no differences, or even the opposite association, if sex hormones play a role in producing the behavioral effect. Another study of adolescent C57/Bl6 mice showed progesterone improved performance of males on a wire grip test, but led to worse performance in females, suggesting a ceiling effect. 38
O’Connor probed spatial memory using the Barnes Maze, and females performed better post injury than males when isofluorane was used as the anesthetic during injury, but no difference between the sexes was detected when halothane was used. This suggests a sex-dependent effect of injury as well as anesthetic. In a subsequent study by the same group, ovariectomized (OVX) SD rats performed similarly to males on rotarod and Barnes maze after injury, but regained their pre-TBI performance advantage over males when treated with progesterone 30 minutes after injury. 39 Females also were shown to be less impaired in the novel context mismatch task, a test of working memory, after repetitive mTBI. 37 Other groups have also shown a similar sex-dependent difference in spatial memory using the Morris Water Maze (MWM), where females performed better than males following a controlled cortical impact (CCI). 33 However, in a previous study from the same group, MWM performance of males and females was similar, but only males improved their performance when housed in an enriched environment. 40 Still, others have shown the exact opposite association of sex with MWM performance after TBI, with males showing shorter latencies to find the platform. 41 Of note, this last study was the only one reviewed to use a model that accelerated the animal into a fixed object to produce TBI, rather than an object accelerated into the animal’s head. In summary, cognitive deficits have largely been shown to be more pronounced in male animals than females, but vary across TBI models.
A few groups have examined sex-dependent effects of TBI on depression- and anxiety-like phenotypes using the open field test and the elevated plus maze (EPM). Mychasiuk et al found that both male and female rats exhibit reduced locomotion in the open field following TBI produced using the modified weight drop model, but male locomotion was depressed more than that of females. 42 They also showed females to have more exploration of the open arms of the EPM than males, indicating greater anxiety in the males, or loss of inhibition in the females. In a subsequent study by the same group, adolescent rats subjected to repetitive mTBI showed sex differences in the forced swim task, where females showed a greater depressive phenotype than males. 37 Other groups have also found greater activity of female mice in the open field following CCI, 43 whereas others have found no difference between males and females.44,45 The few studies that have reported sex dependence of depression- and anxiety-like behaviors following TBI seem to show a protective effect of female sex, but may this may depend on the TBI model, including number of impacts. The paucity and diversity of studies to date precludes any definitive conclusions.
Finally, one group reported on social behavior in juvenile rats following TBI by using a social play fighting test. Following induction of TBI by modified weight drop, females showed reduced levels of play compared with males, and there were sex-dependent changes in the social interaction of sham animals with TBI animals. Sham females were less likely to interact with TBI females than sham males with their male TBI counterparts. 35 In a follow-up study, the same group investigated attention deficit hyperactivity disorder–like behavior, following experimental TBI, and found males exhibited reduced inhibitory control compared with females. 45
Sex-Dependent Changes to Pathophysiology
Quantifying the extent of tissue damage is important for assessment of TBI pathology. Depending on the severity of TBI, however, frank tissue loss, necrosis, or apoptosis may be absent. In studies of more severe injuries that produce macroscopic brain lesions due to TBI, females consistently exhibit a smaller lesion than males receiving the same TBI, across various species and TBI model mechanisms.30,31,46–52 Moreover, in studies of the most severe TBI, females generally have lower mortality than males,53,54 with one exception. 55 Limited reports on neuropathological aspects have detected no divergence of neurodegeneration and cell death by sex, using a silver stain, 56 while another identified greater apoptosis in males compared with females using TUNEL staining. 57 Structural magnetic resonance imaging (MRI) studies have also been limited and their results were mixed, such as reduced prefrontal cortex (PFC) volume after mTBI in female SD compared with males in one study and reduced curvature of the corpus callosum in males after repetitive experimental mTBI in another.36,37 Lesion recovery after injury was found to diverge between the sexes, demonstrated by labeling with BrdU (bromodeoxyuridine), a marker of newly proliferated neurons. Female mice showed greater cortical, but not hippocampal neurogenesis following CCI than males, indicating regional specificity of the sex dependence. 32 Further investigation of pathways governing the sex dependence of cell death after TBI has found that X-linked inhibitor of apoptosis (XIAP) is upregulated in female rats after fluid percussion injury (FPI), possibly protecting females from cell death. 58 Another group showed that male and OVX female rats treated with progesterone after TBI showed reduced expression of caspase-3, a key component of the apoptotic pathway, and produced a reduction in dark cell change in the hippocampus, suggesting less cell death. 39 Overall, studies indicate females have a protective advantage over males when considering numerous pathologic features such as lesion size, cell death, and recovery/neurogenesis. However, others have shown no sex difference, or an advantage for males over females, effects that appear to be model dependent.41,44,55
Deeper investigations into the root causes of sex-dependent injury and recovery of tissue have revealed secondary physiologic changes that may prove informative. Some of the earliest studies of sex dependence investigated TBI-induced edema by measuring cerebral water content. In a series of studies, Roof et al59,60 showed that adult SD rats elaborate edema after CCI, with males showing a greater increase than females. This difference was attributed to the action of progesterone, because males who received progesterone 1 hour following injury exhibited levels of edema similar to females. 60 Another group followed the evolution of post-injury edema attributed to changes in blood brain barrier (BBB) permeability. Male rats elaborated greater edema than females following a weight drop TBI. 61 In addition, the peak of edema occurred at 5 hours post injury for males, but was delayed to 24 hours for females. Edema resolved by 5 days post injury in females, whereas it persisted in males at 5 days post injury, the last day assayed. Studies from the same group showed administration of estrogen or progesterone 30 minutes after injury reduced brain water content following TBI. 62 Sex divergence of post-TBI hyperthermia has also been reported. Males remained hyperthermic for 5 days post TBI, whereas females recovered normal body temperatures after 1 day. 63 Post-injury brain edema and hyperthermia are important prognosticators of impairment64,65 that affect males to a greater extent than females.
In association with brain swelling and edema, hypoperfusion with reduction of cerebral blood flow (CBF) is a hallmark of severe TBI that confers significantly worse outcome. 66 One group has reported sex differences in CBF using the FPI model of TBI in piglets. They were able to show that pial arteries in the parietal cortex constricted in a sex-dependent manner following TBI, where males were more constricted than females. 67 In subsequent studies, a sex-dependent increase in adrenomedullin was shown to dilate the pial artery to a greater extent in females, by acting on arterial potassium channels.67,68 Various pressors were then administered to assess restoration of CBF. Sodium nitroprusside was shown to increase CBF compared with vehicle-treated TBI animals of both sexes but failed to improve outcome. 69 Phenylephrine was shown to upregulate endothelin-1 (ET-1) in a sex-dependent manner and to act on potassium channels and regulate vasodilation.70–72 ET-1 itself was upregulated in a sex-dependent manner following TBI, and phenylephrine further worsened CBF in males, but improved in females.71,73 Dopamine was investigated in the same model and blocked TBI-induced upregulation of ET-1 in both males and females, protecting them from hypoperfusion. 74 Norepinephrine and epinephrine were shown to have differential effects, with norepinephrine augmenting recovery of CBF in females but not males, whereas epinephrine improved CBF for both infants, but failed to confer the same protection for juvenile males.75,76 The choice of pressors used to manage cerebral perfusion in severe TBI may be greatly influenced by sex, a finding with important translational implications.
Many groups have reported on the neuroinflammatory response of the brain after TBI, which is recognized as a crucial component of injury, repair, and recovery. However, relatively few studies have reported sex divergence of TBI-related neuroinflammation. 77 Widespread elevation of the neuroinflammatory markers IL-6, TNFα, and MCP-1 and activated microglia were reported in both sexes following CCI. 78 When OVX female mice were resupplemented with exogenous estrogen, reduction of proinflammatory IL-6 and MCP-1, an elevation in anti-inflammatory IL-4 was shown in comparison with males, but it made no difference in TBI lesion size. 78 Taylor et al 63 also profiled TNFα, IL-6, and IL-1β after TBI, and found female rats expressed more cortical IL-6 than males, but males expressed more cortical TNFα and IL-1β. A study by Villapol et al 52 did more detailed profiling of microglial morphology and activation state showing male mice exhibited more activated microglia 1 day following CCI than females, as well as greater peripheral macrophage infiltration at 1 and 3 days post CCI. In addition, more astrogliosis was seen by glial fibrillary acidic protein (GFAP) immunofluorescence in males compared with females at 1 and 7 days post CCI in the same study. Jullienne et al, 44 however, demonstrated greater GFAP expression in female mice at 1 day post CCI but no difference between the sexes at 7 days post injury.
Differences seen in neuroimmune cell populations following injury (above) may have resulted from, and contributed to, differential cytokine expression following injury. Females expressed more IL-1β at 4 hours post injury than males, whereas males expressed more anti-inflammatory TG-Fβ than females at 1 day post injury. 52 In addition to cytokines, proinflammatory enzymes COX-2 and iNOS play important roles in neuroinflammation and oxidative stress in the brain. Gunther et al 57 found that TBI induces a greater increase of COX-2 in male rats, and a greater increase in iNOS in females. Although females produced more iNOS, they did not report a sex-dependent difference in oxidative stress by measuring 3-nitrotyrosine. However, Lazarus et al 79 measured protein carbonylation, a quantifiable consequence of oxidative stress, after CCI and found region-specific increases in males that were significantly greater than females. Although the neuroinflammatory response following experimental TBI is highly dependent on injury model, sex appears to play a crucial role in nature and evolution of the post-TBI neuroinflammatory response.
Few studies have explored sex differences in TBI pathogenesis related to individual genes and pathways associated with brain injury and recovery. Brain-derived neurotrophic factor (BDNF) is an important trophic factor that helps maintain existing neuronal function, and supports new neuron growth, among many other functions. 80 Chen et al 81 investigated BDNF expression in adult rats following CCI and found spatial and temporal sex-based differences in BDNF protein levels. Males and females showed significantly different increases in BDNF within frontal cortex ipsilateral to the impact site and in the contralateral hippocampus, respectively, while both males and females showed reductions in BDNF in the ipsilateral hippocampus.
Wagner et al 82 examined the sex-dependent changes to the dopamine system following TBI and showed the dopamine transporter (DAT) expression to be reduced in cortical-striatal circuits in rats of both sexes, but relatively more so in males.
Hormonal status can have profound effects on behavior and pathology. 83 To test the effects of TBI on hormone levels in the brain, Meffre et al 84 performed CCI to produce diffuse TBI in adult rats and measured endogenous hormones by gas chromatography/mass spectrometry. They found increases in progesterone and its metabolites following TBI that varied by sex, location, and time following CCI. The authors suggest that progesterone is reduced into its metabolites locally within the brain following injury, and that it may contribute to differences in recovery between males and females.
Conclusion
It is still largely unknown what features underlie variation of risk and susceptibility to long-term adverse outcome among concussion patients as well as those who experience repetitive head impacts (RHI). However, epidemiologic and animal studies have given us clues regarding specific pathologies and molecular pathways to explore. The heterogeneous nature of injury complicates the situation, but there remains optimism toward finding better treatment strategies. 85
At the moment, there is clinical and preclinical evidence that sex may play a role in susceptibility to worse outcomes following brain trauma. Although it is not yet known what aspect of biological sex confers this increased risk, the knowledge of its existence provides a path for investigation into its molecular underpinnings. However, most animal studies have studied males only.86,87 In the relatively few studies that have investigated sex in relation to experimental TBI, sex-specific hormones have mainly been the focus. Progesterone has been most commonly associated with beneficial effects, but translation to human studies has not been shown beneficial in any clinical trial. 85 With the National Institutes of Health (NIH) acknowledging and mandating research address sex as a biological variable, hopefully, the discovery of new mechanisms to target will be expedited.
Although post-concussion syndrome (PCS) can be assessed quite readily in the clinic, it is still very much unclear why some people recover rapidly, but others do not. One aspect that has been consistently shown to exacerbate outcome is repeated injury.19,88–90 However, only 2 studies that have examined sex differences in experimental TBI employed more than a single impact.36,37 Moreover, a recent systematic review of experimental RHI found all published studies exclusively used male animals. 91 Studies of human subjects show that PCS is more likely to persist after multiple concussions, and mounting evidence suggests RHI may contribute to long-term disease and dysfunction, even in the absence of recognized concussion.92–94 In light of these clinical realities, there seems to be some degree of disconnect from current preclinical research approaches.3,92,95 Uncovering mechanisms underlying RHI pathology and dysfunction is an imperative goal because of the tremendous “window of opportunity” that exists to treat the disease, or possibly prevent it altogether. Identifying risk factors on the population and molecular level should provide legitimate targets to intervene and steer at-risk individuals toward recovery from head trauma.
To progress toward these goals, we suggest the following:
Inclusion of both sexes in all preclinical TBI research to determine sex dependence of injury and recovery.
Increased use of newer and more translatable models for mild TBI that do not require craniotomy and direct brain impact.
Increased focus on multiple impact models to reveal how behavioral and pathophysiological sequelae differ from isolated concussion.
Increased use of advanced imaging techniques to facilitate reverse translation of the human condition into animal models for more robust and relevant findings.
Footnotes
Author Contributions
TGR performed all searches and all research related to the manuscript, as well as wrote the main manuscript text and table. MLL reviewed and edited the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the National Institutes of Health, National Institute of Neurologic Disorders and Stroke (R01 NS082432 and F31 NS105450-03).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
