Abstract
Stem cells are ubiquitously found in various tissues and organs in the body, and underpin the body’s ability to repair itself following injury or disease initiation, though repair can sometimes be compromised. Understanding how stem cells are produced, and functional signaling systems between different niches is critical to understanding the potential use of stem cells in regenerative medicine. In this context, this review considers kynurenine pathway (KP) metabolism in multipotent adult progenitor cells, embryonic, haematopoietic, neural, cancer, cardiac and induced pluripotent stem cells, endothelial progenitor cells, and mesenchymal stromal cells. The KP is the major enzymatic pathway for sequentially catabolising the essential amino acid tryptophan (TRP), resulting in key metabolites including kynurenine, kynurenic acid, and quinolinic acid (QUIN). QUIN metabolism transitions into the adjoining de novo pathway for nicotinamide adenine dinucleotide (NAD) production, a critical cofactor in many fundamental cellular biochemical pathways. How stem cells uptake and utilise TRP varies between different species and stem cell types, because of their expression of transporters and responses to inflammatory cytokines. Several KP metabolites are physiologically active, with either beneficial or detrimental outcomes, and evidence of this is presented relating to several stem cell types, which is important as they may exert a significant impact on surrounding differentiated cells, particularly if they metabolise or secrete metabolites differently. Interferon-gamma (IFN-γ) in mesenchymal stromal cells, for instance, highly upregulates rate-limiting enzyme indoleamine-2,3-dioxygenase (IDO-1), initiating TRP depletion and production of metabolites including kynurenine/kynurenic acid, known agonists of the Aryl hydrocarbon receptor (AhR) transcription factor. AhR transcriptionally regulates an immunosuppressive phenotype, making them attractive for regenerative therapy. We also draw attention to important gaps in knowledge for future studies, which will underpin future application for stem cell-based cellular therapies or optimising drugs which can modulate the KP in innate stem cell populations, for disease treatment.
Keywords
This review builds on our previous contributions in this area in stem and developing cells for example,1-11 by integrating the latest discoveries in the rapidly expanding and important fields of tryptophan (TRP) metabolism through the kynurenine pathway (KP), and it’s known functions in stem cells. Where relevant and significant, some sections summarise key physiological roles of serotonin/melatonin in specific stem cells, however the substantial focus is on the KP. Subsections explore the latest knowledge of KP in different types of stem cells, which are of significant interest and attention to the research community in regenerative medicine, precision medicine and therapies to modulate the effects of prolonged inflammation.
Kynurenine Pathway
TRP in the diet and initial catabolism
TRP is 1 of 9 essential amino acids that cannot be synthesised by cells and must therefore be acquired elsewhere, typically through dietary intake. Once TRP has been ingested and absorbed, it travels through the peripheral circulation in its free form or bound to albumin. 12 TRP is incorporated into proteins but is also a precursor for a variety of metabolic pathways that can result in KP metabolites, and a separate pathway culminating in formation of the neurotransmitter serotonin and sleep-wake regulator melatonin, and tryptamine13,14 (Figure 1). However, the KP accounts for the metabolism of more than 95% of TRP, 15 and of that 95%, approximately 90% occurs in the liver and 5% in distal sites.

Schematic diagram of the Kynurenine pathway. Tryptophan (TRP) is primarily metabolised by the KP (~95%), with only a small amount converted into serotonin and melatonin (~3%), tryptamine or utilised in protein synthesis (~1%). The metabolism of TRP through the KP is initiated by transformation to N’formylkynurenine followed by kynurenine (KYN). This first step is regulated by the enzymes indoleamine-2,3-dioxygenase (IDO-1 and 2) and tryptophan-2,3-dioxygenase (TDO2). N’formylkynurenine is degraded by arylformamidase (AFMID) to the main metabolite KYN. KYN is then converted to either anthranilic acid (AA), kynurenic acid (KYNA) or 3-hydroxykynurenine (3-HK). These conversions are controlled by the kynureninase (KYNU), kynurenine aminotransferases (KATs) and kynurenine 3-monooxygenase (KMO) enzymes, respectively. There is some evidence that KYNA can be further transformed into quinaldic acid, likely via dihydroxylation, although the majority remains unmetabolised.16,17 3-HK is converted to either 3-hydroxyanthranilic acid (3-HAA) by the action of KYNU, or to xanthurenic acid by KAT enzymes. 3-HAA is also formed though hydroxylation of AA. 18 3-HAA is an antioxidant (i.e. protective) towards certain types of reactive oxygen species (ROS),19 -22 but when exposed to intracellular metal ions (e.g. copper II+) it can be a pro-oxidant, forming ROS such as OH•. 23 3-HAA is further metabolised into 2-amino-3-carboxymuconic-semialdehyde by the enzyme 3-hydroxyanthranilate-3,4-dioxygenase (3-HAO), or cinnabarinic acid via autoxidation.24,25 2-amino-3-carboxymuconic-semialdehyde is catabolised down two pathways, being (1) metabolised to 2-aminomuconate-6-semialdehyde by 2-amino-3-carboxymuconate-semialdehyde decarboxylase (ACMSD), and then non-enzymatically to (neuroprotective) picolinic acid (PIC), or (2) non-enzymatically to (neurotoxic) quinolinic acid (QUIN), which is further metabolised toward the essential cofactor nicotinamide adenine dinucleotide (NAD) by quinolinate phosphoribosyltransferase (QPRT; several subsequent reactions are needed to produce NAD). Importantly, the physiological function and effects on cells of all the metabolites in the KP as a result of TRP metabolism are not same. There are both neuroprotective (KYNA, PIC, and cinnabarinic acid) and neurotoxic (3-HK and QUIN) metabolites. In the figure neuroprotective metabolites are marked in blue whereas neurotoxic metabolites are marked in red.
KP enzymes catabolise TRP into a variety of metabolites
As shown in Figure 1 IDO-1 is an enzyme that metabolises TRP into n-formylkynurenine, and whose expression is generally induced by the pro-inflammatory mediator interferon-γ (IFN-γ),26,27 LPS 28 and other cytokines. IDO-1 uniquely can regulate blood pressure via the formation of singlet oxygen, 29 and it’s expression is generally regulated by inflammation and the capacity of activated immune cells like T and Natural killer cells to release IFN-γ. Although other triggers of it’s expression have been described for example, interleukin-1β, 30 interleukin-631,32 and interleukin-10 (downregulation). 33 IDO-1 is typically found monocytes, 3 macrophages, and microglia, and has also been shown in non-immune cells such as neurons. 7 Another variant, IDO-2, is expressed in a range of tissues and like IDO-1 also metabolises TRP. It’s regulation is less described, but recent evidence shows it is upregulated in SARS-CoV-2 infection, 34 so whether other types of viruses and infections induce it’s expression, perhaps to augment IDO-1, is an open question. Another enzyme, tryptophan-2,3-dioxygenase (TDO2) is also capable of the same enzymatic reaction, yet crucially the enzyme expression tends to be constitutive and does not respond greatly to IFN-γ; conversely it can be induced by corticosteroids and tryptophan 35 and inhibited by several things including IDOs. 36 TDO2 is typically expressed in the liver 35 but has also been detected in the central nervous system37 -40 and placenta.
Catabolism of n-formylkynurenine by enzyme arylformamidase (AFMID) forms KYN, the first KP metabolite capable of being metabolised to 3 potential products – AA, 3-HK or KYNA. The action of KMO here is critical because it drives the formation of 3-HK, a QUIN precursor, whereas formation of metabolites AA and KYNA divert substrate away from QUIN production – a potent neurotoxin. 41 The enzyme QPRT mediates the first step of QUIN’s breakdown towards product for entry into the de novo pathway for essential co-factor NAD production. However QPRT is easily saturated at levels above 50 nM (shown in neurons 11 but not yet ascertained in other cells), therefore in circumstances of chronic inflammation, where QUIN levels are likely to continue to rise through KP activation, are therefore likely to pose a challenge to susceptible tissues and organs as QPRT will not be able catabolise QUIN fast enough to prevent its accumulation. This, and the aforementioned inability of the pathway to rebalance itself in chronic inflammation, mostly mapped in the context of neurodegenerative diseases, 42 pose a challenge to susceptible stem cell populations. While QUIN does not readily cross from the bloodstream into tissues such as the brain, 43 it can be carried into tissues by cells capable of synthesizing it for example, monocytes, and released locally, or alternatively QUIN can be synthesized in tissues expressing the required suite of enzymes by uptake of TRP, KYN or 3-HK, which readily cross into tissues.
Expression of KP enzymes in various cells of the body
Few cells are capable of expressing the entire suite of KP enzymes (Figure 1) – these include monocytes, 3 macrophages 44 and brain microglia, 45 although these will typically have negligible basal expression and require an external stimulus to prompt their upregulation, thereby having a typically inflammation or infection-associated response. Microglia are slightly different, in that they don’t appear to express TDO2, 46 and hence rely on indoleamine-2,3-dioxygenase (IDO-1) for TRP depletion. Foetal neurons also express IDO-1 with IFN-γ treatment as evaluated by PCR, 7 which was later confirmed via immunostaining in those cells and also in SK-NS-H glioblastoma cells and adult hippocampal brain tissue. 47 Expression of other enzymes including KMO, KYNU, KAT-II, ACMSD and presence of intracellular metabolite QUIN) was also confirmed. Cells such as astrocytes lack some enzymes such as KMO and IDO-1.6,48 The liver is the major site of KP metabolism (90% of the 95% that goes down the pathway), while the CNS is one of the secondary sites of the remainder of the metabolism. 49 Sex organs, 50 kidneys 51 and heart 52 are some additional sites where at least one KP enzyme has been found to be expressed.
KP activation to downregulate inflammation
As discussed, the KP is activated typically during inflammation by IFN-γ secreted by activated T and Natural killer cells, which induces the expression of IDO-1 and ultimately leads to an the process of accelerated and sustained depletion of TRP. Creation of kynurenines, thereby reducing bioavailable TRP for protein synthesis and proliferation of immune cells are critical in modulation of the immune response and restoring homeostasis.53,54 However KP activation has also been reported to contribute to cancer cells ability to escape the immune system. 55 Those KP metabolites that are ligand agonists of the Aryl hydrocarbon Receptor (AhR, see section below) provides a crucial link between KP metabolism and transcription of large numbers of genes. TRP depletion, and an immunosuppressive local microenvironment tends to only provide acute benefits, as chronic inflammation, most studied in the CNS,42,56 dysregulates the KP away from physiological, balanced levels of metabolites, and towards a focussed production of (only) the neurotoxin QUIN, whose levels quickly become supraphysiological.
Physiological actions of KP metabolites are well-studied in the CNS, and far less in other organs and tissues
In other vital organs such as the heart, recent studies are informing that KP metabolites can play important biological roles, as KYN was shown to positively regulate cardiomyocyte proliferation, and hence may play a regenerative role. 52 Future fundamental discoveries in other organs and tissues therefore appear likely. However due to clear dysregulation of the pathway in various diseases, most evidence of the physiological actions of KP metabolites comes from studies of the CNS. A competitive non-specific L-type amino acid transporter (leucine-preferring system L1 large amino acid transporter (LAT1) transfers the free form of TRP across the blood brain barrier.57,58 Once in the CNS, TRP may be further metabolised by resident cells, or as described above, KP metabolites for example KYN and 3-HK also enter tissues while QUIN is blocked from getting across the BBB. 59
As per Figure 1, TRP metabolites segregate into one of 2 categories – neuroprotective (KYNA, PIC) or neurotoxic (QUIN, 3-HK). Typically the ‘balancing act’ to produce kynurenines at levels where they will not cause localised cell toxicity is impeded by chronic inflammation, which favours 3-HK production (and production of further metabolites towards QUIN) at the expense of metabolites like KYNA, which is protective in the CNS by binding (neurotransmitter) NMDA receptors with higher affinity,53,60,61 thus reducing QUIN-induced toxicity. 56 During inflammation in the brain, it is macrophages, microglia and dendritic cells that produce most of the elevated levels of QUIN.62 -64 Astrocytes and neurons catabolise QUIN to NAD, however saturation of this system leads to the toxic accumulation of QUIN. 65 QUIN is a neurotoxin, gliotoxin, proinflammatory mediator, pro-oxidant molecule and has the ability to affect the integrity of the blood brain barrier. 41 QUIN can cause neuronal cell death,66,67 and even with chronic low levels causes ultrastructural abnormalities in neurons reminiscent of those found in dementia. 10 Among other KP products, PIC is a monocarboxylic acid and a neuroprotectant. 68 AA is important for NAD+ synthesis in neuronal cells but has also been shown to be neurotoxic. 5 3-HK is neuroactive 53 while 3-HAA, which can be derived from the oxidation of AA or from the hydrolysis of 3-HK 53 is a free-radical generator, 17 and can supress T cell proliferation. 69
KP and disease
With the 2 enzymes IDO-1/2 and TDO2 with differing expression profiles, the body has the means to precisely regulate bioavailable tryptophan in homeostasis, however with persistent, chronic inflammation there is a breakdown of this delicate balancing act, and in general, too much of the potent neurotoxin quinolinic acid (QUIN) is produced for the body to be able to be able to convert it to the essential co-factor NAD+. Increased degradation of TRP and an increased kynurenines have therefore been detected in many diseases or disorders such as HIV/AIDS, 70 diabetes, 71 cancer,72,73 coronary heart disease.74,75 In the CNS, KP is heavily associated with Alzheimer’s disease,76,77 amyotrophic lateral sclerosis,78,79 dementia, 70 Huntington’s,80,81 Multiple sclerosis,48,56,82,83 schizophrenia 84 and neoplasia. Depletion of tryptophan is also associated with aging, but substantial depletion of tryptophan is associated with neurodegenerative diseases.65,85 Tryptophan catabolites and their derivatives are utilised as a strategy to treat T helper-1 mediated autoimmune diseases like Multiple sclerosis. 86 Because of the role that kynurenine has in these diseases, modulating this pathway has therapeutic potential. 83 Kynurenines that are involved in the suppression of T cell proliferation appear to target immune cells when they are being activated, 54 in addition to this they likely work together to exert this effect. 87 QUIN and the KP are involved in both inflammatory and non-inflammatory neurological disease. 88
Aryl hydrocarbon receptor
AhR is a cytosolic transcription factor in the basic helix-loop-helix family first identified as being activated upon the signaling induced by dioxin toxicity. Activation occurs through ligands such as indole and several of its derivatives, 89 including KP metabolites xanthurenic acid (lower potency but is blood-brain barrier permeable), cinnabarinic acid, 90 and the potent KP agonist (KYNA).91,92 This results in translocation to the nucleus to bind specific target DNA sequences associated with diverse gene signaling responses for example, proliferation, defence and development. Recent evidence also suggests that contrary to previous belief, KYN does not function as a direct AhR ligand, 93 but instead as a precursor for the formation of highly active derivatives which can activate AhR at picomolar levels. 94 AhR activation regulates a diverse set of biological processes including metabolising environmental toxins, cellular oxidation/antioxidation and immune regulation in the CNS. 95
Immune regulation mediated by AhR can result in both anti- and pro-inflammatory effects,95 -97 as previously reviewed. 98 AhR activation induces IDO-1/2 expression,99 -102 which upregulates the KP by driving TRP catabolism into KYN, the precursor for metabolites that bind with high affinity to AhR. For example, IDO-1 activation leading to KYN binding to AhR which drives immunosuppressive signalling through protective cytokines (e.g. transforming growth factor-beta [TGF-β]) causing differentiation of T cells to regulatory T cells. 103 TGF-β also upregulates the AhR in mouse CD4 T cells, 104 while causing transdifferentiation of pro-inflammatory Th17 cells into regulatory T cells which helped resolve inflammation in 2 mouse models. 105 However, under certain circumstances signalling via the IDO/KYN/AhR can be detrimental by producing pro-inflammatory responses. AhR binds to the HIV-1 5′ long terminal repeat, activating HIV transcription and infection factors. 106 AhR activation by KP metabolites increases HIV-1 activation in blood mononuclear cells, and TRP metabolites stimulate viral reactivation up to ~5-fold compared to unstimulated control. 106
The IDO-KYN-AhR axis has been identified as a potential target for therapeutics. 107 For example, the transplant of dendritic cells overexpressing IDO into pro-atherosclerotic mice was associated with a reduction in vascular inflammation and aortic lesions. 108 This was accompanied by increased KYN levels and AhR expression, and the effects on lesions were negated by the application of an AhR antagonist. 108 This highlights the positive feedback loop of this pathway in which AhR induces IDO but IDO/KP activation also promotes AhR expression which reinforces the discovery that several IDO inhibitors are unexpectedly also AhR ligands, and therefore may paradoxically increase IDO expression.109,110
Stem Cells
Stem cells are defined as undifferentiated cells which: (1) are capable of self-renewal, (2) differentiate into other cell types via asymmetric cell division (potent), (3) usually arise from a single cell and (4) are capable of generating all cells within their lineage(s).1,111 Stem cells can be classified according to their developmental potential within the hierarchy (Figure 2). Totipotent stem cells located in zygotes or morulae can give rise to whole organisms, namely the organs of the body, the extraembryonic tissues (such as the placenta), and the germline. 112 Similar to totipotent stem cells, pluripotent stem cells can give rise to all types of cells of the 3 germ layers except for trophoblasts. Multipotent stem cells can give rise to multiple cell types that may reside within specific tissues or organ. 113 Lineage-restricted precursor cells are committed progenitors that typically reside in a specific tissue/organ and are primarily restricted to generating a specific cell type.
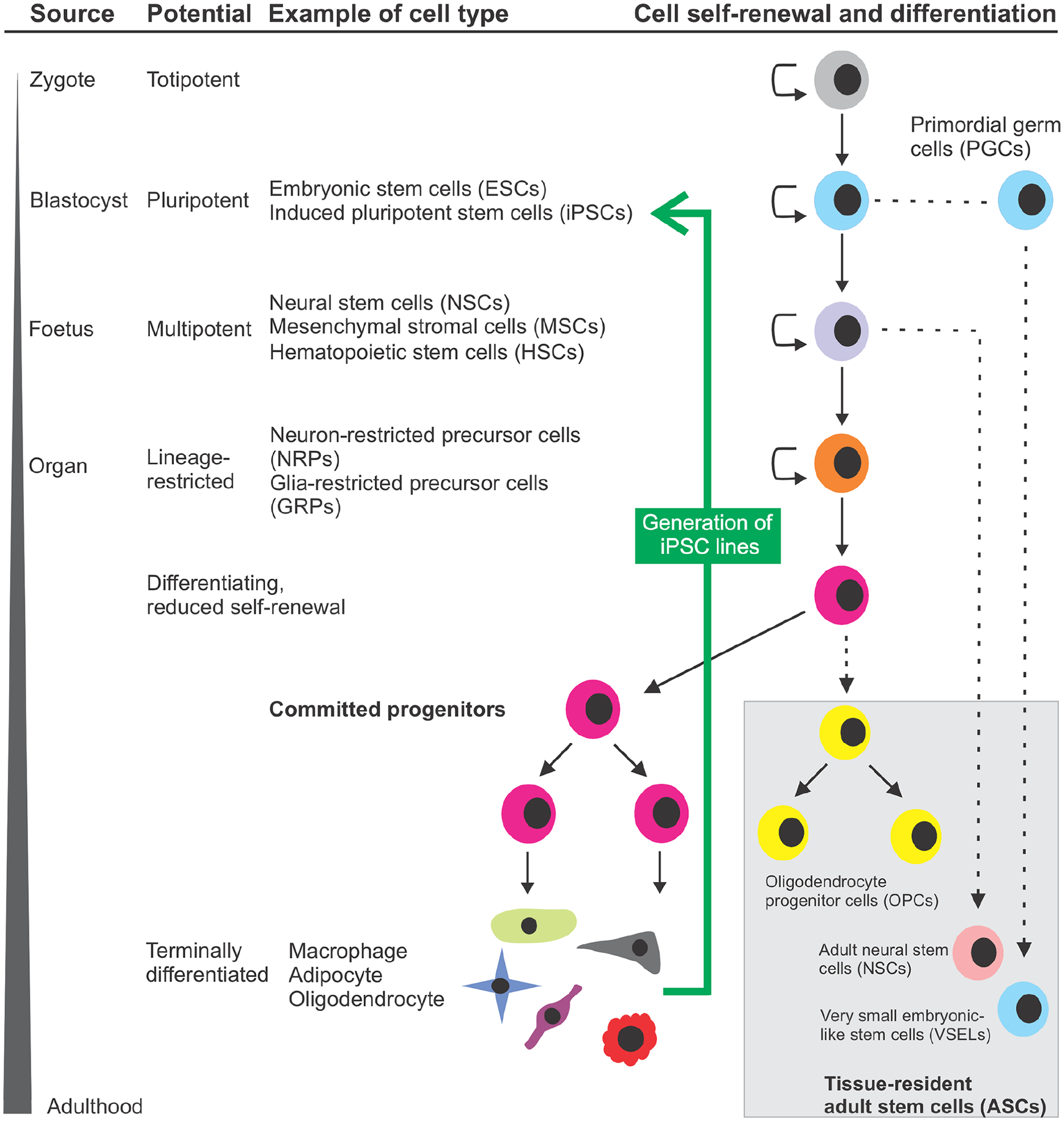
Schematic diagram demonstrating the hierarchy of stem cells occurring in the body during development, and their progressive fate restriction before terminal differentiation into cell types. A zygote is a fertilized egg that has totipotent capability, facilitating differentiation to any cell type and a whole organism. The zygote further develops to form a blastocyst which consists of trophoblast, blastocoel and inner cell mass (ICM). The ICM can give rise to pluripotent embryonic stem cells (ESCs), which after further rounds of symmetric division (indicated by semicircle arrows) can give rise to primordial germ cells (PGCs), which are precursors of gametes and can generate pluripotent Very Small Embryonic-Like Stem Cells (VSELs) which persist into adulthood, express markers of ESCs and are typically quiescent. ESCs are capable of differentiation into any differentiated cell type via progressive fate restriction (asymmetric cell division) to specific, multipotent stem cells, including hematopoietic stem cells (HSCs), mesenchymal stromal cells (MSCs) and neural stem cells (NSCs). These multipotent cells arise during development, can differentiate into limited specialized cells within the specific lineage, and are retained as tissue-resident adult stem cells (ASCs; grey shaded region at the bottom right). ASCs are undifferentiated cells derived from adult tissues or organs, which are characteristically quiescent, reflecting their innate characteristics of genomic stability underlying their long life, typically proliferating or mobilising only when required for replacement of cells lost over the lifetime of the organism or when tissues are injured or diseased. Lineage-restricted precursor cells are committed to differentiating into cell types of a specific lineage, but display reduced multipotency. Examples include neuron and glia-restricted precursor cells, which are derived from NSCs. Committed progenitors tend to differentiate into specific cell types such as macrophages, adipocytes and oligodendrocytes. Examples include oligodendrocyte progenitor cells (OPCs), which are uniformly distributed throughout the parenchyma of the adult CNS and retain the ability to proliferate and terminally differentiate into mature oligodendrocytes, and neuroblasts, which are very primitive, committed neurons. The final cell type considered – induced pluripotent stem cells (iPSC) can be generated from somatic cells (green arrow, intended to demonstrate that this process works against the natural flow of stem cell progressive fate restriction and lineage commitment), via genetic 114 or more recently, non-genetic means. 115 Solid lines with arrows represent progressive fate restriction to defined types of stem cells as development proceeds, whereas dotted lines represent different types of stem cells of varying differentiation capacity. This figure was created using the CorelDraw graphics suite.
Stem cells can be derived from embryos or adult somatic cells within tissues and organs. Embryonic stem cells (ESCs), also known as blastocytes, are derived from the inner cell mass of a developing blastocyst of the embryo and can differentiate into mature cells of all 3 germ layers. Despite their profound developmental potential, there is still reservation in using ESCs for specific purposes including cell-based therapies, because of the associated ethical, moral, and technical issues such as immune-mediated rejection. Therefore, there has been increasing interest in the therapeutic application of adult stem cells (ASCs) and more recently, induced pluripotent stem cells (iPSCs; considered below), particularly where they can be isolated from the patient for potential self-therapy/personalised medicine.
Adult tissues and organs retain small populations of ASCs, which are most relevant to this review, with the most widely studied types including hematopoietic stem cells (HSCs) and mesenchymal stromal cells (MSCs). Significant research attention has been focussed on assessing how ASC populations respond to disease, or how they can be harnessed to fight disease, including replacing dead or dying cells. Most ASCs, including multipotent stem cells, are metabolically quiescent in a normal, healthy state, yet capable to differentiating and proliferating under cellular stress to combat tissue injury. 116 These stem cells are conventionally classified according to the lineage differentiation repositories. 117
In order to further develop the potential of the stem cell therapy in various diseases, it is crucial to understand the metabolic signaling pathways they express. Within these developmental and adult stem cell populations, the KP plays an important role in regulating metabolism of TRP, and is in turn regulated by a diverse repertoire of activating signals. We now consider the characteristics of several types of stem cells and the role(s) and the impact of KP signaling (and possible dysregulation) on stem cell health and function, which will be discussed in the following sections in further detail. We lead off with development and the emergence of embryonic stem cells.
KP in embryonic stem cells
In pregnancy, the KP is critical to maintain an immunosuppressive, tolerogenic microenvironment at the maternal/foetal interface. For example MSCs in the placenta and umbilical cord express KP enzymes, depress TRP concentrations and in turn, T cell proliferation (see section Mesenchymal stromal cells and the KP). While this demonstrates the importance of the KP during embryonic development per se, further consideration needs to be made about KP catabolism in ESCs. ESCs are a pluripotent cell population located in the inner cell mass of the developing blastocyst. Understanding the signaling pathways that regulate the developmental programming that underpins their differentiation and other processes is vital for expanding our understanding of embryonic development. Human ESCs express key KP enzymes including the initial rate-limiting enzymes IDO-1/2 and TDO2.118,119 Activation of the KP in ESCs is linked to their self-renewal/proliferation, with IDO-1 activity associated with their maintenance in an undifferentiated state, and KAT-2 activity (forming due to broad substrate specificity the product 2-aminoadipic acid; 2-AAA) with ectodermal differentiation.119,120 Expression of self-renewal genes in ESCs has been linked to the activation of AhR, which is activated by several KP metabolites. 119 Additionally, KP activation has been identified as a marker of primed pluripotent ESCs, being downregulated as they enter a naïve state. 121 Together, these findings suggest that that regulation of the KP plays an essential role in ectodermal differentiation and embryonic development, and there is evidence that KP dysregulation/metabolite production in ESCs is associated with the development of developmental disorders. For instance, it is hypothesised that KP activation, and the reduced availability of TRP, are associated with aberrant development associated with reduced serotonin synthesis. 118 However, several important questions remain unanswered concerning ESCs and the KP. For example, the response of ESCs to IFN-γ is unknown, a topic that is highly relevant in the context of infection or inflammation during pregnancy.
We now shift to consideration of ASCs or ASC-derived cells, that are most studied or are emerging in the context of studies involving the KP. This covers all of the identified stem cell types discussed below.
Very small embryonic-like cells
Other types of ASCs are less well characterised and possess unique challenges in their identification and quantification using for example, flow cytometry. VSELs derived from adult tissues were recently suggested to hold promise for future regenerative medicine approaches as they are early developmental stem cells that express pluripotency and ESC markers. 122 These unique, rare, very small (5-7 µm in human) cells are also highly quiescent due to the deletion of regulatory sequences for specific paternally imprinted genes such as at the Igf2/H19 locus. Therefore, these cells are protected from insulin and insulin-like growth factor stimulation, preventing them from proliferating and differentiating in a normal, non-stressed state. 123 A recent study has shown that VSELs successfully differentiated into cardiomyocytes when treated with 5-Azacytidine in vitro. 124 Although VSELs have been utilised in emerging fields of clinical applications, the potential of these cells for bulk propagation in vitro, development into a variety of differentiated cell types, and ability to fully integrate with cells within the site of tissue injury are yet to be investigated, as is the possible expression of the KP in these cells.
Induced pluripotent stem cells – significant promise yet significant questions remain
iPSCs differ dramatically from all the other stem cell types described, in that they are artificially-derived from adult somatic cells that have been genetically reprogrammed in order to acquire pluripotent properties mimicking those of ESCs. This approach, first reported in mouse cells 125 before human cells in 2007 by Shinya Yamanaka and colleagues, 114 has revolutionised the study of diseases, as once produced, iPSCs can then be differentiated into a desired cell type for further study.
Previously, rare diseases presented a considerable challenge to researchers seeking to understand pathogenesis, underlying risk factors, and potential genetic underpinnings, and their study often required co-operation from researchers across the globe for access to rare samples. Now, for research purposes, it is possible to derive iPSC lines from patients in standard laboratories, drive them to commit to development into cell types of interest affected in disease, and perform analysis comparative to relevant aged-matched controls. 126 In many cases, this has allowed cell types of interest to be cultured that would be otherwise difficult to isolate. 127 The use of iPSCs also overcomes the limitation of cultivating cells or cell lines from post-mortem patient tissues, that due to their nature may only reflect the end state of the disease, and therefore may not yield sufficient definitive biological clues to the initial underpinnings of disease. iPSCs can also be used as the basis for development of advanced 3D cell culture models including organoids for example, of the brain. 128
Production of new iPS lines for use in research is commonplace, but requires testing prior to use in research. This typically focuses on demonstrating pluripotency, karyotype, and differentiation capability of each line. To improve rigor and reproducibility of iPS lines produced for research, the International Society for Stem Cell Research (ISSCR) in June 2023 published a set of standards and core principles. 129 Despite considerable advances in the cell culture and genetic technologies underlying their production, iPSCs or iPS-derived cells are still limited in clinical application due to stringent additional regulatory frameworks surrounding their use. These clinical therapy requirements commonly add additional quality control measures including investigating genomic stability, human leukocyte antigen (HLA) testing, single nucleotide polymorphism (SNP) analysis and increasingly RNA sequencing (RNAseq) analysis. These requirements need to be carefully considered by manufacturers of potential Good Manufacturing Practice (GMP) grade cells, and solutions thoroughly tested, therefore requiring considerable financial, labour and resources to satisfy typically multiple regulatory bodies,130-133 as well as consideration as to how procedures can be standardised across different sites 134 for larger studies.
Species-specific differences in biology are well-recognised 135 and may underpin why translation of exciting findings from pre-clinical animal models to human clinical trials often fail,136,137 prompting attempts to improve and standardise procedures for the conduct of pre-clinical testing regimes. 138 Until recently most studies investigating KP expression using primary human cells have been limited to those using easy-to-access, minimally-invasive sources of cells, like blood 3 or using developing brain cells,4,6 -10,63,139 however potential differences in signaling system expression upon differentiation and maturation of cells, and also between organism embryonic or foetal development (stem/precursor cells within), and adult stem cells residing in niches and iPS-derived cells, need to be appreciated and not assumed there is no difference. The use of high-throughput transcriptomics and other genetic approaches has helped researchers in various fields towards answering these questions,140 -142 however it should be recognised that there can be intrinsic biases favouring detection of highly expressed transcripts at the expense of modestly-expressed ones; and basal KP expression in many cells may fall into this latter category. Also, RNA from heterogenously-expressing samples/derived from multiple cell types will essentially be averaged in output datasets. 143
Considering these limitations, genomic analyses alone will be an insufficient readout as to how a cell type physiologically functions. It will still be important for researchers using iPS technology to confirm that their cell lines express functional signaling systems in the way that existing, well-characterised primary isolated cells do. This characterisation can be cell type-appropriate for example, for neurons, electrophysiology is appropriate while for microglia, demonstration of phagocytotic ability is important. Despite the technology for producing iPSCs being approximately 15 years old, being constantly enhanced and broadly available, there are very few published studies of the KP in iPS or iPS-derived cells, and they have mostly been disparate, typically focusing on certain components of the pathway rather than undertaking an entire screen. Of those, IDO-1 expression has been demonstrated in human primary and iPS-derived microglia. 144 Components of the AhR signaling were confirmed in iPSCs and iPS-derived motor neurons, and both KYN and KYNA were found to regulate AhR expression. 145 The most comprehensive study compared human pluripotent stem cells with their iPS counterpart, finding in iPSCs that TRP supplementation (to several hundred µM) enhanced proliferation through the formation of n-formylkynurenine, and levels of intermediate for example, KYN and end stage (e.g. QUIN) were decreased or unchanged, respectively. 146 As significant basal protein expression of IDO-1 (TDO2 not tested) was observed in these cells and negligible AhR, it suggests that continual KP activation, and the likely production of specific non-AhR ligand metabolites like n-formylkynurenine is important to their physiological functioning.
Hematopoietic stem cells
Residing in the developing foetus and primarily localised in adult bone marrow (but also umbilical cord blood) is a population of multipotent stem cells, HSCs, which can also be mobilised into the blood.147 -149 They are a rare cell type, comprising ~0.01% of bone marrow cells, 150 yet can transplanted cells are capable of reconstituting the entire blood lineage of lethally irradiated organisms. In humans, CD34 is a primary marker for adult bone marrow HSCs,151,152 with other markers including VE-cadherin, CD117, CD90, CD45, and CD105.151,153 The main function of HSCs is in haematopoiesis, the process in which they differentiate into all classes of blood cells including megakaryoblasts, erythroblasts, myeloblasts, lymphoblasts, and their respective lineages (Figure 3). As precursors of cells comprising the immune system, understanding the biology of HSCs is of significant clinical interest in autoimmune diseases, transplantation biology, and myelodysplastic syndromes. 154 Due to their relatively non-invasive accessibility and knowledge built up over the past few decades which led to their use in stem cell transplants, they have been one of the most intensively studied stem cell types.

Schematic diagram demonstrating the hierarchy of bone marrow-derived HSCs occurring in the body during development and throughout adulthood, and their progressive fate restriction before terminal differentiation into myeloid and lymphoid-derived cell types. HSCs are the only cells within the niche that are capable of both multipotent differentiation (into all blood cell types) and self-renewal. Multipotent progenitors can be further subdivided based on their proliferation type. Long term self-renewing HSCs are quiescent cells that self-renew indefinitely to maintain the pool of blood cells needed throughout life due to turnover of differentiated cells. Short term HSCs are progeny generated from long term HSCs, and tend to be highly proliferative and generate multipotent progenitors that have lost their self-renewal capability 155 but ultimately produce the repertoire of differentiated blood cells depicted. Figure adapted and altered from the original source 156 and used under creative commons BY 4.0 license (http://creativecommons.org/licenses/by/4.0).
Tryptophan and the KP in HSCs
KP metabolites have myriad effects on HSCs and their lineages, and modulation of the bidirectional interactions between these is relevant in treating various medical conditions. Given the role of HSCs in haematopoiesis (Figure 3), a process that is vital for the generation of immune cells and an effective immune response, we will now discuss the known involvement of HSCs in TRP metabolism, and the impact of KP modulation on HSC behaviour in healthy conditions and in disease.
The effects of IFN-γ and IDO on HSCs
Very little is known about the basal expression of KP enzymes in HSCs. Unstimulated HSCs do not appear to play a major role catabolising TRP into KYN, with one study detecting no IDO mRNA or protein in adult human-derived CD34+ cells in vitro. 157 However, two independent studies have shown that cultured CD34+ HSCs produce tiny amounts of KYN,158,159 suggesting either low-level TDO2 or IDO-2 expression, with the more recently identified IDO-2 isoform likely to be undetected in earlier studies. TDO2 and IDO-2 expression in HSCs must be directly assessed to establish this, as the HSC cultures in these studies were between 85% and 92% pure, and it is, therefore, possible that other contaminating cell types produced some or all the detected KYN.
While HSCs basally have no/low-level basal IDO/TDO2 expression, IFN-γ potently stimulates KYN production and induces an elevated KYN/TRP ratio in cultured CD34+ HSCs, 158 through induction of IDO. 157 Interestingly, one study observed that while IFN-γ stimulated the generation of IDO mRNA in CD34+ HSCs, this was not accompanied by IDO protein expression, suggesting post-transcriptional regulatory mechanisms that are not understood. 157 In addition to the direct effects of IFN-γ in the activation of the KP within HSCs, the associated elevated levels of various KP metabolites also modulates HSC behaviour, having an active role in maintaining the equilibrium between the initiation and suppression of the immune response through their effects on HSC proliferation, differentiation, and apoptosis.
IFN-γ has a marked impact on hematopoietic differentiation. In steady-state haematopoiesis, HSC self-renewal and differentiation are in balance to maintain HSC quiescence and differentiation into various types of blood cells through a combination of lymphopoiesis, myelopoiesis, and erythropoiesis. Exposure of HSCs to IFN-γ signaling can alter this hematopoietic balance by biasing differentiation towards a myeloid lineage at the expense of self-renewal and differentiation to the lymphoid and erythroid lineages. 160 However, neither IDO-1−/− or IDO-2−/− mice exhibited defects in embryonic development of hematopoietic differentiation, 161 suggesting that IDO induction may not play a major role in this cellular differentiation process, however this would not there being important roles at an adult stage.
Influence of the 5-HT pathway on hematopoietic stem cells
TRP is also catabolised by another enzyme, tryptophan-hydroxylase (TPH) in the serotonin (5-HT) pathway to form 5-HT and melatonin. The resulting 5-HT, in turn, exerts effects on progenitor cells during haematopoiesis. 162 While only a minor fraction of available TRP is catabolised in this pathway, 163 evidence is emerging demonstrating the indirect effects of these 5-HT pathway end products on both the KP and haematopoeisis associated with a reduction in available TRP concentrations, and through a direct effect of 5-HT on HSCs. One such study, using a TPH−/− mice model, demonstrated the function of 5-HT in positively influencing HSC survival during embryonic development. 164 Similarly, in vitro stimulation of cultured HSCs derived from the human umbilical cord blood with 5-HT increased proliferation while also exerting anti-apoptotic effects. 165 Chronic infections have been shown to divert available TRP towards the KP via IFN-γ upregulating IDO-1 resulting in reduced 5-HT production, modulating these effects of 5-HT on HSCs.166,167 However, while these studies demonstrate the importance of 5-HT on HSCs, they primarily focus on embryonic HSCs during early development and therefore further research needs to be undertaken on the role of 5-HT in influencing the activity of HSCs in the adult human bone marrow niche.
Disease relevance
The interactions of the KP with HSCs are increasingly being understood in the context of hematopoietic diseases. This is particularly relevant in the context of immune diseases, as hemopoiesis is integral in the production of immune cells (Figure 3). One such study showed that cytopenias, an umbrella term encompassing several conditions characterised by low blood cell count, are accompanied by KP activation and elevated levels of KP metabolites, and that KYN, KYNA, and 3-HAA potently inhibited HSC proliferation in vitro. 159 This may be at least partially driven by KP agonism of AhR, which is expressed by HSCs, 168 as the AhR agonist 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) inhibits the proliferation of HSCs and immature hematopoietic progenitor cells. 169 Conversely, applying the AhR antagonist StemRegenin1 promotes potent CD43+ HSC proliferation and differentiation into NK cells in vitro.170 -172 This finding is supported by observations of increased HSC proliferation in AhR knockout mice,173,174 and in their derived primary HSC cultures, 174 with this abnormal hyperproliferative phenotype severe enough to cause premature depletion of the HSC niche, with an overall phenotype of premature aging. 175 AhR therefore has a functional role in HSCs of negatively regulating proliferation, thus promoting their quiescent state.
The KP also plays a key role in successful HSC transplantation, which involves the transplantation of donor HSCs, either autologous or allogenic, for the treatment of certain haematological malignancies including leukaemia and Fanconi anaemia. 176 A potential complication common to allogenic HSC transplantation is GVHD, which is classified into acute (aGVHD) and chronic (cGVHD) forms. GVHD occurs when the donor T cells initiate an immune response against the recipient’s healthy cells. 177 The mechanisms affecting the severity of cGVHD are not fully understood; however, a recent study found that altered KP metabolic profiles occur in individuals with cGVHD, with several KP metabolites comprising the top-ranked 30 in severe disease cases. 178 Another study investigated if metabolic profiles identified before HSC transplantation influence the risk of developing aGVHD post-transplant and found that lower levels of KYN and IDO-1 activity pre-transplant function to identify patients at higher risk of developing aGVHD. 179 Similarly, it has been shown that lowering levels of TRP and KYN through the use of statins to induce IDO and enhance KYN catabolism reduces severity of cGVHD, indicating that pharmacologically targeting KP metabolites may be a therapeutic option post-HSC transplantation. 180 This is likely associated with the immune-dampening effects of KP induction.
Endothelial progenitor cells
Endothelial progenitor cells (EPCs; also called endothelial precursor cells) have been extensively investigated in the context of cardiovascular disease. Initial studies identified EPCs as hematopoietic CD34+/Vascular Endothelial Growth Factor Receptor 2 (VEGFR-2+) from bone marrow cells that primarily function to form new blood vessels. 181 Given their localisation in the bone marrow, similarities in cell surface markers, and evidence that they originate from a common progenitor, it is still debated if EPCs are a subtype of HSCs. Further review has shown that a subpopulation of EPCs do not originate from hematopoietic cells, and display clonal potential; these have been termed endothelial colony-forming cells (ECFCs).182,183 ECFCs are believed to play a large role in vascular repair.184,185
As endothelial dysfunction has been implicated in various cardiac diseases such as atherosclerosis, myocardial infarction and hypertension, 186 new approaches to improve endothelial repair are crucial to manage myocardium damage and prevent heart failure. Thus, there is growing interest in repurposing EPCs to replace damaged blood vessels. Clinical studies, although still in the early stages have shown promising results. For example, Losordo et al. 187 reported that intramyocardial injection of EPCs in a cohort of 167 patients with refractory angina, significantly reduced disease recurrence. A study by Vrtovec et al., 188 using a smaller group of patients with dilated cardiomyopathy demonstrated that stem cell transplantation resulted in increased left ventricular ejection fraction and improved ventricular remodelling. One of the primary challenges in the study of EPCs is their rarity, with estimates that they account for as few as 0.01%-0.0001% of PBMCs.189,190 This low abundance makes the isolation of sufficient numbers of EPCs challenging, particularly from blood samples.
Lack of characterisation of KP expression in EPCs
There are currently no studies that have characterised the KP in EPCs, or the direct effects of KP metabolites on EPC function. However, EPC proliferation has been shown to correlate with circulating KYN levels, with EPC number significantly decreased in both wild-type mice administered a selective IDO-1 inhibitor and in Ido−/− mice, that was rescued and increased above baseline by oral KYN administration. 191 Additionally, cultures of blood outgrowth endothelial cells containing ECFCs displayed aberrant network formation in the presence of tumour necrosis factor-α (TNF-α), for which KYN was partially protective. 191 It remains unclear by what mechanisms these effects occur, or if EPCs have the capacity to metabolise TRP or KYN, or subsequently KYN further down through the KP.
KP modulation is also associated with endothelial cell dysfunction and an altered capacity for endothelial remodelling/repair. IDO-deletion induced in pregnant mice resulted in the development of preeclampsia and these mice exhibited endothelial dysfunction and renal disorder. 192 Accordingly, Xiao et al. 193 demonstrated that enhanced endothelial IDO expression rescued endothelial apoptosis and attenuated vascular remodelling in pulmonary hypertension. An interesting study by Ryu et al. 194 found that intrastriatal injection of QUIN in vivo, resulted in increased vessel formation after 1 day, but led to the development of abnormal vascular remodelling after 7 days, and where striking microgliosis and astrogliosis was evident. These studies clearly demonstrate that the KP is an essential regulator of endothelial cell function. In this regard, regulating the expression of these TRP catabolites is indeed an attractive area for future investigations. KP catabolites could be key to the balance of endothelial apoptosis in vascular remodelling, and recruitment of EPCs in promoting angiogenesis for cardiac repair.
Mesenchymal stromal cells
MSCs are multipotent progenitors that can be isolated from a variety of developmental and adult tissues including adipose, umbilical cord, bone marrow, and tooth root.1,195 They have the ability to differentiate into a range of cell types; classically osteoblasts, chondrocytes, and adipocytes,196,197 although more recent studies indicate they are not restricted to a mesodermal fate, and can differentiate into other cell types including endothelial and neural cells.195,198-200
Debate still exists as to the most accurate name for these cells, with alternative/previously used names including mesenchymal stem cells, multipotent stromal cells, and medicinal signaling cells.201 -203 Additional difficulties exist in comparing studies based on differing approaches to isolate, expand and characterise cells. The International Society for Cell and Gene Therapy (ISCT) in 2006 published a position statement and criteria for defining the preferred name – mesenchymal stromal cells – and their functional properties. 197 The criteria included a combination of the presence and absence of specific markers (must express CD105, CD73 and CD90 while lacking CD45, CD34, CD14 or CD11b CD79α or CD19 and HLA-DR surface molecules, demonstrated adherence to plastic culture dishes, and differentiation capability to osteoblasts, adipocytes and chondroblasts in vitro. 197 These criteria were minimal, and hence it was not surprising that consideration of other criteria was later examined, especially for those cells grown under Good Manufacturing Practice (GMP) and intended to be used for clinical transplantation. For those purposes, there should ideally be a robust suite of marker characterisation that defines the stemness and lack of differentiation but also the immunological profile of the cells, which must be closely matched between host and graft cells. HLA-DR isotype expression is one such critical marker whose expression level was profiled between 130 clinical batches of bone marrow MSCs from two different facilities. 204 While lack of HLA-DR was included as a marker of MSCs, it was expressed when stimulated, such as by IFN-γ. 197 Lines expressing HLA-DR were found to maintain a mesenchymal phenotype, including multipotency and immunomodulatory capacity, which was independent of the HLA-DR expression level, which was variable between lines. Induction of HLA-DR expression with IFN-γ treatment may therefore be necessary for initiating the therapeutic properties of MSCs post-transplant.
The in vivo origin of MSCs remains a mystery and a point of contention within the research community. While some studies indicate MSCs are derived from perivascular mural cells, more commonly known as pericytes, cells associated with the vasculature,205 -209 in vivo fate tracing suggests that pericytes do not generate MSCs under normal or pathological conditions. 210 Regardless of their origin, their ability to differentiate into a myriad of cell types identifies MSCs as a promising therapeutic target. They possess the additional benefit of being easily obtained from many adult tissues without ethical issues which restrict the use of ESCs, and have low immunogenicity and tumorigenicity in comparison to other readily available stem cell types. 211 However, perhaps the most attractive function of MSCs is their unique ability to modulate both the local adaptive and innate immune systems, with the capacity to inhibit the proliferation, development, and function of immune cell types including NK cells, macrophages, dendritic cells, neutrophils, B, T and mast cells.212,213 These properties of MSCs have been exploited in several preclinical animal models and human clinical trials.214 -222 For example, Angiotensin-converting enzyme 2 negative MSCs transplanted into patients infected with severe acute respiratory syndrome coronavirus (SARS-Cov-2) and presenting with COVID-19-related pneumonia was shown to be a promising clinical management strategy. 223
Given their broad presence in various tissues throughout the body, multipotency, and innate ability to modulate the immune system, MSCs remain a leading candidate as an endogenous stem cell target in disease therapeutics. We will now discuss the role of the KP in modulating MSCs.
Evidence against IFN-γ as a prerequisite for the immunomodulatory effects of Mesenchymal stromal cells
Most of what is known about MSC regulation of the immune response has come from in vitro studies, which have shown the inhibitory effects of MSCs on T cell proliferation. 224 MSCs are also capable of inducing apoptosis of activated T cells, a process associated with converting TRP into KYN, and altering T cell activation and differentiation. 212 Interestingly, it has been suggested that MSCs cannot undertake immune cell suppression unless pre-stimulated by specific inflammatory cytokines, predominantly IFN-γ.225 -227 Conversely, MSCs with a mutated IFN-γ receptor can still suppress the immune response without IFN-γ stimulation. 228 Other studies also showed that pre-treating MSCs with other inflammatory cytokines including interleukin-1β, IFN-β, and transforming growth factor-α can induce their immunosuppressive effects.229 -231 Additionally, the molecules required for the priming of MSC immunosuppressive behaviour are species dependent, with inducible nitric oxide synthase (iNOS) being the most potent activator of these behaviours in murine MSCs. 2
Mesenchymal stromal cells and the KP
At the core of MSC-induced immunosuppression in adult tissue is the KP, and in particular IDO-mediated TRP depletion. 212 This mechanism appears to be conserved from development, as this immunosuppressive effect is also associated with MSCs in the placental and umbilical cord. IDO-1 is a crucial determinant of the immunomodulatory abilities of MSCs. 109 For example, MSCs suppress liver Th17 cells in an IDO-dependent manner, leading to attenuation of pathophysiology in a mouse model of liver fibrosis. 232 These researchers also demonstrated that IDO-1 is integral in the capacity of MSCs to alter the ratio of IL-17-producing Natural killer T cells (NKT cells) and regular NKT cells, and therefore suppress hepatotoxicity of NKT cells, a potential therapeutic target for IL-17-driven liver inflammation. 233 MSCs express the complete KP, 2 IDO-1 is upregulated in human MSCs in response to inflammation, 212 and MSCs express high levels of IDO-1 when stimulated by IFN-γ.2,234 Interestingly, IFN-β also induces IDO-1 in human bone marrow-derived MSCs, which produces an immunosuppressive phenotype. 231
Degradation of TRP into KYN may subsequently inhibit the proliferation of activated T cells. 234 The interplay between MSCs and T cells has been extensively studied, but MSCs have a significant impact on all kinds of immune cells, and this property gives them the potential to treat many diseases that display abnormal immune regulation. For example, Luk et al. 235 demonstrated that MSCs can inhibit B cell proliferation through IDO-mediated TRP depletion. They showed that the ability of MSCs to induce Interleukin-10 (IL-10) producing B cells was lost when MSCs were pre-treated with IFN-γ but could be recovered when TRP was supplemented to the culture, indicating that IDO activity plays a significant role not only on T cells but also on B-cells. 235 We will now discuss examples of the interplay between MSCs and the KP.
Most of what is known about MSCs has come from studying bone marrow-derived MSCs (BMSCs). 214 KYN production increases with age and plays a vital role in a number of age-related pathophysiological changes, including bone loss. 236 Within the bone marrow, KYN disrupts BMSC autophagy and increases cellular senescence in a dose-dependent fashion, a process that can be blocked by inhibiting ligand binding to AhR. 237 Furthermore, an age-related increase in KYN has been observed in murine BMSCs, where KYN reduces bone formation via modulating levels of CXCL12 and its receptors, and histone deacetylase 3 (Hdac3). 238 Similarly, KYN promotes osteoblast formation, possibly resulting from its inhibition of myeloid MSC proliferation and the consequent loss of osteoblast precursors. 239 On the other hand, there is evidence that the renal deficiency that results from kidney damage or loss increases the expression of AhR in osteocytes and elevates plasma levels of a range of metabolites including KYN and 3-HK.240,241
KYN can be further metabolised into the KP branch product KYNA, a metabolite well known for its neuroprotective role in the CNS. 242 The beneficial effects of KYNA extends outside of the CNS, with pre-treatment of human MSCs with KYNA shown to enhance immunosuppression in a mouse model of lipopolysaccharide-induced acute lung injury. 243 In contrast to KYNA, the KP metabolite QUIN is widely known for its excitotoxicity as a potent NMDA agonist.56,244,245 MSCs have been investigated as a potential therapeutic for QUIN-associated pathology. Transplantation of BMSCs into the damaged hemisphere of adult Wistar rats with QUIN-induced partial cerebellar degeneration was associated with improved motor performance and the prevention of memory deficit caused by cerebellar degeneration. 246 This neuroprotective action of MSCs against QUIN was also observed by Lin et al., 247 who demonstrated that human BMSCs enhance neural differentiation potential, provide neurotrophic support and produce an anti-apoptotic effect, making them a potential candidate as a therapeutic for Huntington’s disease.
MSCs have also been implicated with NAD-associated pathologies, with MSCs derived from progressive supranuclear palsy (PSP) patients exhibiting mitochondrial dysfunction resulting in inhibited NADH-dependent respiration. 248 Pi and colleagues 249 found that NAD is down-regulated during MSC replicative senescence, and could be used as a targeting strategy for the severe shortage of cells for MSC-based therapies. Additionally, in the MRL/lpr mouse model of systemic lupus erythematosus, the transplantation of human umbilical cord-derived MSCs alleviated lupus disease-associated pathologies through the promotion of splenic CD4+ T cell senescence via Sirt1, an NAD-dependent deacetylase signaling pathway. 250 These studies suggest that alterations to the KP and reduced NAD production may be associated with various disease-related pathologies.
Although not metabolised via the KP, serotonin and melatonin are also TRP metabolites involved in the regulation of MSCs. Melatonin-treated MSCs protect against endoplasmic reticulum (ER) stress-induced ischaemic diseases, and could be a key therapeutic strategy in ameliorating ER stress and autophagy. 251 Additionally, it was suggested that melatonin protects MSCs against replicative senescence during ex vivo expansion of MSCs for clinical application. 252 The protective effects of melatonin/serotonin on MSCs may be inhibited by KP upregulation, as their production may be reduced if an increased proportion of TRP is catabolised into KYN.
Mesenchymal stromal cells in disease and therapeutics
MSCs represent a clear therapeutic target due to their relative immune evasiveness, immune dampening activities, and their ability to be utilised in an allogenic setting, making them promising seed cells for cell therapy and tissue engineering.196,212 There has been considerable excitement around the use of MSCs in the treatment of neurodegenerative diseases, although several challenges remain including general inaccessibility and complex, incompletely understood pathologies, difficulties assessing disease stage and determining the optimal therapeutic time window. These challenges have contributed to roadblocks in the development of robust therapies. Additional advantages of using MSCs as stem cell therapies in the CNS arise from their ability to differentiate into neurons and glial cells 195 while being significantly more accessible than NSCs (which also have a restricted localisation). For example, MSC transplantation has been proven as a safe and effective stroke treatment associated with improvements in neurological deficits, motor function, spatial learning and memory, and quality of life. 253 MSCs also represent a potentially viable therapeutic target for MS treatment, with a study utilising EAE-induced mice showing that activation of IDO by IFN-γ in MSCs creates a TRP-depleted milieu that promotes immunosuppression and ameliorates disease progression. 2
MSCs are also therapeutic targets in cancer treatment due to their innate capacity to home/migrate directly to specific tissues. 196 However, ongoing preclinical trials suggest that the anti-tumour effects of MSCs are still controversial. For example, MSCs have tumoricidal effects on the liver, lung cancer cell lines and pancreatic tumours both in vitro and in vivo,254 -256 but can enhance progression and metastasis in breast and colon cancers.257 -260 The role of the KP in cancer cells is discussed later in this review.
Inflammation is a major pathogenic factor in liver fibrosis. 261 Since MSCs considerably impact the immune system, the interaction between MSCs and immune cells has been widely investigated in liver fibrosis. MSC treatment was found to promote Treg expansion and to significantly suppress the proliferation of Th17 cells in the liver of carbon tetrachloride-treated mice via the production of IDO, leading to attenuation of pathology. 232 Furthermore, Milosavjevic and colleagues, 233 using 2 well-established murine models of neutrophil and NKT-cell mediated acute liver failure, investigated the molecular mechanisms involved in MSC-mediated modulation of Interleukin-17 (IL-17) signaling during acute liver injury. They showed that a single intravenous injection of MSCs attenuated acute hepatitis and hepatotoxicity of NKT cells in a paracrine, (IDO)-dependent manner, demonstrating that MSCs have potential as a new therapeutic approach in IL-17 driven liver inflammation.
Despite the interest in their therapeutic properties, some pathogens like the Respiratory syncytial virus (RSV) utilise MSCs to their advantage. RSV has been reported to infect human MSCs and modulates their immune regulatory functions. RSV infection of MSCs alters their immune regulatory function by upregulating IFN-β and IDO, affecting immune cell proliferation, which may account for the chronic nature of RSV-associated lung diseases and lack of protective RSV immunity. 262
Challenges and opportunities for modulating the KP in transplanted MSCs
The use of MSCs in therapeutics has also faced challenges due to MSC heterogeneity, and the lack of standardised protocols for their generation and use in stem cell therapies. Discrepancies between studies may be attributable to the plasticity of MSCs, and therefore standards in relation to source, timing, dose, infusion route and pre-treatment, which need to be established to guarantee efficacy and comparability of MSCs for future studies and use as clinical therapeutics. 212 For example, exposure to inflammatory cytokines could help MSCs gain immunomodulatory functions, whereas they may show a pro-inflammatory phenotype in a quiescent environment. Pre-treatment of MSCs with IFN-γ enhanced their anti-inflammatory ability via induction of IDO and resulted in better amelioration of experimental colitis than pure MSCs. 263 Additionally, the prolonged culture expansion required for developing MSC-therapy can lead to replicative senescence, which can be prevented by the applying AhR antagonists.237,264 Therefore, the KP may also be investigated as a way to promote the longevity of MSCs in culture before administration. Taken together, the KP plays an essential role in multiple essential functions of MSCs, including differentiation, senescence, and self-renewing abilities, and is a prominent influence on their immunomodulatory phenotype.
Multipotent adult progenitor cells
Considered by some to be a subpopulation of MSCs, transcriptomic analysis has shown multipotent adult progenitor cells (MAPCs) are significantly different to MSCs. 265 MAPCs are a rare population (0.01%-0.001%) of nucleated cells266,267 derived from adult bone marrow and other tissues including brain, muscle, dental pulp, and adipose tissue.268 -271 They are non-haematopoietic, adherent cells well-known for their pro-angiogenic and immunomodulatory properties, 272 which includes their secretome, which promoted wound healing when tested in cell cultures and also in skin wounds of mice. 273 Their marker expression profile is currently defined as CD13+/44+/49d+/73+/90+/105+/34−/45−/117−/140ab−/309−/MHC II−. 274 While they share some, but not all CD markers with MSCs, both MAPCs and MSCs express a core set of markers defined by The International Society for Cell and Gene Therapy (ISCT) as those of MSCs. 197 Compared with MSCs, MAPCs are seen to be more biologically primitive and have greater differentiation potency (termed stem cell plasticity), underlined by the requirement for hypoxic cell culture conditions (which reduces telomerase shortening) as well as supplementation of culture media with epidermal growth factor (EGF) and platelet-derived growth factor (PDGF), which are not required by most MSCs.274,275 Their differentiation potential includes to endothelium and smooth muscle cells,276,277 HSCs, 278 osteoblasts, 279 hepatocytes280,281 and neuroectodermal cells, including mouse neurons, 282 as well as upregulating genes upon differentiation for human neurons (neurofilament-200, microtubule-associated protein-2, MAP2 and Tau), astrocytes (glial fibrillary acidic protein, GFAP) and oligodendrocytes (oligodendrocyte transcription factor-1, Olig-1). 283
KP activation and MAPCs are protective in controlling autoimmune signaling in type I diabetes
Type 1 diabetes is mediated by autoimmune destruction of the pancreatic β-cells of the islets of Langerhans by T-lymphocytes. Specific T cell induction by dendritic cells (DCs) has been implicated as a mechanism of immunomodulation. IDO present in DCs is activated in response to inflammatory cytokines such as IFN-γ, and plays a role in controlling inflammatory and autoimmunity responses, 284 and therefore may be a valuable target.
Various pro-inflammatory cytokines such as IFN-γ trigger IDO expression in islets in vitro, which could arise through local inflammation (insulitis), 285 while viruses are emerging as causative agents for some kinds of type 1 diabetes. 286 Increased IDO activity, and hence tryptophan catabolites induced by IFN-γ promotes immunotolerance in the NOD type 2 diabetes mouse model,287,288 suggesting that IDO has a protective tolerance role in therapy of autoimmune diabetes in mice non-genetically prone to disease. Furthermore, transfection of IDO-1 into β cells prolongs graft survival in NOD mice. 289 Using engineered 3D allografts another study concluded that local expression of IDO, conferred by adenoviral-mediated gene transfer to bystander syngeneic fibroblasts, enhances the tolerance to islet allografts and significantly prolongs allograft survival. 290 Isolated human islets and other endocrine cells showed a strong immunoreactivity to IDO, which was enhanced when the islets were treated with IFN-γ. IDO has profound effects on reducing islet allograft rejection in these cells through inhibition of T cell proliferation, differentiation, effector function, and viability. 291 Collectively, this shows that IDO activation can trigger protective effects on autoimmunity and islet health in individuals with autoimmune diabetes.
While manipulation of islets and their microenvironment, resulting in IDO activation and TRP depletion (discussed above) is 1 strategy for diabetes therapy, harnessing the innate anti-inflammatory properties of MAPCs is another. Two studies reported the potency of MAPCs in suppressing autoreactive T cell expansion and cytokine production in patients with type 1 diabetes, which was found to be dependent on via IDO-mediated tryptophan catabolism.292,293 IDO activation enhanced the proliferation, and differentiation of MAPCs. It decreased T cell expansion, in agreement with 2 other studies that illustrated that human MAPCs suppressed T cell expansion in an IDO-dependent manner, downstream of IFN-γ signalling during inflammation.234,294 In conclusion, considering the potential role of IDO in the amelioration of human autoimmunity and allograft rejection, its impairment might be involved in that undesirable cascade, leading to β-cell dysfunction. Future studies could explore the possibility of combinatorial therapies to harness the anti-inflammatory potential of MAPCs (via KP modulation) in conjunction with replacement islets in the development of future targeted therapies.
Other studies of the KP in MAPCs
Although MAPCs are a scarce stem cell population, 266 their biologically primitive phenotype and enhanced differentiation potency 274 makes them a promising and versatile target for therapeutics. While their study in the context of the KP is limited, with no comprehensive characterisation of their KP enzyme expression, several studies have shown KP modulation of MAPC function. MAPCs were found to express IDO, enabling them to catabolise TRP down the KP,295 -297 and IFN-γ potently increases IDO expression.294,295,298 In vitro studies indicate a role for the KP in MAPC-associated immune suppression, with MAPCs able to inhibit IFN-γ production, 295 and induce IDO-dependent inhibition of CD4+ and CD8+ T cell proliferation,295,297 Treg induction, 298 and NK cell proliferation. 300 This opens the potential for targeting MAPCs in therapeutics for diseases such as graft versus host disease (GVHD), where immune system suppression is vital for the successful grafting of donor cells.
Neural stem cells
NSCs are a resident stem cell population of the CNS, and a stem cell class that continues to gain traction in the literature as a potential therapeutic target for neurodegenerative diseases. NSCs are found in the adult brain within the subventricular zone (SVZ) adjacent to the ventricles, and the subgranular zone of the hippocampus. 301 NSCs have been utilized therapeutically to clinically manage CNS diseases including stroke and spinal cord injury,302,303 and can give rise to astrocytes, neurons, and OPCs able to differentiate into myelinating oligodendrocytes. While persisting into adulthood, hippocampal neurogenesis well known to decline with ageing to a level that, until recently, was difficult to determine.304,305
The KP in neural stem cells
NSCs remain a relatively understudied population in the context of the KP. Analysis of KP enzymes has largely focused on RNA, with limited study of how this translates to protein expression. For example, research from the Brew lab has shown that unstimulated cultured embryonic mouse NSCs contain RNA transcripts that encode the majority of KP enzymes, including IDO-1/2, TDO-2, KMO, KYNU, kynurenine aminotransferase (KAT-1/2), arylformamidase (AFMID), and QPRT, and that levels of several transcripts, including KAT-1/2 and QPRT, are modulated by 72-hours exposure to IFN-γ. 2 Interestingly, this study observed no significant impact of IFN-γ exposure on initial KP rate-limiting enzymes, IDO-1/2 or TDO-2. However, our understanding of IDO expression in NSCs remains inconclusive, with alternative studies showing no IDO transcripts or protein in rat foetal NSCs, 306 while the conditionally immortalized CTX0E03 human NSC cell line 307 shows basal IDO-1 expression, which is potently induced by IFN-γ, and IDO-2 and TDO-2 are absent or present in very low abundance in unstimulated and IFN-γ-treated cells. 308 Variation between these studies may be associated with factors including innate differences between primary NSCs and the CTX0E03 cell line, the developmental stage of NSCs (embryonic/foetal versus adult), interspecies variability, or the exposure time and/or concentration of IFN-γ. The effect of KP modulation or KP metabolites on NSCs also remains largely unclear. Interestingly, KYN, 3-hydroxy-KYN and anthranilate were all identified from a screen for bioactive metabolites capable of epigenetic modulation of hypothalamic-linked loci in iPS-derived NSCs, 309 suggesting KP metabolites can modulate gene expression as well as exert conventional physiological actions.
Inflammation inhibits NSC proliferation, 310 and neuronal differentiation 311 in vivo. Similar effects have been observed in NSCs exposed to IFN-γ both in vitro and in vivo, which displayed inhibited proliferation and enhanced neuronal differentiation, with an overall net reduction in neurogenesis.312 -314 While activation of the STAT1 313 and JNK 315 pathways were implicated in these changes, it is also plausible that effects could be associated with KP induction. Supporting this, cultured hippocampal progenitor cells exposed to IL-1β express increased transcripts for IDO, KMO and KYNU, and display reduced neurogenic capacity. 20 Interestingly, KYNA has also been shown to inhibit the proliferation and neuronal differentiation of human iPSC-derived NSCs in a 3D culture model of Alzheimer’s disease. 316 This was subsequently confirmed in adult zebrafish, and was mechanistically linked to KYNA activating AhR. 317
Regulating the KP in NSCs may therefore be a therapeutic target, both in stimulating the generation of new neurons and glia, and in suppression of the immune system. Immune suppression is relevant in inflammatory diseases of the CNS such as MS. A study utilising NSCs genetically modified to express high levels of IDO protein, showed IDO-associated inhibition of T cell proliferation in vitro, immune suppression and reduced symptoms when transplanted in EAE-induced mice. 306 An alternative study found that while intravenous injection of IL-10 over-expressing NSCs into EAE mice had an immunosuppressive effect by inhibiting T cell activation; this effect was independent of IDO. 318 The discrepancy in findings may be due to cytokine-specific effects on IDO-linked pathway regulation.
Together, these studies show a role for the KP in modulating NSCs. However, significant gaps remain in our understanding of their full KP enzyme expression, the factors that modulate this, and the effects of crucial KP metabolites on NSC function. Exploring these will be important areas for developing future therapeutics.
Cancer stem cells
Cancer, which results from the uncontrolled division of abnormal cells, is a leading cause of death worldwide. 319 Treatment is dependent upon factors including tumour type, histologic grade and cancer stage, 320 and can involve surgical removal, radiotherapy, chemotherapy, monoclonal antibody therapy, or combinations of these treatments.320,321 Combined chemotherapy can be curative in some forms of advanced cancer, including acute lymphoblastic and acute myelogenous leukemias, and in Hodgkin’s and non-Hodgkin’s lymphomas. 321 However, for tumours, such as those of the prostate, the advanced disease is associated with a high rate of mortality despite aggressive treatment.320,322
Typically developing from a single mutated cell, tumours rarely remain homogenous, containing cancerous cells in various states of differentiation and with differential proliferative capacity. The cancer stem cell (CSC) theory posits that tissue expansion within many tumours is primarily regulated by a small population of highly proliferative cancer cells that can generate all cancerous cell types that form the primary tumour. These cells are termed CSCs, as they have many of the defining features of stem cells including self-renewal capacity, potency, and the ability to generate all cells within their lineages. They also display resistance to chemotherapeutic drugs and radiation therapy, indicating they may be significant players in treatment failure.323,324
The KP in cancer stem cells and cancer therapeutics
There is significant interest in the KP in cancer cells and cancer pathogenesis due to its role in immunosuppression, angiogenesis, and the generation of bioactive and pro-inflammatory metabolites. For example, CSCs rely on NAD to facilitate the generation of cellular energy though glycolysis and oxidative phosphorylation to maintain rapid proliferation.325 -328 While cancer cells are believed to generate NAD primarily via the salvage pathway, 329 NAD is also a primary end product of the KP, produced by the catabolism of QUIN by the QPRT enzyme (Figure 1). Tummala and colleagues 330 utilised a genetic knock-in mouse model in which KP NAD synthesis was specifically inhibited in hepatocytes through an induced expression of unconventional prefoldin RPB5 interactor, which inhibits AhR and estrogen receptor mediated transcription of KP enzymes. They showed that NAD depletion caused DNA damage leading to hepatocellular carcinoma; a process that could be inhibited though NAD supplementation. Similarly, inhibition of NAD generation through alternative splicing of AFMID, the enzyme that functions directly downstream of IDO-1/2 and TDO2 to catabolise N-formyl-kynurenine into KYN, has been identified as an early event in the development of hepatocellular carcinoma. 331 This suggests that NAD acts as a double-edged sword, as it provides energy for CSC proliferation to promote cancer expansion, while its depletion can trigger DNA damage which can cause cells to become cancerous. The balance of NAD levels within cells is, therefore, essential to maintain homeostasis. In general, cancer cells have much higher NAD levels than normal cells, as a result of increased synthesis from the salvage, nicotinic acid and de novo pathways (Tummala et al.,330,332 reviewed in Kennedy et al. 329 ). This higher storage pool in turn enhances their survival, and ultimately cancer progression.
Role of AhR signaling in cancer stem cells
AhR is upregulated in many cancer cells, and constitutive activation of AhR has been observed mainly in high-grade tumours.333,334 Antibodies against programmed cell death receptor-1 (PD-1) have successfully been used in the clinic for cancer therapy. 335 T cells can be harnessed to clear cancerous cells, CD8+ T cells that secrete IFN-γ provide the necessary stimulus for KP activation. Resultant KYN production is then shuttled to T cells via transporters SoLute family Carrier 7 member 8 (SLC7A8) and Proton-coupled Amino acid Transporter 4 (PAT4), where it activates AhR and PD-1 expression in CD8+ T cells. This produces an immune environment that facilitates tumour growth, while inhibiting the KYN-AhR pathway enhances anti-tumour therapies. 336 Additionally, blocking AhR inhibits the rapid migration of oral squamous cell carcinoma cells, reduces chemoresistance, reduces tumour growth and increases survival. 333 Alternatively, activation of AhR with IKK alpha induces stemness genes including the ATP-binding cassette sub-family G member 2, and c-Myc in radioresistant cell lines. 324 However, the ability of AhR agonists to inhibit breast cancer stem-like cells 337 suggests that activation of AhR may have multiple effects that are cell type, cancer, and ligand-specific. Indeed, a study by Cheng and colleagues showed that in embryonic carcinoma cells and ESCs, AhR mRNA negatively correlated with the stem cell marker Oct4 during stem cell differentiation due to the direct binding of AhR to response elements in the POU5F1 gene. 338 They also showed that culturing U87 cancer cells (a human primary glioblastoma cell line) with L-TRP and natural AhR ligand 2-(1′H-indole-3′-carbonyl)-thiazole-4-carboxylic acid methyl ester (ITE) resulted in reductions in POU5F1 mRNA, and this activity was AhR-dependent as reducing AhR using shRNA reduced the suppressive effect on POU5F1. Following hypoxia and L-TRP depletion, POU5F1 transcription increased, but this could be blocked by incubation with ITE. 338 Recently, it has been suggested that AhR antagonises K-RasG12D driven non-small cell lung adenocarcinoma by restricting the number of cancer-initiating stem cells, and was also posited as a potential prognostic marker to determine disease progression. 339 Han and colleagues 340 observed a similar phenomenon when they demonstrated that AhR signaling is protective in genetically-induced colon tumorigenesis via the suppression of Wnt signaling, suggesting a possible therapeutic target for colorectal cancer.
IDO-1 inhibitors for cancer therapy
IDO-1 up-regulation and increased TRP metabolism have been observed in tumour microenvironments and are thought to contribute to disease progression. 341 IDO-1 inhibitors have demonstrated efficacy at reducing tumour growth in mouse xenografts due to activation of the immune system and subsequent depletion of CSCs. 342 Due to this success, IDO-1 inhibitors are currently being investigated in clinical trials. Phase 1 studies of IDO-1 inhibitor Epacadostat showed that the drug had an appreciable side effect profile, inhibited IDO-1 activity, and normalized KYN levels. 343 However, as a standalone treatment in phase II clinical trials, IDO-1 inhibitors have not demonstrated efficacy in human cancers such as recurrent ovarian, peritoneal and fallopian tube cancers, and some trials were terminated. 344 In kidney cancer cells, IDO-1 inhibitors were only effective at reducing tumour growths following co-treatment with TNF-α, 341 suggesting that IDO-1 inhibitors should be trialled in combination with other immunomodulatory drugs or therapies. In agreement with this, a mouse xenograft study revealed that IDO-1 was up-regulated via Notch1 activation in cervical CSCs, which decreased their radiosensitivity. 345 Blocking IDO-1 was able to enhance the efficacy of radiotherapy. 345 Some recent clinical trial results from phase II studies of IDO-1 inhibitors in combination therapy have been completed, and suggest that such polytherapeutic approaches may be promising.346,347 Newer inhibitors with higher potency are also being developed. 342 For example, a novel prodrug potently suppressed CSCs by inducing immunogenic cell death and inhibiting IDO in triple negative breast cancer 4T1 tumours. 348 While IDO inhibition represents a promising therapeutic for the treatment of cancer, many of the available inhibitors also activate the AhR109,110 and may subsequently upregulate IDO expression. 110 This complex interplay between AhR and IDO and similarity of inhibitors to known AhR ligands may explain the mixed success of human cancer trials using IDO-1 inhibitors.
While the focus of the KP in cancer has traditionally been on IDO-1, it is becoming progressively evident that there is broad dysregulation of the KP in cancer pathogenesis, pointing towards several other KP enzymes as targets of interest. IDO-2, for example, is upregulated in cancers of the pancreas 349 and lung, 350 and IDO-2 knockout 351 and silencing 352 inhibits tumour expansion. TDO2 has also been implicated in the production of an immunosuppressed environment that promotes cancer cell survival.60,353 -355 Its overexpression is correlated with tumour grade and poor prognosis in triple-negative breast cancer and brain tumours, with TDO2 inhibition suggested as a therapeutic target in oesophageal cancer. 353 Upregulation of other downstream KP enzymes including AFMID, 356 KMO,357 -360 KYNU 361 and QPRT,362 -364 has also been observed in various cancers. KYNU facilitates tumour growth by catabolising 3-HK to 3-HAA, with 3-HAA inhibiting ferroptotic death of tumour cells, a process that can be negated by HAAO-mediated catabolic depletion of 3-HAA. 365 Highlighting the diversity and heterogeneity of cancer cells, QPRT, HAAO, and KMO downregulation has been shown in renal cell carcinoma. 366 In summary, dysregulation of the KP is a pathogenic feature of various cancers, stressing the importance of targeting TRP metabolism in cancer therapeutics.
Cardiac stem cells and the KP – little known but significant promise in regenerative medicine?
Cardiac disease remains a significant cause of mortality worldwide despite advancements in understanding of it’s pathogenesis and surgical treatment options. Cardiac stem and progenitor cells (CDSCs), of several types and residing in several niches of the heart,367 -369 are an attractive potential therapy for example, acute myocardial infarction patients, however progress in this area has had challenges due to their immune recognition, targeting and their rapid clearance from transplant sites.
Currently there is a dearth of studies to examine KP expression in these cells. Only one study, by Sebastiao and colleagues, 370 showed that in vitro CDSCs could have immunosuppressive functions via modulation of T lymphocytes by their expression of IDO-1 and causing TRP depletion, in addition to other mechanisms. Whether other physiological actions exist is unclear, and could be investigated in the future. Further actions are suggested by evidence showing KYN acts on differentiated cardiomyocytes to positively regulate their proliferation by AhR. 52
Conclusions
Stem cell therapy, whether through leveraging endogenous niches or transplantation, offers a promising avenue for addressing tissue damage, immune system modulation, regulation of bioactive molecule release, and targeted drug delivery. Central to these endeavours, the KP emerges as a critical biochemical signaling route, pivotal for steering stem cell responses that contribute to systemic homeostasis. Hence, unravelling the intricate interplay between cell biology and development of effective therapeutics necessitates a comprehensive understanding of the KP. This review underscores that the KP's regulation varies among distinct stem cell types and niches (summary schematic Figure 4), marked by shifts in KP enzyme expression and disparate responses to specific KP metabolites. Furthermore, this diversity hinges on the developmental stages of cells (e.g. embryonic versus adult) as well as potent modulation by inflammatory cues and essential cytokines.

Schematic diagram of the key features of KP metabolism in stem cell types covered in this review. A saggital plane through a pregnant human female is shown in the centre of the schematic, in order to depict the localisation and known functions of the KP in pluripotent, embryonic, stem and precursor cells from the different tissues and organs depicted, which are then enlarged in adjacent expanded boxes either side to illustrate the major known KP phenotype in those cells. In each box, a cartoon representation of the major reactions in the KP is shown in the top left corner of each expanded box, confirmed metabolites produced shown in green text, and therefore enzymatic pathways likely to be active indicated by green arrows. Where studies have not investigated KP expression in that cell type, the pathways are shown in gray. Where studies have shown conflicting evidence for example, between mouse and human or between primary cells and cell lines, this is indicated in orange. TRP is shown in black as the key metabolite from which all downstream metabolites are derived. AhR is shown with a green background (to represent proven expression in that cell type and where relevant, functionality with respect to KP ligands) versus red (uncertain/not investigated). A plus ‘+’ inside of a circle represents pathways or outcomes that are positively enhanced, while a minus ‘−’ sign indicates negative/reduced pathways/outcomes. A ‘T’ symbol is used when referring to the actions of an inhibitor. ‘1’ and ‘2’ in circles represent separate outcomes as indicated and referred to in the heading of each box. Question marks appear where unresolved questions arise or where more evidence would be helpful to substantiate the phenotype. Summary of individual boxes: Human CDSCs inhibit T lymphocyte growth via IDO-1 mediated TRP depletion, 370 however the expression of other KP enzymes and metabolites produced is currently unknown, as is the activation of AhR. iPSCs, can be isolated from somatic cells like skin. Their growth is regulated by extracellular TRP concentration, where high basal IDO-1 expression promotes production of metabolites including n-formylkynurenine which upon secretion is pro-proliferative (and may act in an autocrine or paracrine manner), while AhR is not involved and NAD synthesis is not altered. 146 While many metabolites have been detected, expression levels of their corresponding enzymes has not been investigated. KP expression or AhR function in EPCs is unknown, however in vitro evidence shows that KYN can regulate their proliferation, and circulating EPC numbers reduced when a selective IDO-1 inhibitor was given to mice, suggesting KP metabolites are protective, 191 while AhR function is unknown. HSCs synthesise little KYN basally, however synthesis significantly rises with IFN-γ treatment.157,317 KP metabolites KYN, KYNA, and 3-HAA potently inhibit HSC proliferation in vitro. 159 Whether this occurs via AhR (confirmed expressed in HSCs) is unclear. qPCR analysis has proven mouse NSCs express major KP enzymes and respond to IFN-γ 2 however secreted metabolites have not investigated and enzyme expression varies in for example, cell lines. KYNA negatively regulates iPS-derived NSC proliferation and promotes neurogenesis via AhR.316,317 CSCs synthesise high NAD levels from the salvage pathway needed for proliferation, 329 while activation of interplay between KP activation in CSCs, secretion of metabolites and AhR activation via secreted ligand KYN in T cells is important for immune evasion. 336 Inhibiting AhR signaling does not always give a beneficial outcome in all types of cancer, as discussed in the text. Placental MSCs are immunosuppressive by limiting T cell proliferation through TRP depletion via IDO-1, 371 though much about their KP enzyme expression beyond that is not known. Bone marrow MSCs express most KP enzymes when examined by PCR. 2 ESCs are regulated by KP activation through IDO-1, and activation of AhR by KP metabolites to maintain self-renewal and pluripotent phenotype, while KAT-2 activity and production of metabolite 2-AAA is a biomarker for ectodermal differentiation. TDO2 is not basally expressed but can be induced by a neurodevelopmental toxin, while KMO is not expressed.118,119 Expression/detection of many KP enzymes and metabolites is currently unknown.
A primary and well-established function of KP activation is immune system suppression, largely mediated by cytokine-induced IDO expression. 372 This manifests in diminished proliferation and inactivation of immune cells, including T and NK cells.212,233,371,373 Simultaneously, the activation boosts the generation of vital bioactive KP metabolites, culminating in the metabolically-indispensable cofactor NAD. Various stem cell subpopulations are directly engaged in KP-associated immunosuppressive roles, a phenomenon observed in ESCs, HSCs, MSCs, MAPCs, CSCs, and to some extent, NSCs. This influence likely extends to other stem cell populations whose range of functions are as yet unexplored. Moreover, KP activation spurs the synthesis of metabolites such as KYNA, which, in turn, impedes the proliferation of NSCs and HSCs,316,317 subsequently hampering the generation of immune cells within the circulatory system and CNS.
In numerous instances, this immunomodulatory response, including AhR activation, manifests as a broadly positive impact, contributing to inflammation resolution and facilitating medical treatments including cellular transplants. Nonetheless, KP can play a role in pathological processes. For instance, in certain cancers, it creates an environment that supports tumour expansion. 372 Prolonged activation of the KP may also elevate levels of several KP metabolites, some of which are known to have deleterious effects. Neurotoxic QUIN, for instance, which could play a role in advancing several CNS and periphery diseases by generating reactive oxygen species and functioning as both as an excitotoxin at NMDA receptors, and perturbing synaptic glutamate uptake and recycling. 59
In light of these considerations, the future of the trajectory of the KP and its interplay with stem cells in the field of regenerative medicine necessitates a focus on 2 pivotal research avenues. First and foremost, a fundamental question arises: Are stem cells acting as beneficial agents or potentially adverse contributors when assessed through the lens of the KP metabolic profile and enzyme expression, and does this dynamic shift in the context of disease? Notably, significant gaps persist in comprehending the expression (and regulation thereof) of KP enzymes and metabolites within various stem cell niches (summarised in Figure 4), including the intricacies of its regulation in response to inflammation. Addressing this knowledge gap holds the promise of furnishing essential insights into the direct impact of stem cells on diverse pathologies and, by extension, the potential approaches for modulated for modulating their behaviour in therapeutic contexts.
Secondly, a critical exploration beckons into the mechanisms governing how fluctuations in KP metabolite concentrations within specific niches exert control over the mobilization, proliferation, and differentiation capacities of the resident stem cell. Several KP metabolites wield substantial influence over stem cell function, underscoring the importance of understanding this intricate interplay comprehensively. Unveiling the nuances of these biochemical activities may yield a promising avenue for targeting these metabolites as part of a therapeutic strategy. Collectively, advancements in our understanding of these areas have the potential to propel the field of stem cell biology toward the realization of precise and effective treatments spanning a spectrum of pathologies.
Footnotes
Acknowledgements
None.
Author Contributions
BSS: Secondary editing of text contributed by primary authors, reference updating and final approval of manuscript. STB: Writing – original draft, final approval of manuscript. TWRP: Writing - original draft, figure preparation and final approval of manuscript. HDM: Writing – original draft and final approval of manuscript. NKB: Writing – original draft and final approval of manuscript. NCT: Writing – original draft and final approval of manuscript. MLN: Writing – original draft and final approval of manuscript. MY: Writing – original draft and final approval of manuscript. MKS: figure preparation, secondary editing and final approval of manuscript. SS: Writing – original draft and final approval of manuscript. MWW: Secondary editing and final approval of manuscript. AC: Secondary editing and final approval of manuscript. CKL: Secondary editing and final approval of manuscript. MDL: Study conceptualization, secondary editing, figure preparation, data curation, project administration, final approval of manuscript. BJB: secondary editing, project administration, final approval of manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B.J.B, M.D.L and B.S.S acknowledge the generous support and funding of the Peter Duncan Neurosciences Research Unit at St. Vincent’s Centre for Applied Medical Research (AMR). M.D.L and B.J.B acknowledge the financial support of this work by Multiple Sclerosis Research Australia (MSRA), The University of New South Wales (UNSW), Curran Foundation and St. Vincent’s Clinic Foundation. M.K.S acknowledges a Careers in Immunology Fellowship awarded from the American Association of Immunologists.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Prof. Bruce Brew is a member of the International Journal for Tryptophan Research journal editorial board. The other co-authors declare that they have no conflict of interest.
