Abstract
Type 1 diabetes (T1D) is characterized by anomalous functioning of the immuno regulatory, tryptophan-catabolic enzyme indoleamine 2,3 dioxygenase 1 (IDO1). In T1D, the levels of kynurenine—the first byproduct of tryptophan degradation via IDO1—are significantly lower than in nondiabetic controls, such that defective immune regulation by IDO1 has been recognized as potentially contributing to autoimmunity in T1D. Because tryptophan catabolism—and the production of immune regulatory catabolites—also occurs via the gut microbiota, we measured serum levels of tryptophan, and metabolites thereof, in pediatric, diabetic patients after a 3-month oral course of Lactobacillus rhamnosus GG. Daily administration of the probiotic significantly affected circulating levels of tryptophan as well as the qualitative pattern of metabolite formation in the diabetic patients, while it decreased inflammatory cytokine production by the patients. This study suggests for the first time that a probiotic treatment may affect systemic tryptophan metabolism and restrain proinflammatory profile in pediatric T1D.
Keywords
Introduction
Intestinal microbial homeostasis inhibits growth of pathogens, improves the gut mucosal barrier and modulates local and systemic immune responses. Any perturbation in the host-microbiota crosstalk can be an initiating or reinforcing factor in autoimmunity. 1 Probiotic treatment of any gut dysbiosis in type 1 diabetes (T1D) patients can shape the host immune response and interfere with the metabolic pathways of the host.2,3 Growing evidence suggests that gut dysbiosis plays a role in the rising incidence of T1D, yet the mechanisms whereby perturbation of the gut microbiota may favor T1D onset have been unclear. Technologies are now available to better dissect the interplay between the host’s microbiome and systemic immunity 4 ; at the same time, large cohorts of children at increased risk for T1D development have been monitored over time for gaining insight into the role of the microbiota in T1D prevention versus pathogenesis. 5 Recently, the TEDDY (The Environmental Determinants of Diabetes in the Young) study provided some evidence that early administration of probiotics (mostly, Lactobacillus and Bifidobacterium spp.) is associated with a reduced risk of islet autoimmunity.6,7 These data, in combination with additional results examining different human T1D cohorts8,9 as well as T1D mouse models,10,11 call for in-depth investigation of whether and how specific gut microbial components may have a predictive, protective, or conversely, a causal role in T1D pathogenesis.
Tryptophan (Trp) metabolism is more than a mere metabolic pathway and plays an important role in immune homeostasis. Specifically, it represents a powerful means of restraining autoimmunity, and the so-called “kynurenine pathway” of Trp degradation is now being recognized as a major immune checkpoint.
12
Indoleamine 2,3 dioxygenase 1 (IDO1), the enzyme that catalyzes the oxidative degradation of
The alternative fate of Trp in eukaryotic cells is the “serotonin pathway,” by which the amino acid is sequentially transformed in 5-hydroxytryptophan (5-OH-Trp), 5-hydroxytryptamine (5-HT or serotonin), N-acetylserotonin (NAS), and melatonin. In peripheral tissues, serotonin plays several important functions in gastrointestinal motility, secretion, and inflammation.17-20 It can act as a real immune modulator on various immune cells expressing 5-HT receptors, such as lymphocytes, monocytes, macrophages, and dendritic cells. 21 A specific Trp-metabolomic signature characterized by increased levels of 5-OH-Trp and NAS—in addition to further altered Trp metabolites—was found in the cerebrospinal fluid (CSF) of autoimmune patients affected by multiple sclerosis (MS). 22 Recently, NAS has been described as a positive allosteric modulator of IDO1 enzyme in experimental autoimmune encephalomyelitis (EAE), an animal model of MS. 23 As a result, increased Kyn will activate the aryl hydrocarbon receptor (AhR), mediating antinflammatory and immunoregulatory effects. A possible interplay between the two Trp metabolic pathways has been also suggested in depressed diabetic rats in which the hippocampal activation of IDO1 enzyme contributes to the reduction of serotonin levels associated with depressive-like behavior of the animals. 24
Gut bacteria can also affect Trp metabolic fate by using the amino acid for the production of indole derivatives—namely, indole-3-aldehyde (IAld), indole-3-acid-acetic (IAA), indole-3-propionic acid (IPA), indole-3-acetaldehyde (IAAld), and indoleacrylic acid—as ligands of the receptor AhR, resulting in immune regulatory effects. 25 Activated Trp-AhR axis in the gut can induce the expression of downstream cytokines such as interleukin-22 (IL-22) and interleukin-17 (IL-17), thereby regulating intestinal homeostasis.26,27 Under pathologic conditions in which eukaryotic Trp-degrading pathways are overactivated, decreased gastrointestinal Trp availability may contribute to deplete AhR agonists otherwise utilized by the gut microbiota. In different experimental models, Trp supplementation or inhibition of the eukaryotic metabolic pathways rebalanced Trp metabolism between the host and the microbiota, improving insulin sensitivity, gut mucosal barrier, chronic inflammation. 28 Overall, these findings indicate that Trp-focused metabolomic analysis in different biological matrix (i.e., sera, CSF, urine, feces) will implement the use in the clinic of such metabolites as predictive biomarkers. A deeper knowledge of the causal-effect relationship regulating Trp metabolism by gut microbiota in several pathogenic settings will facilitate the targeting of the microbiome and the use of metabolome as an actor in therapeutic strategies.
The current study evaluated the impact of a probiotic treatment with Lactobacillus rhamnosus GG in pediatric T1D patients by analyzing serum levels of Trp-derived metabolites and the cytokine profile secreted by PBMCs. For the first time, these data provide support to the effect of a probiotic treatment on Trp metabolism and cytokine secretion in a pediatric T1D cohort.
Materials and Methods
Subjects
Serum and PBMCs isolated from 61 young (aged 3-18 years) T1D patients were collected and analyzed. These patients represent a subcohort of a larger cohort recruited at the Pediatric Clinic of the Perugia University (Italy), as described elsewhere. 2 Patients affected by concomitant chronic diseases other than T1D, infections or subjected to immunosuppressive therapy were excluded from the study. Patients have been randomly assigned to two experimental groups on the basis of a computer-generated list. Specifically, 36 patients (the “probiotic group”) received Lactobacillus rhamnosus GG (LGG, Dicoflor 60 Immuno D3, Dicofarm, Italy) daily for 3 months, each dose containing 1 billion LGG/drop. Twenty-five patients (the control group) were treated with placebo (namely, a similar formulation not containing the probiotic). The baseline characteristics are summarized in Table 1. The protocol, as approved by the Ethics Committee of the Umbria Region, was in accordance with the Declaration of Helsinki and the standards of Good Clinical Practice for trials of medicinal products in humans. For the collection of samples and subsequent analysis, written informed consent was obtained from the parents/legal guardian of individual patients aged <8 years and from each subject aged ⩾8 years.
Baseline characteristics of T1D patients.

Biological sample preparation and treatment
At the end of probiotic treatment, peripheral venous blood (3-5 mL) was obtained from T1D patients. To limit the effects of diet on systemic Trp level, blood samples were collected after overnight fasting. Samples were immediately centrifuged at 450 g for 5 minutes for collecting the sera (upper phase), which were stored at −80°C until analysis. PBMCs were isolated by a Ficoll-Hypaque (GE healthcare, PA, USA) gradient applied to the residual cell fraction (lower phase) after dilution with phosphate buffer.
Serum metabolomics analysis
Circulating tryptophan and its derived metabolites, listed in Table 2, were detected in the serum of T1D patients by mass spectrometry. All reagents and standard metabolites used in the analysis were LC-MS grade and purchased from Sigma-Aldrich (Saint Luis, MO, USA). The quantification was carried out through a 6-point calibration curves in the appropriate concentration range, dissolving the analytes in a mixture of H2O/MeOH/formic acid 95/5/0.1 (v/v/v). Metabolites were extracted from 100 µL of serum using 300 µL of acetonitrile. The solution was vortexed for 30 seconds and left to stand for about 15 minutes. The sample was then centrifuged, the entire volume of the supernatant was evaporated at 30°C and reconstituted in 100 µL of a mixture H2O/MeOH/formic acid 95/5/0.1 (v/v/v). Finally, 1 µL was injected into the LC system. Analyses were performed with an UHPLC Dionex Ultimate 3000 system coupled to a hybrid quadrupole-orbitrap mass spectrometer (LC-Q-Exactive, Thermo Scientific, San Jose, CA, USA). Chromatography separation involved a Kinetex XB column (3.0 × 100 mm, 2.6 μm, Phenomenex, Torrance, CA, USA) thermostated at 40°C. The mobile phase were A (water) and B (methanol), both containing 0.1% formic acid at a flow rate of 0.3 mL/min. The following gradient was applied: 0 to 2.0 minutes, 2% B; 2.0 to 37.0 minutes, 2% to 100% B; 37.0 to 42.0 minutes, 100% B; 42.0 to 42.1 minutes, 100% to 2% B; 42.1 to 46.0 minutes, 2% B. A heated electrospray ionization source (HESI-II) (Thermo Scientific, San Jose, CA, USA) in positive mode was applied to ionize target compounds using an ionization voltage equal to +4.0 kV. Source conditions were optimized as follows: capillary temperature 250°C, heater temperature 270°C, S-lens RF level 50, sheath gas flow rate 40 and auxiliary gas flow rate 15 (arbitrary units). The acquisition was achieved in full MS/dd-MS 2 mode. The m/z range was from 100 to 900 with a resolving power equal to 70 000 FWHM (m/z 200). The automatic gain control target and maximum injection time were set at 1 × 106 and 300 ms, respectively. Mass accuracy tolerance was lower than 5 ppm. An inclusion list for dd-MS 2 experiments was used including the m/z precursor and the expected retention time (±1 minute) of each metabolite. The precursor ion was filtered by the quadrupole with a mass isolation window of ±0.9 Da. Resolving power, AGC, and IT were set at 17 500 FWHM (m/z 200), 1 × 105 and 80 ms, respectively. Stepped normalized collision energy (NCE): 10, 25, and 50 eV. All the parameters of the LC-HRMS system were controlled through Xcalibur software version 4.0 (Thermo Scientific, San Jose, CA, USA). The retention times (RTs), the monitored ion species and the limits of quantification (LOQs) of each analyzed metabolite in the serum samples of T1D patients are reported in Table 2.
LC-MS analysis parameters of the detected metabolites.

Abbreviations: RT, retention time; ND, not determined for metabolites present in the samples at µM concentration.
Quantifier ion (full scan experiment).
Qualifier ions (dd-MS2 experiment).
LOQ, limit of quantification for metabolites present in the samples at nM concentrations.
Kynurenine and cytokine analyses in culture supernatants from T1D PBMCs
Freshly isolated PBMCs (1 × 106 cells/mL) were cultured in vitro in Roswell Park Memorial Institute (RPMI) medium supplemented with 10% Fetal Calf Serum (FCS), 1 mM glutamine and penicillin/streptomycin (Gibco, Invitrogen CA, USA) at 37°C in a humidified 7% CO2 incubator, before harvesting culture supernatants for analysis. For Kyn concentration measurements, cells were left untreated or exposed to 100 U/mL interferon (IFN)-γ (Novus Biologicals LLC) for 48 hours, as described. 15 Kyn concentration in the culture supernatants was measured by HPLC, as described.29,30 Briefly, a Perkin Elmer (series 200 HPLC) instrument and a Kinetex C18 column (250 × 4.6 mm, 5 μm, 100 Å, Phenomenex, USA) were used for the analysis. The column was maintained at the temperature of 25°C and a pressure of 1800 PSI. Samples were eluted with a phase containing 10 mM NaH2PO4 (pH = 3.0; 99%) and methanol (1%) (Sigma-Aldrich, MO, USA), with a flow rate of 1 mL/min. Kyn was detected at 360 nm by an UV detector. For assessment of cytokine production, T1D PBMCs were activated in vitro with 1 μg/mL phytohemagglutinin (PHA; Sigma-Aldrich, MO, USA) for 48 hours, as described, 2 before harvesting culture supernatants. A 15-plex immunoassay (Bio-Plex Pro Human Th17 cytokine panel, Cat N. 171aa001m; Bio-Rad, CA, USA) and a MAGPIX system (Luminex Corporation) were used for the detection of IFN-γ, IL-17F, IL-31, IL-6, and sCD40L.
Statistical analysis
Serum metabolite concentrations were represented as box and whiskers (Min to Max) and data were analyzed by two-tailed unpaired Student’s t test (probiotic vs control). Kyn and cytokine concentrations (µM and pg/mL, respectively) in PBMCs supernatants were represented as the mean ± standard error mean (SEM). Kyn concentrations were analyzed by one-way ANOVA, followed by post hoc Bonferroni’s test. Cytokine concentrations were analyzed by unpaired multiple Student’s t test per row (probiotic vs control), corrected for multiple comparisons using Sidak-Bonferroni’s method. Differences were considered statistically significant when p < 0.05 (*), p < 0.01 (**), p < 0.001 (***). GraphPad Prism Software, version 6.00 for Windows (La Jolla California, USA) was used for representation and statistical analyses.
Results
Serum tryptophan-focused metabolomic analysis of T1D patients treated with the probiotic LGG
A serum Trp-focused metabolomic analysis was performed in a subcohort of young T1D patients recruited for a larger study. 2 A total of 61 pediatric T1D patients were analyzed, 25 of which received placebo (as a control group) and 36 (as a probiotic group) daily received the Lactobacillus rhamnosus GG (LGG) for 90 days. The baseline characteristics of T1D cohort are reported in Table 1. At the end of the probiotic treatment, sera from T1D patients were used for Trp-focused metabolomic analysis, in order to assess any systemic metabolic perturbation induced by probiotic treatment. Specifically, we focused the analysis on Trp and the downstream catabolites along the two main Trp-degrading metabolic pathways, namely, the IDO1-dependent kynurenine pathway and the IDO1-independent serotonin pathway, as reported in Figure 1. A statistically significant increase in circulating Trp levels was observed in the probiotic group, as compared to control samples (p = 0.024) (Figure 2a). Concomitantly, probiotic treatment induced a significant decrease in 5-hydroxytryptophan (5-OH-Trp) levels, the first metabolite along the serotonin pathway, and a decrease in the production of the downstream product 5-hydroxytryptamine (or serotonin), although statistical significance was not reached, suggesting a down-modulated Trp degradation along the serotonin pathway (Figure 2b). Undetectable levels of the downstream NAS and melatonin were found in pediatric sera (limit of quantification, LOQ <0.005 µM, <0.002 µM, respectively) (data not shown). Circulating levels of Trp-derived metabolites along the kynurenine pathway, namely kynurenine, 3-OH-kynurenine, kynurenic acid, and quinolinic acid did not significantly differ between the two groups (Figure 2c), while anthranilic and xanthurenic acids in the sera of pediatric T1D patients were below the LOQ (<0.04 and <0.02 µM, respectively) (data not shown). Thus, the probiotic treatment did not affect kynurenine pathway, not even the circulating levels of indole derivatives generated by the intestinal microbes, since 3-indolepropionic acid (IPA) and indole-3-aldehyde (IAld) serum levels did not change between the 2 groups (Supplemental Figure 1). Overall, these findings are compatible with the hypothesis that the significant increase of circulating Trp after probiotic administration in T1D patients could be ascribed to a lowered amino acid degradation along the serotonin pathway.
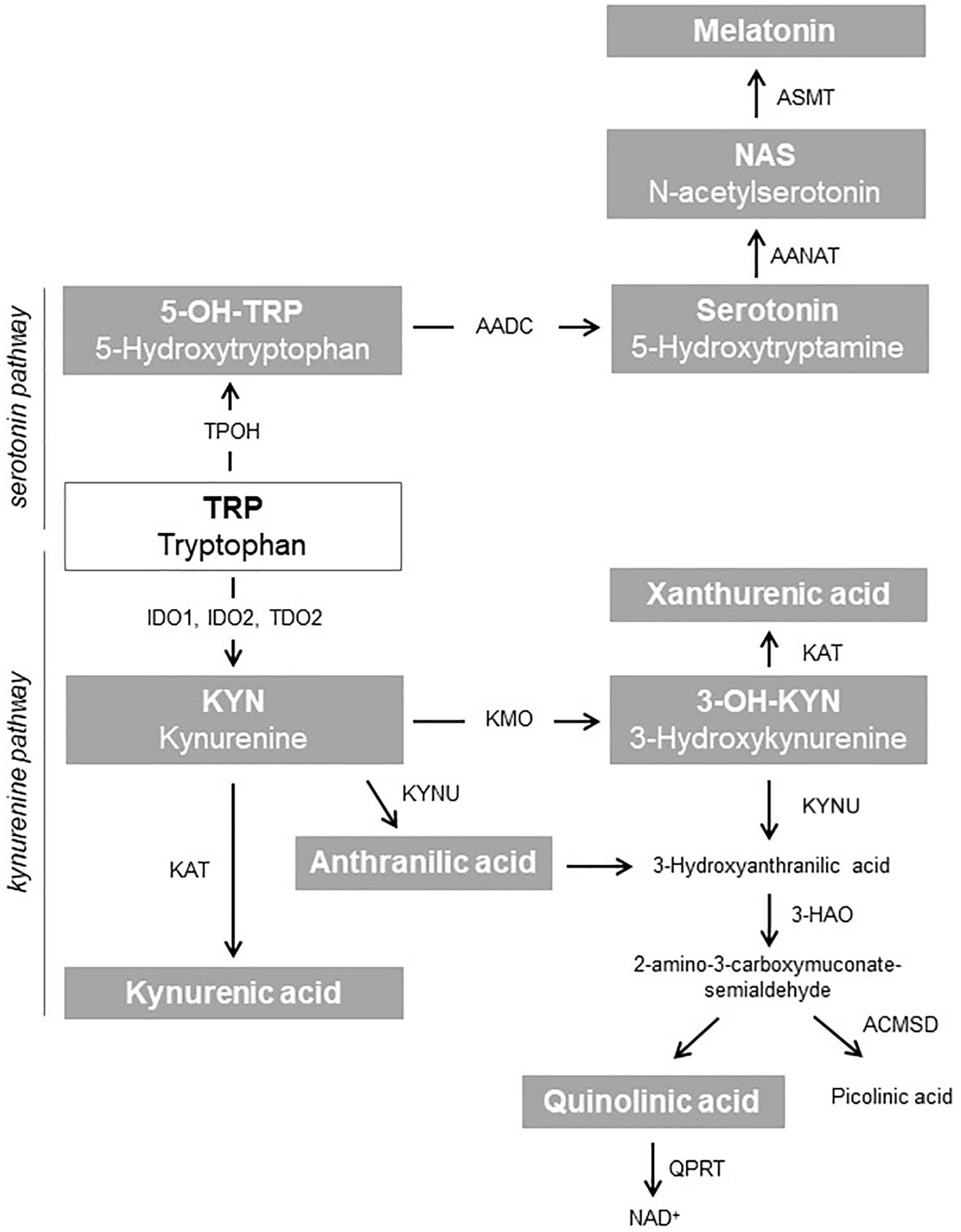
Tryptophan metabolism along the kynurenine and serotonin pathways. In the gray boxes are indicated the Trp-derived metabolites analyzed in the T1D patients serum.
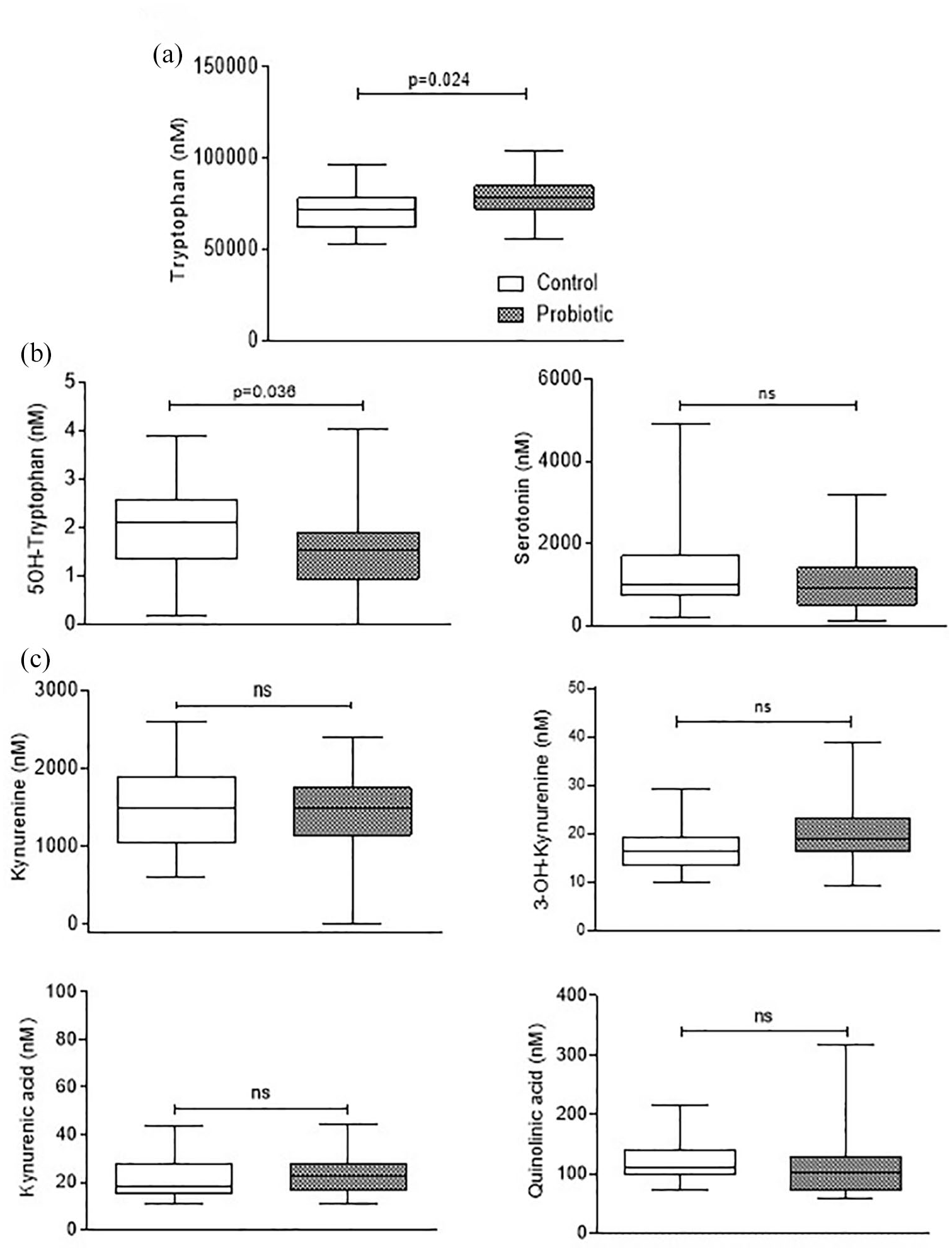
Quantification of tryptophan metabolites in the serum of T1D patients. Concentrations of tryptophan (a) and detectable metabolites generated in both serotonin (b) and kynurenine (c) pathways in the sera of control (white box) and probiotic (dark box) groups. Metabolite concentrations were represented as box and whiskers (Min to Max) and analyzed by two-tailed unpaired Student’s t test (probiotic vs control).
Analysis of kynurenine and cytokine secreted by PBMCs from T1D patients treated with the probiotic LGG
Defective tryptophan catabolism mediated by IDO1 has previously been observed in IFN-γ-stimulated PBMCs from a majority (66%) fraction of pediatric T1D patients, as compared to healthy controls. 15 To assess whether probiotic treatment of T1D patients might affect IDO1 activity in PBMCs, secreted Kyn was measured in the culture supernatants of either untreated or IFN-γ-stimulated PBMCs from both groups of T1D patients. No differences were detected in Kyn levels produced by untreated PBMCs from both groups, nor did IFN-γ stimulation induce differential effects in the two groups of samples (Figure 3a), suggesting that the increased serum concentration of Trp induced by probiotic treatment does not translate into increased IDO1 activity in T1D PBMCs. However, T1D patients regularly treated with the probiotic LGG showed a less inflammatory profile in the cytokine secretion pattern of PBMCs. The use of a multiplex panel to analyze mediators (ie, cytokines and soluble CD40 ligand (CD40L)) secreted in culture supernatants by PHA activated-PBMCs in vitro revealed an overall restrainment of proinflammatory mediators in PBMCs from the probiotic group, as compared with controls (Figure 3b). In particular, a significant decrease was observed in IFN-γ and IL-17F productions by PBMCs from probiotic-treated patients; a decrease, albeit not statistically significant, in IL-6 levels was likewise detected in the same cells (Figure 3c).

Levels of Kynurenine and cytokines secreted by PHA-activated PBMCs from T1D patients. (a) Kyn levels in supernatants of T1D PBMCs, isolated from control and probiotic groups, stimulated in vitro with 100 U/mL IFN-γ for 48 hours. Kyn levels were represented as the mean ± SEM of the concentrations (µM) and analyzed by one-way ANOVA, followed by post hoc Bonferroni’s test. ***p < 0.001. (b) Heat map showing cytokine levels in culture supernatants from PHA-activated PBMCs of control and probiotic groups, as indicated at the bottom of the heat map. Each row corresponds to an individual cytokine expressed as mean concentration (pg/mL). Color codes are as follows—red for the highest levels (>200 pg/mL) and green for the lowest levels (<100 pg/mL). (c) Cytokine levels were determined in culture supernatants of PHA-activated T1D PBMCs, isolated from control and probiotic groups. Cytokine levels are expressed as the mean ± SEM of the concentrations (pg/mL) and are analyzed by multiple Student’s t test per row (probiotic vs control), corrected by Sidak-Bonferroni’s method. **p < 0.01.
Discussion
Perturbation of Trp metabolism in dysbiotic gut has been described in different pathogenic settings, 25 however very few data report the possibility of modulating Trp metabolism in human disorders, specifically in pediatric T1D, by manipulating gut microbiota. Preliminary observations in our laboratory had indicated the possibility of increasing serum Trp levels in pediatric T1D patients via a 3-month probiotic course with LGG. Increased circulating Trp was not employed in the serotonin pathway for the synthesis of 5-OH-Trp and serotonin, whose levels decreased in T1D patients after probiotic administration. Trp did not even take the road of the kynurenine pathway, as the levels of detectable metabolites along this pathway did not differ between the two experimental groups. These observations support the hypothesis that the probiotic treatment could increase circulating levels of Trp by down modulating the serotonin pathway. Apparently, the main Trp-derived metabolic branches—the kynurenine and the serotonin pathways—seem not to be interconnected in the pediatric T1D cohort, differently from previous observations in preclinical models of depression associated diabetes and EAE.23,24 Since Trp is also employed by gut bacteria for the production of immune active indole derivatives, 26 we also searched for the indole metabolites IAld and IPA. No significant differences were detected in circulating levels of IAld and IPA between the two groups. A deeper metabolomic analysis of stool samples probably would have contributed to a better refinement of the Trp fate after the probiotic treatment. As expected, circulating Trp levels increased by the probiotic treatment did not translate into higher IDO1 activity in T1D PBMCs, where the same pathway was previously demonstrated defective because of the inflammatory milieu that accelerates IDO1 protein turnover, via proteasome degradation.13,15 Nevertheless, a concomitant restrainment of the inflammatory profile of PBMCs was observed in probiotic-treated patients, with significant lowering of the cytokines IFN-γ and IL-17F. Overall, a 3-month probiotic treatment can change the Trp-focused metabolomic profile in the sera of pediatric T1D patients, by lowering the serotonin pathway with increased serum Trp levels, and by decreasing the secretion of proinflammatory cytokines in T1D PBMCs. For the first time, this exploratory study demonstrates the possibility of manipulating Trp metabolism in pediatric T1D by a probiotic treatment and represents an encouraging starting point for searching in-depth, perhaps multiple, correlations between tryptophan metabolism and diabetic dysbiotic gut in pediatric T1D patients. A correct repartioning of Trp fluxes in the multiple metabolic pathways could represent a promising strategy for preventing or treating T1D. Trp metabolism in the gut can become an actionable actor from a therapeutic perspective, using either probiotics for directing Trp metabolic fluxes or circulating Trp-derived metabolites as predictive biomarkers.
Supplemental Material
Supplementary_material – Supplemental material for Effect of Probiotic Administration on Serum Tryptophan Metabolites in Pediatric Type 1 Diabetes Patients
Supplemental material, Supplementary_material for Effect of Probiotic Administration on Serum Tryptophan Metabolites in Pediatric Type 1 Diabetes Patients by Giada Mondanelli, Elena Orecchini, Claudia Volpi, Eleonora Panfili, Maria Laura Belladonna, Maria Teresa Pallotta, Simone Moretti, Roberta Galarini, Susanna Esposito and Ciriana Orabona in International Journal of Tryptophan Research
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Italian Ministry of Education, Universities and Research (PRIN2017- 2017BA9LM5 to CO).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s Contributions
GM, EO, MTP, MLB performed in vitro experiments. CV, EP performed ex vivo analysis on PBMCs. SM, RG performed sera metabolomics analysis. SE recruited T1D patients. CO researched data, wrote the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
