Abstract
Background:
BeaT-2/Sugarburner is a nutritional supplement based on a co-existing microbial consortium of bacterial strains. The purpose of this study was to investigate potential beneficial effects of the supplement versus placebo on glycemic control and on biomarkers of inflammation, insulin resistance and ß-cell dysfunction.
Methods:
A total of 40 Patients with type 2 diabetes (31 male, 9 female, age:65.5 ± 8.0 years, BMI: 33.6 ± 5.5 kg/m², HbA1c: 7.2 ± 0.9%) were included into the study. They were randomized to receive 2 capsules of either BeaT-2 or placebo once daily for 6 weeks. Observation parameters were time in normoglycemia as assessed by continuous glucose monitoring, and biomarkers of glycemic control, ß-cell function, insulin resistance and chronic systemic inflammation.
Results:
Time in range was stable with BeaT-2 between weeks 0 and 2 versus weeks 4 and 6, while it slightly impaired with placebo (−4%, n.s.). At endpoint, there were significant improvements versus baseline with BeaT-2, but not with placebo for biomarkers of glycemic control (BeaT-2 vs Placebo, HbA1c: −0.3% vs 0.1%, fasting glucose: −14% vs +8%, both P < .05), insulin resistance (insulin: −17% vs +14%, HOMA-IR: −26% vs +21%, both P < .05), ß-cell dysfunction (intact proinsulin: −40% vs −2%, P < .05) and chronic systemic inflammation (adiponectin: +8% vs −8%; P < .05, hsCRP: −31% vs +27%, n.s.). In addition, observed changes in the lipid profiles and other parameters of metabolic syndrome were more favorable with BeaT-2 than with placebo. There were no differences between the groups with respect to number and type of adverse events.
Conclusion:
The results observed with BeaT-2 were comprehensively indicative for improvements in the cardiometabolic situation. BeaT-2 may be a valuable supplement to any existing treatment combination in patients with type 2 diabetes.
Keywords
Introduction
There is increasing evidence that gut microbiota is involved in the development of human diseases, including but not limited to disorders of immune system, energy metabolism, lipid metabolism, and glucose metabolism.1,2 Appropriate development of the metabolic functionality of the human body is strongly driven by this symbiosis between intestinal microbiota and the host organism. The amounts of microorganisms in a human-being is estimated to be around 3.8 × 1013, with a total mass between 200 g and 1 kg of the total body weight of a person.3 -5 A wide range of biological processes is influenced by the gut microbiota, such as energy balance homeostasis or digestion of nutrients. The trigger for this function is the production of short-chain fatty acids (butyrate, propionate, and acetate), which are considered as signaling molecules in gut and extraintestinal tissues. 6 Other functions are a participation in the transport and metabolism of carbohydrates and amino acids, in the production of fat-soluble and water-soluble vitamins, 7 in elimination of xenobiotics and drugs, and in bile acid metabolism. 8 Disturbances in composition and subsequently also in the functionality of the host-microbiota interaction can result in a chronic systemic inflammation induced by bacterial fragments, which appear in the circulation as consequence of a malfunctioning gut barrier. Hence, a malfunctioning gut barrier, has considerable trigger effects for host adiposity and insulin resistance. 9
Recent evidence suggests that there are certain bacterial species that may interact with host metabolism, for example, through metabolite-mediated stimulation of enteric hormones. Modulation of the gut microbiota by means of external supplementation with bacteria and probiotics may offer an interesting opportunity for supplementary treatment of diabetes, obesity and other inflammation-related diseases. 10 By definition probiotics are substances contained in certain foods, which may modulate the composition of gut microbiota, thereby influencing short-chain fatty acid production, satiety, weight control, energy homeostasis, and suppressing the growth of pathogens and immunomodulatory actions.11,12 Indeed, probiotics have been shown to be effective in varied clinical conditions - ranging from infantile diarrhea, necrotizing enterocolitis, antibiotic-associated diarrhea, inflammatory bowel disease to cancer, female urogenital infection, surgical infections, insulin resistance, obesity, and oxidative stress.13 -15
A nutritional supplement comprised of bacterial and yeast strains, which constitute the probiotic properties of the product, has been used for more than 10 years by more than 1400 volunteers without observing any relevant side effect, when taken alone with dietary interventions or in addition to other common drugs, for example, cardiovascular drugs, anti-diabetic drugs, anti-inflammatory drugs, cytostatic drugs, antibiotics and others.
The purpose of this study was to initiate the generation of scientific evidence for BeaT-2 by verifying and quantifying the anecdotally reported beneficial effects on glycemic control and on biomarkers of inflammation, insulin resistance and ß-cell dysfunction. In this double-blind study, BeaT-2 or placebo was given as a nutritional supplement in addition to standard of care treatment interventions for 6 weeks in patients with type 2 diabetes mellitus in late disease stages.
Patients and Methods
This prospective double-blind placebo-controlled monocentric parallel study was conducted in accordance with international and local ethical and scientific standards. The protocol was approved by the responsible ethical review board (Landesärztekammer Rheinland-Pfalz, Mainz, Germany), and reported to the responsible national authority (Bundesamt für Verbraucherschutz und Lebenmittelsicherheit). The study was registered at the German Registry for Clinical Studies (DRKS No. DRKS00023745).
Primary objective was the impact of BeaT-2 on glycemic control (time in normoglycemia) as compared to placebo and as assessed by continuous glucose measurement by means of the Freestyle Liber 2 device (Abbott Diagnostics, Wiesbaden-Delkenheim, Germany) after 2, 4, and 6 weeks of treatment. Secondary objectives were the effects of the dietary supplement versus placebo on HbA1c, lipids, uric acid, insulin sensitivity (as assessed by HOMA-score 16 ), ß-cell dysfunction (insulin and intact proinsulin17 -20), chronic systemic inflammation and cardiovascular risk (total adiponectin and hsCRP,19 -21), and the tolerability of the nutritional supplement (type and nature of (serious) adverse events).
To be eligible for the study, patients had to be adult and diagnosed with Type 2 diabetes (HbA1c between 6.0% and 9.9 %, while treated with any kind or combination of standard of care treatments). They were not allowed to suffer from type 1 diabetes, any acute or chronic gastrointestinal disease, anemia, acute hyperthyreosis, known allergy against probiotic nutritional supplements, or any major fatal disease.
Prior to enrollment the participants signed written informed consent. Thereafter, blood was drawn for the safety analysis and to identify potential exclusion criteria. The randomization into the 2 study arms and blood draws for determination of efficacy parameters took place at the baseline visit (V1). The entire observation period was 6 weeks. A subcutaneous sensor for continuous glucose monitoring (CGM, FreeStyle Libre, Abbott) was provided for the next 2 weeks. Three follow-up visits (V2, V3, V4) were scheduled to happen in 2-week intervals to exchange the CGM sensor and to draw blood for the efficacy analysis. Conclusion of visit 4 terminated the participation of the individual patient in the study.
All safety and efficacy parameters were measured at a central laboratory. Immunoassays were used for determination of insulin, intact proinsulin and total adiponectin (TecoMedical, Sissach, CH). Lipids, hsCRP, glucose, and HbA1c were determined with a standard laboratory analyzer (Cobas c513, Roche Diagnostics, Basel, CH). At each treatment visit, the patients were asked for occurrence of any (serious) adverse events.
BeaT-2 (also referred to as “Sugarburner”; de Faire Medical AB, Uppsala, Sweden, and Arminius Pharma, Frankfurt, Germany, www.beat-2.de) is a nutritional supplement based on a co-existing microbial consortium of bacterial strains, which constitute the probiotic properties of the product. The product composition is provided in Table 1.
Composition of Beat-2/Sugarburner (Per Capsule = 500 mg).

The product action is driven by excreting bioactive substances, such as enzymes, that break down complex organic molecules into nutrients, reactants and energy. The bacterial strains included into the product (eg, Bacillus subtilis, Bacillus coagulans, and Pediococcus pentosaceus) preferably metabolize glucose and other more complex carbohydrate molecules and degrade them directly into CO2 and water. In consequence, the degraded glucose does not reach the blood anymore and cannot contribute to energy balance. BeaT-2 has no active hypoglycemic action and does not affect normal glucose levels in healthy people or patients with diabetes.
The data was evaluated using methods of standard exploratory and descriptive analyses to gain an understanding of the qualitative and quantitative nature of the collected data. For quantitative variables arithmetic means, medians, standard deviations, and minimum and maximum values were determined. Appropriate parametric and non-parametric statistical tests were used to compare the collected results. For normally distributed data sets, students t-test was used to compare the changes from baseline to endpoint, and to compare the changes from baseline between the groups (Software: Statistica, Version 14.1.0, TIBCO Software Inc., Palo Alto, CA). A P-value < .05 was considered to be statistically significant.
Results
A total of 46 patients were screened of whom 40 meeting the inclusion/exclusion criteria could be enrolled into the study (31 men, 9 women, mean age: 65.4 ± 8.0 years (range: 44-89 years.), BMI: 33.3 ± 5.3 kg/m² (range: 21.5-42.8 kg/m²). HbA1c: 7.2 ± 0.9% (55 ± 10 mmol/mol; range: 6.0%-9.4%/42-79 mmol/mol)). All included patients performed the study per protocol. After randomization, there were slightly more women in the placebo arm. The vast majority of the patients were on combination treatments with multiple anti-diabetic drugs indicating an advanced disease stage. The patient characteristics at baseline for both treatment arms and the nature and distribution of the diabetes treatments are provided in Table 1.
Primary measurement variable was time in normoglycemia as determined by means of the FreeStyle Liber analysis software between weeks 0 and 2 and week 4 and 6. It must be noted that there was no data available from the time before the patients started to take the study supplements. Therefore, a true baseline to understand the immediate impact of BeaT-2 supplementation on time in normoglycemia for the comparison is missing. The comparison between the first and the last sensor analysis in this trial is provided in Figure 1.

Changes in the continuous glucose monitoring analysis between the first (week 0-2) and the last 2 weeks (week 5-6) of the study observation period.
It can be seen that patients taking BeaT-2 had stable glycemic control, while a slight but non-significant impairment could be observed in the placebo group.
The differences that were seen in the majority of the clinical examination parameters (body weight, blood pressure, waist circumference) were numerically more favorable for BeaT-2 but not significant. Only an observed reduction in hip circumference was significantly more pronounced with BeaT-2 (from 121 ± 11 cm-116 ± 11 cm, P < .005) as compared to placebo (116 ± 11 vs 116 ± 10 cm, n.s.).
To explore the impact of BeaT-2 on biomarkers of glycemic control and the underlying diabetes deteriorations (insulin resistance, ß-cell dysfunction and chronic systemic inflammation) a panel of biochemical parameters were measured at baseline and endpoint. The results are provided in Table 2 and the percent changes from baseline in both groups are provided in Figure 2 (Table 3).
Patient Characteristics at Baseline in Both Treatment Arms.

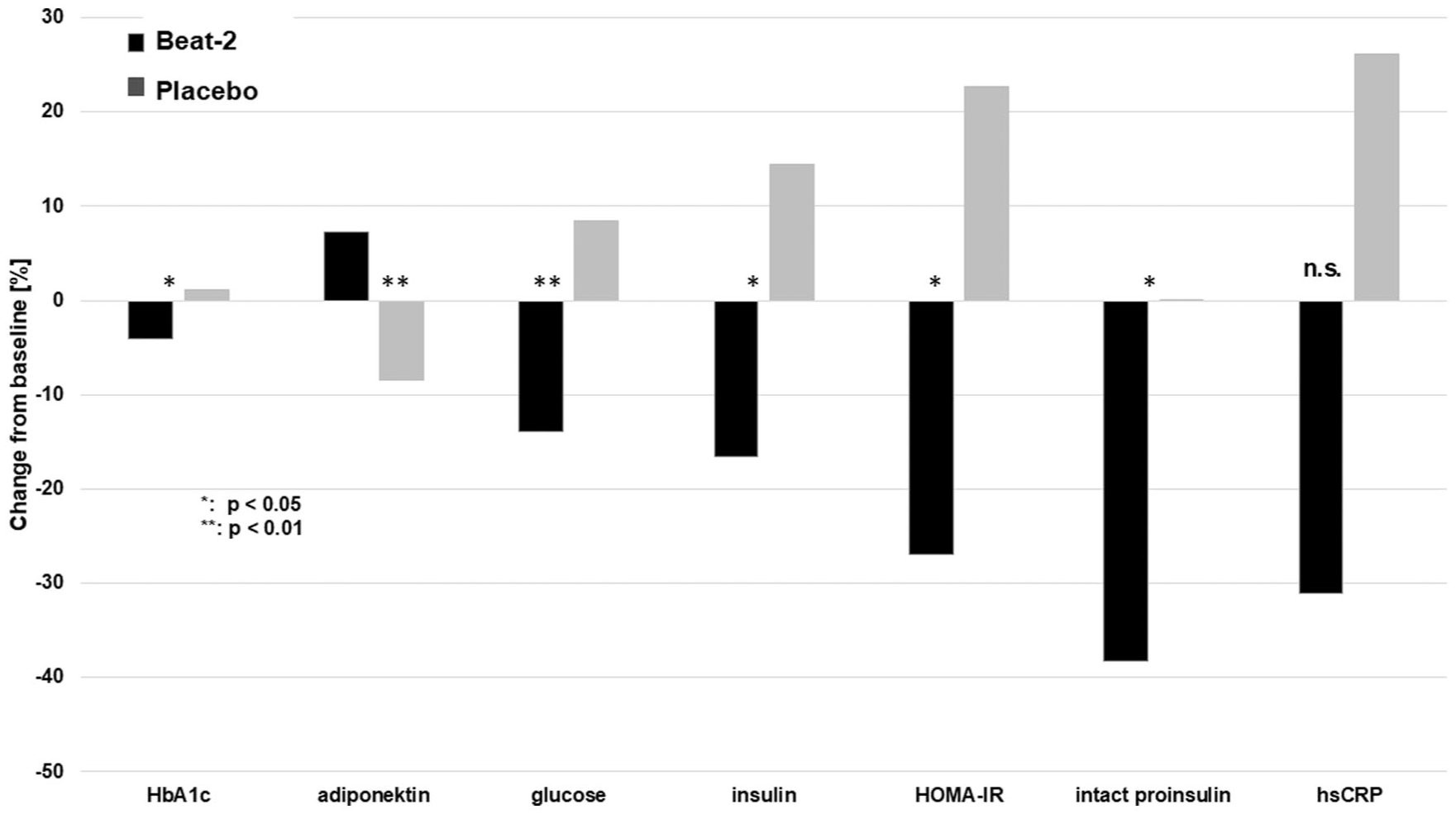
Changes from baseline after 6 weeks of treatment of the measured biomarkers for glycemic control and for the underlying diabetes deteriorations (n = 40).
Results for the Glycemic and Cardiodiabetes Parameters at Baseline and Endpoint.

P versus baseline.
P < .05. **P < .01. (*): strong trend = P < .1.
Hemoglobin A1c as well as fasting glucose improved significantly with BeaT-2, while no change (HbA1c) or an impairment (fasting glucose) was seen with placebo.
Intact proinsulin, insulin and the HOMA-IR score (were determined as indicators for insulin resistance and ß-cell dysfunction.16 -18 All of them improved significantly with BeaT-2, while they further deteriorated with placebo. All changes from baseline with BeaT-2 as well as the differences between the groups reached the level of statistical significance. These findings are indicative for a pronounced positive impact of BeaT-2 on insulin resistance in this trial.
Biomarkers indicative for chronic systemic inflammation determined in this study were total adiponectin and hsCRP.19 -21 Adiponectin increased significantly with BeaT-2 and decreased with placebo. These observations were also highly significant for changes between the groups (P < .001). BeaT-2 lead to a numerical reduction in hsCRP by more than 30%, while it increased by more than 20% with placebo. However, due to a high variability observed, the changes between the groups did not reach the level of statistical significance.
With respect to other laboratory biomarkers of metabolic syndrome (triglycerides, cholesterol, and uric acid) an almost similar picture could be observed (see Table 2). There was a statistically significant improvement in the triglyceride concentrations with BeaT-2 but not with placebo. There were strong trends in favor of BeaT-2 for the total cholesterol and HDL-cholesterol levels. Observed impairments for LDL-cholesterol and uric acid were less pronounced with BeaT-2 than with placebo. These findings are in line with the overall positive laboratory picture observed with BeaT-2 in comparison to placebo.
The supplements were well tolerated, and there were no serious adverse events reported in this trial. A total of 33 adverse events were reported, of which 18 occurred with placebo and 15 occurred with BeaT-2. Symptoms associated with problems occurring in the gastro-intestinal tract were seen in 6 cases with placebo and only one case with BeaT-2. Hypoglycemia was reported 6 times from 2 patients with BeaT-2 and 4 times from 4 patients with placebo. From the reported adverse events it can be concluded that the nutritional supplement had no special side effects in this trial. In addition, there were no clinically relevant changes during the study in any other value of the safety biochemistry panels taken before and after the study.
Discussion
Diabetes mellitus is a chronic systemic disease associated with complications in most of the body organs. It is a pandemic, which affects ~9% of the population in the world. 22 Changes in pancreatic beta-cell function and insulin resistance lead to relative insulin deficiency and impaired cellular response to insulin. A significant symptom of the disease is hyperglycemia, which can cause several severe secondary complications.23,24 Anti-diabetic drugs are commonly used in accordance with national and international treatment guidelines25,26 and the most commonly used drug, metformin, can have limited effect and can cause adverse effects on the composition of the bacterial microfauna in the intestine tract. Recently, the gut microbiota was found to be altered in diabetic patients, for example, with increased ratios of Bacteroides to Clostridium species in the gut, and increased numbers of various opportunistic pathogens.27 -30 Circulating gram-positive gut bacteria were detected in blood samples of diabetic patients. 29 Changes in gut microbiota may lead to metabolic endotoxemia through the release of lipopolysaccharides, consequently stimulating inflammation and insulin resistance. 31 The epithelial enteroendocrine L cells play a role in producing inflammation, and their number was positively or negatively correlated with the abundance of 25 bacterial taxa in the intestine.32,33
In light of these findings, the effect of probiotic supplementation in diabetes and diabetes-related complications gets into the focus of modern research. Animal studies showed positive results, such as reductions in levels of blood glucose, hemoglobin A1c (HbA1c), and insulin resistance.34,35 Also, first studies in humans were performed to investigate the clinical effect of probiotics on glycemic control.14,36,37 In these placebo-controlled trials, probiotic fermented milk, probiotic yogurt, bread, and kefir mainly containing different Lactiplantibacillus strains and Brewer’s yeast were administered for 6–12 weeks. In a meta-analysis of these studies, significant improvements were reported for HbA1c, fasting glucose and the HOMA-IR score, while non-significant improvements were seen in insulin and fasting proinsulin levels. 38
The BeaT-2 development is different to these interventions so far, as special bacterial strains have been selected, which preferably metabolize glucose and other carbohydrates, and which should therefore provide an even stronger impact on glucose levels and insulin resistance. Within the limitations of this study protocol, this hypothesis of an even more pronounced effect seems to be confirmed by the observed results, in particular when employing a laboratory biomarker panel, which describes the diabetes phenotype by indicating the severity of the underlying diabetes deteriorations.20,39
The net result of BeaT-2 supplementation seems to be a lower absorption of glucose through the intestine tract as postulated. A similar effect and corresponding biochemical findings were seen, when drugs belonging to the class of SGLT-1 inhibitors (α-glucosidase inhibitors, eg, acarbose or voglibiose) are used in routine diabetes treatment. The difference between the 2 approaches is that SGLT-1 inhibitors actively withhold the glucose molecules in the intestine tract, where they finally get metabolized by bacteria populating the large bowel, with methane gas as the final end product. In consequence, patients treated with SGLT-1 inhibitors were usually complaining about major problems with flatulence and bloated belly.40,41 In contrast, BeaT-2 bacteria metabolize glucose and other carbohydrates into CO2 and water in the upper part of the ileum. CO2 formation in this part of the GI tract appears to be less problematic as the patients treated with BeaT-2 reported less GI problems than patients treated with placebo in this study. The majority of the patients in both treatment arms were already using insulin in different treatment regimens, indicating a somewhat advanced disease stage. The positive effects of BeaT-2 on biomarkers of the underlying diabetes deteriorations must be quite pronounced because consistent positive and significant results were seen for almost all of the investigated indicators.
The nutritional supplement was shown to have an excellent tolerability in particular when taken in combination with many drugs and drug combinations used for treatment of chronic diseases, such as diabetes, hypertension, lipid disorders etc. There may even be the possibility that the regulation of the gut microbiome by means of the product may help to reduce side effects from other treatments, and a number of positive effects were picked up from the safety field tests, such as alleviated intestinal inflammations, stabilized glucose levels, reduction of alcohol digestion and detoxification and more. These observations are in line with similar results from a previous study where we investigated the impact of related bacterial strains on ethyl alcohol absorption.42,43
There were several limitations in our study, which needs to be addressed. Firstly, the baseline measurement of the time in normoglycemia started after the patients also started to use the nutritional supplements. We have therefore missed any immediate effect of BeaT-2 on this CGM-based parameter and can only determine that a slight impairment of time-in-range was seen with placebo but not with BeaT-2 at the end of the study. Secondly, the study duration of 6 weeks is not long enough to draw final conclusions about the impact of BeaT-2 on HbA1c. Still the significant reduction observed in this pilot study is an encouraging result to expect similar results in subsequent studies with longer study duration. Finally, the randomization resulted in a slight misbalance of the genders with a higher percentage of females in the placebo arm. As female patients are usually expected to be more adherent to pharmaceutical treatments and lifestyle recommendations than men, 44 this may have even influenced the differences observed between the groups in favor of placebo.
Conclusions
The results observed with BeaT-2 were comprehensively indicative for improvements in the cardiometabolic situation, while the opposite was seen for all parameters with placebo. Considering the intensive background diabetes treatments taken by the participating patients, these results are a strong indicator that BeaT-2 is a valuable supplement to any existing treatment combination in patients with type 2 diabetes. Appropriately designed long-term comparative clinical studies are now required to confirm the potential value of BeaT-2 when added to drug and lifestyle treatments at the different disease stages.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by de Faire Medical AB.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JDF is founder and shareholder of de Faire Medical AB. PCP is employee of de Faire Medical AB, APF has received a study grant from de Faire Medical AB. The other authors have no conflict of interest to report.
