Abstract
Background:
Neonatal hyperinsulinemia is a rare condition, but poses a significant increase in the risk of recurrent and persistent hypoglycemia in newborns, which can lead to long-term neurodevelopmental complications if not identified early. This condition may be due to a congenital disorder, or arise transiently due to perinatal stress, and may develop as a consequence of interventions such as total parenteral nutrition (TPN). Infants diagnosed with SBO following extensive intestinal resection are particularly at higher risk.
Case Presentation:
This report is of a 2-week-old male neonate, delivered at 36 weeks gestation with a birth weight of 2.5 kg, the patient presented with jaundice, bilious emesis, and poor weight gain. Surgical intervention showed a midgut volvulus with extensive ischemic necrosis. A near-total resection of the small intestine performed. As a result, the patient developed short bowel syndrome as a result and required total parenteral nutrition (TPN). Even with administration of high-dose intravenous glucose (up to 13 mg/kg/min), the patient continued to experience recurrent, persistent hypoglycemia. Elevated insulin levels alongside normal cortisol, ACTH, growth hormone, and ammonia are revealed by laboratory evaluation, the profile suggest a stress-induced hyperinsulinemia. Management included careful glucose monitoring, optimization of TPN, and initiation of pharmacotherapy including diazoxide with hydrochlorothiazide. This approach led to stabilization of blood glucose and gradual transition to partial enteral feeding.
Conclusion:
This case highlights the complexity of diagnosing and managing hyperinsulinemia in neonates with short bowel syndrome who totally depend on TPN. Persistent hypoglycemia despite high glucose infusions should raise the concern of hyperinsulinism. Early identification is important. A multidisciplinary team approach is required to avoid serious complications.
Keywords
Introduction
Short bowel syndrome (SBS) is considered as the most common cause of intestinal failure in neonatal intensive care units, this possibly due to multiple surgical intervention that frequently required because of life-threatening intestinal conditions. 1 SBS can occur due to different causes in neonates and young infants including necrotizing enterocolitis, gastroschisis, intestinal atresia, or malrotation with volvulus.1 -3 Among these etiologies, volvulus represents a particularly acute and severe event in which the twisting of the intestine disrupts vascular supply, causes rapid tissue death, and requires immediate surgical intervention.1,4,5
After an extensive resection of the intestine due to volvulus, the remaining gastrointestinal tract undergoes a complex physiological process called intestinal adaptation, aiming to optimize absorption from the remaining gut. 1 Residual intestinal length is an important prognostic factor. infants with less than 50 cm experience a particularly poor prognosis, and are considered to be at higher risk for dependence on parenteral nutrition. 6
Residual intestinal length considered as one of the most important prognostic factors in predicting the outcomes of neonatal short bowel syndrome. However, not only residual intestinal length influence SBS outcomes, according to recent studies, several additional factors play an important role. Factors include the presence or absence of the ileocecal valve, residual colon length, early initiation of enteral nutrition, and the efficiency of intestinal adaptation processes such as mucosal remodeling and epithelial proliferation.7,8 all these factors influence how well nutrients get absorbed and how long a child might need parenteral support.
Total parenteral nutrition (TPN) represents a significant improvement in the management of short bowel syndrome, enabling patients to maintain essential growth and development even when enteral feeding is inadequate or impossible. Nevertheless, TPN associated with some major long-term risk to watch for, one of the most important risk factors is line infections. Other risk factors include sepsis, liver complications, and cholestatic disease which can all threaten survival if not addressed immediately.6,9 One particularly challenging issue is the development of persistent hypoglycemia. Even when clinicians optimize glucose delivery via TPN, some patients continue to experience low blood sugar levels, which underscores how it’s very complex to manage these cases. 9
Neonatal hyperinsulinemia is a condition characterized by excessive insulin secretion, leading to persistent episodes of hypoglycemia. The condition could be transient usually caused by perinatal stress, nutritional excess, or metabolic adaptation in the early days of life. Alternatively, it can be permanent which result from congenital abnormalities affecting pancreatic β-cell regulation.10,11 Total parenteral nutrition (TPN), particularly when high glucose infusion rates (GIR) are used, can provoke significant hyperglycemia. The pancreas often responds by increasing insulin secretion, it sometimes fails to return insulin levels again to normal even when blood glucose dips. This disrupts the normal interplay between glucose levels and insulin release. 12
Management of these scenarios include frequent monitoring of glucose level, carefully adjusting GIR, and if needed, adding pharmacological agents like diazoxide. Diazoxide functions through opening pancreatic ATP-sensitive potassium (KATP) channels, effectively suppressing excessive insulin secretion and helping to bring some order back to the system.13,14
In this case, we report a neonate who underwent a substantial small bowel resection and subsequently required TPN. The patient became TPN-dependent and developed recurrent hypoglycemia due to hyperinsulinemia. This case underlies the complexities of diagnosis such cases, challenging therapeutic decisions.
Case Presentation
The patient is a 2 week old male neonate who was delivered at 36 weeks gestation by cesarean section with a birth weight of 2.5 kg. Despite being fed both breast milk and formula, infant remained hungry, failed to gain weight, and developed prolonged jaundice accompanied by dark yellow, bilious vomiting. At the initial presentation, the patient present with nonspecific symptoms and actually got better with supportive care, also, abdominal examination did not demonstrate peritoneal signs. Because the early episodes of emesis were intermittently and happened along with jaundice, dehydration, and feeding intolerance, the clinical suspicion for obstructive pathology was initially low. Things changed when the patient present with worsening abdominal distension, persistent bilious vomiting, elevated inflammatory markers, and abnormal radiographs, at that point, urgent imaging and immediate surgical exploration were undertaken. Laboratory evaluation of the patient at presentation showed a markedly elevated total bilirubin of 204 μmol/L (reference: 3.4-22.3), and a direct bilirubin of 8.6 μmol/L (reference: 0-5.1). Also, random glucose level was 5.6 mmol/L (reference: 2.6-5.6). Additional findings included green nasogastric aspirate, abdominal X-ray evidence of significant bowel dilation with increased intraluminal gas with mild dehydration.
Initial management included initiate proton pump inhibitor therapy and erythromycin, alongside with starting of a hypoallergenic, amino acid-based formula in order to minimize allergenic triggers and reduce gastrointestinal workload. On postnatal day 17, the clinical status of the infants worsened and deteriorated, developing an abdominal distension with increased abdominal tension, and diminished bowel sounds. A follow-up abdominal radiograph obtained and showed air-fluid levels consistent with an obstructive process. Laboratory findings revealed increase CRP level to 214 mg/L, alongside with thrombocytosis (platelets 351 × 10^3/μL), and anemia (hemoglobin 10.4 g/dL).
Exploratory laparotomy revealed a midgut volvulus resulting in segmental gangrene involving approximately 10 cm of proximal jejunum. Surgical intervention included lysis of adhesions congenital bands, widening of the mesentery, and formation of a defunctioning jejunostomy. Despite these interventions, serial evaluations over the next 48 hours demonstrated ongoing ischemia and additional bowel necrosis, necessitating a second operation for resection of additional gangrenous bowel. Also, despite this intervention, the clinical status of the patient deteriorated, and a third emergency laparotomy revealed near total small bowel gangrene, sparing only about 5 cm of proximal jejunum and 6 cm of distal ileum. Aggressive resection of gangrenous tissue was performed, and antimicrobial therapy was initiated include piperacillin-tazobactam, metronidazole, and fluconazole. TPN was initiated by postoperative day 3 to manage the SBS.
Blood glucose level monitoring showed recurrent episodes of symptomatic hypoglycemia, although GIR were steadily increased and reach to 13 mg/kg/min (see Table 1). Endocrinology consultation was sought for further evaluation. During a hypoglycemic episode (lowest glucose recorded at 1.7 mmol/l), a normal levels of growth hormone, ammonia, cortisol, and adrenocorticotropic hormone (ACTH) are revealed through sampling. Insulin was moderately elevated at 8.9 μU/mL (reference: 2-20), all these findings collectively are compatible with stress-induced hyperinsulinemia.
“Trend of Glucose Infusion and Blood Glucose Responses Post-Bowel Resection in a Neonate.”
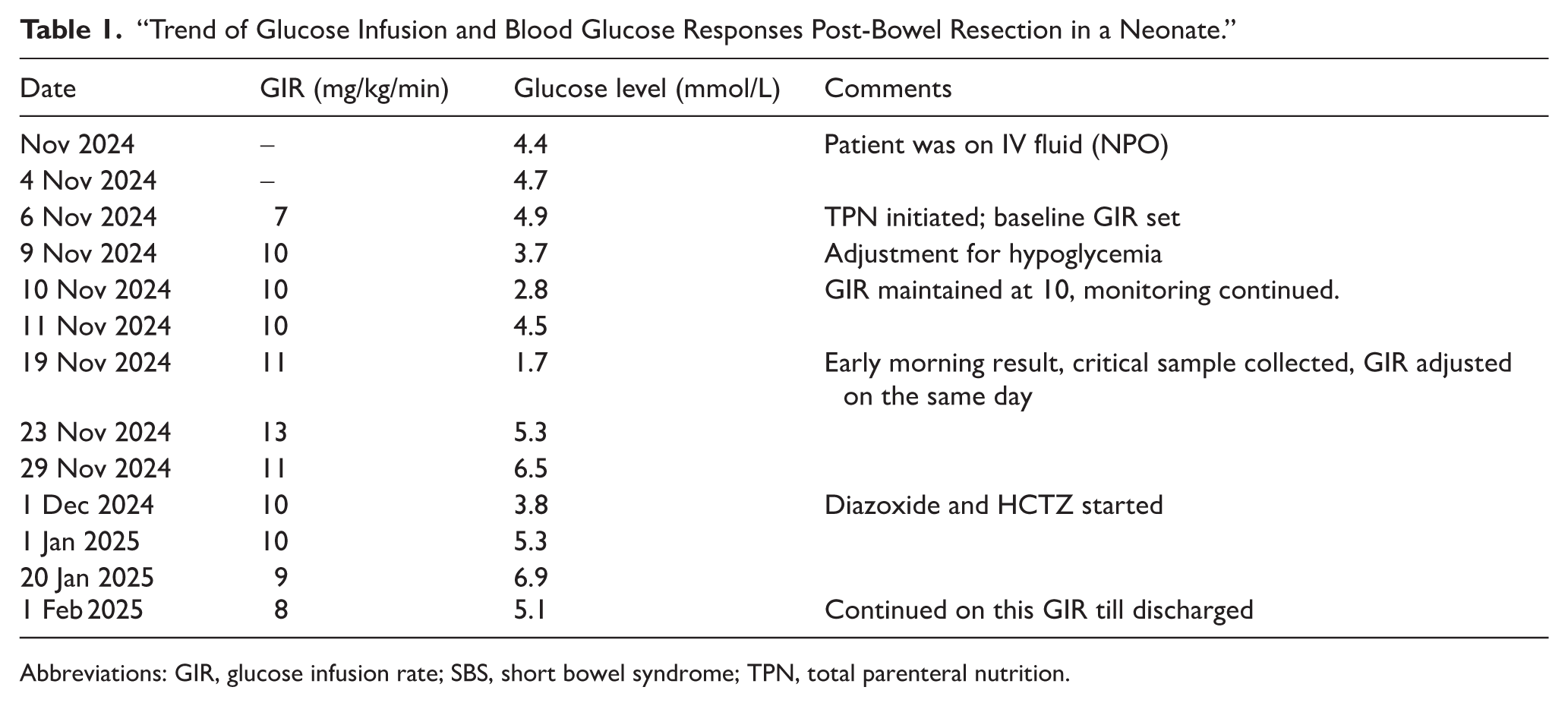
Abbreviations: GIR, glucose infusion rate; SBS, short bowel syndrome; TPN, total parenteral nutrition.
The therapeutic approach is a multidisciplinary approach involved strict blood glucose monitoring, through utilization of both sensor-based technology and traditional glucometer assessments. Pharmacotherapy such as Diazoxide was initiated and the dose was increased to reach 10 mg 3 times daily, alongside with hydrochlorothiazide that was introduced at 3 mg/kg/day. Adjustments to the TPN formulation were made periodically to optimize metabolic control. As a result, gradual improvement in glucose level was observed, enabling reduction of the GIR to reach 8 mg/kg/min without further episodes of hypoglycemia. Nutritional advancement continued alongside ongoing intestinal adaptation, with cooperation of a multidisciplinary team, a facilitated transition toward partial enteral feeding.
Discussion
This case underscores the complex, overlapping mechanisms than can lead to hypoglycemia in neonates with significant small bowel resection and those who relies on TPN. One cause is surgical stress (ischemia, reperfusion injuries, systemic inflammatory response) which serves as a potent factor for triggering excessive insulin secretion, usually out of proportion to actual blood glucose levels. Stress induced hyperinsulinism is a well-documented in neonates who’ve experienced perinatal distress and it’s associated with poor neurodevelopmental outcomes if diagnosis delayed, underscoring the urgency of timely intervention.15,16
Although patient insulin level was 8.9 μU/mL, which falls within the normal range. This remains inappropriate for a plasma glucose of 1.7 mmol/L. The pediatric endocrine society states that the diagnosis of hyperinsulinemia is based on insulin secretion that is proportionate to the glucose concentration. Detectable insulin during hypoglycemia, low ketones and free fatty acids, normal cortisol and growth hormone responses, and an increase in glucose immediately after glucagon administration, all collectively are considered as a supporting criterion.17,18 Thus, the biochemical profile in this instance was in line with hyperinsulinism brought on by stress.
In our case, persistent and recurrent hypoglycemia even with a high GIR suggests inadequate suppression of insulin, supporting the idea that the mechanism isn’t simply an insufficient glucose substrate. Moreover, TPN itself complicates the presentation, this is attributed to the rapid intravenous delivery of glucose and nutrients which can lead to exaggerated insulin response. Also, the absence of enteral feeding diminish incretin signaling glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide (GLP-1, GIP) and alter β-cell responsiveness to glucose through disruption of the usual gut-pancreas hormonal interplay. 12
According to recent guidelines, diazoxide remains the cornerstone, its currently the only FDA-approved pharmacologic option for managing most forms of hyperinsulinemic hypoglycemia in neonates. The recommend initiating dose is 10 to 15 mg/kg/day in divided doses. Careful monitoring is important to assess efficacy over several days and also to detect potential adverse effects such as pulmonary hypertension and fluid retention that may occurred.13,19 Recent literature mention that neonates treating with diazoxide may have increased risk of severe adverse events, especially in preterm infants, those with low birth weight, or pre-existing risk factors for pulmonary hypertension. 13
In our patient, close monitoring including assessment of fluid balance, respiratory status as indicated. In addition to pharmacotherapy, optimizing enteral nutrition, minimizing the need for high GIR, and continuous glucose monitoring. These measures can improve safety and allow earlier weaning off diazoxide therapy.10,14
Perinatal stress-induced hyperinsulinism (PSIHI) is associated with notable increase neurodevelopmental delays particularly when hypoglycemia is prolonged or recurs. According to recent data from published cohorts, over 40% of affected infants score more than 1 standard deviation below the mean on developmental assessments, and the deficits are more pronounced in motor and cognitive domains. 15 Early detection, prevention of severe hypoglycemic episodes, and careful management to minimize potential adverse drug effects are all important factors for improving outcomes. 16
This case contributes to the expanding literature on neonatal hypoglycemia, particularly in the context of intestinal failure and TPN dependence. In the literature, stress-induced hyperinsulinemia is underrecognized, even though it led to persistent hypoglycemia in many critically ill newborns. 10 When infants lose most of their small intestine, fluctuations in glucose homeostasis will occur. The reasons include disrupted incretin signaling, poor nutrient absorption, and the fact that TPN formulas are loaded with glucose. 20
Our report adds new insight, we show that hyperinsulinemia may occur even when insulin levels look normal or only slightly elevated. The case also highlights how vital it is to get endocrinology involved early when a TPN-dependent infant keeps having low blood sugar, even with aggressive glucose support. Furthermore, this case supports using diazoxide, in the right patients. Put simply, our findings give clinicians clearer signals for when to suspect hyperinsulinemia, how to interpret labs, and how to act when they see this pattern in the NICU.
Conclusion
This case demonstrate how complicated hyperinsulinemia can be in neonates especially in those undergone significant small bowel resections and are relying on total parenteral nutrition. Even with high GIR, persistent and recurrent hypoglycemia occurred, driven by stress-induced hyperinsulinism. Early identification is important and it’s linked to better prognostic outcomes and associated with decrease the risk of neurodevelopmental issues.
Management of this situation require a multidisciplinary approach, combining surgical expertise, endocrinology, neonatology, and nutritional support. All these collaborative efforts were essential for both glycemic regulation and gradual intestinal adaptation in the patient.
Footnotes
Abbreviations
Ethical Considerations
All procedures performed in this case report involving human participants were conducted in accordance with the ethical standards of the institutional and national research committees and with the principles of the Declaration of Helsinki (1964). Ethical approval was not required for this case report in accordance with institutional policies.
Consent to Participate
Written and verbal informed consent was obtained from the patient’s legal guardians for participation in this case report and for the use of relevant clinical data and images. A copy of the written consent is available for review by the Editor-in-Chief of this journal upon request.
Consent for Publication
The institution to which this case was admitted does not require approval for writing this case report.
Author Contributions
MRT and BHE were involved in the clinical management of the patient and data acquisition. DAR contributed to data interpretation and literature review. BA conceptualized the case report, coordinated the manuscript preparation, and served as the corresponding author. MA contributed to manuscript drafting and critical revision. All authors read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be provided on request from the editor-in-chief due to limitations.
