Abstract
Obesity is a complex metabolic disorder associated with chronic low-grade inflammation, insulin resistance, and heightened risk of comorbidities such as cardiovascular diseases and type 2 diabetes mellitus (T2DM). The Cluster of Differentiation 44 (CD44), a transmembrane glycoprotein, interacts with hyaluronic acid (HA), a major extracellular matrix (ECM) component, to regulate adipogenesis, immune cell infiltration, and metabolic dysfunction. Dysregulation of the CD44-HA signalling axis promotes adipose tissue hypertrophy, macrophage infiltration, and activation of pro-inflammatory pathways, including nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinases (MAPKs), which further aggravate metabolic disturbances. This review provides a comprehensive analysis of CD44-HA signalling in the pathophysiology of obesity and evaluates therapeutic strategies targeting this axis. Small-molecule inhibitors, such as CD44 antagonists and hyaluronan biosynthesis modulators (eg, 4-methylumbelliferone), offer advantages in synthesis, bioavailability, and cost-effectiveness, but face challenges related to limited specificity, systemic toxicity, and potential resistance. In contrast, Nanobioconjugates engineered nanoparticles conjugated with biomolecules such as HA enable targeted delivery to CD44-overexpressing adipose tissues, enhance therapeutic efficacy, prolong drug release, and minimize off-target effects. Examples include lipid-based nanoparticles, polymer-based nanocarriers, and HA-functionalized systems, all of which show promise in preclinical models of obesity. Comparative analysis underscores the superior specificity and bioavailability of nanobioconjugates, though issues of large-scale production, immunogenicity, and regulatory approval remain. Combining nanobioconjugates with small molecules may optimize treatment outcomes by leveraging synergistic mechanisms. Emerging strategies including high-affinity HA ligands, monoclonal antibodies, RNA-based therapeutics, and artificial intelligence (AI) guided drug design, offering new opportunities for precision obesity management. Future directions highlight the importance of personalized and combinatorial therapies, supported by biomarker profiling and gene editing technologies such as CRISPR, to overcome current limitations in CD44-HA-targeted interventions.
Keywords
Introduction
Obesity is a complex metabolic disorder characterized by excessive adipose tissue accumulation, leading to systemic inflammation, insulin resistance, and various comorbidities such as cardiovascular diseases and type 2 diabetes,1,2 and musculoskeletal diseases (Osteoarthritis (OA), Low Back Pain, Intervertebral Disk Degeneration, Gout, Tendinopathies, Osteoporosis and Sarcopenia, Carpal Tunnel Syndrome, Plantar Fasciitis, and Bone Fractures). 3 The public health crisis of obesity continues to grow worldwide because type 2 diabetes mellitus (T2DM). together with cardiovascular diseases (CVD) and particular cancers appear with this condition.1,4 The World Health Organization considers obesity as when a person achieves a Body Mass Index at or above 30 kg/m². 5 The multiple causes that lead to obesity create a complex physiological process starting from genetics and environment and ending in disturbed energy regulation. 6
The fundamental explanation of obesity is when energy input exceeds energy expenditure which creating a positive energy balance that leads to too much body fat storage.7,8 Body weight regulation happens through multiple central and peripheral systems which control hormonal signals together with neural pathways as well as inflammation. 9 The hypothalamus function as the central hub of the central nervous system to monitor hormonal and nutritional information for controlling metabolic actions and appetite behavior. White adipose tissues (WAT) features as the storage unit for energy together with lipid hormone production while brown adipose tissue (BAT ) functions by using uncoupling protein (UCP1) to generate heat.9,10 The expansion of white adipose tissues (WAT) beyond normal levels results in tissue malfunction which shows itself through enlarged adipocytes together with tissue inflammation, hypoxic conditions and fibrosis while causing metabolic problems. 11 Stronger weight gain in obesity drives WAT enlargement while it causes a metabolic dysfunction through this transformation of insulin-sensitive cells into insulin-resistant cells.12,13
Hypothalamic inflammation persists from obesity since excessive food consumption activates brain inflammation which causes the body to resist satiation signals and drives continued eating.14,15 Endurance of inflammatory responses weakens the blood-brain barrier efficiency for metabolic signal passage. The swelling of adipose tissues from excessive expansion creates long-term low-grade inflammatory conditions which trigger the body to produce inflammatory cytokines including tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and C-reactive protein (CRP). When macrophages penetrate WAT they intensify inflammation which leads to a destructive pattern of metabolic dysfunction. 16 The condition of being overweight leads to dyslipidemia which produces high triglyceride levels together with reduced highi density lipoprotein (HDL) and elevated low density (LDL).17,18 Pharmaceutical and biophysical defects from excess free fatty acids (FFAs) spill into tissues outside adipose tissue cause lipotoxicity and compel metabolic pathways to malfunction. 19 Endoplasmic reticulum tension and mitochondrial dysregulation increase as a consequence. Hepatic steatosis (also known as fatty liver disease) together with pancreatic beta-cell dysfunction occur because of lipotoxicity which makes insulin resistance and metabolic syndrome worse.20,21
The entire population of microorganisms inside the human gut actively controls both metabolic procedures and energy distribution. 22 The rise in Firmicutes/Bacteroidetes ratio along with endotoxemia and short-chain fatty acids imbalance linked to obesity results in more severe obesity-related health problems. 23 The risk factors for becoming obese include genetic and epigenetic elements because the genes Fat Mass and Obesity-Associated Gene (FTO), Melanocortin-4 Receptor Gene (MC4R), and Leptin Receptor Gene (LEPR) regulate appetite and energy processing functions. 24 The physiological breakdown of mitochondria alongside increased oxidative stress conditions create fundamental problems in metabolic dysfunctions of obesity. 25 The investigation of these mechanisms requires urgent study for creating treatment methods to manage obesity-related diseases while creating better global health results.
The pathology of obesity extends beyond a simple imbalance between calorie intake and expenditure, involving intricate neuroendocrine, genetic, epigenetic, and environmental factors. 2 Central to obesity is the dysregulation of energy homeostasis primarily orchestrated by the hypothalamus, which governs appetite and satiety through signaling pathways involving hormones such as leptin, insulin, ghrelin, and neuropeptide Y. 26 In obese individuals, leptin resistance impairs satiety signaling, perpetuating hyperphagia. Adipose tissue, once considered a passive fat reservoir, is now recognized as an active endocrine organ that secretes adipokines such as adiponectin, resistin, and pro-inflammatory cytokines like Tumor Necrosis Factor (TNF-α) and Interleukin-6 (IL-6). 24 These mediators contribute to systemic low-grade inflammation, insulin resistance, endothelial dysfunction, and a heightened risk for comorbid conditions including type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD), non-alcoholic fatty liver disease (NAFLD), obstructive sleep apnea, and certain cancers. 27
Despite the increasing recognition of obesity as a disease with profound systemic consequences, there remain numerous unmet clinical needs in its diagnosis, treatment, and long-term management. Current clinical approaches often rely on body mass index (BMI) as a diagnostic tool, which fails to distinguish between fat and lean mass or account for fat distribution parameters that significantly influence metabolic risk. 28 Moreover, many healthcare systems lack standardized protocols for routine screening and early detection of obesity-related complications, particularly in pediatric and geriatric populations.29,30 Pharmacologic interventions are limited by modest efficacy, high cost, and concerns regarding long-term safety, while bariatric surgery, though effective, is invasive, costly, and inaccessible to many due to socioeconomic and healthcare disparities. Additionally, obesity is often underdiagnosed and undertreated due to stigmatization, inadequate provider training, and societal perceptions that place sole responsibility on individual behavior rather than acknowledging the biological and structural determinants of obesity. 31 The need for a more comprehensive and personalized approach to obesity care is urgent. Emerging research into the molecular underpinnings of obesity such as alterations in the gut microbiome, brown adipose tissue activation, metabolic reprograming, and CD44-HA signaling pathway, offers promising therapeutic targets.
Role of CD44-Hyaluronic Acid (HA) Signaling in Obesity
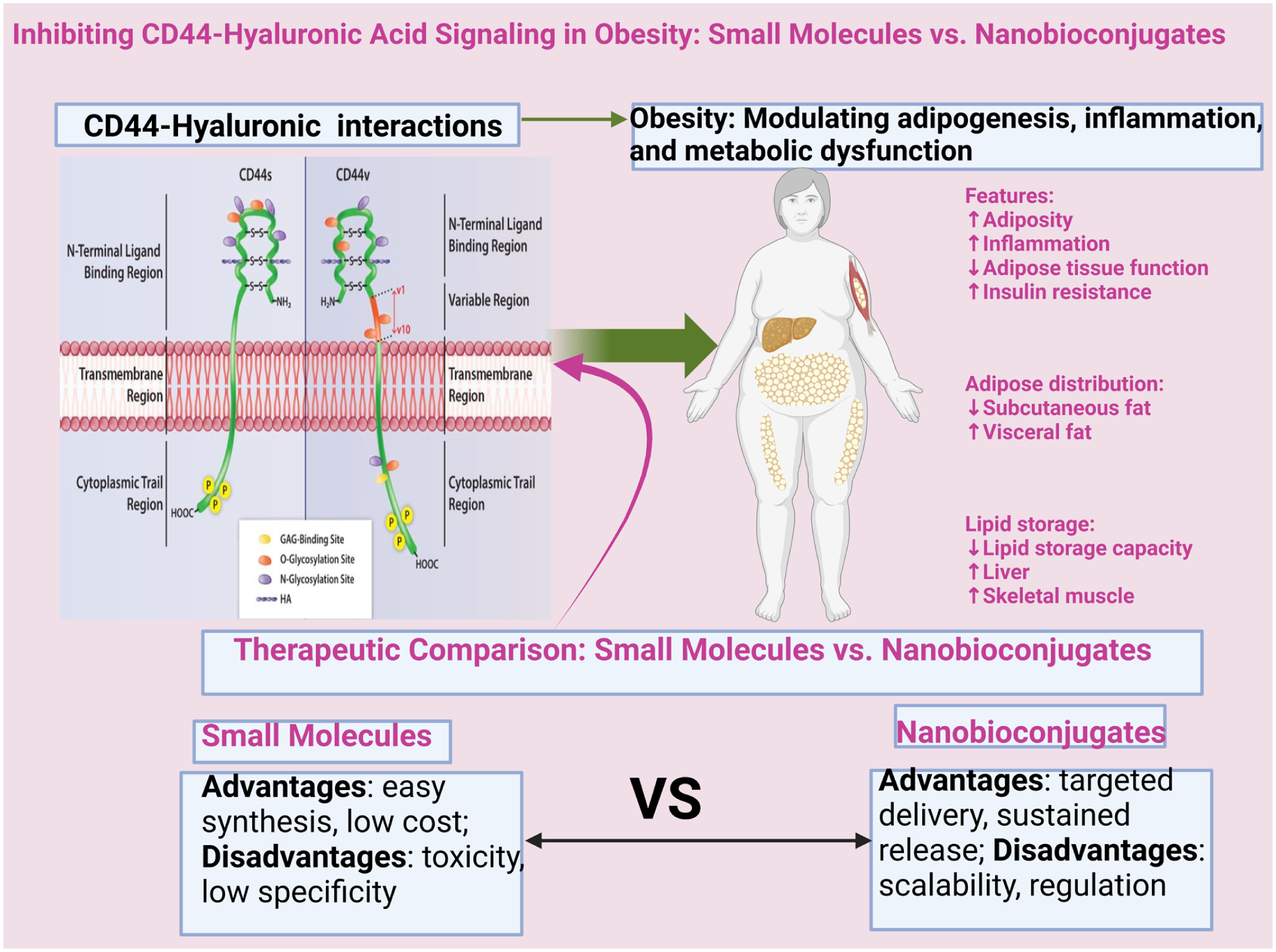
The interactions between CD44 and Hyaluronic Acid (HA) at the signaling level function as the central controller of adipogenesis alongside inflammation control and metabolic conditions during obesity 32 (Figure 1). The cell surface glycoprotein functions as a transmembrane mediator which sustains both cell-matrix bond formation and inflammatory responses as well as tissue remodeling activities. 33 The signaling axis operates incorrectly in obesity which creates problems with adipose tissue structure together with metabolic irregularities. CD44 functions as a type I transmembrane glycoprotein that occurs in immune cells together with endothelial cells and adipocytes. 34 The molecule functions as a bridge connecting cells with extracellularmatrix elements by binding to glycosaminoglycan HA which makes up a principal ECM part. The biological activities of high-molecular-weight HA (HMW-HA) differ from those of low-molecular-weight HA (LMW-HA) which belong to different HA weight classes. 35

The interactions between CD44 and Hyaluronic Acid (HA) at the signaling level function as therapeutic target. Adapted from Kotla et al. 36 Under a free open access license: https://creativecommons.org/licenses/by/4.0/. Interaction between CD44 and hyaluronic acid. The CD44 receptor exists in 2 forms: variant (CD44v) and standard (CD44s). It has 4 segments: an N-terminal hyaluronic acid-binding loop (BX7B), a stem region, a transmembrane domain, and a cytoplasmic C-terminal domain.
The CD44-HA signal plays an essential role in various physiological events together with pathological occurrences of tissue remodeling as well as inflammation and metabolic regulation. The cluster of differentiation (CD44)–hyaluronic acid (HA) pathway is necessary for maintaining communication between cells, cellular integrity, the immune system, and metabolism. 37 Large numbers of CD44 proteins can be found on the surfaces of adipocytes, immune cells and endothelial cells, among others. The matrix contains hyaluronic acid as its main ligand, making it an important structure of the extracellular matrix (ECM.) In a normal, healthy situation, CD44 and HA must interact to help with tissue remodeling, cell sticking together and cell migration. 37 In fat tissue, this interaction ensures the structure and function of the tissue are normal. However, when weight increases and an individual approach obesity, the CD44-HA pathway is switched from normal to pathological (Figure 2). 37 The body of an obese person tends to make and remove large amounts of HA, leading to the build-up of low molecular weight HA. 38 Experts have discovered that these small pieces turn into damage-associated molecular patterns (DAMPs) which join with CD44 and start inflammatory signaling pathways such as factor-kappa B (NF-κB) and mitogen-activated protein kinases (MAPKs) 38 (Figures 3 and 4). The outcome is an amplified inflammatory response that attracts macrophages and other immune cells to the adipose tissue, thereby initiating chronic inflammation and metabolic dysfunction.

CD44-hyaluronic acid signaling in obesity-induced adipose tissue dysfunction and metabolic disturbances: This scientific illustration visualizes the mechanistic pathway through which the interaction between CD44 (Cluster of Differentiation 44) and hyaluronic acid contributes to obesity-induced adipose tissue dysfunction and systemic metabolic disturbances. With weight gain and obesity, adipocytes (fat cells) undergo hypertrophy (increase in size), triggering overexpression of CD44. When CD44 binds to hyaluronic acid, it activates pro-inflammatory signaling pathways. This leads to the infiltration of macrophages, hypoxia, apoptosis (programed cell death), and extracellular matrix (ECM) remodeling, including fibrosis, all of which result in adipose tissue dysfunction.The chronic inflammation resulting from this local tissue damage causes the release of inflammatory cytokines into circulation. These cytokines negatively affect peripheral tissues such as the liver and skeletal muscle, contributing to metabolic complications. The liver becomes prone to Non-Alcoholic Fatty Liver Disease (NAFLD), and systemic inflammation promotes insulin resistance and dyslipidaemia (abnormal lipid levels in the blood), thereby linking adipose tissue dysfunction to broader metabolic diseases.
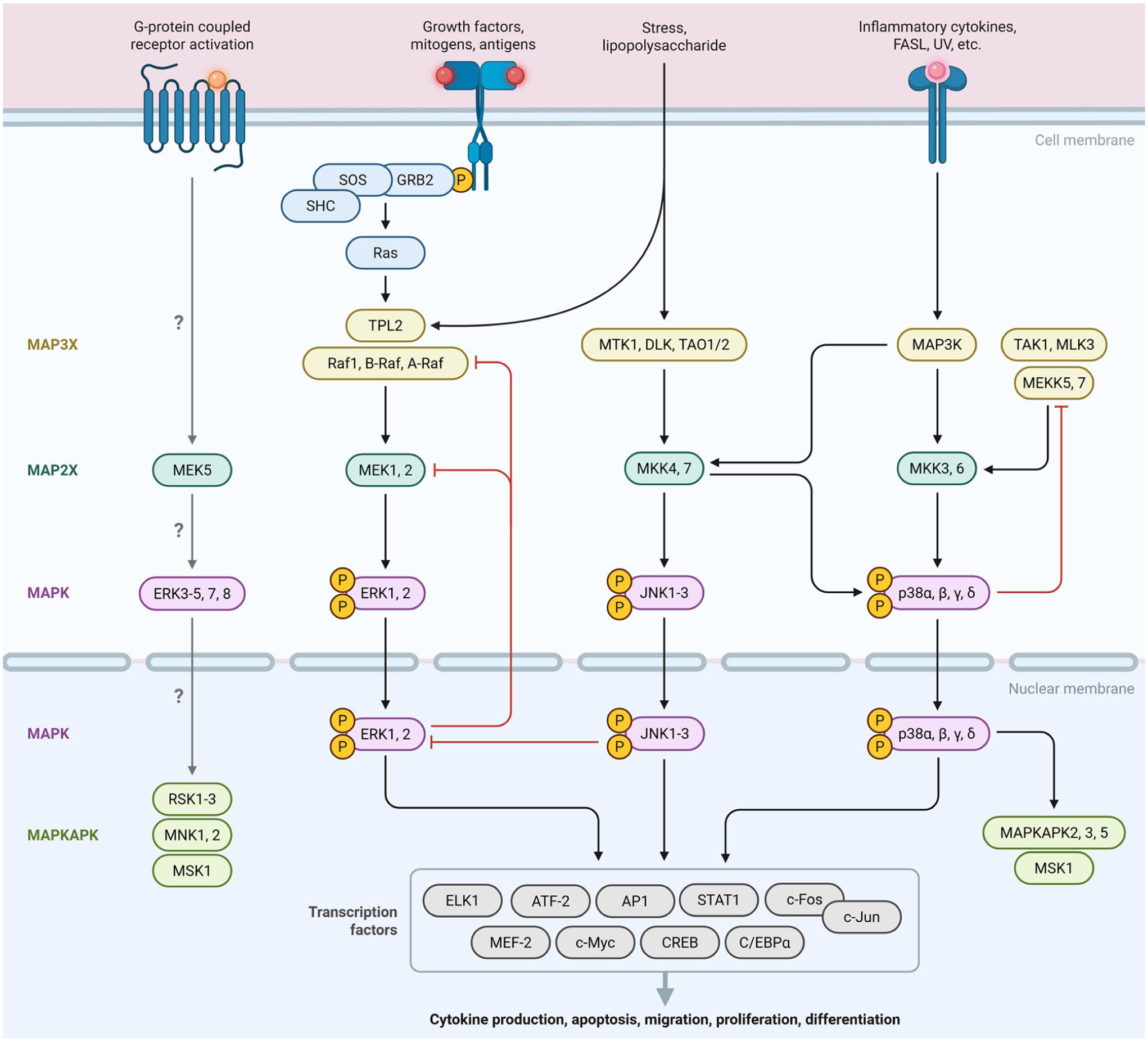
MAPK pathway: This figure illustrates the Mitogen-Activated Protein Kinase (MAPK) pathway. It shows the signaling cascade activated by various extracellular stimuli (such as growth factors, cytokines, and stress signals) that leads to the sequential phosphorylation and activation of MAPK kinases (MAPKKs) and MAPKs. The end result is the regulation of gene expression, cellular growth, differentiation, and inflammatory responses MAPK: Mitogen-Activated Protein Kinase – A family of protein kinases involved in cellular responses such as growth, proliferation, and differentiation. MAPKKK (or MEKK): MAPK Kinase Kinase – The upstream kinases that activate MAPKKs. MAPKK (or MEK): MAPK Kinase – These kinases phosphorylate and activate MAPKs. ERK: Extracellular Signal-Regulated Kinase – A type of MAPK often involved in cell growth and survival. JNK: c-Jun N-terminal Kinase – A MAPK involved in stress responses and apoptosis. p38 MAPK: A stress-activated MAPK that responds to inflammatory cytokines and environmental stresses (Created in https://BioRender.com).

NF-κB pathway: This figure depicts the NF-κB signaling pathway, a central regulator of immune and inflammatory responses. Under normal conditions, NF-κB is sequestered in the cytoplasm by inhibitor proteins (IκBs). Upon activation by stimuli such as cytokines or stress, IκBs are degraded, freeing NF-κB to translocate to the nucleus, where it promotes transcription of genes involved in inflammation, immunity, and cell survival NF-κB: Nuclear Factor kappa-light-chain-enhancer of activated B cells – A transcription factor that regulates genes involved in inflammation and immune responses. IκB: Inhibitor of κB – Proteins that inhibit NF-κB by keeping it in the cytoplasm. IKK: IκB Kinase – A complex that phosphorylates IκB, leading to its degradation and NF-κB activation. TNF-α: Tumor Necrosis Factor-alpha – A cytokine that activates the NF-κB pathway. IL-6: Interleukin-6 – A pro-inflammatory cytokine regulated by NF-κB signaling (Created in https://BioRender.com).
CD44-Hyaluronic Acid Signaling in Obesity
As the CD44-HA interaction becomes dysregulated in the context of obesity, it contributes significantly to structural and functional impairment of adipose tissue. 39 As the bodyweight increases, the expanded fat tissue promotes reorganization of the extracellular matrix by raising CD44 expression and HA build-up.40,41 Too much of HA in the ECM makes tissues stiffer, puts mechanical stress on them and lowers their elasticity as well as their ability to be stretched when storing fat. This environment promotes adipocyte hypertrophy, in which fat cells become enlarged and more prone to hypoxia (oxygen deficiency), apoptosis (programed cell death), or necrosis (uncontrolled cell death). 41 Dying adipocytes leave cell fragments and active molecules that make the body’s immune cells more responsive and draw new macrophages into the area. It is notable that when faced with inflammation, macrophages can change into M1 phenotype, which secretes cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and monocyte chemoattractant protein-1 (MCP-1) which both increase inflammatory reactions and and interfere with insulin signaling. 41 Another outcome from this imbalance is that blood vessels do not grow as needed, leading to reduced oxygen and supplies reaching adipose tissue cells which increases cell stress and tissue failure.
The pathological consequences of abnormal CD44-HA signaling extend well beyond local adipose tissue, leading to widespread systemic metabolic disturbances. Chronic inflammation in adipose tissue contributes to the development of insulin resistance by disrupting the insulin receptor substrate (IRS) signaling cascade and inhibiting key molecules such as phosphoinositide 3-kinase (PI3K) and protein kinase B (Akt), which are essential for glucose uptake in cells. 42 Furthermore, dysfunctional adipocytes release elevated levels of free fatty acids (FFAs) into the bloodstream, leading to lipid abnormalities such as elevated triglycerides and reduced high-density lipoprotein (HDL) cholesterol, a condition known as dyslipidemia.2,43 The persistent presence of FFAs and inflammatory cytokines also affects other organs. In the liver, this can lead to hepatic steatosis and inflammation, which are characteristic features of non-alcoholic fatty liver disease (NAFLD). 44 Similarly, chronic insulin resistance contributes to impaired glucose homeostasis, paving the way for the development of type 2 diabetes mellitus (T2DM). 42 The cumulative effect is a vicious cycle in which dysfunctional adipose tissue driven by abnormal CD44-HA signaling promotes systemic metabolic dysfunction, further reinforcing adipose inflammation and tissue injury.
Given the central role of CD44-HA signaling in adipose tissue pathology and metabolic disease, this axis has emerged as a promising therapeutic target. Experimental studies using CD44-deficient animal models or pharmacological inhibitors of CD44 have demonstrated improvements in insulin sensitivity, reductions in adipose tissue inflammation, and enhanced metabolic outcomes. 45 Additionally, approaches aimed at modulating HA metabolism – such as inhibition of hyaluronidase enzymes that break down HA or therapeutic agents that influence HA synthesis have shown potential in reducing fibrosis and restoring ECM balance. Targeting the HA-CD44 pathway could, therefore, provide a multi-faceted approach to treating obesity and its associated complications by simultaneously reducing inflammation, restoring insulin signaling, and improving lipid metabolism. 46 As research continues to unravel the complex biology of this signaling network, further understanding of its molecular underpinnings may lead to innovative therapies for combating the global burden of obesity and metabolic disorders.47,48 The CD44-HA signal pathway affects all major processes related to adipogenesis and the development of inflammation as well as insulin resistance and changes to the extracellular matrix. 49 Positive evidence from studies indicates that lack of CD44 protein saves mice from developing obesity due to poor adipogenesis activation. 1 Adipocyte differentiation depends on CD44-mediated CD44 signaling to regulate the important transcription factors peroxisome proliferator-activated receptor gamma (PPAR-γ) as well as C/EBPα. 50 The disturbance between HA synthetic and degradative processes leads to excessive lipid deposition within tissues and enlarged adipocytes which intensify metabolic dysfunctions.51,52
CD44 shows high expression levels on macrophages together with T cells leading to their enhanced ability to dock and move into adipose tissue. 33 The persistent inflammation resulting from CD44-HA signaling patterns leads to insulin resistance which represents the core characteristic of metabolic disorders linked to obesity. 53 The connection between CD44-HA binding leads to insulin resistance by first lowering insulin receptor signaling performance through stress kinase activation and secondly intensifying gluconeogenic processes and lipolysis in both fat tissue and liver cells which subsequently enhances blood sugar regulation and insulin responsiveness. 54
The CD44-HA signaling axis demonstrates potential value for obesity care through treatments involving CD44-blocking agents and hyaluronidase therapy along with HA-modification solutions and therapeutic eating habits. 1 Upcoming research must reveal how CD44-HA interactions proceed at a molecular level in obesity patients while exploring particular treatment options to control this pathway effectively. The integration of CD44-HA regulation systems with current obesity treatment options will create fresh methods to address obesity alongside its related health issues. During adipose tissue remodeling, CD44 directs processes of adipogenesis and adipocyte hypertrophy that results in metabolic complications and enhanced fat deposition.33,55 CD44 expression levels rise during obesity and this increase shows potential relationships with problems in insulin signaling pathways and rises in tissue inflammation levels and oxidative damage. 56 CD44 expression controls the proportion between M1 pro-inflammatory and M2 anti-inflammatory macrophages found in adipose tissue therefore affecting metabolic stability. 57 The dual benefits of targeting CD44-HA axis include both inflammation reduction along with controlled adipogenesis and enhanced metabolic health through lowered oxidative stress and inhibited inflammatory responses. 1 Scientists have investigated 2 main methods to affect this axis by studying small-molecule inhibitors and nanobioconjugates.58,59 Low-molecular-weight compounds which specifically target biological elements function effectively as small molecules to modify their biological targets. The synthesis of these compounds remains simple while they also demonstrate good bioavailability when taken by mouth and are reasonably priced with very short onset times. These therapeutic compounds display several weaknesses including poor selective binding and short-lived duration together with drug-naive resistance.60,61
Engineered nanoparticles which are biomolecule-conjugated nanobioconjugates have proven to be a progressive approach to manage obesity.59,62 The designed nanoparticles have the ability to reach adipose tissue, macrophages, and inflammatory sites to deliver drugs with enhanced therapeutic precision. Nanobioconjugates enable precise drug delivery along with sustained drug release and better drug accessibility and multiple functionalities. Organizations using these products encounter manufacturing difficulties coupled with immunological effects and regulatory obstacles.63 -65
The evaluation in this review analyzes the therapeutic benefits for obesity management through CD44-HA axis target strategies by analyzing small molecule and nanobioconjugate treatments. The study aims to explore obesity pathophysiology mechanisms involving the CD44-HA axis while reviewing existing CD44-HA targeting therapeutics and evaluating small molecule performance against nanobioconjugates and discussing research barriers for obesity CD44-HA treatment and proposing translational methods for clinical trial readiness of CD44-targeted therapies. The objectives discussed in this review will enhance obesity therapeutic knowledge while establishing a framework for manufacturing advanced interventions aimed at targeting the CD44-HA axis more effectively.
CD44: Structure, Function, and Expression in Adipose Tissue
The multifunctional transmembrane glycoprotein CD44 maintains essential roles in cellular adhesion, migration, and immune regulation as well as signal transduction.66,67 Additionally, CD44 occurs in broad distribution across different tissues including fat tissue where it manages biochemical control as well as inflammatory responses and weight-related medical conditions. 68 This section investigates CD44 structure and function in adipose tissue along with their metabolic impacts in health and disease scenarios.
Structure of CD44
CD44 exists as a transmembrane protein with extensive glycosylation that functions within the hyaluronan receptor family because of its structure. The CD44 gene exists on chromosome 11p13 in humans where multiple alternative spliced versions emerge from this location. As a key part of its construct CD44 has 3 fundamental domains which form its structure69,70 (Figure 5). The extracellular domain functions as a hyaluronic acid (HA) and osteopontin and collagen and matrix metalloproteinases (MMPs) binding area. Post-translational modifications such as glycosylation and sulfation influence its binding affinity and function. The transmembrane domain secures the protein in the cell membrane while enabling signal transduction operations through its connection to cytoskeletal proteins and signaling complexes. The intracellular section of CD44 binds signaling molecules called ERM proteins together with ezrin, radixin, and moesin to control cell responses involving cell proliferation and differentiation as well as inflammation pathways.

Structure of the CD44 protein. CD44 primarily comprises 3 regions: the extracellular domain, the transmembrane domain, and the intracellular domain. In contrast to CD44s, the extracellular region of the CD44v protein has an additional variable domain. Adapted from Li et al. 74 Under a free open access license https://creativecommons.org/licenses/by/4.0/.
Function of CD44 in Adipose Tissue
The adipose tissue uses CD44 as an essential controller for various biological and harmful processes. CD44 performs different tasks which fit into 3 distinct groups1,33 (Figure 6).

Functions of CD44 in adipose tissue: This figure illustrates the multifaceted roles of CD44 in adipose tissue. CD44, a cell-surface glycoprotein, regulates processes such as adipogenesis, immune cell recruitment and inflammation, extracellular matrix (ECM) remodeling, insulin sensitivity, glucose metabolism, lipolysis, and lipid metabolism. Its expression levels vary in white and brown adipose tissues, influencing metabolic health and inflammation, and making CD44 a potential therapeutic target in metabolic diseases (Created in https://BioRender.com).
Regulation of Adipogenesis
The differentiation process of adipocytes depends on CD44 because it connects with extracellular matrix (ECM) elements and growth factor signals. The preadipocyte maturation process requires both PI3K/Akt and MAPK pathways which result from Hyaluronan-CD44 signaling that drives preadipocyte proliferation together with differentiation.41,71
Inflammation and Immune Cell Recruitment
CD44 plays a role in adipose tissue inflammatory responses which intensifies during metabolic dysfunctions that arise from obesity. The protein recruits immune cells especially macrophages with T cells to adipose tissue. Mice deficient in CD44 show decreased inflammation together with better insulin sensitivity as evidence indicates CD44 plays a role in obesity-related persistent low-grade inflammatory condition. 39
Extracellular Matrix Remodeling
CD44 uses attachment to hyaluronan and collagens within the ECM which enables tissue remodeling in adipose storage depots. MMP activity control through CD44 affects the deterioration and restructuring processes of ECM tissue that promotes adipose tissue expansion while regulating fibrosis development. 45
Insulin Sensitivity and Glucose Metabolism
According to research CD44 facilitates insulin signal transmission and regulates the metabolic pathway of glucose within adipocyte cells. The expression of CD44 rises during obesity while also directly associating with insulin resistance. Its role in inflammatory signaling pathways, such as NF-κB activation, links it to metabolic dysfunction. 33
Lipolysis and Lipid Metabolism
The CD44 molecule controls lipid metabolism through its management of both lipolytic enzymes and proteins which associate with lipid droplets. The CD44 knockout research revealed modifications in energy metabolism processes together with changes in fat storage and release activities.72,73
Expression of CD44 in Adipose Tissue
The CD44 protein shows different levels of presence throughout distinct adipose tissue areas like white adipose tissue (WAT) and brown adipose tissue (BAT). The expression of CD44 increases in obese obesity-related WAT tissue that has become hypertrophic. 33 Fibroblast activation protein is primarily located within adipose stromal cells and immune cells with endothelial cells where it generates inflammation and ECM remodeling. 75 The expression levels of CD44 in Brown Adipose Tissue (BAT) remain low but this protein supports thermogenic actions during metabolic adaptation. Experts need more scientific inquiries to establish BAT functional effects linked to CD44 activity.76,77 The relationship between CD44 and metabolic diseases treatment should be explored because CD44 controls both inflammatory processes and extracellular matrix (ECM) remodeling and insulin resistance development. 77 Researchers should explores CD44 inhibitors along with hyaluronan-blocking agents and monoclonal antibodies as possible treatments for obesity-related clinical manifestations. The elevated manifestation of CD44 during obesity as well as metabolic conditions makes it both a suitable diagnostic indicator and a viable treatment candidate. Additional scientific investigation will help map out the complete functions of CD44 while creating specific treatments for metabolic health conditions.
Hyaluronic Acid: Biosynthesis, Degradation, and Its Role in Adipose Biology
The glycosaminoglycan (GAG) known as hyaluronic acid is a natural substance which essential for biological functions including cell signaling and tissue hydration along with maintaining structural integrity. The biosynthesis of hyaluronic acid starts with the phosphorylation of glucose by hexokinase, yielding the primary precursor, glucose-6-phosphate. The HA synthesis process may be bifurcated into 2 separate routes that produce the 2 precursors of HA: glucuronic acid and N-acetylglucosamine 78 (Figure 7). Hyaluronic acid (HA) participates in extracellular matrix (ECM) arrangement inside adipose tissue as well as supporting adipocyte differentiation while regulating metabolic functions and facilitating inflammatory processes. 33 The 3 hyaluronan synthases (HAS) enzymes located at the plasma membrane produce hyaluronic acid through disaccharide unit polymerization of D-glucuronic acid and N-acetyl-D-glucosamine. 79 The mammalian cells express 3 HAS enzymes which produce distinct HA lengths: HAS1 synthesizes high-weight HA chains while HAS2 generates longer HA ranges but HAS3 synthesizes short-chain HA and links to inflammatory events. Hyaluronan synthases (HAS) biosynthetic activity depends on different factors including growth factors and cytokines and cellular energy levels.80,81 The synthesis of hyaluronan requires the participation of UDP-sugar precursors as essential components. The degradation of HA occurs through enzymatic pathways performed by hyaluronidases and through non-enzymatic pathways that cause fragmenting of HA molecules resulting in changes to its bioactivity. High levels of HA production in the body result in tissue scarring and damaged cellular function of fat tissue. 81

Hyaluronic Acid Biosynthesis. Modified from Sze et al. 78 (Created in https://BioRender.com). Glucose is first metabolized by hexokinase into glucose-6-phosphate, which then enters one of 2 different routes to produce UDP-glucuronic acid or UDP-N-acetylglucosamine. The precursors are then linked by the activity of hyaluronic acid synthase, or HAS, to produce hyaluronic acid.
The adipose ECM contains HA as its main component which drives adipocyte differentiation and supports tissue expansion. The composition of insulin response depends on HA molecular weight configuration because high-weight molecules activate signaling but metabolic dysfunction emerges from HA fragments.82,83 Body tissue inflammation along with metabolic system dysregulation develops from abnormal HA metabolic processes in obesity cases. 84 The disrupted metabolism of HA leads to fibrosis in adipose tissues because it limits tissue plasticity and disrupts metabolic equilibrium. The metabolic increase of HA concentration in obesity creates rigid ECM which limits adipocyte growth and intensifies insulin resistance. 85
The polyelectrolyte HA functions as a vital modulator which affects adipose tissue development as well as metabolic stability and inflammatory reactions. Metabolic disorders that include obesity along with insulin resistance show a direct link to improper HA synthesis and degradation control found in adipose tissues. Further study on how to control HA metabolism will have promise as a new therapeutic method for managing metabolic diseases.
CD44-HA Interaction and Its Impact on Inflammation, Adipogenesis, and Insulin Resistance
The CD44-Hyaluronic Acid (HA) binding acts as an essential regulator for cellular operations that manage inflammation together with adipogenesis and insulin resistance pathways. 33 The transmembrane glycoprotein CD44 serves as an HA receptor which binds to the large glycosaminoglycan HA. The molecular interaction between CD44 and Hyaluronic Acid controls vital cellular events for moving cells as well as adhesive mechanisms and growing behaviors along with cell multi-facets development. CD44-HA axis is critically involved in the modulation of key processes such as inflammatory responses, adipocyte formation, and the development of insulin resistance.33,34 This interaction matters when creating therapeutic options toward the treatment of obesity, type 2 diabetes, and cardiovascular conditions.
CD44 and Hyaluronic Acid (HA) Interaction
CD44 appears in different tissues at high levels but shows its densest concentration in immune cells together with endothelial cells and adipocytes. 86 The CD44 receptor links to HA which functions as a major component of the extracellular matrix so it intervenes with tissue hydration functions and cellular elasticity and cell signaling operations. Multiple HA forms exist but CD44 binds to high-molecular-weight HA as its main form. 87 Various intracellular signaling cascades begin after the binding occurs and affect how cells behave. Absolute binding between HA molecules and CD44 receptors activates multiple signaling cascade pathways in cells through the sequence of MAPK (mitogen-activated protein kinase), PI3K (phosphoinositide 3-kinase), and Akt (protein kinase B). Cells use these signal pathways to perform essential cellular functions of survival as well as migration differentiating into different cell types and promoting inflammation situations. 87
Impact of CD44-HA Interaction on Inflammation
Acute inflammatory mechanisms strongly associate with the CD44-HA molecular interaction during chronic inflammatory disease states that include obesity with metabolic syndrome. Numerous obesity patients develop excessive amounts of HA within their adipose tissues while these deposits maintain a pro-inflammatory condition which describes this disease. 88 When CD44 combines with HA it activates inflammatory pathways inside cells that resulted in the release of pro-inflammatory TNF-α (tumor necrosis factor-alpha) and IL-6 (interleukin-6) as well as MCP-1 (monocyte chemoattractant protein-1). 89 Cytokines activate a recruitment process that brings macrophages into adipose tissues to worsen the inflammatory state. 90 Obese patients show increased levels of HA and CD44 along with pro-inflammatory macrophage infiltration in adipose tissue which develops an inflammatory spiral that harms insulin sensitivity. Inflammation at a low constant level caused by this tissue-cell connection functions as a major contributor to insulin resistance development according to research.
Role of CD44-HA in Adipogenesis
CD44-HA interaction influences the process which transforms mesenchymal stem cells (MSCs) into fat cells referred to as adipogenesis. 33 CD44 serves as a regulatory factor which affects the MSC transition into adipocyte development. When combined with HA molecules CD44 activates transcription factors PPAR-γ and C/EBP-α which serve as essential activators of adipocyte differentiation. Insufficient cell service occurrence in adipose tissue contributes to an increase of fat cell numbers and formation of new adipocytes. A high HA concentration facilitates pre-adipocyte maturation into adipocytes thus extending the size of adipose tissue in obesity. 91 The knowledge derived from this method helps explain obesity pathophysiology because excessive adipogenesis creates worse metabolic issues.
CD44-HA Interaction and Insulin Resistance
Type 2 diabetes, and obesity shows insulin resistance that directly correlates with the CD44-HA axis activity. 38 The development of insulin resistance heavily relies on chronic tissue inflammation that starts when CD44 binds HA inside adipose tissue. 33 Released inflammatory cytokines during this process disrupt insulin signaling pathways which prevents insulin from performing its function in promoting tissue glucose uptake in muscle cells and liver cells. 92
Adipose tissue HA levels bind to CD44 which causes a breakdown of insulin signaling pathways. Laboratory studies have shown blood HA levels strongly correlate with insulin resistance thus indicating CD44-HA interaction blocking as a promising therapy to handle insulin resistance and its linked metabolic conditions. 33 The binding of HA to CD44 receptor alters the activation of the NF-κB signaling pathway which maintains inflammatory control functions. 93 Insulin signaling becomes impaired when NF-κB becomes active since this activates pro-inflammatory cytokines that escalate insulin resistance throughout the body.
Therapeutic Implications
The CD44-HA molecular interaction serves as a necessary component in inflammation processes and adipose tissue development plus insulin resistance which shows potential for treating obesity and its related conditions including type 2 diabetes and cardiovascular diseases. 94 Several approaches to disrupt the CD44-HA interaction involve creating CD44 blockers as well as regulating adipose tissue HA content and active intervention in inflammatory pathophysiological processes. CD44 agonist molecules serve to decrease inflammation and stop fat cell development thus leading to enhanced insulin function. 95 Regular modification methods of HA enable treatment of pro-inflammatory effects and HA control through enzymatic degradation or blocked synthetic processes. 95 The clinical management of insulin resistance alongside metabolic disorders should involve the treatment of inflammatory cytokines and NF-κB pathway activities. Research into adipose tissue CD44-HA mechanics at cellular and molecular levels could identify new therapeutic goals for insulin resistance together with obesity-related problems. Research has shown that blocking the CD44-HA axis presents potential as a strategy for handling type 2 diabetes together with obesity. Future studies about how this interaction happens at a molecular level must proceed to enable developing specific therapeutic approaches.
Experimental Evidence Supporting CD44-HA in Obesity Pathogenesis
The study of CD44 in weight-related inflammatory diseases alongside HA interactions has become a major scientific focus during the last few years. Multiple research investigations have explored the connection between CD44 expression levels and obesity through the analysis of clinical samples and experimental study designs for clarifying its therapeutic potential. Table 1 gathers research findings from various studies which present type, success rate, clinical and experimental data alongside limitations and future-planned approaches. The different success levels across multiple study types including clinical trials and experimental models are shown in this table to emphasize the necessity of additional research that addresses discovered study limitations. CD44 exhibits potential as both a diagnostic indicator and treatment element but more extensive testing with human clinical samples must occur to confirm its lasting clinical benefits and treatment effect.
Experimental Evidence Supporting the Role of CD44-HA in Obesity Pathogenesis.

Differential Expression of CD44 in Adipose Tissue Types and Depots
White Adipose Tissue (WAT)
White adipose tissue (WAT) is the primary energy storage site in the body and plays a significant endocrine role by secreting various adipokines. It is also a major site for chronic inflammation in obesity. 101 CD44, is highly expressed in WAT, particularly within the stromal vascular fraction (SVF), which includes preadipocytes, macrophages, endothelial cells, and fibroblasts. In obesity, WAT experiences hypertrophy and hypoxia, which lead to macrophage recruitment and activation. 102 These macrophages especially the proinflammatory M1 subtype express high levels of CD44, which facilitates their adhesion to the extracellular matrix and interaction with hyaluronic acid. 2 Elevated CD44 expression has been linked to increased secretion of proinflammatory cytokines such as TNF-α, IL-6, and MCP-1, all of which contribute to insulin resistance and systemic metabolic dysfunction. CD44 also appears to mediate signaling pathways that exacerbate adipose tissue fibrosis and impair adipogenesis, further compounding metabolic complications. 103 In sum, CD44 in WAT is a key player in the development of obesity-induced inflammation and insulin resistance, and its expression levels may serve as both a biomarker and therapeutic target in the management of metabolic diseases such as type 2 diabetes and non-alcoholic fatty liver disease (NAFLD).
Brown Adipose Tissue (BAT)
Brown adipose tissue (BAT) is specialized for non-shivering thermogenesis and is metabolically active due to its high mitochondrial content and expression of uncoupling protein 1 (UCP1). 104 Unlike WAT, BAT contributes to energy expenditure and is associated with improved glucose and lipid metabolism. 104 Studies have shown that CD44 expression in BAT is significantly lower than in WAT.105,106 This is partly due to the inherently low inflammatory state of BAT, which contains fewer immune cells such as macrophages. Additionally, the anti-inflammatory environment of BAT likely suppresses the expression of CD44 and its ligands. 107 The minimal CD44 activity may contribute to the preservation of BAT’s metabolic functionality by limiting immune cell infiltration and extracellular matrix remodeling, processes that are common in WAT during obesity. 107 Furthermore, the signaling pathways active in BAT, such as those involving PGC-1α and PRDM16, favor mitochondrial biogenesis and oxidative metabolism rather than inflammatory responses. 108 These pathways may inherently downregulate CD44 transcription or prevent its upregulation. Given BAT’s beneficial role in energy homeostasis and metabolic regulation, the absence or low expression of CD44 may be protective. 108 This highlights the potential for targeting CD44-related pathways to preserve BAT function or mimic its effects in metabolic disease therapies.
Beige Adipose Tissue
Beige adipose tissue, also known as “brite” (brown-in-white) fat, consists of thermogenically active adipocytes that emerge within WAT depots under specific stimuli such as cold exposure, β-adrenergic stimulation, or exercise. 109 These adipocytes express thermogenic markers such as UCP1 and resemble BAT in function, although they originate from distinct progenitor cells. Emerging studies suggest that CD44 expression in beige fat is significantly reduced compared to typical WAT. 110 The process of WAT browning is often accompanied by a reduction in inflammation and a shift toward an anti-inflammatory immune profile, which includes increased M2 macrophage polarization. 110 Since CD44 is closely associated with proinflammatory signaling and immune cell adhesion, its downregulation appears to be a hallmark of beige adipocyte development. 50 Furthermore, experimental models using CD44-deficient mice have shown enhanced WAT browning and improved insulin sensitivity, reinforcing the idea that CD44 negatively regulates thermogenic programing. 111 The suppression of CD44 may also facilitate extracellular matrix (ECM) remodeling, allowing for cellular flexibility required for beige adipocyte recruitment and differentiation. Overall, low CD44 expression in beige adipose tissue suggests a functional antagonism between CD44 signaling and thermogenic reprograming, indicating that CD44 inhibition may be a viable strategy for combating obesity and related metabolic disorders. 112
Visceral Adipose Tissue (VAT)
Visceral adipose tissue (VAT) is located within the abdominal cavity and surrounds internal organs such as the liver, pancreas, and intestines. 113 Compared to subcutaneous fat, VAT is more metabolically active and exhibits a higher propensity for inflammation, contributing significantly to cardiometabolic diseases. 113 CD44 expression in VAT is markedly higher than in subcutaneous depots, particularly in obese individuals. 2 This elevated expression is associated with an increase in infiltrating immune cells, especially proinflammatory M1 macrophages and T cells. 114 These cells rely on CD44 for migration, retention, and interaction with hyaluronic acid in the extracellular matrix, processes that drive chronic inflammation. 114 VAT inflammation is a major contributor to systemic insulin resistance and has been implicated in the pathogenesis of metabolic syndrome, atherosclerosis, and non-alcoholic steatohepatitis (NASH). 115 Moreover, CD44 expression in VAT has been correlated with increased fibrosis and impaired adipogenesis, further exacerbating metabolic dysfunction. In murine models, CD44 knockout or blockade leads to reduced VAT inflammation, improved insulin signaling, and decreased hepatic fat accumulation. 115 Therefore, CD44 appears to be a critical mediator of the pathogenic characteristics of VAT. Targeting CD44 signaling in VAT may offer a novel approach to reduce visceral adiposity-associated inflammation and improve systemic metabolic outcomes. 116
Subcutaneous Adipose Tissue (SAT)
Subcutaneous adipose tissue (SAT) lies beneath the skin and is generally regarded as a more metabolically protective fat depot compared to VAT. SAT serves as a major energy reserve and plays a role in cushioning and insulation. 117 CD44 expression in SAT is present but lower than in VAT, reflecting the lower inflammatory burden in this depot. In lean individuals, SAT contains relatively fewer immune cells, and the resident macrophages are predominantly of the anti-inflammatory M2 phenotype. 117 The reduced CD44 expression in SAT correlates with diminished leukocyte adhesion, limited ECM remodeling, and decreased proinflammatory signaling. Although SAT does contribute to systemic adipokine secretion, such as leptin and adiponectin, its inflammatory cytokine profile is less harmful than that of VAT. Nonetheless, in obesity, SAT can also expand and become inflamed, leading to an upregulation of CD44, though typically still at levels lower than in VAT. 118 The role of CD44 in SAT may involve modulation of local immune cell activity and tissue remodeling rather than promoting widespread metabolic dysfunction. Its expression dynamics in SAT may also influence the tissue’s capacity to undergo browning. 119 Thus, while SAT is relatively benign, CD44 still contributes to depot-specific responses to metabolic stress, inflammation, and adipose tissue plasticity.
Small Molecules Targeting CD44-HA Signaling in Obesity
The interaction between CD44, a cell surface receptor, and hyaluronic acid (HA), a key component of the extracellular matrix, plays a crucial role in obesity-related inflammation, adipogenesis, and metabolic dysregulation. A therapy built on small molecule inhibitors that target the CD44-HA axis shows great promise in obesity treatment because it modifies both cell binding processes and chronic inflammation while altering how the extracellular matrix changes. 120
As shown in Table 2 the small molecule inhibitors which block CD44-HA interaction receive detailed analysis according to their functional mechanisms together with their preclinical testing results and evaluation of potential therapeutic use for obesity treatment. These prevention methods divide themselves into 3 groups: CD44 antagonists and hyaluronidase modulators and inhibitors that regulate HA biosynthesis pathways. The table illustrates the benefits of these inhibitors because they target specific areas effectively while allowing lowered toxicity levels but details their obstacles from reduced bioavailability and resistant development. This table provides important research information about small molecule inhibitors as a tool to fight obesity via CD44-HA regulation.
Small Molecule Inhibitors of CD44-HA Interaction: Types, Mechanisms, Studies, and Therapeutic Potential in obesity.

Small Molecules: Chemistry, Interaction, and Biological Role
Small molecules are a class of low molecular weight organic compounds, typically characterized by a molecular mass of less than 900 Da. 58 Unlike macromolecules such as proteins, nucleic acids, and polysaccharides, small molecules possess the unique capability to diffuse across cellular membranes. 58 This property grants them a functional advantage in modulating biological activities from within the cell. Their small size and diverse structural characteristics make them ideal candidates for interaction with intracellular targets, including enzymes, receptors, ion channels, and nucleic acids. 58 These interactions are critical in influencing a wide array of cellular pathways and physiological functions, often with high specificity and potency. In the realm of drug discovery and development, small molecules have long held a dominant position due to their pharmacokinetic properties, including favorable absorption, distribution, metabolism, and excretion (ADME) profiles. 58 Their ease of synthesis, modification, and structural optimization enhances their therapeutic potential, allowing researchers to fine-tune their interactions with biological targets. Small molecules are foundational not only in therapeutics but also in diagnostic and research applications, where they are used as probes to understand biological mechanisms and disease pathology. Their adaptability and multifunctional roles underscore their significance across various scientific disciplines. 149
Moreover, small molecules are pivotal in bridging chemistry and biology, as they enable a molecular-level understanding of complex biological systems. This has facilitated the exploration of signaling pathways, gene regulation, and metabolic processes with unprecedented detail. Their contribution extends to personalized medicine, where they are used to develop targeted therapies based on individual genetic profiles. As the field of chemical biology continues to evolve, the importance of small molecules in therapeutic innovation, biomarker discovery, and disease modeling is set to grow even further, reinforcing their indispensable role in modern science and medicine. 149
What is the chemistry behind small molecules?
The chemistry of small molecules is defined by several structural and physicochemical characteristics that influence their biological activity and therapeutic potential. These molecules generally fall below the 900 Da molecular weight threshold, which facilitates their ability to traverse lipid membranes and reach intracellular sites of action. A defining feature of small molecules is the presence of diverse functional groups, such as hydroxyl (-OH), carboxyl (-COOH), amine (-NH₂), ketone (=O), and halogens. 150 These functional moieties contribute significantly to the chemical reactivity, solubility, and binding interactions of the molecules with their biological targets. The hydrophilic and hydrophobic regions within small molecules also dictate their solubility in aqueous and lipid environments, influencing their pharmacokinetics and pharmacodynamics. 150 A crucial tool in the assessment of small molecule drug-likeness is Lipinski’s Rule of Five, which provides a set of guidelines that predict the oral bioavailability of a compound. 151 According to this rule, an ideal drug-like small molecule should have a molecular weight less than 500 Da, a logP (partition coefficient) value under 5, no more than 5 hydrogen bond donors, and no more than 10 hydrogen bond acceptors. 151 While not absolute, these guidelines help researchers in early-stage drug development to select candidates with favorable pharmacological profiles. Other considerations such as polar surface area, number of rotatable bonds, and metabolic stability also contribute to the molecule’s potential as a drug candidate. 151
Small molecules are classified based on their origin and synthesis into 3 major categories: natural, synthetic, and semi-synthetic. 152 Natural small molecules include plant-derived alkaloids like morphine, microbial metabolites such as streptomycin, and peptide fragments found in hormones. 153 Synthetic small molecules are designed and synthesized in laboratories, often using combinatorial chemistry techniques, to yield pharmaceuticals like aspirin, ibuprofen, and imatinib. 154 Semi-synthetic molecules are derived from natural compounds that are chemically modified to enhance efficacy, stability, or bioavailability – examples include semi-synthetic penicillins. 155 Each class offers unique advantages and serves different purposes in medicine, biotechnology, and research, reflecting the versatility of small molecules in chemical and biological sciences.
Interaction of Small Molecules
Small molecules exert their biological effects by interacting with macromolecules through a variety of non-covalent interactions. These include hydrogen bonding, hydrophobic interactions, van der Waals forces, ionic bonding, and specialized interactions such as pi-stacking and cation-pi bonding. 156 Each type of interaction contributes to the binding affinity and specificity of the small molecule to its target. Hydrogen bonds, for instance, play a critical role in determining the orientation and strength of binding by forming specific interactions between donor and acceptor atoms. 157 Hydrophobic interactions arise when non-polar regions of the molecule associate with similarly non-polar regions on a target protein, often contributing to the stabilization of the ligand-protein complex in the hydrophobic core. Van der Waals forces, though individually weak, collectively contribute to the overall molecular complementarity between the small molecule and its target. Ionic bonds, formed between oppositely charged groups, enhance the electrostatic attraction, and strengthen the overall interaction. 157 Aromatic molecules may also participate in pi-stacking interactions with aromatic amino acid residues like phenylalanine, tyrosine, and tryptophan, or engage in cation-pi interactions with positively charged groups. 157 These intricate interactions are crucial for the pharmacological activity of small molecules, determining their efficacy, selectivity, and potential side effects.
The primary targets of small molecules are diverse and include enzymes, receptors, ion channels, DNA, RNA, and protein-protein interactions. Enzyme inhibitors can block catalytic activity, as observed with statins that inhibit HMG-CoA reductase to lower cholesterol levels.158,159 Receptor modulators, such as beta-blockers, bind to adrenergic receptors to manage hypertension. Ion channel modulators, like calcium channel blockers, affect ion flux across membranes, altering physiological responses. 160 Small molecules can also intercalate with DNA or form covalent adducts, thereby influencing gene expression and replication processes, as seen with anticancer drugs like doxorubicin. 160 Furthermore, recent advances focus on targeting protein-protein interactions, a frontier in drug discovery aimed at disrupting or stabilizing essential complexes in disease pathways.
Biological Role of Small Molecules
Small molecules play critical roles in regulating a broad range of biological functions. One of their primary roles is in signal transduction, where they function as ligands, second messengers, or modulators of signaling pathways. For instance, cyclic AMP (cAMP) acts as a second messenger in G protein-coupled receptor (GPCR) signaling, activating protein kinase A and regulating downstream effects such as metabolism, transcription, and cell growth. 161 Nitric oxide (NO) is another small molecule that influences vascular tone and neurotransmission by stimulating guanylate cyclase and increasing cyclic GMP levels. Calcium ions (Ca²⁺), although inorganic, mimic the role of small organic molecules in signal cascades by regulating processes such as muscle contraction, secretion, and gene expression. 162 In metabolic regulation, small molecules serve as key intermediates and regulators of essential biochemical pathways. Molecules such as glucose and pyruvate are central to glycolysis and energy production, while amino acids like tryptophan play both structural and regulatory roles, including serving as precursors for neurotransmitters like serotonin. 163 Lipids and fatty acids function not only as structural components of cell membranes but also as signaling molecules involved in inflammation, apoptosis, and metabolic homeostasis. Their dynamic roles in these pathways illustrate the central position small molecules occupy in maintaining cellular and systemic balance. Furthermore, many small molecules act as hormones, governing physiological processes such as growth, metabolism, and immune response. Steroid hormones like testosterone and estradiol influence reproductive development and metabolic functions. Thyroid hormones such as thyroxine regulate metabolic rate, while eicosanoids, including prostaglandins, mediate inflammation, pain, and immune responses. 164 In therapeutics, small molecules form the backbone of most pharmaceutical drugs. They are utilized to treat a range of diseases, including cancer (eg, imatinib), infectious diseases (eg, penicillin, oseltamivir), neurological disorders (eg, L-DOPA, SSRIs), and cardiovascular conditions (eg, ACE inhibitors, beta-blockers), highlighting their indispensable role in modern medicine.165 -167
Challenges with Small Molecules
Small molecules, despite their significant therapeutic potential, present notable challenges such as off-target effects and toxicity. 168 Their inherent ability to interact with multiple biological targets can result in unintended side effects, necessitating careful optimization of selectivity and ADME (Absorption, Distribution, Metabolism, and Excretion) characteristics to enhance safety and efficacy. Another pressing concern is the development of resistance, particularly in antimicrobial and anticancer applications. Resistance may arise through various mechanisms, including target mutations, overexpression of efflux pumps, or enzymatic degradation, all of which diminish the long-term effectiveness of small molecule therapies. 168
At the same time, emerging fields are expanding the utility and scope of small molecule research. Chemical biology leverages these compounds to probe and manipulate biological systems, offering deeper insights into cellular processes. Innovations such as PROTACs (Proteolysis-targeting chimeras) represent a paradigm shift by promoting the degradation of target proteins rather than merely inhibiting them. 169 Fragment-Based Drug Discovery (FBDD) further enhances drug design by using small chemical fragments to identify key binding motifs. In conclusion, small molecules continue to play a foundational role in biomedical science. 170 With the convergence of chemistry, biology, and computational methods, new opportunities are emerging to refine small molecule design, uncover novel targets, and push the boundaries of therapeutic development (Figure 8).

Overview of small molecules: structure, function, and applications.
Nanobioconjugates Targeting CD44-HA Signaling
Nanobioconjugates are hybrid structures which result from covalent or non-covalent combinations between biomolecules with nanomaterials. 171 Nanobioconjugates unite nanomaterial physical properties with biomolecular characteristics for implementing drug delivery systems and biosensing devices and medical imaging techniques and nanomedical applications.41,171 There are several types of nanobioconjugates, including metallic nanoparticles (AuNP), carbon-based nanobioconjugates (Glene and Graphene Oxide), polymeric nanoparticles (Polymeric Nanoparticle Conjugates), lipid-based nanobioconjugates (Liposome Conjugates), solid lipid nanoparticles (SLN), quantum dots (QD), protein and peptide conjugates (Antibody-Nanoparticle Conjugates), peptide-Nanoparticle Conjugates, nucleic acid-based conjugates (DNA/RNA-Nanoparticle Conjugates), and aptamer-Nanoparticle Conjugates. 41 Nanobioconjugates serve multiple purposes including targeted therapy and molecular recognition as well as cancer therapy and immunodiagnostics and biosensing applications and extended uses in nanovaccines and gene therapy. 41
Three types of nanobioconjugates including lipid-based nanoparticles and polymer-based nanocarriers with HA-functionalized nanoparticles demonstrate promising capability in treating obesity by targeting CD44-HA interactions according to Table 3. The high stability and biocompatible lipid-based nanoparticles of liposomes and solid lipid nanoparticles encounter obstacles owing to their fast clearance from the body. The production of large-scale PEGylated polymers along with PLGA-based systems faces challenges because they require more development to optimize their drug release mechanisms and toxicity reduction rates. HA-functionalized nanoparticles use their selective bond with CD44 receptors to stop HA-CD44 interactions which prevent adipose tissue growth.
Overview of Nanobioconjugates in CD44-HA Inhibition and Obesity Treatment.

The high stability and biocompatibility of lipid-based nanoparticles, such as liposomes and solid lipid nanoparticles, encounter obstacles owing to their fast clearance from the body. Large-scale PEGylated polymers and PLGA-based systems also face challenges in optimizing drug release mechanisms and reducing toxicity. By contrast, HA-functionalized nanoparticles provide selective binding to CD44 receptors, but their clinical translation is limited by production cost and preclinical-to-clinical gaps.
Nanobioconjugates: Chemistry, Interaction, and Biological Role
Nanobioconjugates represent a ground-breaking method in nanobiotechnology, integrating the versatile properties of nanomaterials with the biological specificity of macromolecules such as proteins, nucleic acids, and antibodies. 189 These hybrid systems, developed by chemical conjugation, integrate nanoscale structures with biologically active compounds, therefore improving performance and specificity.59,190 The advancement of nanobioconjugates mitigates shortcomings in conventional medicinal and diagnostic methods, including inadequate solubility and fast disintegration in biological environments. 191 They facilitate site-specific delivery of therapies, augment signal amplification, and increase the stability and half-life of therapeutic substances. 191 The modular characteristics of nanobioconjugates provide several capabilities inside a singular construct, permitting real-time assessment of therapy effectiveness. The creation of nanobioconjugates requires a comprehensive grasp of nanomaterial chemistry and biomolecular interactions. Ongoing research indicates that nanobioconjugates are set to transform contemporary medicine and biotechnology by enabling more accurate and efficient treatments at the molecular scale.
Chemistry of Nanobioconjugates
Nanobioconjugates are a category of nanomaterials engineered for targeted biological purposes. They consist of inorganic or organic nanomaterials, including metal nanoparticles such as gold, silver, and iron oxide, which exhibit distinctive electrical, optical, and magnetic characteristics. These nanoparticles are optimal for imaging, medicinal applications, and biosensing. 192 The interplay between nano and biological components is essential for optimizing performance, minimizing toxicity, and improving targeted specificity.
Biological elements attached to nanoparticles provide specificity, recognition, and bioactivity. Proteins, enzymes, antibodies, peptides, nucleic acids, aptamers, and carbohydrates may selectively target certain cell receptors, facilitating uses in immunotherapy and vaccine development. 192 Conjugation techniques guarantee the retention of biomolecule bioactivity while maintaining the stability and functionality of the nanoparticle surface under physiological settings. Covalent bonding is the predominant way used, although non-covalent methods including electrostatic contacts, hydrophobic interactions, click chemistry, and biotin-streptavidin coupling. 192 Surface functionalization is crucial for enhancing solubility, colloidal stability, circulation duration, and targeting efficacy. PEGylation, silanization, polymer coatings, and zwitterionic surfaces are used to improve biocompatibility, release characteristics, and mucoadhesion. 193 The chemical methods and surface alterations used in nanobioconjugates are designed to guarantee their stability, biofunctionality, and efficacy in intricate biological settings. 193
Interactions of Nanobioconjugates
Nanobioconjugates are essential in biomedical applications because of their interactions at molecular, cellular, and systemic levels. These interactions regulate the manner in which the conjugates engage with target molecules, traverse biological environments, are internalized by cells, and perform their designated tasks. 193 The interactions are influenced by factors such as the characteristics of the nanomaterial, the conjugated biomolecule, surface modifications, dimensions, charge, and microenvironmental circumstances. 193 Molecular recognition is fundamental to the operation of nanobioconjugates, enabling the biological component to preferentially attach to a particular target molecule. Targeted delivery is a revolutionary use of nanobioconjugates, enabling the precise transport of therapeutic medicines to designated cell types, tissues, or pathological areas. 193 Intracellular interactions are vital for payload delivery, whereas biodistribution and clearance are critical for preventing premature elimination by the mononuclear phagocyte system. Particle size, surface charge, and hydrophobicity influence biodistribution; hence, long-term biocompatibility and clearance mechanisms must be evaluated to prevent chronic toxicity or buildup in non-target organs. 193
Biological Role of Nanobioconjugates
Nanobioconjugates serve as adaptable platforms in biomedicine, integrating the distinctive physicochemical properties of nanomaterials with the biological efficacy and specificity of biomolecules (Figure 9). They are pivotal in drug delivery, diagnostics, therapies, immunomodulation, and antimicrobial applications. Nanobioconjugates provide targeted, regulated, and stimuli-responsive drug release, tackling issues such as inadequate solubility, fast clearance, off-target toxicity, and restricted capacity to traverse biological barriers.59,192 They provide unparalleled sensitivity and specificity in diagnostics, establishing the foundation for novel biosensors capable of identifying disease biomarkers at very low concentrations.

Multifunctional biomedical applications of nanobioconjugates (Created in https://BioRender.com).
Nanobioconjugates actively engage in therapeutic approaches, including cancer treatment, gene therapy, and immunomodulation. 194 They may be designed to either stimulate or inhibit immune responses, contingent upon therapeutic objectives, and may be used to obstruct immunological checkpoints or provide immunosuppressive medicines in cases of autoimmune illnesses or transplant rejection. 194 Antimicrobial uses are under investigation, with silver nanoparticles being augmented by conjugation with antibiotics, antimicrobial peptides, or enzymes. This combination has a synergistic impact, enhancing bactericidal action and circumventing resistance mechanisms in multidrug-resistant bacteria. Their use include wound dressings, coatings for medical equipment, and the treatment of infectious illnesses, especially in hospital settings with resistant strains. 194
Challenges in Nanobioconjugates Application
Nanobioconjugates, although its promise in biological applications, encounter several hurdles that must be resolved for their extensive use. This encompasses stability and biocompatibility, scalability and standardization, legal and ethical considerations, as well as the incorporation of new technologies like as artificial intelligence (AI), personalized medicine, and high-throughput screening. 195 Stability and biocompatibility are essential for clinical acceptability, since nanoparticles may agglomerate, disintegrate, or lose their functional coatings in biological settings. Scale-up and standardization are crucial, since nanobioconjugates need the meticulous integration of nanoscale components with biological entities, demanding rigorous control over reaction conditions, purification methods, and formulation parameters. 195 Regulatory and ethical concerns are continuously developing, with organizations like as the FDA and EMA necessitating thorough toxicological evaluations, extensive biodistribution studies, and substantial data on pharmacokinetics and pharmacodynamics. 195 Personalized nanomedicine is a promising development, since advancements in genomes, transcriptomics, and proteomics provide customized therapeutics based on unique patient profiles. Artificial intelligence and machine learning algorithms may enhance nanoparticle design, forecast interactions, and tailor therapy protocols. 195
Comparative Analysis: Small Molecules Versus Nanobioconjugates
The therapeutic landscape for obesity and metabolic disorders has traditionally relied on small-molecule agents, which, despite their efficacy, are often limited by poor tissue specificity, rapid clearance, systemic toxicity, and the need for frequent dosing. Recent advances in nanotechnology have introduced nanobioconjugates as promising alternatives capable of improving drug delivery, enhancing therapeutic precision, and minimizing adverse effects. Nanobioconjugates leverage tunable surface chemistry, targeted ligand conjugation, and controlled release kinetics to optimize pharmacokinetics and biodistribution.
Table 4 provides a comprehensive comparative analysis of small molecules and nanobioconjugates across 7 critical dimensions, including efficacy, pharmacokinetics, surface chemistry, immunogenicity, and clinical translatability. By integrating specific examples, targeting ligands, and discussion of surface modifications, this table highlights the unique advantages of nanobioconjugates, such as improved adipose tissue targeting, reduced systemic toxicity, and potential for sustained delivery. Moreover, it underscores the ongoing clinical translation efforts and regulatory considerations associated with nanocarrier-based therapies. Collectively, Table 4 emphasizes the paradigm shift from conventional pharmacotherapy to precision nanomedicine approaches, offering a clear roadmap for future research and clinical development in obesity and metabolic disease management.
Comparative Analysis of Small Molecules and Nanobioconjugates in Obesity and Metabolic Disorder Management.
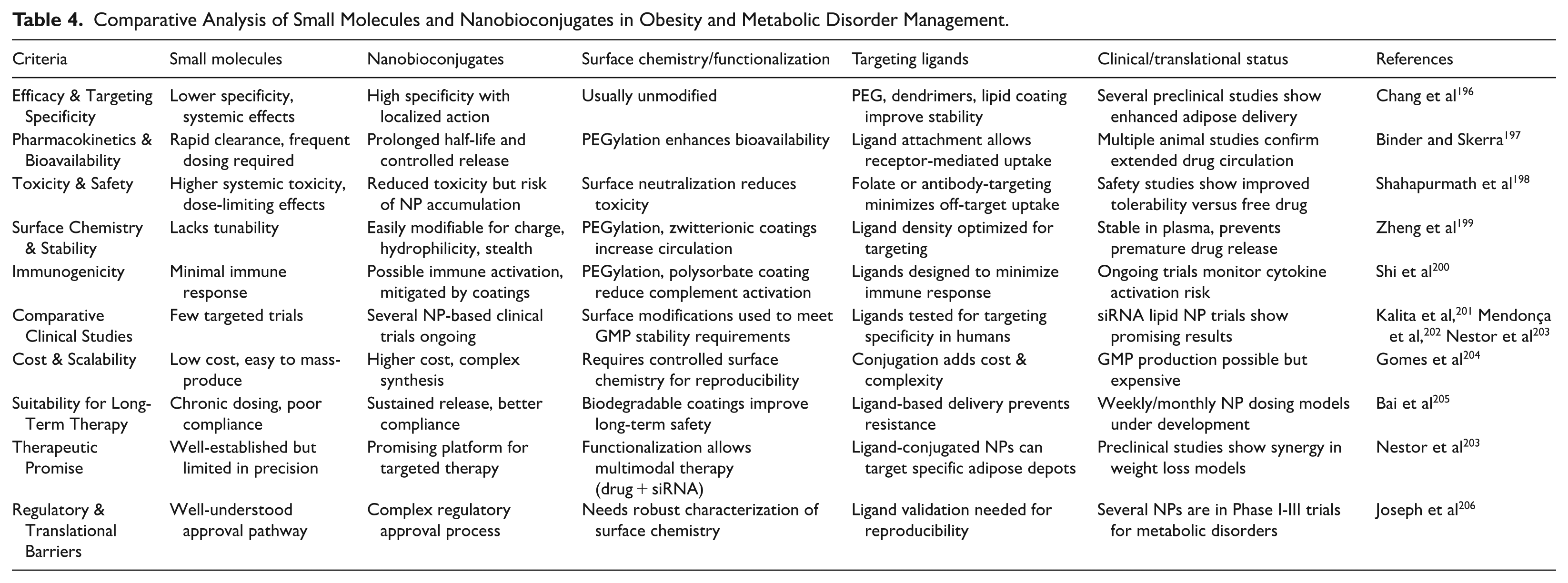
Despite their translational promise, nanobioconjugates face hurdles such as complex regulatory approval processes, the need for robust surface chemistry characterization, and reproducibility of ligand validation. Scale-up remains problematic, as conjugation steps add cost and complexity, while controlled surface chemistry is required to meet GMP standards. These factors make clinical implementation slower compared to conventional small molecules, which have a well-understood approval pathway but remain limited in precision
Future Perspectives and Research Directions in CD44-HA Targeting for Obesity
Emerging Strategies for Enhancing CD44-HA Targeting in Obesity
The transmembrane glycoprotein CD44 holds essential roles during adipose tissue metabolism and inflammation as well as in obesity development. Researchers analyze the connection between CD44 and hyaluronic acid for metabolic disorder therapy development. The goal to enhance CD44-HA targeting in obesity involves developing high-affinity HA ligands as well as monoclonal antibodies against CD44 and RNA-based therapeutics for CD44 expression regulation and biomimetic nanoparticles that reproduce the adipose microenvironment and peptide-based CD44 inhibitors. The targeting strategies work to enhance targeting while controlling inflammation in adipose tissues with improved metabolic functions. The selective binding power of HA-coated nanoparticles toward CD44-expressing cells serves to improve drug delivery as well as to reduce systemic toxicity.
Combination Therapies: Small Molecules and Nanobioconjugates in Obesity Treatment
The therapeutic outcomes of CD44-HA-targeted obesity treatments can become more efficient by combining small molecules with nanobioconjugates. Four treatment approaches for CD44-HA-targeted obesity treatment include synergistic drug combinations as well as nanocarrier-based drug delivery and photothermal and photodynamic therapy and immune modulation strategies. Combination drug synergies involve metabolic regulator-loaded HA-functionalized nanoparticles and nanocarrier-based drug delivery through liposomes and dendrimers, micelles along with genetic material and small-molecule inhibitors. The application of HA-coated nanoparticles with photosensitizers through photothermal and photodynamic therapy meets similar purposes as CD44-targeting nanobioconjugates which integrate anti-inflammatory agents for immune modulation strategies.
Personalized and Precision Medicine Approaches in Obesity Management
Standards of personalized medicine treatment for metabolic disorders have advanced through multiple advanced approaches. These treatment methods combine biomarkers with CRISPR-based genetic editing innovations together with single-cell genomic analysis and AI-powered drug development platform and 3D printing technology. Biomarker-driven therapy enables medical practitioners to determine which patients will obtain the best results from CD44-HA-targeted treatments. CRISPR-Cas9 technology enables the modulation of CD44 expression in adipose tissues which provides a treatment solution specific to individual patients for metabolic conditions. Research at the single-cell level can identify CD44 expression variety which would enable developers to create more accurate therapeutic approaches. Mechanisms that use AI to create computational models demonstrate their ability to estimate which patients should receive CD44-targeted treatment.
Overcoming Current Challenges in CD44-HA Therapeutic Development for Obesity
CD44-HA-targeted therapies face multiple barriers to treating obesity because of divergent CD44 expression levels, and drug toxicity as well as complexity of adipose tissue environment and restricted treatment penetration and emergence of resistance factors. CD44 appears in various forms which leads to expression variations across distinct adipose tissue stores thus blocking the development of universal targeting strategies. Administering HA systemically consists of undesired tissue effects that call for innovative methods to target adipose tissue specifically. The performance of CD44-targeted therapies faced limitations in obese adipose tissue because of its dynamic characteristics together with inflammatory responses which requires new drug delivery systems.
Clinical translation further requires addressing nanoparticle stability and biocompatibility, since these systems may agglomerate, disintegrate, or lose functional coatings in biological environments. Standardization and scale-up are equally critical, as rigorous control over reaction conditions, purification methods, and formulation parameters is essential to produce reproducible, regulatory-compliant products. Without overcoming these toxicity, delivery, and production barriers, promising preclinical findings will struggle to advance into consistent clinical outcomes
Potential for Clinical Translation and Regulatory Considerations
The clinical trials for CD44-HA-targeted metabolic disorder treatments continue while researchers build plans to enhance treatment dosages and partnership treatments along with improved patient inclusion criteria. Organizations that regulate pharmaceutical products demand thorough assessments of both safety and efficacy for preclinical testing purposes. The development of consistent HA-based formulations requires standardization since it ensures both therapeutic performance and reliability of treatment. Technical applications in personalized medicine require essential ethical attention because personalized therapies need to solve patient privacy concerns as well as genetic database management and fair treatment distribution standards. The continuous supervision of CD44-targeted drugs must occur to detect possible damaging effects while verifying their long-term performance. CD44-HA-targeted therapies for obesity management will advance through the adoption of modern technologies together with combined therapeutic approaches and patient-specific medicinal strategies. Progresses in 3 areas of nanotechnology biomarkers for treatment selection and genetic manipulations suggest they will enable the best possible therapeutic results.
Conclusion
The review examines how CD44-Hyaluronic Acid (HA) signaling affects obesity pathogenesis while concentrating on its effects on adipogenesis, inflammation and insulin resistance with subsequent metabolic dysfunction. The review demonstrates that CD44-HA binding mechanisms substantially drive both adipose tissue growth and sustained low-grade inflammatory processes which makes it suitable for therapeutic targeting. The analysis indicates that small molecule inhibitors and nanobioconjugates demonstrate different benefits along with their limitations where small molecules possess both quick drug absorption and economical production but nanobioconjugates deliver better targeted delivery and controlled drug duration.
Relevant to obesity treatment is the essential role of CD44-HA signaling blockade as a new therapeutic target to reduce adipose tissue inflammation and metabolic disturbances. Anatlysis with small compounds including CD44 blocking elements and HA-building inhibitor substances demonstrate ability to stop the growth of adipocytes while limiting production of inflammatory signaling compounds. Nanobioconjugates particularly those based on HA-functionalized nanoparticles provide an accurate method to break CD44-HA bonds without causing major systemic harm. Implementing these treatment strategies as part of obesity management would help produce more effective personalized obesity treatments while using conventional metabolic therapies. The analysis indicates that small molecule inhibitors, and nanobioconjugates demonstrate different benefits along with their limitations where small molecules possess quick absorption and economical production, nanobioconjugates deliver superior targeted delivery, and sustained action. Therefore, future therapeutic design must critically weigh these strengths and weaknesses to achieve optimal clinical translation.
While CD44-HA inhibition demonstrates strong preclinical potential, its translation to human application will depend on solving key barriers in toxicity mitigation, scalable production, and navigating complex regulatory frameworks. A critical future direction lies in integrating safety testing with scalable nanomedicine design, ensuring that therapeutic efficacy aligns with clinical feasibility.
The field requires additional research for effective clinical implementation of CD44-HA targets. Progress in biomaterials science combined with artificial intelligence in drug development processes as well as personalized medical techniques will improve the performance of CD44 inhibitor treatment methods together with nanomedicinal approaches. Truly effective CD44-targeting therapies require the resolution of several important obstacles which include tissue specificity requirements and immune responses as well as regulatory compliance requirements to reach human trials from preclinical trials. CD44-HA inhibition shows great potential as a progressive therapeutic treatment method for metabolic diseases by managing obesity.
Footnotes
Acknowledgements
NA.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Conceptualization; DEU, IJA, EUA, EGE, writing—original draft preparation: DEU, WAO, MAW, RIO, OPCU, writing— review and editing, all authors, and All the authors have read and agreed to the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All used data is fully presented in the manuscript.
Clinical Trial Number
Clinical trial number: not applicable.
