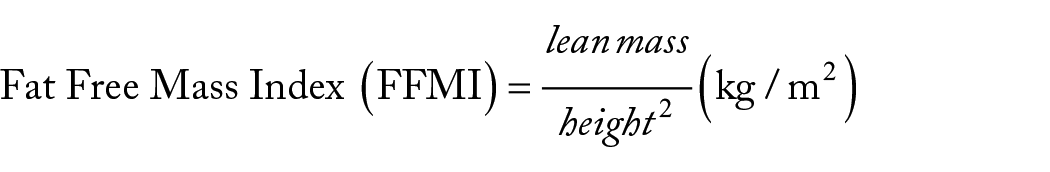Abstract
Introduction:
Any alteration in nutritional status can compromise the progression of breast cancer, as well as the tolerance and efficacy of chemotherapy. The aims of our study were to assess the nutritional status of breast cancer patients treated with chemotherapy and to identify nutritional factors that may exacerbate chemo-induced digestive toxicity.
Methods:
This descriptive cross-sectional study was conducted within the medical oncology department of the National Institute of Salah Azaiez. A Mediterranean Diet Score (MDS) was used to evaluate the level of adherence to the Mediterranean Diet. Nutritional status was assessed using 2 methods; body composition evaluation utilising the TANITA BC 601 impedance metre and High authority of health (HAS) criteria of malnutrition. Digestive effects were assessed with World Health Organization (WHO) grades ranging from 0 to 4. High grades 3 and 4 signified toxic grades.
Results:
We enrolled 107 adult women diagnosed with breast cancer undergoing the same chemotherapy protocol. The participants had an average age of 52.56 ± 9.75 years and an average BMI of 29.85 ± 5.72 kg/m2 with 78.5% classified as overweight. Malnutrition was observed in 41% of patients, while sarcopenia was present in 12%. Chemotherapy-related adverse effects were predominantly constipation (80%), diarrhea (73%), nausea (70%) and vomiting (63.4%) with one-third experiencing high-grade toxicity. Strong adherence to Mediterranean Diet was observed in 48.6% of cases and higher MDS scores were inversely correlated with the severity of nausea (P = .01) and constipation (P = .02). Malnutrition was significantly associated with all digestive toxicities except with constipation. Sarcopenia was associated only with vomiting (P = .005).
Conclusion:
There is a relationship between nutritional status and digestive toxicity, justifying early nutritional treatment to improve the tolerance and efficacy of chemotherapy.
Introduction
Breast cancer remains one of the leading causes of morbidity and mortality among women worldwide. According to the World Health Organization (WHO), there were around 2.3 million new cases of breast cancer in 2020, which accounts for 11.7% of new cases of cancer. It ranks first in terms of both incidence and mortality among women worldwide. 1 The Number of deaths exceeds 450 000 per year.
Nutritional status is emerging as a crucial element in this complex equation. Undernutrition is often associated with this neoplastic disease, affecting its progression. However, with the rapid rise of obesity, evaluating nutritional status has become more complex. This requires the use of more rigorous diagnostic criteria than just body mass index (BMI). Current data indicates that overweight and obesity at diagnosis, well as weight gain during the disease’s progression, are associated with unfavourable outcomes for breast cancer. 2
The abnormalities detected, particularly sarcopenia, can impact the tolerance and efficacy of anti-cancer treatments and consequently survival and quality of life.3,4 Malnourished breast cancer patients often endure fewer cycles of chemotherapy and radiotherapy, making them more vulnerable to treatment toxicities, treatment failures and rapid disease progression. 5
Despite significant advancements in therapeutic methods, chemotherapy remains the core treatment. However, individual tolerance to chemotherapy can vary considerably, according to the nutritional status of breast cancer patients who are subject to unique nutritional challenges whose pathophysiological mechanisms remain unsolved. 6 The side effects of these treatments can indirectly affect food intake and nutrient absorption. To minimise nausea and vomiting that occur after chemotherapy sessions, breast cancer patients often engage in prolonged fasting. However, fasting for more than 48 hours can lead to muscle mass loss, further worsening their nutritional status. 5
Some studies have focused on the nutritional status of breast cancer patients in Africa,5,7 but none have specifically addressed North Africa, particularly Tunisia so the objectives of our study were to evaluate the nutritional status of breast cancer patients undergoing chemotherapy treatment exclusively and identify the nutritional factors associated with chemotherapy-related digestive toxicity.
Methodology
Study design
This was a cross-sectional study conducted over a four-month period (May 2023–August 2023), with breast cancer patients at the medical carcinology department of Salah Azaiez institutes in Tunis.
Population
The study included consent patients aged between 18 and 69, diagnosed with stage I, II or III luminal A or B or triple-negative breast cancer). 8 These patients were undergoing sequential chemotherapy (Anthracyclines-Taxanes). This study did not include breast cancer male patients, pregnant and breastfeeding women, patients with stage IV (metastatic), patients undergoing immunotherapy or associated targeted therapy, patients who have received radiotherapy, patients with recurrent breast cancer, or patients with renal, cardiac, or hepatic insufficiency or dysthyroidism. Around a third of patients (35%) were obese before being diagnosed with breast cancer.
The number of patients required was calculated based on a 95% confidence interval (CI) and an estimated prevalence of early stage of breast cancer in Tunisia of 6%, according to the North Tunisian Cancer Registry. 9 The minimum number of women required was 87, as determined by the following formula:
where:
N: is the sample size,
z: is 1.96 for a confidence level of 95%
p: is the estimated proportion of the population
d: is the margin of error tolerated at 5%.
Data collection
Data related to sociodemographic characteristics and neoplastic disease (including the histological type of cancer and duration of disease progression) were collected for each patient. The TNM stage was determined according to the regional referential for breast cancer management in France 2018. 10 This information was gathered on an individual form for each patient, based on medical records and questioning. The patient was interviewed during chemotherapy (after at least 2 sessions).
All patients were asked about chemotherapy-related digestive toxicity, including symptoms such as nausea, vomiting, diarrhoea and constipation. Digestive side effects were classified according to WHO grades, which range from 0 to 5. Grade 0 signifies the absence of any event, while grades 1 to 4 describe manifestations with an increasing gradient of severity. Grades 3 and 4 reflect the toxicity associated with this effect. 11
Assessment of adherence to the Mediterranean diet
All patients completed a questionnaire to assess their adherence to the Mediterranean Diet Score (MDS), which is based on the latest update of the Mediterranean Diet Pyramid. 9 This validated questionnaire uses the recommended frequency of consumption for various food groups, including vegetables, fruit, olive oil, meat, dairy products, oilseeds and fast sugars. 10 The score typically ranges from 0 to 14; 12 a score of 10 or above indicates strong adherence to the Mediterranean diet, a score between 6 and 9 indicates moderate adherence to the Mediterranean diet and a score below 6 indicates poor adherence.
Anthropometric measurements
All patients underwent anthropometric measurements. Height was measured in centimetres using a graduated measuring tape. Patients were asked to remove their shoes, stand with their heels together, legs straight, arms at their sides and shoulders relaxed. The measurement was taken at the end of inspiration.
The TANITA BC 601 impedance metre with ISO 9001 certification was used to measure body weight (kg) and directly calculate the body mass index (BMI) in kg/m². It has been properly calibrated before use. Body weight and height, variables necessary to record the analysis of body composition, were measured with an accuracy of 0.1 kg and 0.01 m, respectively. The obesity class was determined by referring to the classification adopted by the World Health Organization (WHO) in 1998. 13 With patients fasting, shoeless and lightly clothed, this device also enabled the determination of body composition by calculating fat mass (%), lean mass (%), muscle mass (kg), bone mass (kg), total body water (%) as well as visceral fat levels. Normal values for fat mass, muscle mass and water mass are dependent on age and sex. They were assessed based on the reference values mentioned in the manual of the impedance metre used, which were consistent with the literature.14,15
Diagnosis of undernutrition
The diagnosis of undernutrition, according to the High Authority of Health (HAS 2021), 16 is based on the presence of at least 1 phenotypic criterion and 1 etiological criterion. In this case, we selected breast cancer as the etiological criteria. The phenotypic criteria included weight loss of 10% or more within 6 months compared to the usual weight before the onset of the disease, a BMI of less than 18.5 kg/m² and/or a quantified reduction in muscle mass and/or muscle function. 17
The reduction in muscle mass was calculated with reference to the bioimpedance metric Fat Free Mass Index (FFMI) using the following formula 18 :
A FFMI of less than 15 in women indicates a reduction in muscle mass.
Reduced muscle strength was assessed by measuring grip strength in the dominant or non-implanted hand using a CAMRY-type electric dynamometer. The patient was asked to hold the dynamometer with their arm in a resting position and exert maximum grip for 5 seconds on 3 trials separated by rest periods of at least 10 seconds. The highest value (in kg) was selected. A grip force below 16 kg indicates a reduction in muscle strength.
Diagnosis of sarcopenia
According to HAS 2021 and the European Working Group on Sarcopenia in Older People (EWGSOP 2019), the diagnosis of sarcopenia is based on the combination of reduced strength and muscle mass. 19 Strength was determined by dynamometer and muscle mass by FFMI.
Statistical analysis
The collected data were entered and processed using SPSS version 23 statistical analysis software. Initially, a descriptive analysis of the study population and the various data collected was conducted. The results were expressed for quantitative variables following the normal distribution in the form of mean and standard deviation of the mean if not they were expressed in median with the interquartile range (IQR) [Q25–Q75], and in number and percentage for qualitative variables.
Subsequently, a bivariate analysis was performed to investigate associations between nutritional status and various chemotherapy-induced digestive toxicities. The chi-square test was used to compare frequencies or means between categorical variables, if conditions were met. Otherwise, the Fischer test was used. The correlation test was used to compare quantitative variables. Comparisons between quantitative and qualitative variables were made using Student’s t-test if the quantitative variable followed a normal distribution. If not, the non-parametric Mann-Whitney test was used. A significance level for P ⩽ 5% was retained.
Finally, a multivariate analysis using binary logistic regression with 0.2 as the threshold was performed to determine the independent predictive nutritional factors for chemotherapy digestive toxicity in the model. The odds ratio (OR) was expressed by its 95% confidence interval (CI).
Conflicts of interest
All study participants were informed in advance of the type and purpose of the study and gave their consent for the use of their clinical and paraclinical data for the conduct of this study. We declare no conflict of interest in relation to this work. The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the National Institute of Nutrition and Food Technology (N 03/2021). Date: 5 April 2021.
Results
We have included 107 patients. The mean age of the study population was 52.56 ± 9.75 years with extremes ranging from 30 to 69 years. The mean duration of the disease was 8.71 ± 3.36 months, with a maximum duration of 1 year. Ductal carcinoma was the dominant histological type (92.5%). All patients received the same sequential chemotherapy protocol (anthracyclines-taxanes). Around 80% of patients have presented at least one of the chemo-induced digestive effects studied. The frequency of digestives effects was respectively; constipation (80%), diarrhea (73%), nausea (70%) and vomiting (63.4%). Levels of digestive toxicity are shown in Figure 1. Digestive toxicity (grade 3 and 4) was observed in 35% of cases.

Digestive toxicity according to WHO grades.
The mean Mediterranean diet adherence score (MDS) was 8.83 ± 2.71. Moderate and strong adherence were noted in 30.8% and 48.6% of patients respectively.
Table 1 shows the patients’ anthropometric measurements. The majority of the population (78.5%) were overweight (BMI ⩾ 25 kg/m2).
Anthropometric measures.

Abbreviations: BMI, body mass index; FFMI, fat free mass Index.
Referring to the phenotypical criteria for undernutrition of the High Authority of Health (HAS), 31% of the population had a decrease in grip strength; 13% had a FFMI below the thresholds; 12% lost at least 10% of their weight in 6 months and only 1.8% had a BMI of less than 18.5 kg/m².
In all, 41% of patients met the HAS criteria for diagnosis of undernutrition. Sarcopenia was diagnosed in 12% of cases.
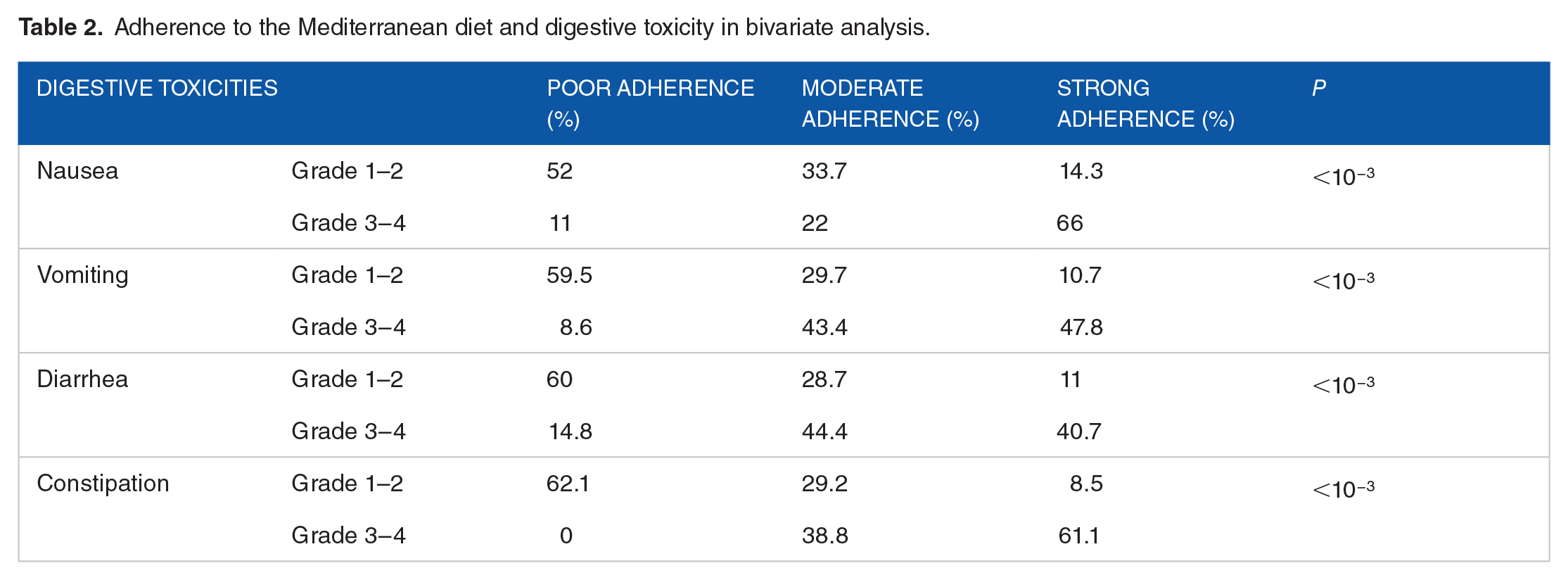
Adherence to the Mediterranean diet and digestive toxicity
In bivariate analysis, better adherence to the Mediterranean diet (MD) was significantly and inversely associated with all digestive toxicities (Table 2). In multivariate analysis, adherence to the Mediterranean diet was found to be a protective factor against nausea (OR = 0.13 [0.0–0.89]; P = .01) and constipation (OR = 0.7 [0.6–1.8]; P = .02) (Figure 2).
Adherence to the Mediterranean diet and digestive toxicity in bivariate analysis.

Adherence to the Mediterranean diet and digestive toxicity in multivariate analysis.
Anthropometric parameters and digestive toxicity
In our bivariate analysis, we noted a significant association between different digestive toxicities and anthropometric parameters which are summarised in Table 3.
Anthropometric parameters and digestive toxicities in bivariate analysis.

Abbreviation: BMI, body mass index.
However, in multivariate analysis of this study, we demonstrated a significant association between weight loss greater than 10% over 6 months and chemotherapy-related nausea (P = .02) and constipation (P < 10−3). A decrease in muscle strength, as assessed by handgrip, was significantly related to all digestive toxicities except with nausea. Similarly, the severity of nausea, vomiting and diarrhoea was significantly associated with undernutrition. As for sarcopenia, a significant association was noted only with vomiting (P = .005).
Table 4 summarises the associations between anthropometric parameters and digestive toxicity in the multivariate analysis.
Anthropometric parameters and digestive toxicities in multivariate analysis.

Abbreviation: BMI, body mass index.
Discussion
The main findings of our study were marked by a particular anthropometric profile; the mean BMI was 29.85 ± 5.72 kg/m2 with 78.5% of the population was overweight. Undernutrition was found in 41% of patients, while sarcopenia was found in only 12%. Adverse events related to chemotherapy toxicity were dominated by digestive effects observed in around 80% of cases. Toxicity was observed in 35% of cases.
Strong adherence to Mediterranean diet was noted in 48.6% of cases. The MDS score was inversely correlated with chemotherapy-induced Gastro-intestinal toxicities.
Mediterranean diet and digestive toxicity
In our study, around half of the patients had strong adherence to the Mediterranean diet (MD). As the Mediterranean Diet Score (MDS) increased, the severity of digestive toxicity decreased. As a result, digestive toxicity linked to nausea and constipation was inversely associated with adherence to the MD. Few studies have been able to conclude on the effect of nutrients specific to the Mediterranean diet on the digestive effects of chemotherapy. Due to its specific intake of fibre and olive oil, the MD was associated with better digestion and easier transit. 20 This was explained by its effect on modulating the intestinal microbiota by increasing microbial diversity and altering the proportion of certain bacteria. 21 In fact, through the fermentation of fibres, fatty acids are produced, promoting beneficial bacteria that have an anti-inflammatory effect and modulate intestinal dysbiosis. 22 Its effect on the efficacy of anti-cancer treatment and disease progression has been well documented in a literature review published in 2021, showing the ability of its nutritional components, notably resveratrol, retinoids, polyunsaturated fats and omega-3, to affect breast cancer growth. 23
Anthropometric parameters and digestive toxicity
In order to investigate the association between digestive toxicity and anthropometric parameters, the majority of studies refer to BMI. However, the toxic effects of chemotherapy depend more on muscle mass and body composition than on weight, body surface area and BMI. This is explained by the pharmacokinetics of therapeutic agents. 24
In this study, we demonstrated a significant association between weight loss of over 10% in 6 months and chemotherapy-related digestive toxicity. To our knowledge, few studies have debated this topic. Nevertheless, nausea, vomiting or even loss of appetite can disrupt food intake, affecting energy intake and subsequently leading to weight loss and even undernutrition. 25 To the best of our knowledge, few studies have discussed this subject. An early study published in 1998 investigated the effect of weight loss on chemotherapy tolerance in gastrointestinal cancer. 26 The results showed that those who lost weight during treatment were at greater risk of developing high-grade chemo-induced toxicity. There is controversy about the weight loss recommended by medical staff, which may reduce the risk of chemotherapy toxicity.27,28
Undernutrition and digestive toxicity
Reduced tolerance has been observed in malnourished patients in several studies of cancer patients.26,30,31 In our study, 41% of patients were diagnosed as undernourished. Around one third of the population had developed WHO grade 3 or 4 digestive toxicities. The severity of nausea, vomiting, and diarrhoea was significantly associated with digestive toxicity. Our results are consistent with those reported in the literature. 29 A prospective study published in 2010 30 showed that 50% of patients were malnourished prior to chemotherapy. These patients presented significantly more toxicity after chemotherapy. In contrast, other studies found that undernutrition was strongly associated with the development of grade 3 and 4 oral toxicities (stomatitis and mucositis) but did not conclude that there was an association with digestive toxicity. 26 The study by Hébuterne et al. found that the adverse effects of chemotherapy, notably taste disorders and digestive toxicities, 31 were at the root of anorexia. The latter is the main cause of undernutrition.
Sarcopenia and digestive toxicity
The relationship between sarcopenia and chemo-induced toxicity has been discussed in the literature and remains a subject of debate due to the inherent effects of this condition, which is still under-diagnosed.32,33 In our study, we noted a significant association between toxicity due to vomiting, transit disorder and sarcopenia. Similar findings reported in the literature indicate that sarcopenia was strongly associated with chemotherapy-induced nausea and vomiting.32,34 A cohort of 49 studies showed that being sarcopenic at inclusion was associated with a higher risk of developing severe digestive toxicities. 35 Similarly, for patients with metastatic disease, a decrease in skeletal mass index was significantly associated with high-level digestive toxicities, as well as post-operative complications.27,36 Other studies in women with early-stage breast cancer undergoing chemotherapy found a significant association between skeletal mass index and non-haematological toxicity. 26 This association has been explained by the pharmacokinetic characteristics of chemotherapeutic agents (narrow therapeutic index). 37 The impact of sarcopenia is not limited to the initial stage of treatment. As chemotherapy cycles progress, sarcopenia tends to increase. A significant and parallel increase in digestive toxicities, leading to frequent hospitalisations and a lower overall survival rate, is observed with taxane-based chemotherapy. 38 These findings underline the need for appropriate management of sarcopenia throughout the therapeutic course, with important implications for the planning and personalisation of canXcer therapy. 39
Limits and strengths of the study
Using a larger sample could improve the rigour of the analytical study by producing more significant results. Cross-sectional study that does not allow us to analyse the imputability link between the various nutritional factors examined and digestive toxicity (causality bias). The study has several strengths that enhance its impact. It identified significant associations between weight loss and chemotherapy-related digestive toxicity, providing valuable insights into how nutritional status affects treatment outcomes. Additionally, the study’s comprehensive analysis covered a wide range of nutritional factors, such as undernutrition and sarcopenia, and their association with various chemotherapy-induced toxicities, offering a holistic understanding of the issue.
Conclusion
Breast cancer is a multifactorial pathology with increasing incidence, impacting both the nutritional status of patients and their quality of life. Changes in body composition, in turn, affect tolerance to chemotherapy treatments. An altered nutritional status, often masked by overweight, is closely linked to the digestive toxicity of chemotherapy. We believe that intervening on the associated nutritional factors could reduce the severity of chemotherapy’s digestive effects in this population. Therefore, we recommend a thorough and comprehensive nutritional assessment of all patients with or at risk of breast cancer, along with individualised and targeted management to restore and maintain optimal nutritional status. Additionally, while the Mediterranean diet shows promise, further studies are necessary to address potential causality biases before clinical recommendations Cbv an be made.
Footnotes
Acknowledgements
There is no acknowledgement.
Author Contributions
B.O, S.M and B.O.R: writing, B.M.H and K.H: statistics, J.H and M.A: supervision
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All study participants were informed in advance of the type and purpose of the study and gave their consent for the use of their clinical and paraclinical data for the conduct of this study. We declare no conflict of interest in relation to this work. The study was conducted in accordance with the Declaration of Helsinki and approved (removed for blind peer review).
Data Availability
All data used in this study is available upon request to the corresponding author.
Ethical Statement
This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving research study participants were approved by the ethical commitee of salah azaiz hospital of Tunis under number ISA/2023/04. Written informed consent was obtained from all subjects.
Informed Consent
This study was taken for all patients