Abstract
Background:
Foodborne illnesses are the main cause of morbidity and mortality in the twenty-first century, and food handlers are the main source of these illnesses.
Objective:
The study aimed to assess the prevalence of intestinal parasites, enteric bacteria, antimicrobial susceptibility patterns, and associated factors among food handlers working in a food and drinking establishment in Yabelo town, Borena zone, Southern Ethiopia, in 2022.
Methods:
A facility-based cross-sectional study design was employed to assess the food handler’s state of health. Data were gathered using semistructured questionnaires and observational checklists, and 396 regular stool samples were collected. Samples were examined with microscopy and bacteriological culture to isolate enteric bacteria. Statistical Package for the Social Sciences, version 26 The factors that had a P-value of <.05 were considered statistically significant.
Result:
Of the 396 stool samples collected, 164 (41.4%) were tested positive for intestinal parasites, while 40 (10.1%) were positive for enteric bacteria (Salmonella and Shigella). Among intestinal parasites, the most predominant parasite was Ascaris lumbricoides 48 (12.1%). All isolates of Salmonella and Shigella showed resistance to ampicillin. In multivariable analysis, not trimming fingernails regularly (AOR = 2.02, 95% CI 1.97-5.37), not washing hands with soap after the toilet (AOR = 3.02, 95% CI 2.37-5.62), and eating raw food (AOR = 2.1, 95% CI 1.79-6.51) have a significant association with the prevalence of bacteria and parasites.
Conclusion:
The majority of food handlers had poor hygiene practices, and the prevalence of parasites and bacteria was high in the study setting. Educational status, eating raw vegetables or fruit, hand washing after the toilet, and fingernail trimming have associations with the health status of food handlers.
Background
Foodborne illnesses have developed into a global public health issue in this century. 1 Food-borne pathogens are a class of pathogenic microorganisms that infect people through food or food-borne pathways. 2 Street food is a daily meal for an estimated 2.5 billion people worldwide. Consequently, foodborne illnesses resulting from pathogenic bacteria and parasites are increasingly becoming a significant health concern linked to street food.3,4
Worldwide, intestinal parasite infections are common; however, in less developed countries, the incidence is higher because of factors such as inadequate personal cleanliness, unsanitary environments, socioeconomic status, demographics, and health-related practices.4,5 Intestinal parasite infections are primarily disseminated by tainted food and drink, although they can also be passed from person to person by fecal-oral transmission. 1 Approximately 3.5 billion individuals worldwide are impacted, of whom 450 million have symptoms. Each year, there are about 200 000 recorded deaths due to this parasite.5,6 Ascaris lumbricoides, hookworm, and Trichuris trichiura were the main causes of the infections.7,8 Intestinal parasites (IPs) are incredibly common in Ethiopia. Ascaris lumbricoides, Trichuris trichiura, and hookworm, respectively, afflict 1 in 3 (26 million), 1 in 4 (21 million), and 1 in every 8 (11 million) Ethiopians.
Microbes that cause enteric bacterial infections (EBIs), specifically Shigella (Shigellosis) and Salmonella (Salmonellosis), are the main causes of food-borne illnesses. Worldwide, 230 000 people died from non-typhoidal Salmonella enterica, and 52 472 people died from Salmonella typhi. 9 According to estimates from the World Health Organization (WHO), 31 food-borne diseases were responsible for 420 000 deaths and 600 million illnesses in 2015. Following the death toll of 52 472 from Salmonella typhi, 230 000 individuals died from Salmonella enterica. 10
Food handlers are those who handle food that may contain gastrointestinal, parasitic, and bacterial diseases. They can be major sources of intestinal parasites and enteric bacterial infection transmission to the general public if they maintain poor personal hygiene. 11
Studies demonstrate that enteric bacteria and intestinal parasites are common among Ethiopian food workers. The prevalence of IPs and EBIs among food handlers was found to be influenced by a number of factors in Ethiopia, including eating raw meat and vegetables, washing hands after using the toilet, getting medical cheek-ups, including stool exams, food hygiene training, and knowledge of enteric parasites and bacteria.11-13
Numerous studies have been conducted in Africa to investigate the prevalence of intestinal parasitic and enteric bacterial infections among handlers of food. In Ethiopia, the risk of Salmonella and Shigella infection among food handlers ranged from 1% to 8.85%.2,14,15 On the other hand, the prevalence of intestinal parasites ranges from 14.5% to 44% in restaurants and cafeterias.12,16
Although there is little information available, there were 280 458 outpatient instances of foodborne illness in Ethiopia, accounting for 9.39% of the population. 17 In terms of pain and suffering, lost productivity, and medical costs, the estimated annual economic cost of food-borne illness ranges from $10 to 83 billion. 18 People began eating more frequently outside of their homes as a result of rising urbanization and improvements in living standards, which allowed for the unrestricted creation of food establishments. 19 The frequency and antibiotic susceptibility profile of enteric bacterial infections, intestinal parasites, and their determinants are, however, poorly known in Ethiopia, and no research has been done there. Therefore, the purpose of this study was to assess the prevalence and antimicrobial resistance profile of intestinal parasites, bacteria, and associated factors among food handlers in a food and drinking establishment in Yabelo town, Borena Zone, Southern Ethiopia, in 2022.
Methods and Materials
Study area and period
The study was conducted in Yabelo town from November to December 2022. Located northwest of Moyale in the Borena Zone of Oromia, This town has a latitude and longitude of 4°53’N and 38°5’E and an elevation of 1857 m above sea level. It has served as the administrative center of the Borena Zone since its division from the Guji Zone in September 2002, as well as Yabelo woreda. Based on figures from the Central Statistical Agency in 2005, Yabelo has an estimated total population of 18 478, of whom 9551 are men and 8927 are women. In the town, the number of food and drinking establishments is 212, with 59 hotels, 70 restaurants, and 83 cafeterias.
Study population and design
A facility-based cross-sectional design study was conducted with select food handlers among food and drinking establishments of Yabelo town.
Inclusion and exclusion criteria
Food handlers who were working in the food and drinking establishment were willing to participate in the study, age ⩾18 years were included in the study. Food handlers who have taken anthelminthic, antiprotozoal, and antimicrobial drugs 2 weeks prior to sample collection were excluded. 1
Sample size determination and sampling techniques
The sample size was calculated using a single population proportion formula with the assumptions of a 95% confidence interval and 5% marginal error; n = (Z1-α/2) 2 P (1 – P)/d2,
Where “n” is the required sample size. Taking P = 44.6% (.446) (the prevalence of intestinal parasites among food handlers in Metu town, South West Ethiopia) 20 was used after assessing the proportion that can give the maximum sample size from all variables under study.
The final sample size was 416, including a 10% non-response rate. Study participants were selected by a simple random sampling technique after a proportional allocation of participants from all food and drink serving establishments and observed for assessment of personal hygiene, interviewed to assess practice, and examined for the presence of food-borne pathogens.
Data collection and laboratory processing
For collecting data, a standardized and structured questionnaires were employed. A combination of observation and in-person interviews were used to get the data. Each chosen food handler’s stool was collected in clean, dry, leak-proof stool containers. The stool samples were sent for analysis as soon as they were collected. In order to test for enteric bacteria, 1 g of stool samples were immediately transferred into the Cary Blair transport medium, tagged, and sent to the examination area (Blue Hora University Laboratory) in an ice-packed cold box (4°C) within an hour of collection. 1
Microscopic examination of stool
The stool was observed macroscopically (physically) for the presence of adult stage of some intestinal helminthes, consistency, and color. After visual assessment of the stool, intestinal parasites were examined microscopically mounted on each stool specimen using both direct smears mounted in saline and formal ether concentration procedure as recommended. 1
Culture and identification of bacteria
The selenite F broth was then mixed with a loop-full feces solution and incubated for 18 hours. Following incubation, Xylose Lysine Deoxycholate Agar (High Media, India) was inoculated with a loop of the suspension colonies. It was then incubated aerobically at 37°C for 24 hours. Salmonella and Shigella species’ growth after overnight incubation was determined by the color of their colonies on XLD agar (Salmonella: red with or without the black center; Shigella: red or pink colonies). The bacterial isolate was finally identified using 5 series of biochemical tests: Klinger iron agar (KIA), Simmons citrate agar, Sulfide indole motility test (motility, H2S generation, indole), lysine iron agar (LIA), and urease test.1,21
Antimicrobial susceptibility test
The Clinical Laboratory Standards Institute’s 22 Kirby-Bauer disc diffusion method was used for antimicrobial susceptibility testing. Briefly stated, the pure culture was added to a tube holding 5 ml of sterile normal saline (0.85% NaCl) and gently mixed until it created a uniform solution. The suspension’s turbidity was modified to have an optical density that is equal to 0.5 McFarland standards. The surplus was then removed by gently rotating a sterile cotton swab on the tube’s surface after dipping it into the suspension. The Mueller-Hinton agar (Oxoid, UK) surface was equally covered with the swab. The inoculated plates were allowed to dry for 3 to 5 minutes at room temperature. Ampicillin (10 g), trimethoprim-sulfamethoxazole (23.75/1.25 g), ciprofloxacin (5 g), gentamicin (10 g), ceftriaxone (30 g), and doxycycline (30 g) are the 8 antibacterial discs (Oxoid, UK) that are available. Using sterile forceps, tetracycline and nalidixic acid were applied aseptically to the inoculated plate. The zone of inhibition, which includes the disc, was measured with a digital caliper after a 24-hour incubation at 370°C and classified as sensitive, moderate, or resistant based on 18 interpretive breakpoints.
Data analysis
For data cleaning and analysis, the obtained data were examined, coded, put into Epi-data version 3.1, and exported to SPSS version 26.0. The data were analyzed using bivariate logistic regression, and variables with a P-value of .25 were chosen for the multivariate logistic regression analysis. To assess the variables associated with the prevalence of intestinal parasites and enteric bacteria, multivariable logistic regression was conducted. The variables were considered statistically significant if their P-value was less than .05. The Hosmer-Lemeshow test was used to assess the fitness of the models.
Data quality control
Data collectors received training to ensure the quality of the data. Throughout the data gathering, the supervisor provided daily oversight. Prior to the actual data collecting period, the questionnaire was pretested with a pilot survey of a comparable study sample. The questions have been translated into the regional tongue. Through all stages of data collection, entry, and analysis, data consistency was guaranteed.
The specimen’s quality, collection methods, and serial number were examined. The collected sample was promptly transported to the lab following collection. Stool was cultured and examined under a microscope by skilled laboratory personnel. Each medium was given a sterility test by being incubated at 37°C overnight. After an overnight incubation, the media that exhibited growth were discarded, and new media that didn’t show any growth was used in their stead. Throughout the study, the reference strains of American-type culture collections, including E. coli (ATCC 25922) and P. aeruginosa (ATCC 27853), were employed as a quality control for culture and testing for antibiotic susceptibility.
Result
Socio demographic characteristics of food handlers
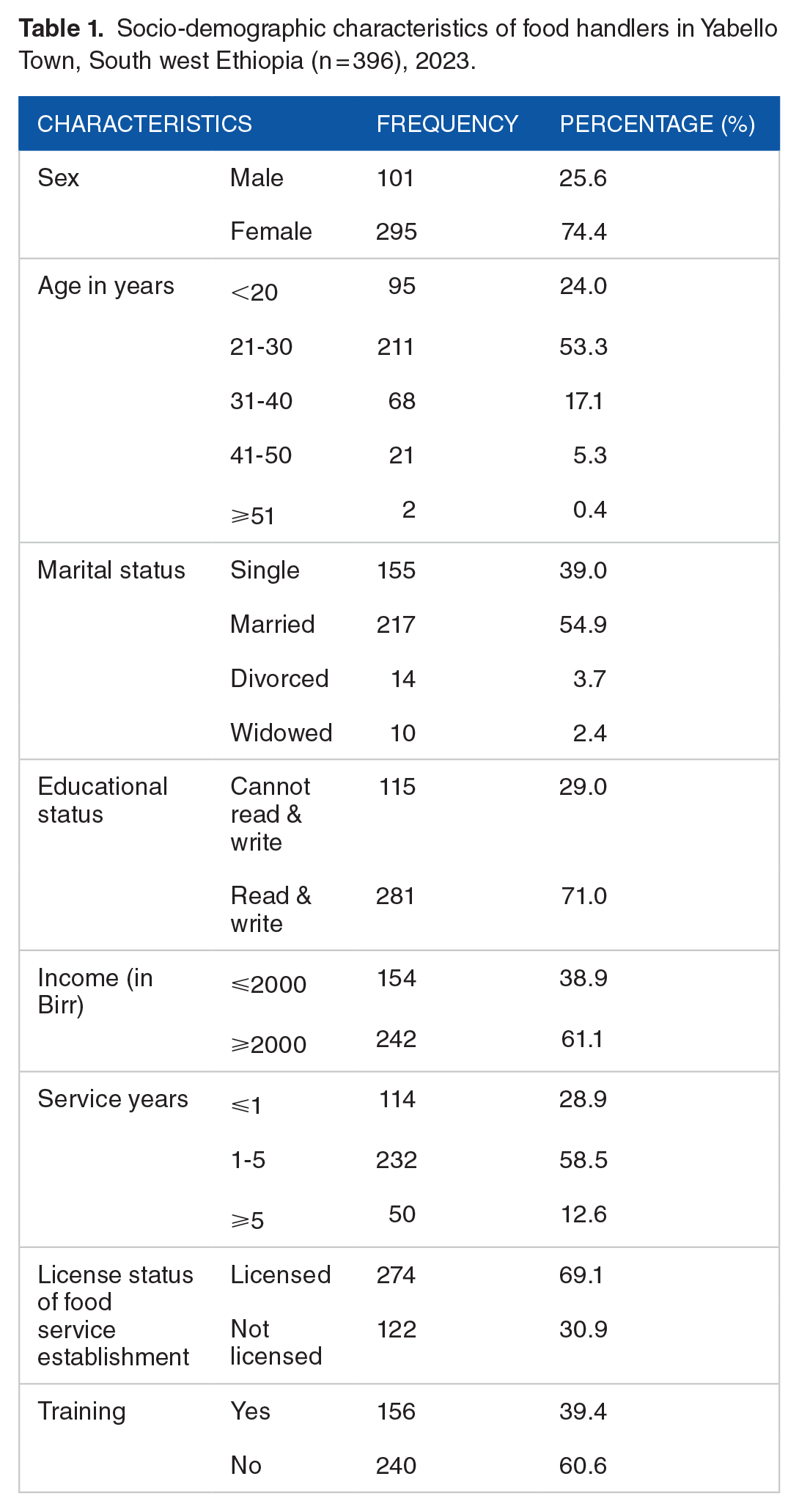
This study included 396 food handlers in total, with a response rate of 95.2%. Two hundred ninety-five people, 74.4% of the study participants were women. The participants are between the ages of 18 and 56. The majority of the participants in the study, 211 (53.3%), were between the ages of 21 and 30. 217 (54.6%) of the study’s participants, the majority, were already married. Of the study’s participants, 236 (58.5%) had worked as food handlers for 1 to 5 years. 281 (71%) of the study’s food handler participants were literate in both reading and writing. A total of 274 (69.1%) food service businesses had licenses. One hundred fifty-six food handlers (39.5%) have received training in safe food handling (Table 1).
Socio-demographic characteristics of food handlers in Yabello Town, South west Ethiopia (n = 396), 2023.
Prevalence of intestinal parasite infections
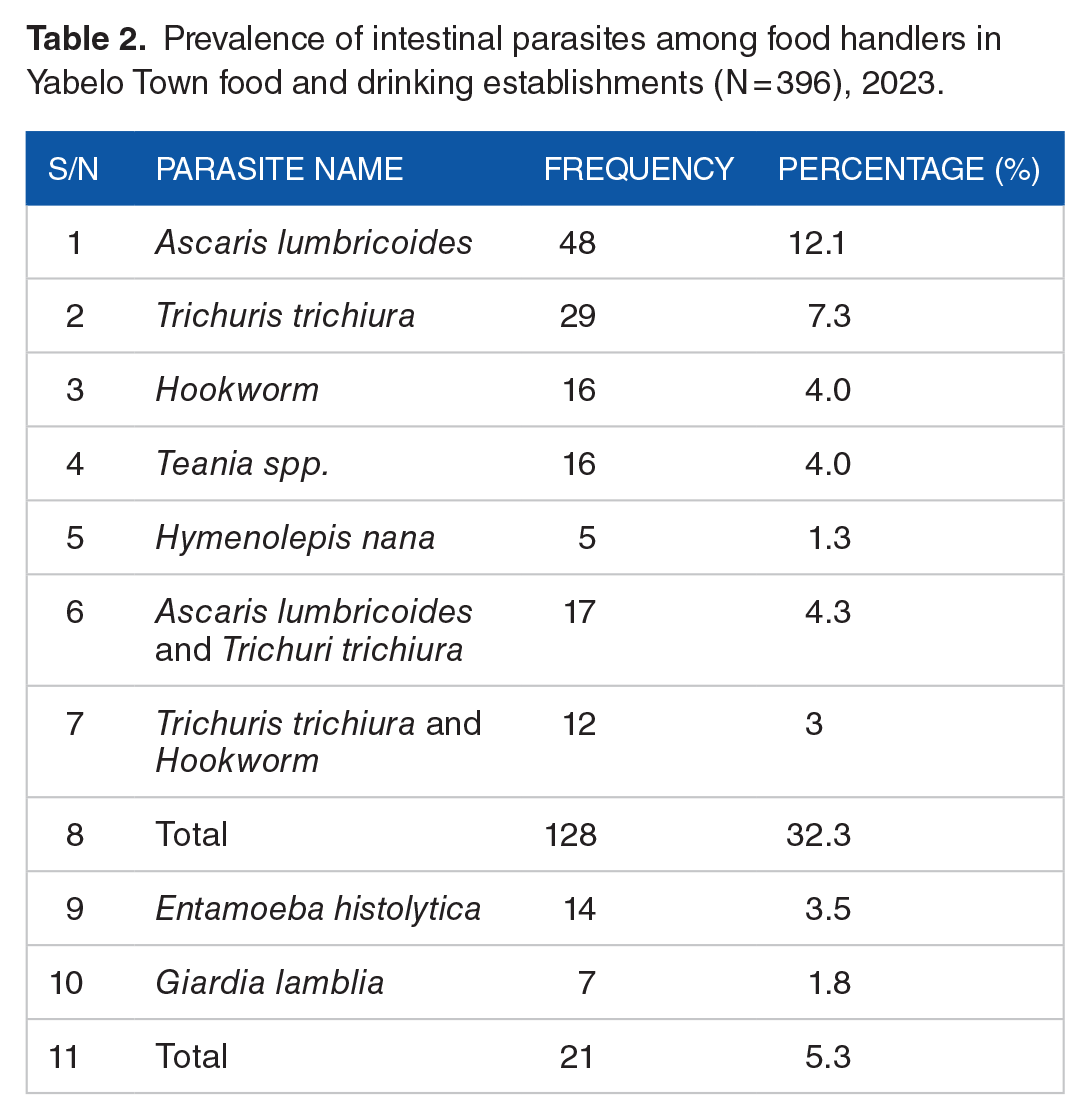
A total of 164 stool samples out of the 396 that were investigated were positive for various intestinal parasites, translating to a prevalence of 164 (41.4%). 29 (7.3%) of the positive subjects had mixed infections, while 135 (82.3%) had single infections. There were found to be 7 distinct intestinal parasites. Ascaris lumbricoides 48 (12.1%) and Trichuris trichiura 29 (7.3%) were the 2 most common intestinal parasite infections. In cases with combined infections, Ascaris lumbricoides and Trichuris trichiura predominated in 17 (4.3%) cases, followed by Trichuris trichiura and hookworm in 12 (3%) cases. Helminthes 143 (36.1%) are more prevalent than Protozoa 21 (5.3%) (Table 2).
Prevalence of intestinal parasites among food handlers in Yabelo Town food and drinking establishments (N = 396), 2023.
Bacteria identification
From 396 stool samples cultured for Salmonella and Shigella, 40 (10.1%) food handlers were positive. Salmonella was isolated from 37 (9.3%) study participants while shigella isolated from 3 (0.76%) of the study participants (Figure 1).
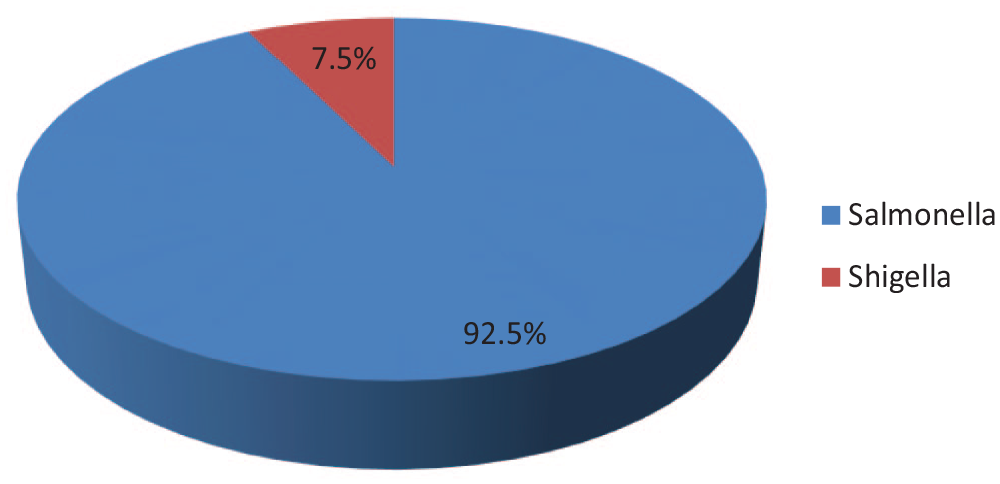
Prevalence of enteric bacteria among food handlers in Yabelo Town food and drinking establishments.
Antimicrobial susceptibility patterns of Salmonella and Shigella isolates
In this study, 8 antimicrobial drugs were tested against 37 Salmonella and 3 Shigella isolates. The Salmonella isolates had a 100% (37/37) ampicillin resistance rate, a 67.6% 23 tetracycline resistance rate, a 24.4% (11/37) nalidixic acid resistance rate, a 16.3% (7/37) trimethoprim-sulfamethoxazole resistance rate, a 5.4% (2/37) ciprofloxacin resistance rate, and a 10.9% (4/37) gentamicin resistance rate. Salmonella exhibited complete (37/37) sensitivity to ceftriaxone and doxycycline. While the 3 Shigella isolates were susceptible to ciprofloxacin, doxycycline, ceftriaxone, trimethoprim-sulfomethoxazole, Tetracycline, and nalidixic acid, the remaining 3 Shigella isolates were resistant to ampicillin. Shigella isolates don’t demonstrate resistance to more than one antimicrobial agent, while 7 Salmonella isolates (18.9%) did (Table 3).
Antimicrobial susceptibility pattern of Salmonella and Shigella isolates among food handlers in Yabelo town, Southern Ethiopia, 2023.
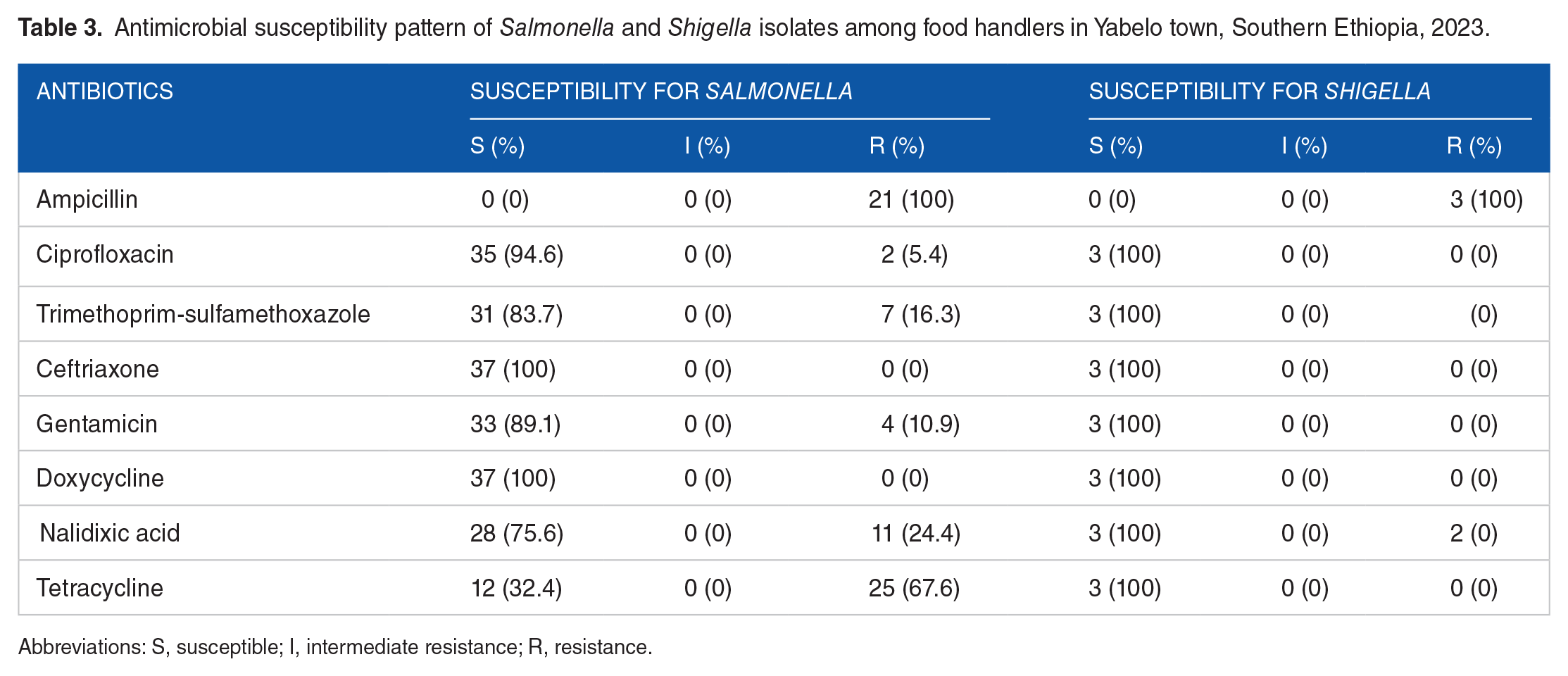
Abbreviations: S, susceptible; I, intermediate resistance; R, resistance.
Factors associated with the prevalence of bacteria and parasites of food handlers
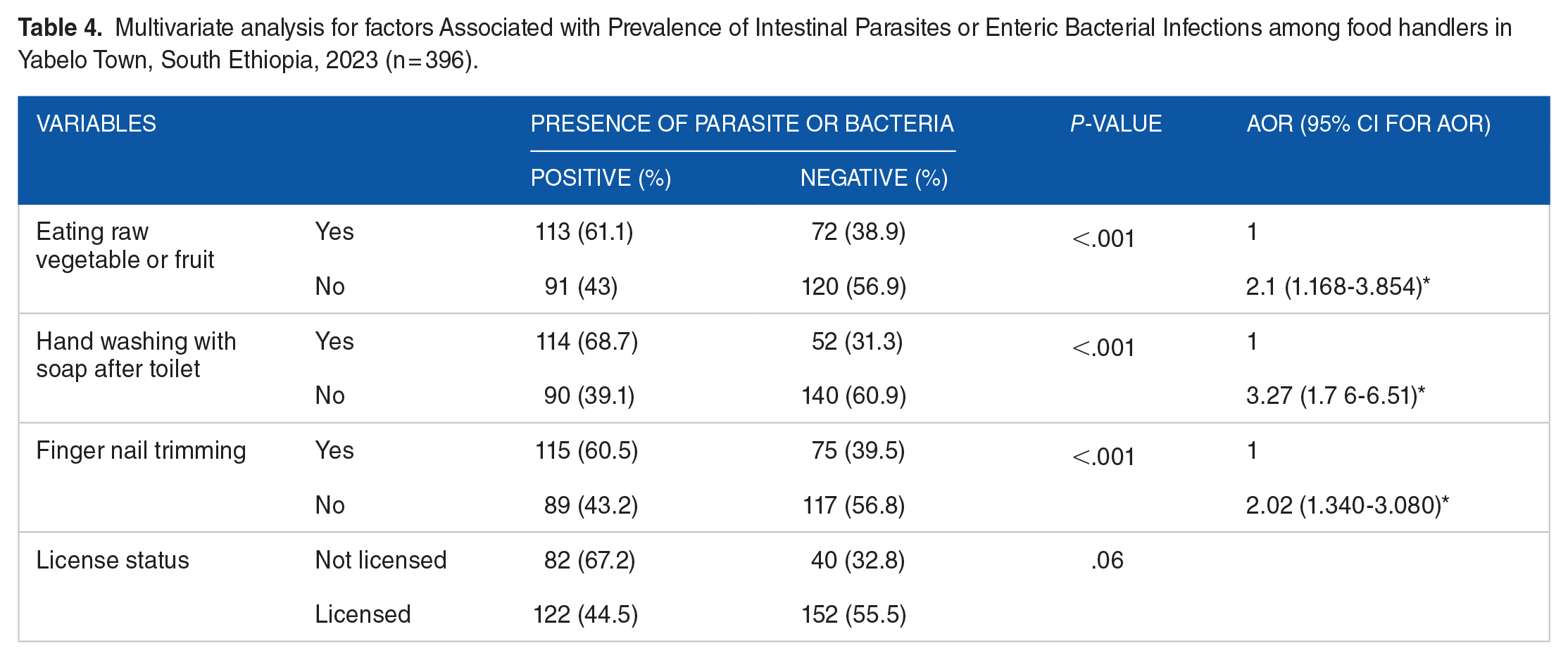
In multivariable analysis, not trimming fingernails regularly (AOR = 2.12, 95% CI 1.97-5.86), not washing hands with soap after toilet (AOR = 3.02, 95% CI 2.37-5.62), and eating raw food (AOR = 2.1, 95% CI 1.79-5.05) have a significant association with the prevalence of bacteria and parasites (Table 4).
Multivariate analysis for factors Associated with Prevalence of Intestinal Parasites or Enteric Bacterial Infections among food handlers in Yabelo Town, South Ethiopia, 2023 (n = 396).
Discussion
Food handlers have a great role in food and drinking establishments (where food is served to a mass of people) in the transmission of diseases since they have close contact with food items. The results of the current study show that 41.4% of food workers have intestinal parasites. Studies conducted in Yebu Town (44.1%), 23 Bahirdar Town (41.1%), 24 and Southern Ethiopia (36%) 25 all supported this conclusion. In contrast to the current study, a lower prevalence of intestinal parasites was found in Ghana (21.5%), 26 Aksum town (14.5%), 27 Chagni town (14.8%), 13 Motta town (26.7%), 28 Sodo University (23.6%), 29 and Sodo town (33.68%). 14 In addition to the current finding, a high prevalence of intestinal parasites was reported from Cameroon (56.7%), 30 Abukato, Nigeria (97%), 31 and Mekele University (52.4%). 32
In a recent study, Ascaris lumbricoides (12.1%) and Trichuris trichiura (7.3%) were the 2 most common parasites found, with helminths having more prevalence than protozoa. This result was in line with research conducted in Yebu Town. 23 But a study carried out in Chagni Town 13 and Nekemte Town 11 identifies Entamoeba histolytica (protozoa) as the main intestinal parasite. The prevalence of mixed parasite infections in this finding was 7.3%, which is higher compared to studies in Jimma Town (1.2%) 2 and Mekele University, which reported 1.3% of mixed intestinal parasite infections. 32 The variation in the incidence of intestinal parasites in various regions may be brought on by variations in personal hygiene habits, frequent medical exams, sociodemographic traits, detection tools used in labs, and geographic dispersion.
Regarding bacteria, the prevalence of salmonella and shigella in the current study was 10.1%, which is comparable to studies in Debre Markos (5.9%), 33 and Haramaya University (5.04%), 34 but higher than those in Dire Dawa city (8.7%), 35 Gondar town (11.3%), 36 and Arbaminch (9.5%). 37 Studies in Abuja, Nigeria (57.8%), 38 and Dila (12.7%), 39 as opposed to the current study, found a greater prevalence of Shigella and Salmonella.
The prevalence of salmonella was 9.3% in the current study, which was comparable with a study from Wolyta Sodo (9.1%) 14 and Jimma Town (8.1%). 2 However, the current finding was higher compared to studies in Dire Dawa city (6%), 35 Arbaminch (6.9%), 37 Motta town (2.5%), 28 Debre Markos (3.6%), 33 and Addis Ababa (3.8%), 40 compared to the current study. These variations in Salmonella prevalence may be due to personal hygiene and geographical distribution differences.
In this finding, about 0.75% of the study participants were carriers of the Shigella isolate. This finding agrees with findings from Dire Dawa City (1.7%), 35 Jimma Town (0.8%), 2 and Motta Town (1.6%). 14 A higher prevalence of Shigella was reported from Gondar town (10.1%) 36 compared to the current study. The prevalence of Shigella isolates in the current study was relatively low. However, a study at Addis Ababa University reported no Shigella isolate. 41 This discrepancy may be due to the cultural media used and geographical distribution.
Salmonella had 100% resistance to ampicillin, 67.6% resistance to tetracycline, 24.4% resistance to nalidixic acid, 16.3% resistance to trimethoprim-sulfamethoxazole, 10.9% (4/37) resistance to gentamicin, and 5.4% (2/37) resistance to ciprofloxacin, according to the antimicrobial resistance profiles of the isolates. This finding was consistent with studies in Bahirdar, 42 Motta, 28 and Addis Ababa. 40 The sensitivity of the isolate was observed for doxycycline (100%), ciprofloxacin (94.6%), ceftriaxone (100%), gentamicin (89.1%), nalidixic acid (73.7%), and trimethoprim-sulfamethoxazole (83.7%). This result was consistent with a Dire Dawa City study, which found that the isolate was very sensitive to ceftriaxone (100%), ciprofloxacin (89.5%), nalidixic acid (75.6%), and gentamicin (84.2%). The current analysis concurs with a study from Addis Ababa University that showed a high (100%) sensitivity of Salmonella to ciprofloxacin and gentamicin. 29 This finding’s sensitivity to trimethoprim-sulfamethoxazole (83.7%) was consistent with research from Addis Ababa (85.7%) 40 and Motta Town (83.3%). 28 However, compared to the current finding, studies in Debremarkos observed a reduced sensitivity of trimethoprim-sulfamethoxazole (62.5%). 33
All Shigella isolates in the current study were ampicillin-resistant. A similar finding was reported from Motta town 28 and Gondar town. 36 This might be because people have been using the antibacterial medicine repeatedly without a prescription. The isolate, however, was completely susceptible to the antibiotic’s doxycycline, ciprofloxacin, ceftriaxone, gentamicin, and trimethoprim-sulfamethoxazole. Findings from Gondar Town, 36 Motta Town, 28 Arbaminch University, 37 and Addis Abeba 40 are in agreement with this one. Our results showed that the medications that should be prescribed for the treatment of Shigella and Salmonella were trimethoprim-sulfamethoxazole, ciprofloxacin, ceftriaxone, gentamicin, and nalidixic acid.
According to the multivariate analysis of the current study, food handlers who consume raw fruits and vegetables have a 2-fold higher risk of testing positive for intestinal parasites and bacteria. This study is comparable to one that was done in Dila. 39 This suggests that consuming raw food increases the chance of contracting bacterial and parasitic diseases.
According to the current study, food handlers’ health is significantly related to how often they wash their hands with soap after using the lavatory. People who did not consistently wash their hands after using the toilet had 3 times the chance of testing positive for intestinal germs and parasites. The results of this study are consistent with research done in Ethiopia. 2 Regular hand washing with soap, which has an antibacterial effect, can minimize intestinal parasites and germs by eradicating or killing them. Therefore, routine hand washing after using the lavatory is advised to lower the incidence of parasites and germs, especially at restaurants and bars where food is given to a large number of customers. Additionally, regular finger trimming is significantly linked to food handlers’ health. Those who don’t frequently clip their fingernails have a 2-fold higher chance of being positive. This result is consistent with research done in Chagni 13 and Nekemte 35 towns in Ethiopia. When a food handler prepares and serves food regularly, untrimmed fingernails can host various intestinal parasites and bacteria and contaminate it.
Limitations of the study
In the current study, the intensity of the parasite, the genes responsible for antibiotic resistance, and the identification of enteric bacteria up to subspecies level were not assessed due to the shortage of resources.
Conclusions and Recommendations
In the study environment, there was a significant prevalence of enteric bacterial illnesses and intestinal parasites among food handler. The existence of an intestinal parasite or enteric bacterial pathogen was substantially correlated with parameters such as eating raw fruit or vegetables, washing hands after using the toilet, and fingernail trimming, indicating poor personal hygiene among food handlers. Consequently, reducing the risk of infection for food handlers and their clients will require regular medical exams, health education, and mass drug administration to all food handlers by a responsible body. High ampicillin resistance was detected in samples of both Shigella and Salmonella. As opposed to this, both isolates showed high sensitivity to other antimicrobials like doxycycline, ciprofloxacin, ceftriaxone, gentamicin, trimethoprim-sulfamethoxazole, and nalidixic acid. Consequently, this medication is suggested for treating Salmonella and Shigella species.
Footnotes
Acknowledgements
First and foremost, we would like to express our gratitude to the Department of Medical Laboratory Sciences at Blue Hora University’s Institute of Health for giving us the opportunity to write this study. We appreciate the Yabelo town Health Bureau and the town government for allowing us to collect the sample from the community. We appreciate the proprietor of the restaurant’s willingness to help us with data collection and extend our gratitude to them. We want to thank food handlers for taking part in the study, but we won’t name names.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
AST, Antimicrobial Susceptibility Test; ATCC, American Type Culture Collection; CLSI, Clinical and Laboratory Standard Institute; MDR, Multi-Drug Resistance; TSI, Triple Sugar Iron; XLD, Xylose-Lysine Decarboxylase.
Author Contributions
All authors contributed significantly to the proposal document’s creation, data collection, analysis, and interpretation, the paper’s critical evaluation, and the choice to submit the work to a prestigious publication.
Availability of Data
Data on which the conclusions relies on was found in the main manuscript.
Ethical Clearance
The Bule Hora University Institute of Health Chief Research and Community Service and technology transfer Directorate granted ethical approval for the study. After a brief discussion of the advantages of the study, the owner/manager of the restaurant and the food handlers gave their informed consent.
