Abstract

Dear Editor,
Varicella, commonly referred to as chickenpox, and Herpes Zoster, known as shingles, are both caused by the Varicella Zoster Virus (VZV), a neurotropic virus that is widely prevalent. This virus is classified within the human Herpesviridae family, specifically under the human alphaherpesvirinae subfamily and the Varicellovirus genus. Varicella, typically experienced during childhood, is characterized by symptoms such as fever and the development of vesicular rashes. It is important to note that VZV is the causative agent for both chickenpox and shingles. Transmission of this virus primarily occurs through direct contact with the sores of chickenpox or through inhalation of droplets exhaled by individuals who are infected. 1 VZV is highly contagious and easily spreads through contact with vesicular fluid and nasopharyngeal secretions, boasting an infectivity rate exceeding 85% to 90%. 2
The VZV nomenclature, derived from the complete genome sequences of Varicella Zoster Virus (VZV), identifies a total of 7 well-established clades (designated as clades 1-6 and 9), while 2 provisional clades (VII and VIII) have also been detected in global circulation. 3 In a comprehensive survey of suspected Monkeypox cases conducted by ICMR-NIV, an analysis of 16 whole genome sequences revealed a predominance of clade 5, with 11 instances, followed by clade 9, which accounted for 3 cases, and clade 1, represented by 2 cases. This investigation primarily aimed to elucidate the prevalence of various VZV clades and scrutinize the significant single nucleotide polymorphisms (SNPs) within them. Notably, this pioneering study marks the inaugural documentation of the presence of clade 9 in India, 4 a strain that notably constitutes the prevailing variant in countries such as Germany, the UK, and the USA. Furthermore, a study centered on patients afflicted with central nervous system (CNS) involvement in VZV infection in the metropolitan region of New York City revealed the prevalence of clade 9 strains in localities exhibiting a higher concentration of immigrants from Middle Eastern and Indian backgrounds. This discernment suggests the potential circulation of clade 9 within India’s demographic landscape. 5
Recent reports have highlighted cases of coinfection involving both chickenpox (Varicella) and Monkeypox (MPX) among children in African regions.6,7 The prevailing hypothesis put forth by earlier investigations leans toward the notion that Monkeypox likely preceded the onset of chickenpox in these children. Monkeypox, an emerging ailment, is attributed to the Monkeypox virus, which falls under the Orthopoxvirus genus. Monkeypox manifests an incubation period spanning from 5 to 21 days, while chickenpox’s incubation period ranges from 10 to 21 days. Distinguishing characteristics between the 2 include the nature of skin rashes, which tend to be more uniform in Monkeypox as opposed to the typically varied rashes seen in chickenpox. Furthermore, Monkeypox exhibits a gradual evolution of rashes for days, contrasting with the swift progression observed in chickenpox. The distribution pattern of rashes also differs, with Monkeypox displaying a centrifugal spread, whereas chickenpox presents a centripetal pattern. Notably, lymphadenopathy, marked by swollen lymph nodes, is a prominent feature in Monkeypox but typically absent in cases of chickenpox. 8 In the United States, a majority of VZV infections are attributed to clades 1 and 3. However, with the introduction of the Varicella vaccine and shifts in immigration patterns, the present distribution of clades and their respective contributions to both CNS and non-CNS-related diseases remain largely uncharted territory, thus paving the way for further research endeavors.
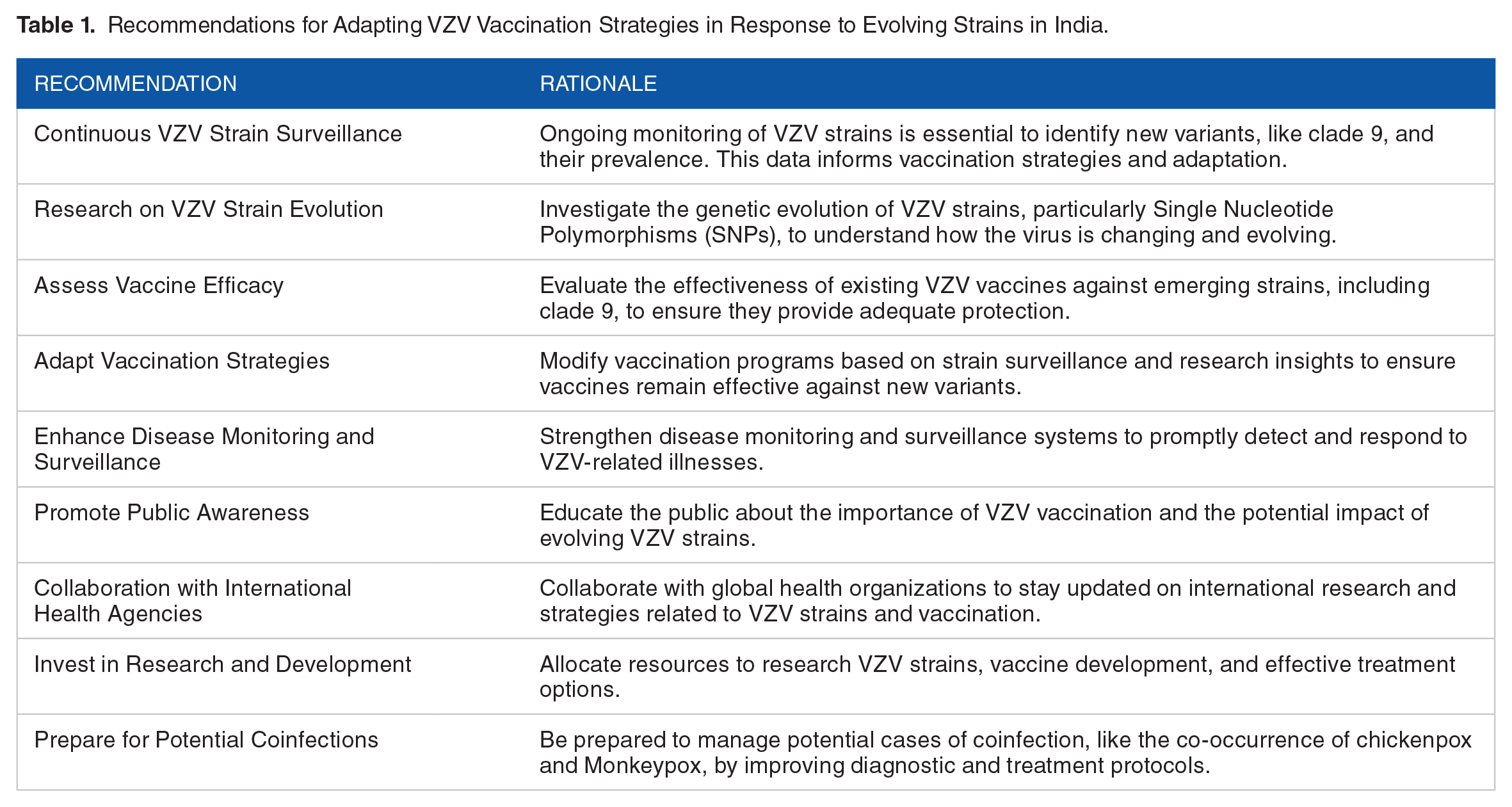
The presence of the VZV clade 9 variant in India instigates multifaceted considerations for the nation’s VZV-focused vaccination campaign. Intriguingly, despite the definitive identification of clade 9, patients did not manifest an augmented disease severity, underscoring the intricate facets of vaccine efficacy. This underscores the imperative for an upswing in research efforts, meticulously scrutinizing recombination dynamics in both indigenous wild-type VZV and vaccinated populations. A profound understanding of the evolutionary nuances of SNPs is pivotal for precise disease monitoring and surveillance, especially within the unique backdrop of VZV infections in the Indian context. These insights assume an indispensable role in refining vaccination strategies for the proficient management and mitigation of VZV-associated diseases (Table 1). Furthermore, this investigative endeavor is integral to buttressing the resilience and adaptability of vaccination campaigns, ensuring they remain robust and agile in the face of an ever-evolving spectrum of VZV strains. This robust stance is essential for steadfastly safeguarding the public health of the Indian populace against the neurotropic virulence of this viral adversary. Thus, an unceasing evaluation of vaccination program effectiveness in light of emerging genetic variants is imperative, enhancing our readiness to combat VZV-related diseases within the dynamic demographic landscape of India.
Recommendations for Adapting VZV Vaccination Strategies in Response to Evolving Strains in India.
Footnotes
Acknowledgements
None.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: SB & AN; Methodology: SB, AN, AA; Formal analysis and investigation: HK; Data extraction and screening: AA,DM; Writing—original draft preparation: SB,HK,NPS; Writing—review and editing: DM & DD.
All the authors have contributed to revising and final approval of the version to be published. All authors agreed and confirmed the manuscript for publication.
Ethics Approval
N/A
