Abstract
Background:
Antimicrobial resistance (AMR) remains a global health challenge, as bacteria display increasing resistance to last-resort antibiotics such as carbapenems. Enterobacter cloacae are evolving and developing high level of resistance to carbapenems. With increasing AMR, availability of antibiotics for treatment dwindles, hence a need to complement antibiotics to enhance activity or reduce the level of resistance. This study explored the use of calcium ions in attenuating bacterial resistance to carbapenems.
Method:
E. cloacae strains isolated from hospital fomites and air were subjected to antimicrobial susceptibility testing with carbapenem antibiotics (imipenem, meropenem, doripenem and ertapenem) using the disc diffusion (E. coli ATCC 25922 as control). Growth profile, Ca-Adjusted assay and time-kill curve of the strains was determined in the presence and absence of carbapenem antibiotics following a calcium stress assay.
Results:
Growth profile showed that all the E. cloacae strains grew markedly well at 37°C relative to ATCC 25922 and all strains displayed 80% to 100% level of resistance to tested antibiotics. The growth rate of the strains in the presence of the antibiotics was comparable to the growth rate in the absence of carbapenems. Conditional growth stress with calcium ions showed a 50% reduction in the level of resistance with doripenem displaying the lowest level of reduction and ertapenem, the highest.
Discussion:
The study showed that E. cloacae strains displayed high levels of resistance to carbapenems, increasing the possibility of treatment failure. Challenging strains with calcium prior to antibiotic treatment led to a significant reduction in level of resistance, indicating that calcium ions could affect bacterial strains during antibiotic activity leading to reduction in level of resistance.
Conclusion:
Calcium supplement could potentiate carbapenem effectiveness and reduce bacterial AMR.
Introduction
Antibiotics are losing effectiveness due to emergence of antimicrobial resistant bacterial pathogens, especially to those described as ‘Critical Global Priority Pathogens’ in the Intensive Care Unit (ICU).1,2 This is compounded by little or no new antibiotics in the development pipeline, facilitating a need to potentiate or repurpose available antibiotics to increase their effectiveness. In developing countries where nosocomial infections are on the rise, some of these pathogens including Enterobacter cloacae are emerging and becoming increasingly resistant to multiple antibiotics. 3 High mortality rates of patients as a result of infections associated with multidrug resistant E. cloacae is becoming a concern. 4 This is aggravated as the pathogen is now developing resistance to last-resort antibiotics including carbapenems. 5 Carbapenem-resistant Enterobacteriaceae (CRE) of which E. cloacae is a notable member demand urgent attention. (CRE) produce carbapenemases, which are β-lactamases that hydrolyse carbapenems and β-lactams. CRE depends on divalent cations such as Ca2+ to activate the production of these enzymes. 6 The mechanism by which this is achieved is unclear, however cationic antibiotics like polymyxin B interact with the lipopolysaccharides (LPS) localised on the outer membrane of Gram-negative by competitively displacing cations from the lipid A component of the LPS. 7 While this is an indulgent escape of bacteria from cationic antibiotics, it is likely that divalent cations might have an influence on the effectiveness of antibiotics against resistant bacteria such as E. cloacae. This study aimed to profile the effects of Ca2+ on carbapenem resistant E. cloacae isolated from ICU.
Experimental Approach
Bacterial strains and growth
This study was conducted in 2020-2021 at the Molecular Biology Lab, Department of Biochemistry, Cell and Molecular Biology (WACCBIP-BCMB), University of Ghana. E. cloacae (ENAB_010, ENAB_011, ENAB_012 and ENAB_013) strains were collected from AbiMosi Bacterial Culture (ABC©) Library, (WACCBIP-BCMB). These strains were isolated from fomites (ENAB_010, ENAB_011) and air (ENAB_012 and ENAB_013) from selected Intensive Care Units of hospitals in Ghana. All strains were cultured under aseptic conditions at 37°C in Luria-Bertani (LB) broth and subsequently recovered on LB agar. Smooth, large, creamy with rounded end, rod shaped, catalase positive and oxidase negative pure colonies of E. cloacae were used for all the experiments.
Antibiotic susceptibility testing
Antibiotic susceptibility testing was performed with Kirby Bauer disc diffusion. Briefly, pure colonies of E. cloacae were cultured overnight at 37°C in Mueller-Hinton broth (MHB) (Oxoid, England) and adjusted to 0.5 MacFarland standard. The culture was seeded on MH agar with a sterile cotton swab and carbapenems antibiotic discs (meropenem (10 µg/mL) – MERO, doripenem (10 µg/mL) – DOR, ertapenem (10 µg/mL) – ERTA, imipenem (10 µg/mL) – IMI; Oxoid, Basingstoke, Hants UK) were aseptically applied on the plate and incubated at 37°C overnight (18-24 hours), ATCC 25922 E. coli was used as a control. The zones of inhibition were interpreted according to CLSI guidelines 8 and presented as percentage resistance or susceptible.
Growth profile, Ca-adjusted assay and time-kill experiment
The resistance profile of the strains was further confirmed using a micro-broth dilution assay. This was performed in 96-well plates by determining the growth rates of the strains in the presence of carbapenems. 9 Ca-adjusted bacterial growth assay was conducted as previously described.10,11 Briefly, MHB was supplemented with Ca2+ (20-25 mg/L; 12 mM) and overnight culture of the strains was diluted 10-fold into sterile Ca-adjusted MHB. 100 µL was transferred into the 96-well plates containing 100 µL of the carbapenems antibiotics and incubated with shaking (150 rpm) at 37°C. Optical density (OD600) was measured by plate reader at indicated time points (1-24 hours). Aliquot (10 µL) was spread on LB agar and recovered colonies were further subjected to susceptibility test against carbapenems as previously stated. Also, similar to the Ca-adjusted assay, the time-kill activity of the calcium supplement on E. cloacae was performed in triplicate according to the microbroth dilution protocol guided by CLSI criteria.8,9 Briefly, the Ca-adjusted culture of the strains at the log phase were adjusted to 106 CFU/mL. 100 µL of the inoculum with 100 µL of the carbapenems in the 96-well plates were incubated at 37°C for 24 hours. To determine the time-kill activity in log10 CFU/mL, 30 to 50 µL was withdrawn at 1 hour intervals for 24 hours, spread on LB plates and incubated as earlier stated.
Data Processing
Data were collected and analysed with SPSS 28.0.195. Percentage antibiotic resistance and OD values presented as graphs using descriptive statistics. The analysed data as graphs represents experiments conducted in triplicate unless otherwise noted.
Results
Antimicrobial resistance profiles and growth rate of strains
The E. cloacae (ENAB_010, ENAB_011, ENAB_012 100 ENAB_013) strains displayed high level (80%-100%) of resistance to all the 4 carbapenems antibiotic tested relative to the ATCC 25922 E. coli control (5%-12%). The highest level of resistance was observed in fomite-resident ENAB_010 to meropenem (100%) and doripenem (97%) (Figure 1A). The standard growth curve indicated that the strains grew appreciably well at 37°C (Figure 1B). This was similar to growth rate of the strains in the presence of meropenem (Figure 1C), doripenem (Figure 1D), iminipenem (Figure 1E) and ertapenem (Figure 1F). There was increase in growth rate as time increases with OD600 range of 0.01 in the first 2 to 1.5-2.0 at 24 hours, indicating that the strains are extremely resistant to the carbapenems relative to the control (⩽0.01, 24 hours).
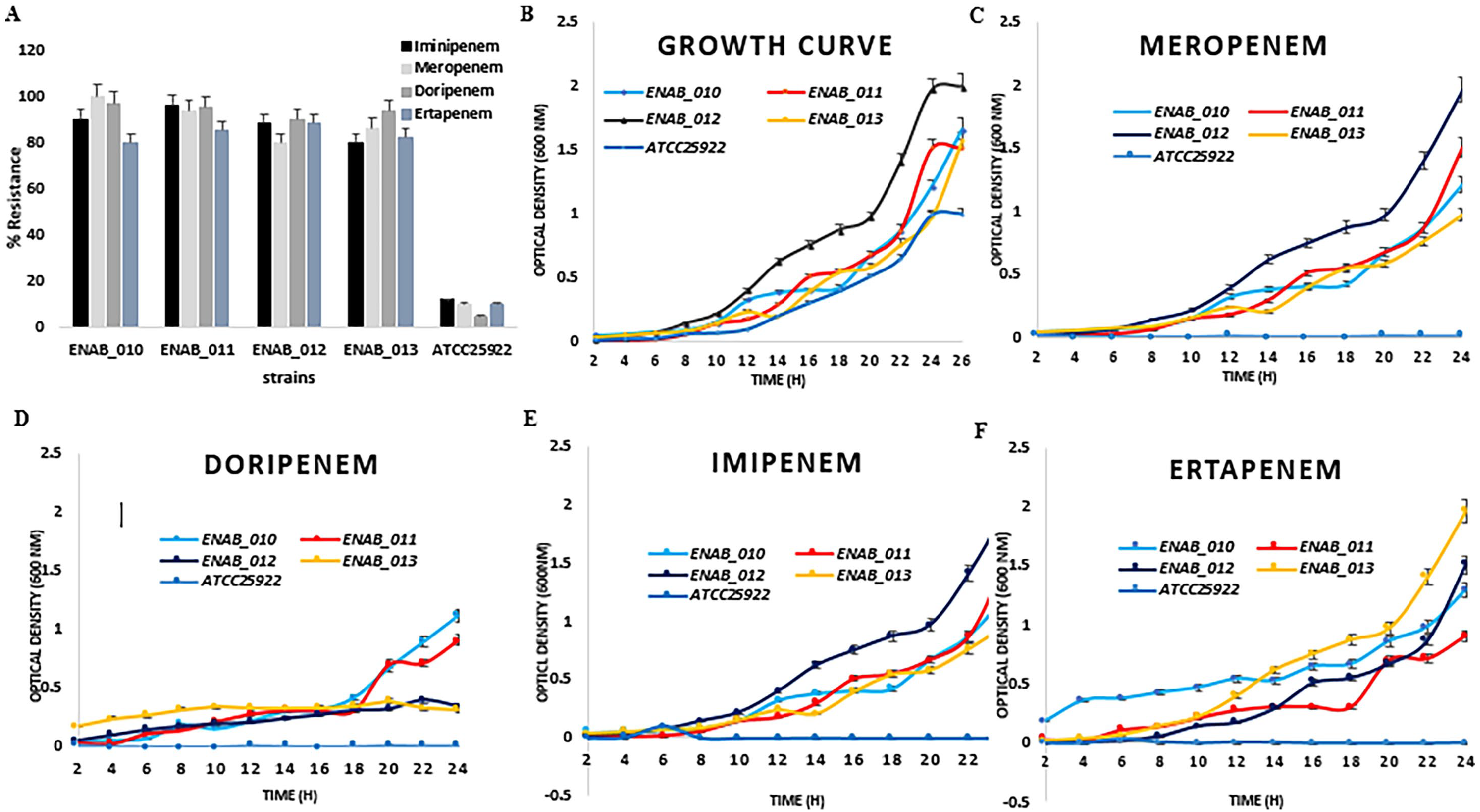
Antimicrobial Resistance Profiles, Bacterial Standard Growth Curve and Growth Rate in the presence of carbapenems relative to the ATCC 25922 E. cloacae control; (A) Percentage resistance of the bacterial strains to imipenem, meropenem, doripenem and ertapenem; (B) Standard growth curve of the bacteria with respect to time; (C-F) Increase in bacterial growth rate with time in the presence of the tested antibiotics. The error-bars indicate percentage resistance and increase optical density with time.
Calcium ions reduced growth profile of strains and resistance to carbapenems
Conditional growth stress of the strains with calcium ions indicated a reduction in the level of resistance of the antibiotics to carbapenems at the indicated time points. ENAB_010 level of resistance to meropenem and ertapenem reduced to 41% and 20% from the initial 100% and 80% respectively (Figure 2A). This rate of reduction in resistance was observed across the strains with 20% to 47% in the presence of calcium ions as compared to (80%-100%) without Ca2+ – an average reduction rate of almost 50%. Overall, the lowest reduction in the level of resistance was to doripenem across the strains, while ertapenem has the highest reduction rate. Relative to the controls (No Ca2+) (OD ⩾ 1.5 at 24 hours), none of ENAB_010, ENAB_011, ENAB_012 and ENAB_013 strains previously resistant to the antibiotics could survive the Ca-adjusted antibiotics (stock, 0.1 v/v) with an average OD of ⩽0.01 at 24 hours (Figure 2B-E). This might indicate that Ca2+ might serve as a good antibiotic potentiator against bacterial resistance to carbapenems.
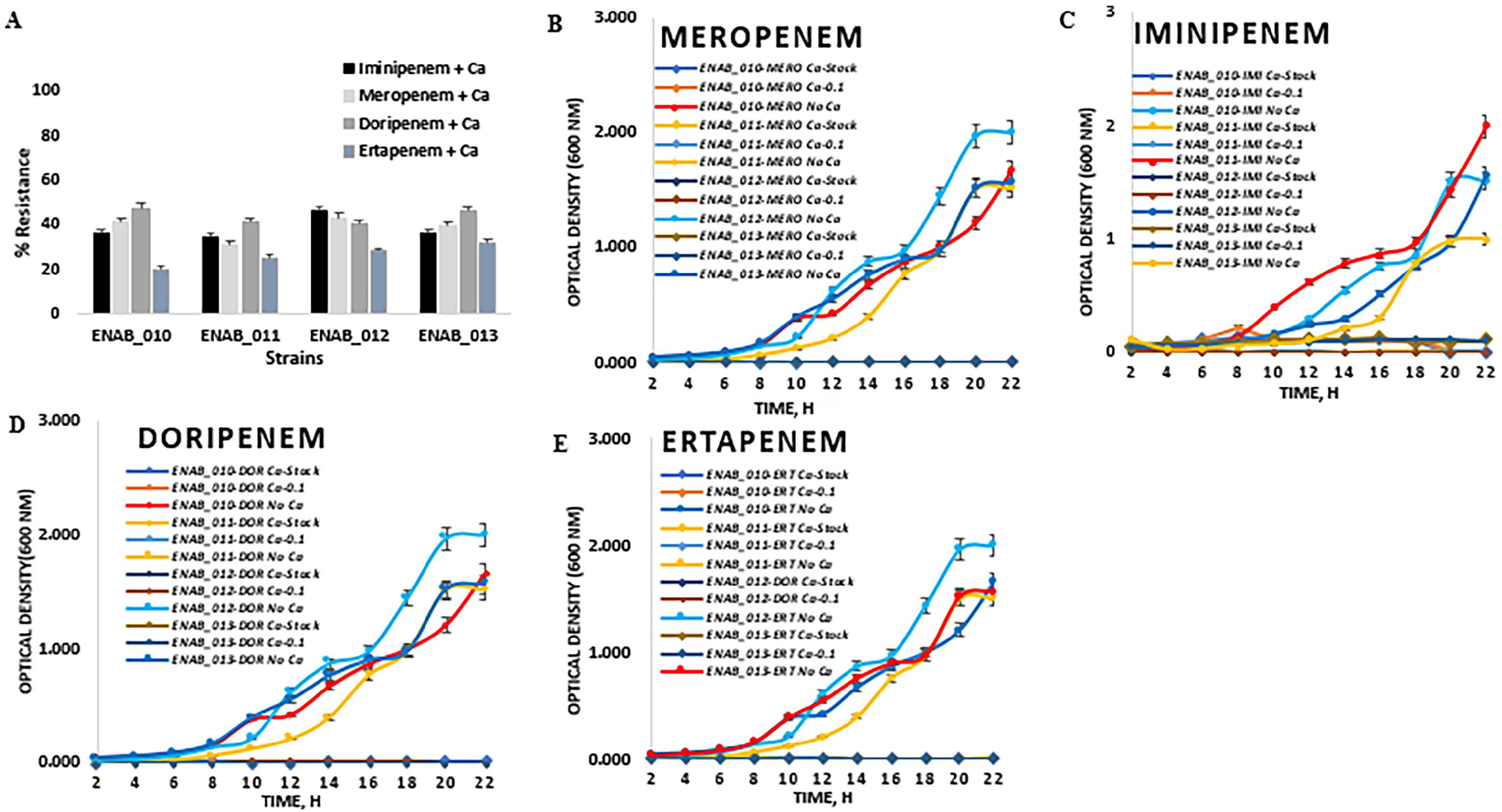
Antimicrobial Resistance in the presence of calcium ion; (A) Ca-adjusted carbapenem showing reduction in the level of resistance; (B-E) Reduction in the level of resistance at stock and 0.1 v/v concentration of calcium ions with respect to time; The error-bars indicate percentage resistance and increase optical density with time.
Time-kill evaluation showed decrease in resistance with time
The bacteria displayed a similar resistance and growth patterns in the presence of the antibiotics alone (Figure 3A-D). There was a gradual increase in bacterial growth in the presence of carbapenems after 2 hours with a steady increase across the time points. Bacterial concentration from 20 log10 CFU/mL to >40 log10 in 2 hours increased to more than 50 log10 in 22 to 24 hours in the presence of carbapenems. However, treatment with Ca2+ showed a general decrease in bacterial concentration with increase in incubation time (Figure 3A-D). In the presence of Ca2+ (stock and 0.1%), there was complete reduction in bacterial concentration between 10 and 18 hours of incubation. Addition of stock concentration of Ca2+ reduced bacteria population relatively faster as compared to 0.1% Ca2+ concentration, although with little difference. The reduction in concentration observed was below 5 log CFU/mL relative to a starting concentration of 20 log CFU/mL. Each of the carbapenem antibiotic was relatively effective with Ca2+ with notable reduction in bacterial population relative to the control (strains with carbapenem antibiotic alone).
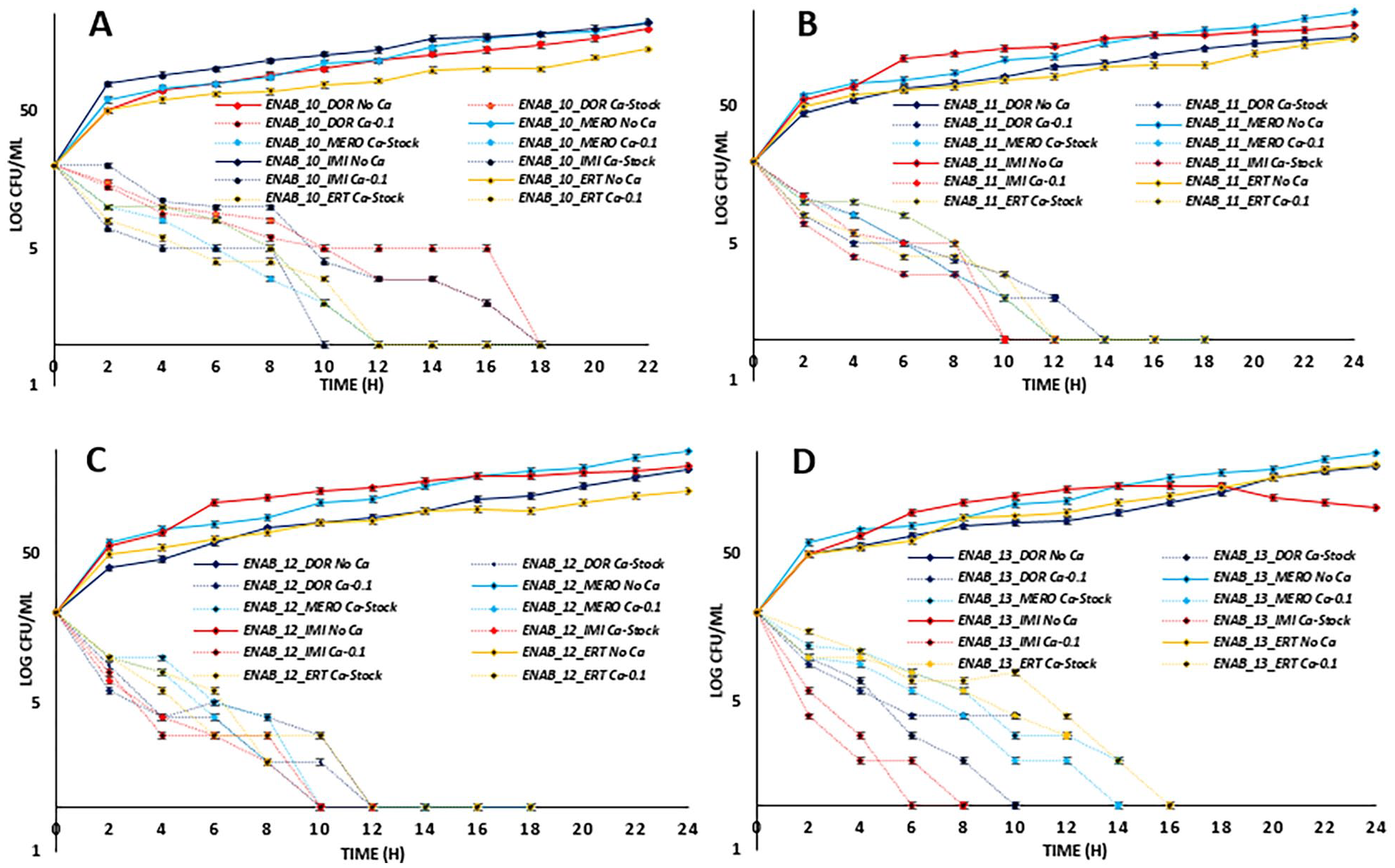
Time-kill curve of the resistant E. cloacae strains in the presence of calcium supplement; (A-D) ENAB_010, ENAB_11, ENAB_12 and ENAB_13 respectively showing a reduction in the level of resistance at stock and 0.1 v/v concentration of calcium ions with respect to time.
Discussion
Four carbapenem antibiotics were tested to determine E. cloacae antimicrobial susceptibility and resistance profiles. More than 80% level of resistance was observed across the strains to carbapenems, suggesting that the strains are potential high-risk bacteria as indicated by WHO in the Global Priority Pathogen list.1,12 This is alarming as these strains were isolated from hospital environments, which might endanger the patients, hospital visitors as well as the healthcare givers. Resistant Enterobacteriaceae such as carbapenem-resistant E. cloacae are described as threats in hospitalised individuals especially with general contact with fomites including medical equipment and invasive devices. 13 Similar studies have equally reported strains with high level of resistance to carbapenems as nosocomial pathogens.14,15 As these prioritised strains are resistant to multiple carbapenem last-resort antibiotics, it calls for quick intervention especially in ICU that house immunocompromised patients. Since some Gram-negative bacteria leverage Ca2+ to trigger some metabolic activities, 6 this study determined resistance profiles of E. cloacae in the presence of calcium.
The effects of calcium ion on carbapenem-resistant E. cloacae strains was tested to determine if resistance will be maintained or strains resistance level will be affected. Responses of the strains in the presence of Ca-adjusted carbapenems showed a rapid decline in growth rate and significant reduction in the level of resistance relative to media without Ca-supplement. There is possibility that the carbapenem-resistant abilities of the strains were compromised with contact with the supplemented calcium ion as the incubation time increases. Contrary to what has been described in previous modelled studies,11,16 extreme resistance to carbapenem dropped to almost half of the initial resistance level across the strains, which might signify gradual loss in its resistance mechanism and consequent metabolic activities. The time-kill curve further showed that bacterial activity is affected by divalent cations in a concentration-dependent manner, as there was rapid decrease in bacterial concentration with Ca2+ stock relative to 0.1% calcium supplement. Studies have also indicated Ca2+ specific activity on bacterial membranes resulting in reduced bacterial growth and multiplication. 17 In line with this, we posit that E. cloacae adapt to carbapenem exposure in the presence of calcium ions, thereby complementary to reduction in its resistance.
Conclusion
This study showed a possible means of reducing E. cloacae resistance to carbapenems, thereby mitigating AMR. Although, there was a considerable reduction in resistance level in the presence of calcium; however, combining 2 different carbapenem antibiotics and possible increase in concentration might have added to reduction in the level of resistance observed.
Footnotes
Acknowledgements
The authors appreciate Mosi Research Lab and AMR Research Group led by Dr. Abiola Isawumi at the Department of Biochemistry, Cell and Molecular Biology, and West African Centre for Cell Biology of Infectious Pathogens at the University of Ghana for supports.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AI conceptualized and designed the study. AI, MKA and EAA processed, analyzed and interpreted data. AI prepared the first draft of the manuscript. All the authors revised the draft for important intellectual content. LM critically reviewed the manuscript. All the authors approved the final draft of the manuscript for submission.
