Abstract
Background:
Otitis media (OM), also known as middle ear infection, is a clinically significant childhood disease. In sub-Saharan Africa, there is a paucity of contemporary reports on its bacterial aetiologies and antimicrobial resistance.
Aim:
To investigate the OM bacterial aetiologies and their antimicrobial resistance patterns among children visiting the Ear, Nose, and Throat clinics of 3 healthcare facilities in Accra, Ghana – Princess Marie Louise Children’s Hospital, 37 Military Hospital, and Mamprobi Hospital.
Methods:
This cross-sectional study involved 100 children below 13 years old with suppurative otitis media. Following standard bacteriological methods, sterile ear swabs were used to take middle ear discharges from the study participants for culture and antimicrobial susceptibility testing. A standard questionnaire was also used to collect data on socio-demographic and clinical characteristics.
Results:
The major OM bacterial aetiologies were Pseudomonas aeruginosa (38.5%), Klebsiella pneumoniae (19.8%), Proteus mirabilis (11.5%), and Staphylococcus aureus (10.4%). The majority of the bacteria demonstrated low to moderate resistance (0%-33.3%) to most of the antibiotics. Eight of the bacteria (4 each of Klebsiella pneumoniae and Escherichia coli) were extended-spectrum beta-lactamase (ESBL) producers; 6 ampicillinase (Amp C)-producing organisms (4 Citrobacter spp. and one each of Morganella morganii and Serratia marcescens) were also identified, and they showed high antibiotic resistance.
Conclusions:
The predominant OM aetiologies were Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus mirabilis, and Staphylococcus aureus, and they were generally susceptible to most of the antibiotics tested. Amikacin, cefepime, ciprofloxacin, and meropenem could be valuable in the empirical management of childhood OM.
Introduction
Otitis media (OM), also known as middle ear inflammation, is a range of diseases of the middle ear.1-3 It comprises acute otitis media, otitis media with effusion, and chronic suppurative otitis media (CSOM).1,2,4 It can also present as non-suppurative. 5 It easily occurs in young children due to the anatomy of their Eustachian tube (ET) – their ET is more flexible, horizontal, and shorter, allowing for easier entry of pathogens of nasopharyngeal origin into the middle ear.6-8 The ET protects the middle ear cavity through ventilation, by clearing middle ear fluid and stopping nasopharyngeal pathogens from entering the middle ear, thus serving as a natural barrier.1,2,6,9 The range of infective agents associated with OM include fungi, viruses, and more predominantly, bacteria.10-15 Key bacterial pathogens implicated in OM include Haemophilus influenzae, Moraxella catarrhalis, Escherichia coli, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus pneumoniae, and Streptococcus pyogenes.10-12,14-17
OM presents a significant public health challenge globally, causing children, who are disproportionately burdened with the disease, to frequently visit the paediatrician.18,19 It may occur as a single or a recurrent infection, and is the leading primary cause of antimicrobial agent prescription and incorrect use, as well as a reduced quality of life in children.1,2,18,20 It ranks second among the causes of hearing loss and fifth in the global burden of disease, affecting about 1.23 billion individuals globally, with the highest incidence occurring in South Asia and sub-Saharan Africa.21-26 Hearing loss may lead to delays in language, speech, and intellectual skills development, and these result in societal challenges later in life.1,2,27 Besides, OM, particularly CSOM, results in complications such as intracranial and extracranial problems, and accounts for a significant proportion of surgical procedures that are performed on children.1,6,20,28-30 In addition, an annual mortality of about 20 000 individuals has been attributed to complications with OM, with the highest death toll recorded in children less than 5 years old.4,25 The economic burden of the disease is enormous. For instance, in the USA, an estimated USD 3 to 5 billion was spent on treatment of OM, and in Australia, an economic burden ranging from AUD 100 to 400 million due to OM has been reported.19,31
One precipitating factor of OM is that its early diagnosis is a challenge in young children, as its signs and symptoms are not easily noticeable, consequently making the management of OM typically empirical.1,6,27,28 Besides the high potential for inappropriate antimicrobial therapy posed by empirical treatment, the frequent use of antibiotics in treating childhood OM makes children important reservoirs for multidrug resistant pathogens. This underscores the need to continuously monitor causative organisms of OM and their antimicrobial resistance patterns so as to contemporarily update OM treatment options available to clinicians, for an improved patient management and outcome. However, such surveillance studies are limited, particularly, in sub-Saharan Africa. In Ghana, there are just 2 reports on OM aetiologies and their antimicrobial resistance patterns. The first publication 32 dates back to more than 3 decades, and the second publication, 10 almost a decade. As the spectrum of OM aetiologies and their antibiotic resistance patterns are dynamic, the absence of current data on OM epidemiology is a major setback to OM management. To help fill these crucial knowledge gaps, this study aimed at determining the OM bacterial aetiologies and their antimicrobial resistance patterns among children visiting the Ear, Nose, and Throat clinics of 3 healthcare facilities in Accra, Ghana – Princess Marie Louise Children’s Hospital, 37 Military Hospital, and Mamprobi Hospital.
Methodology
Study sites
The study was conducted at the Ear, Nose and Throat (ENT) units of Princess Marie Louise Children’s Hospital (PMLH), 37 Military Hospital (37 MH), and Mamprobi Hospital (MH). PMLH, also known as the Children’s Hospital, is one of the few specialist children’s hospitals in Western Africa. It is a government hospital located at Derby Avenue, Accra. It has a 74-bed capacity, and is the second largest paediatric facility in Ghana. It has an ENT clinic that attends to about 25 cases of otitis media on each clinic day. The 37 MH is a specialist hospital located in Accra, on the main road between Kotoka International Airport and Central Accra. It is the largest military hospital in Ghana with an ENT clinic. Mamprobi Hospital is also a government hospital located within the Mamprobi Township in the Greater Accra Region. It has an ENT unit that attends to about 10 children with otitis media daily.
Study design and sampling
This was a cross-sectional study involving 100 children below 13 years of age with otitis media who were consecutively recruited at the ENT units of PMLH, 37 MH, and MH from September 2020 to March 2021. Written consent was obtained from the parents or guardians of younger children and older children. Diagnosis of OM was based on medical examinations done by ENT specialists, with the aid of an otoscope. As part of the medical examinations, interviews of parents, guardians, and older children were conducted to obtain history in relation to the middle ear infection. Children below the age of 13 years who had been diagnosed with otitis media were deemed eligible for the study. The exclusion criteria were presence of craniofacial abnormalities, Down syndrome, and being on antimicrobial therapy.
The minimum number of study participants was determined based on the prevalence of 5.8% 27 using the formula:
Where n = Minimum sample size
z = Z-score for 95% percentile (1.96)
p = Otitis media prevalence (5.8%)
m = Allowable error (0.05)
The minimum sample size was thus determined to be 84 participants.
Specimen and data collection
During the specimen collection, 70% alcohol was used to clean the external canal, and afterwards, sterile swab sticks were used by the ENT specialists to collect the middle ear discharge from the study participants. Each swab specimen obtained was kept in 1 ml of skim milk-tryptone-glucose-glycerin (STGG) medium and labelled with a unique bar code. These specimens were transported on ice packs within 3 hours to the research laboratory of the Department of Medical Microbiology, University of Ghana Medical School. Socio-demographic and clinical data for each child were collected using a standard questionnaire.
Specimen processing
On arrival at the laboratory, each specimen was vortexed for about a minute and stored at −80°C until needed. During microbiological analysis, the stored swab specimens were thawed and vortexed briefly, and then inoculated in about 5 ml of tryptic soy broth for enrichment. After overnight incubation at 37°C, the resultant inocula were cultured on chocolate, blood, mannitol salt (for Staphylococcus aureus identification), and MacConkey agar plates, using a sterile wire loop. The blood, chocolate, and mannitol salt agar plates were incubated at 37°C for 24 to 48 hours in 5% to 10% CO2 achievable by a candle jar for the purpose of isolating potential organisms such as Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. The MacConkey agar plates were incubated at 37°C for 24 hours under aerobic conditions.
The isolates were identified to the species level based on colonial morphology on culture plates, Gram staining, and biochemical tests. Catalase and coagulase tests were performed for Gram-positive cocci. Bile solubility and optochin tests were performed for Streptococcus pneumoniae identification. Aesculin bile agar, 6.5% sodium chloride (6.5% NaCl) broth, and pyruvate, were done to identify Enterococcus species and to differentiate between Enterococcus faecalis and Enterococcus faecium. Indole, oxidase, triple sugar iron, citrate, motility, and urease tests were also done to identify Gram-negative rods. Analytical profile index (API) 20E was further used to speciate Gram-negative rods for which the conventional methods could not provide identification.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was performed by the Kirby-Bauer disc diffusion technique. This was carried out by emulsifying pure colonies of isolates in sterile normal saline using a sterile inoculating loop, and the bacterial suspension was adjusted to 0.5 MacFarland standard. A sterile cotton swab was dipped into the suspension, pressed against the sides of the tube, and then used to inoculate a Mueller Hinton agar plate before the application of single antibiotic discs, which were placed not less than 24 mm apart. The application of the antibiotic discs was done using sterile forceps. Incubation of the agar plates was done aerobically at 37°C for 16 to 18 hours. Mueller Hinton agar supplemented with 5% sheep blood was used for Streptococcus pneumoniae antibiotic susceptibility testing, with incubation done in 5% carbon dioxide at 37°C for 20 to 24 hours. Antibiotic selection was based on the Clinical and Laboratory Standards Institute (CLSI) 33 guidelines, with the following antibiotics: ampicillin (10 µg), cefuroxime (30 µg), amoxicillin-clavulanic (30 µg), ceftriaxone (30 µg), penicillin (10 µg), amikacin (30 µg), ciprofloxacin (5 µg), gentamicin (10 µg), ceftazidime (30 µg), cefepime (30 µg), piperacillin-tazobactam (110 µg), trimethoprim-sulfamethoxazole (25 µg), meropenem (10 µg), tetracycline (30 µg), clindamycin (2 µg), erythromycin (15 µg), and vancomycin (30 µg).
The zones of inhibition around each of the antibiotic discs was interpreted according to the CLSI 33 guidelines.
Data processing
Data obtained were entered into Microsoft Excel 2019, and analysis was done with Statistical Products and Services Solutions (SPSS), version 25. Descriptive statistics, such as counts, frequencies, and percentages were computed for the various variables and presented as tables.
Ethical considerations
Ethical clearance was granted by the Ethical and Protocol Review Committee of the College of Health Sciences, University of Ghana, with protocol identification number ‘CHS-Et/M.7 – 5.3/2020-2021’.
Results
Sociodemographic and clinical information of the study participants
A total of 100 children with otitis media were recruited for the study. Their age distribution ranged from 3 weeks to 12 years old. The majority (74%) of them were below 5 years of age, and a greater proportion of them were males (59%). Acute suppurative otitis media (ASOM) was the most commonly diagnosed infection (68%), and CSOM was recorded among 32% of the study participants. Otorrhea was a major symptom, with a frequency of 100%. Over 80% had otalgia, and a lower frequency of the children (3%) had other medical conditions (Table 1).
Demographic characteristics and clinical information of study participants.
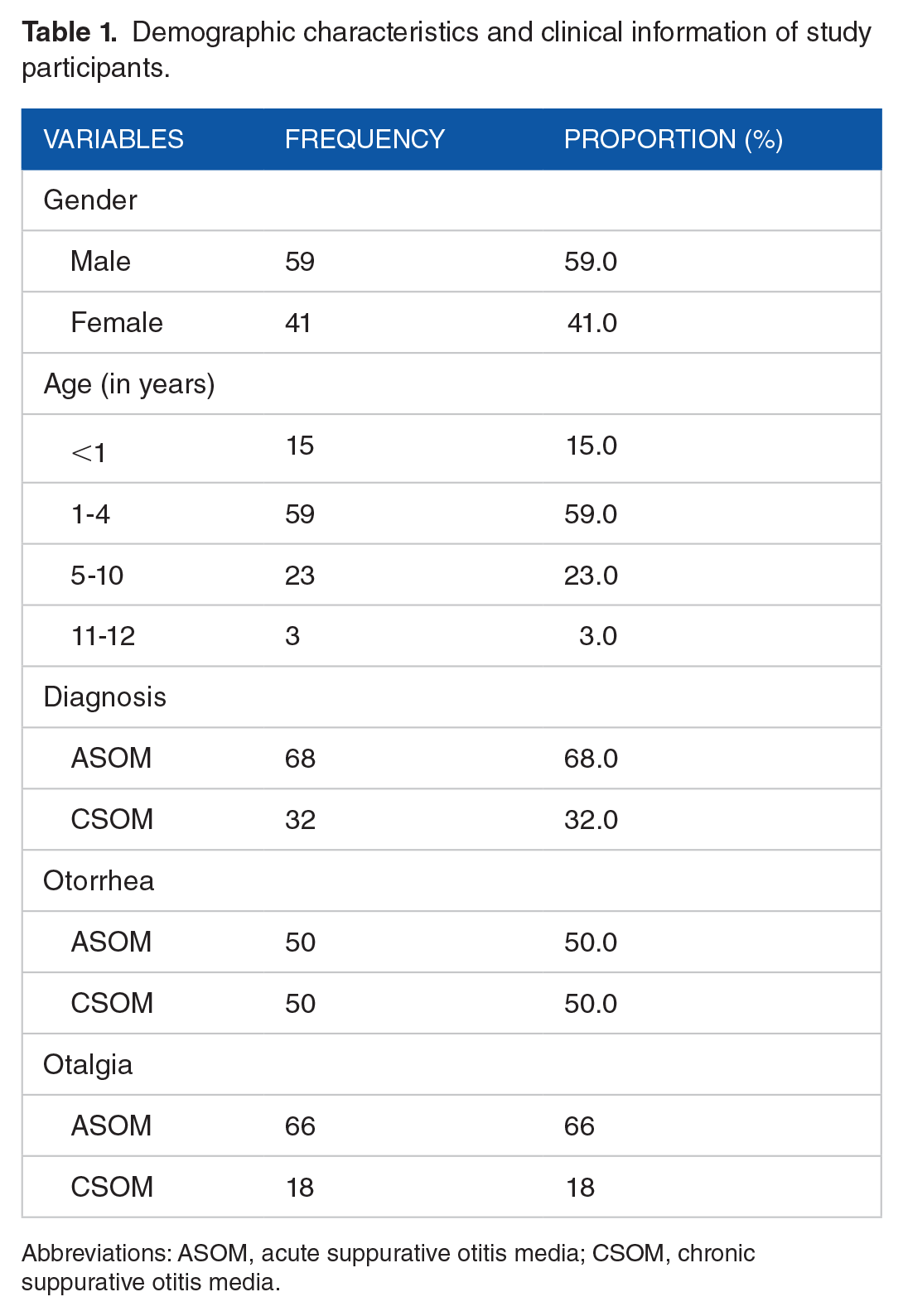
Abbreviations: ASOM, acute suppurative otitis media; CSOM, chronic suppurative otitis media.
Distribution of the bacterial isolates
Out of the 100 children sampled in this study, 97% (n = 97) gave positive cultures, whiles 3% (n = 3) recorded no growth on culture. In total, 112 isolates were obtained from the positive cultures, 12.4% of which occurred as mixed growth; 2% were fungal agents (Candida species), and the remaining 98% were bacteria (among which Gram-negatives dominated – 86.5%). The top 4 bacterial aetiologies of OM were Pseudomonas aeruginosa (38.5%, n = 37), Klebsiella pneumoniae (19.8%, n = 19), Proteus mirabilis (11.5%, n = 11), and Staphylococcus aureus (10.4%, n = 10), whereas the least encountered were Morganella morganii, Serratia marcescens, and Streptococcus pneumoniae, each of which occurred at a rate of 1% (n = 1). The observations regarding the distribution of OM aetiologies, when considered as a composite, were generally similar to those of both ASOM and CSOM (Table 2).
Distribution of the bacterial aetiologies of otitis media.
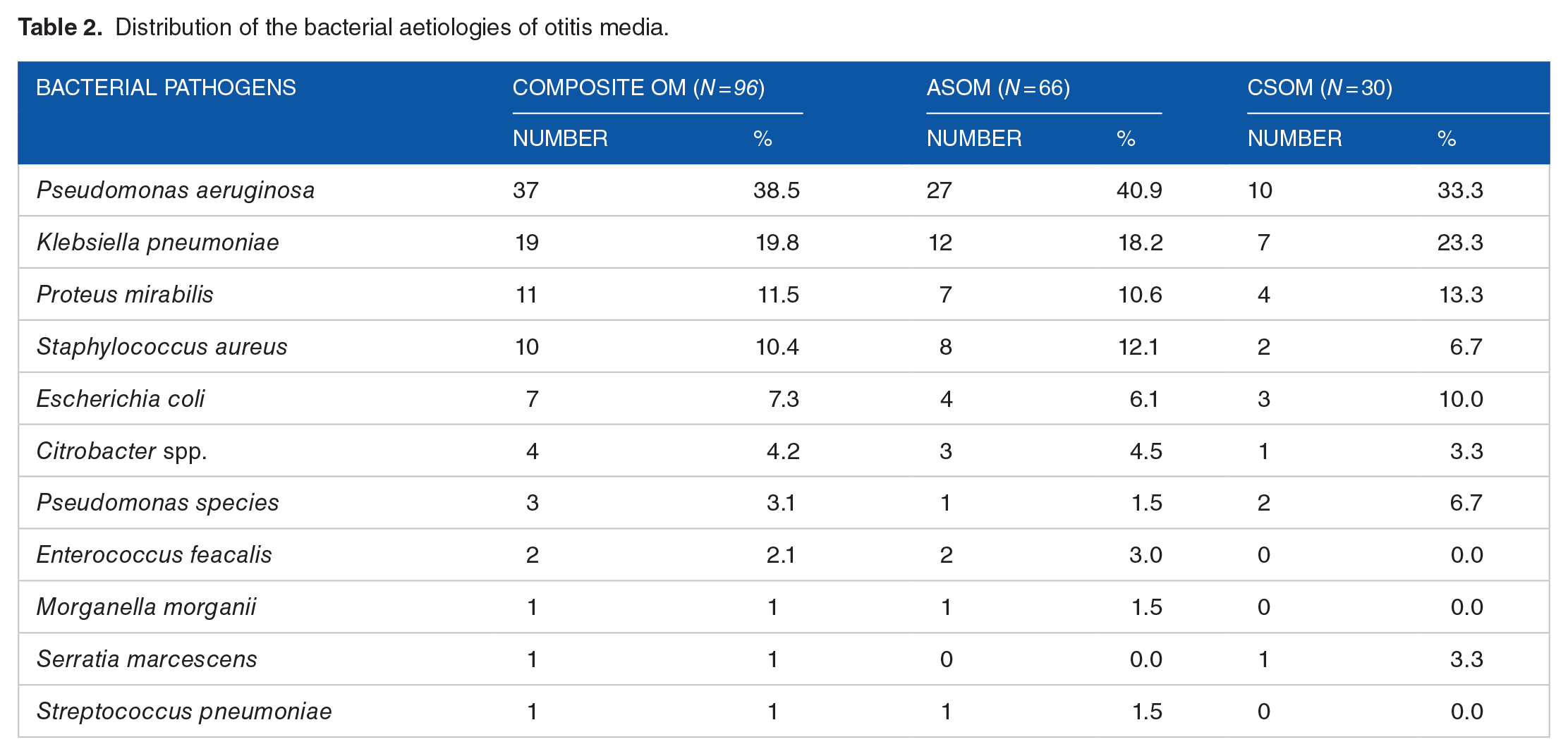
Antimicrobial resistance patterns of the bacterial isolates
The majority of the bacterial pathogens demonstrated low to moderate resistance (0%-33.3%) to most of the antibiotics. Pseudomonas aeruginosa was susceptible to all the antibiotics tested, except gentamicin, to which it recorded a resistance rate of 13.5%. Escherichia coli, Morganella morganii, Serratia marcescens and Citrobacter spp. were highly resistant (mostly 100%) to several of the antibiotics tested against them. Klebsiella pneumoniae was highly resistant (100%) to ampicillin, but demonstrated markedly lower rates against the other antibiotics. Similarly, the single Streptococcus pneumoniae isolated was resistant to trimethoprim-sulfamethoxazole and tetracycline, but not any of cefepime, meropenem, ampicillin, cefuroxime, amoxicillin-clavulanate, clindamycin, erythromycin, ceftriaxone and vancomycin. Also, 8 of the bacterial pathogens were extended-spectrum beta-lactamase (ESBL) producers; this trait was equally shared between Klebsiella pneumoniae (50%) and Escherichia coli (50%). Furthermore, 6 ampicillinase (Amp C)-producing organisms, which included Citrobacter spp., Morganella morganii and Serratia marcescens, were also identified, and they showed high antibiotic resistance. The antimicrobial resistance patterns of the bacterial pathogens are shown in Table 3.
Antimicrobial resistance patterns of the bacterial pathogens of otitis media.
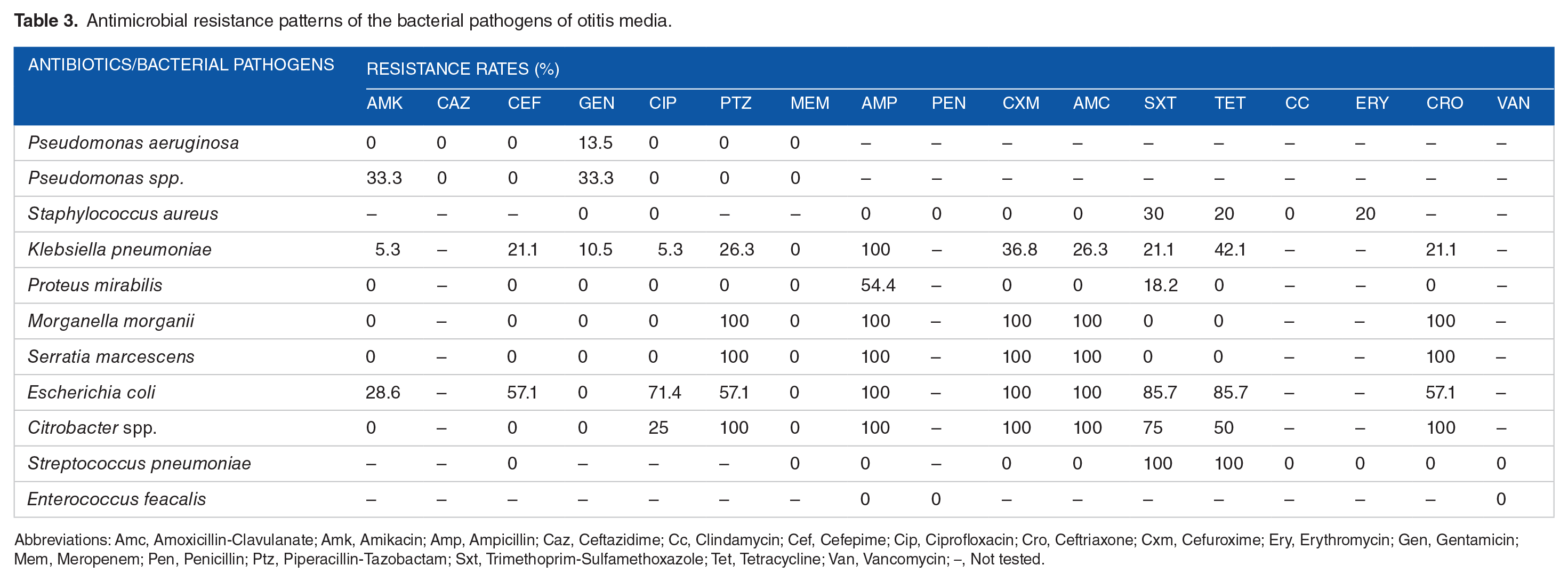
Abbreviations: Amc, Amoxicillin-Clavulanate; Amk, Amikacin; Amp, Ampicillin; Caz, Ceftazidime; Cc, Clindamycin; Cef, Cefepime; Cip, Ciprofloxacin; Cro, Ceftriaxone; Cxm, Cefuroxime; Ery, Erythromycin; Gen, Gentamicin; Mem, Meropenem; Pen, Penicillin; Ptz, Piperacillin-Tazobactam; Sxt, Trimethoprim-Sulfamethoxazole; Tet, Tetracycline; Van, Vancomycin; –, Not tested.
Discussion
This study evaluated the bacterial aetiologies of childhood OM at 3 healthcare facilities in Accra, Ghana, and antibiotic resistance among them. It provides current insights that could prove useful in the clinical management of OM in the country and elsewhere. ASOM was a more common diagnosis (68%) compared to CSOM (32%) among the study participants, an observation supported by other OM reports.1,6,19,25,34 Moreover, the majority of the OM cases (74%) was observed among children under 5 years of age, similar to what have been reported in Ghana (65.1%) 10 and elsewhere (71.7%-84.2%)3,8,35 for childhood OM. This observation could either be due to the immaturity of their immune system, which predisposes them to frequent upper respiratory tract infections, their relatively shorter Eustachian tube that allows for easier entry of nasopharyngeal pathogens into the middle ear, or both.6,36 Similarly, a greater proportion of males (59%) was affected, which is comparable to the proportions of OM cases attributed to males in several studies (51.3%-62.3%),8,34,35,37 albeit in contrast with others (48%-48.5%).3,38
Most of the OM cases in the current study (97%) yielded positive cultures, as was the case for several studies conducted previously (75%-98.2%).8,34,35,38-40 Although the study focussed on bacterial aetiologies, fungal agents – Candida species exclusively – were also recovered, as did Appiah-Korang et al’s 10 study. Moreover, Gram-negative organisms dominated the bacterial aetiologies (86.5%), as is the case in the OM literature (64%-98.6%).5,34,35,37,41 Pseudomonas aeruginosa was the predominantly identified bacterial aetiology of OM, followed by Klebsiella pneumoniae, Proteus mirabilis, Staphylococcus aureus and Escherichia coli. In contrast, in the near 4-decade OM study conducted in Ghana, 32 P. aeruginosa ranked second, with Streptococcus pyogenes, S. aureus and Proteus spp. ranking first, third, and sixth, respectively. In Appiah-Korang et al’s 10 study, however, P. aeruginosa was the principal OM aetiology, followed by S. aureus and Proteus spp., both of which ranked among the top 4 OM aetiologies in the current study. Similarly, in neighbouring Nigeria, P. aeruginosa has been identified as the cardinal OM aetiology.34,36,37 Contrary to what have been reported elsewhere4,16,42-45, Streptococcus pneumoniae, Haemophilus influenzae and Moraxella catarrhalis were rarely encountered in the current study. Especially for S. pneumoniae and H. influenzae, their rare occurrence could be a reflection of the success of decades-long vaccination programmes targeting these organisms in the country. Their rarity may also be due to variations in climatic conditions and geographical locations, a hypothesis that could dually underlie the absence of Moraxella catarrhalis among the spectrum of OM aetiologies observed in the current study.
That P. aeruginosa was susceptible to all of amikacin, ceftazidime, cefepime, ciprofloxacin, piperacillin-tazobactam, meropenem, ampicillin, penicillin, cefuroxime, amoxicillin-clavulanate, trimethoprim-sulfamethoxazole, tetracycline, clindamycin, erythromycin, ceftriaxone, and vancomycin suggests that OM cases that are diagnosed clinically could empirically be managed with all of these antibiotics. Amikacin, cefepime, ciprofloxacin, and meropenem could especially be preferable in this regard, when the resistance patterns are considered as a composite. That notwithstanding, the choice of any of these antibiotics needs to be guided by their stipulation as first-, second-, third- or fourth-line antibiotics. Particularly in the case of meropenem and other carbapenems, it would be of a high clinical relevance to limit their use, in order not to potentiate the prevailing emergence and spread of carbapenem resistance in Ghana and elsewhere.46-50 It is also noted that the therapeutic value of gentamicin in the empirical management of OM in Ghana cannot be ruled out, as its activity is of a broad-spectrum nature, and low resistance rates were recorded against it by P. aeruginosa and the other key OM aetiologies identified in the current study. Nonetheless, given its potential to be ototoxic, its use needs to be restricted to short therapeutic regimens.51,52
Also, that several of the Gram-negative isolates, namely E. coli, M. morganii, S. marcescens, Citrobacter spp., and K. pneumoniae were highly resistant to several of the antibiotics tested against them necessitates a review of OM treatment guidelines in Ghana, especially, as Gram-negative bacteria are major OM aetiologies in the country. The high-level resistance recorded by K. pneumoniae and E. coli could be because some of the isolates were ESBL producers. With regard to Morganella morganii, Citrobacter spp. and Serratia marcescens, their high-level resistance could be due to their being ampicillinase producers. Generally, the antimicrobial resistance patterns observed in the current study are consistent with what were reported by Appiah-Korang et al 10 and others.3,8,34,39 Hence the resistance patterns observed in the current study, particularly, with regard to ampicillin, amoxicillin-clavulanate and cefuroxime, may transcend the possession of ESBLs or AmpCs, and additionally reflect a consequence of the frequent use of these antibiotics in the management of OM and other infections.
Conclusions
It is concluded that the predominant OM aetiologies were Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus mirabilis, and Staphylococcus aureus. The pathogens were generally susceptible to most of the antibiotics tested. Amikacin, cefepime, ciprofloxacin, and meropenem could be valuable in the empirical management of childhood OM, and gentamicin could serve as a substitute if the therapeutic regimen involves shorter periods. Conversely, as the aetiological agents demonstrated high resistance towards ampicillin, cefuroxime, and amoxicillin-clavulanate, it is recommended that their use in managing childhood otitis media in the country is discontinued. It is also important to continuously monitor the range of OM pathogens and their antibiotic resistance patterns (including the molecular basis of the resistance), preferably, using population-based studies. These studies could be designed to address the limitation of the current study – the non-inclusion of anaerobic cultures, which did not enable a prediction of the extent to which anaerobic bacteria are involved in the spectrum of OM aetiologies in Ghana, as well as the non-screening of the OM aetiologies for resistance against antitubercular drugs.
Footnotes
Acknowledgements
The authors appreciate the contributions of the study participants, as well as the staff of the ENT clincs of Princess Marie Louise Children’s Hospital, 37 Military Hospital, and Mamprobi Hospital, as well as the technical staff of the Department of Medical Microbiology, University of Ghana Medical School.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: NTKDD. Design: NTKDD, VB, FCNK, ESD. Sampling: VB, NTKDD, FPD. Validation: NTKDD, VB, FCNK, ESD. Data curation: NTKDD, FCNK, VB. Formal analysis: NTKDD, FCNK. Investigation: NTKDD, VB, FPD, FCNK, ESD. Resources: NTKDD, VB, FPD, ESD. Visualization: NTKDD, ESD, VB. Software: NTKDD, VB. Supervision: NTKDD, ESD. Project administration: NTKDD, ESD, VB. Writing—original draft preparation: NTKDD, FCNK, VB. Writing—review and editing: NTKDD, FCNK, VB, FPD, ESD. All authors have read and approved the final version of the manuscript.

