Abstract
In addition to the ongoing global problem of healthcare-acquired infections, the COVID-19 pandemic continues to pose a serious threat to the health of the global population. This unprecedented pandemic situation has reinforced the need for the development of technologies that can curb the transmission of viruses among human beings and help to control the infection. Existing disinfection techniques using either ultraviolet light or harsh chemicals pose safety risks and are not suitable for use in the presence of humans. Thus, the need for a safe and effective disinfection technique that can be used in the presence of humans to control viral transmission is evident. A technique that can continuously disinfect air and surfaces in indoor environments, where the chances of viral transmission are high, can be an indispensable tool to fight such a pandemic. The Airlens Minus Corona (AMC) device provided by Persapien Innovations has been developed to achieve this goal. In this study, the antiviral functionality and biocompatibility of AMC were evaluated. Activated water mist (AWM) generated from this device was tested in vitro and in vivo for its toxicity to cell lines and in animal model. The AWM was found to be non-cytotoxic to L-929 cell lines and had no sign of clinical toxicity in an animal model (rabbit). This device was further used to inactivate animal viruses and bacteriophages. The AWM was found to be effective in the complete inactivation of influenza A H1N1 virus within 5 minutes of direct treatment. This device was also found to be effective in inactivating >90% of bacteriophage particles.
Keywords
Introduction
The continuous threat of pandemics and epidemics is a major global health concern. The last 20 years have been particularly important due to the emergence of many viral pathogens, including the SARS coronavirus in 2002, Influenza A H5N1 in 2003, Middle East respiratory syndrome (MERS) coronavirus (MERS CoV) in 2012, Ebola virus in 2014-16, Nipha virus in various times and the SARS CoV-2 in 2019.1,2 The world has already withstood several influenza pandemics, including the 1918 pandemic flu and the 2009 pandemic caused by influenza A H1N1 virus.3,4 Moreover, within only the last 10 years, another pandemic struck the world, the COVID-19 pandemic, which originated in Wuhan, China, in December 2019, spread to225 countries globally, and is still ongoing.5,6 As of March 17, 2022, SARS CoV-2, the causative agent of COVID-19, has infected more than 460 million people worldwide which caused 6 050 018 deaths (https://covid19.who.int). In the context of the current situation, disinfection is one of the most important aspects to keep the world safe.7,8 Currently, UV irradiation and chemical disinfection are the key methods of disinfection that are widely used in workplaces, shops, shopping malls, laboratories, hospitals, and major public places affected by SARS CoV-2. However, UV irradiation and chemicals used as disinfectants are not safe for human health. 9 A safe disinfection method should not have any health hazards. In this study, a water-based disinfection technique, namely, activated water mist (AWM), was developed, and it is safe for human beings, effective against viruses and practically applicable.
Water is a potential candidate to develop a safe disinfectant because water is one of the safest compounds available to us. Historically, efforts have been made to convert water into an effective disinfectant. Electrolysis of saline water was used for the generation of “superoxidized water” or “electrolyzed water” containing hypochlorous acid, which showed antimicrobial properties. 10 Adequate Superoxidized water is recognized as a new type of disinfectant by the Centers for Disease Control and Prevention (CDC), USA. 11 The use of saline and electricity to generate disinfectants is inexpensive and environmental friendly. It is safe for humans, and a number of superoxidized water-based products are being used for wound disinfection applications. 12 Although superoxidized water has been proven to be an effective and safe disinfectant, it is mostly used in the liquid state, which limits its application to surface disinfection. However, air disinfection methods are being sought to reduce viral transmission during the COVID-19 pandemic. In another method, containing reactive oxygen species (ROS) were studied as an effective, chemical-free antimicrobial platform for the inactivation of microbes on surfaces or in air.13,14 ROS were generated within nano droplets of water by electrospraying under a high electric field. No harmful effects were found when using these ROS-containing water droplets against animals. To enhance microbial inactivation by this method, electrolysis of water was explored, which helped to increase the amount of ROS within water nanodroplets.14,15 Nanodroplets of water were generated and sprayed by leveraging the Rayleigh effect, and this method was shown to inactivate microbes in a small chamber. This technique established that water droplets can be converted to safe and effective disinfectants by subjecting them to a high electric field. Although this technique was shown to be effective in a small chamber, applying it for the disinfection of large indoor spaces, such as rooms, offices, and hospitals, is challenging due to the inherent design of the electrospray method using capillaries or similar systems with low output. Although it has been established that water can be converted into safe and effective disinfectants, the application of previous techniques is limited to localized or small-scale applications. For example, adequate superoxidized water has been used as a liquid disinfectant, while ROS-containing water droplets have been shown to be effective in small chambers at the laboratory scale. Moreover, the effectiveness of water-based disinfectants against virus particles has not been studied extensively, which is an hourly need. Thus, there is an urgent need to develop a safe technique that can disinfect indoor spaces to prevent the spread of virus.
A new instrument, the Airlens Minus Corona (AMC), has been developed that generates an activated water mist (AWM) using water and electricity. The AWM is a safe and effective disinfectant and highly efficient in inactivating virus particles. The instrument can be designed to produce large AWM quantities as per the requirement of an indoor space. Moreover, mist allows for the easy dispersion of activated water particles. This study shows that virus particles can be deactivated within indoor spaces. Thus, this technology can be used for continuous air and surface disinfection of indoor spaces, such as hospitals and workplaces. Moreover, the AWM biocompatibility has been studied here based on the prescribed guidelines.
Methods
Airlens Minus Corona device and AWM
A small amount of common salt (15 mg) was added to 1 L of pure water, mixed well and poured into the tank of the Airlens Minus Corona device (Figure 1). The device was switched on and allowed to run for 10 to 15 minutes to allow for the generation and outflow of mist in the form of AWM through a hose attached to the device. Samples tested in this study were kept in sterile Petri dishes in the designated chamber. Samples were then allowed to be exposed to the AWM. Viral samples and cell lines treated with charged/ionized water sprayed through the AMC device for different time durations (5, 15, and 30 minutes) were tested to determine their viability. As a control, samples were treated with plain water mist without any treatment. Charged/ionized water spray or activated water mist are referred to as AWM in the manuscript.
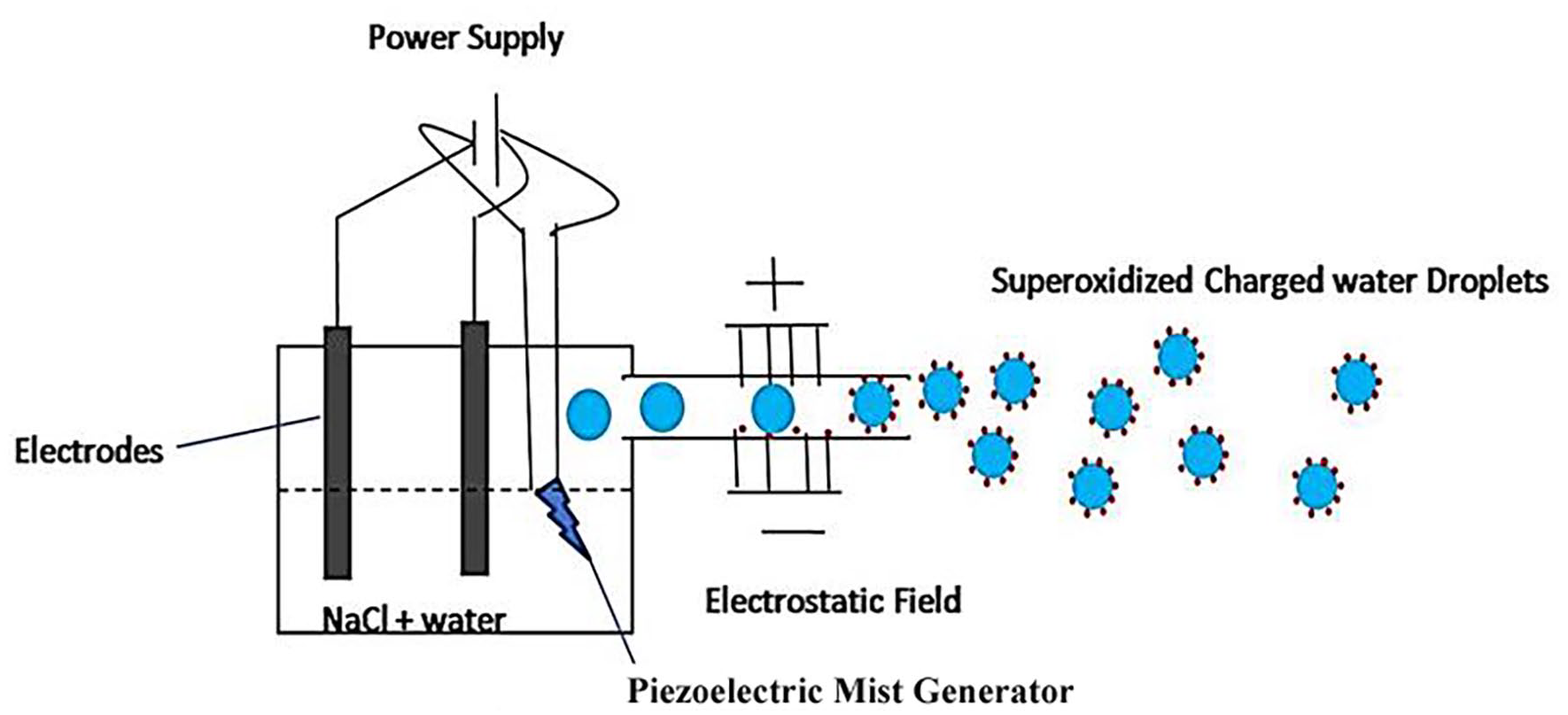
Principle of Airlens Minus Corona technique. AMC device contains a tank containing electrodes and water with small amount of common salt dissolved in it to allow electricity to flow from one electrode to other. A piezoelectric module is used for breaking liquid water in micro-droplets. These water droplets (blue circles) are passed through a high voltage electrostatic field to further charge/ionize water droplets. The mist is allowed to spread in the air through a air circulation motor.
Cell lines
The mouse fibroblast L929, Madin-Darby Canine Kidney (MDCK), and Human embryonic kidney 293T (HEK 293T) cell lines were procured from the National Centre for Cell Science, Pune, India, and maintained in Dulbecco’s modified Eagle’s tissue culture medium (Invitrogen Life Technologies, Carlsbad, CA, USA) containing 10% fetal calf serum, 100 units/ml penicillin, and 100 units/ml streptomycin in tissue culture flasks (Corning, USA) at 37°C in a CO2 incubator. Cell lines grown to monolayer cultures were maintained in Dulbecco’s modified Eagle’s medium (Gibco, Thermo Fisher Scientific, USA) supplemented with heat-inactivated 10% fetal bovine serum (Gibco, Thermo Fisher Scientific, USA) and 2 mM L-glutamine (Sigma-Aldrich, India), 100 units/ml penicillin and 100 μg/ml streptomycin and maintained at 37°C in an atmosphere of 5% CO2 incubator at 95% air humidified. The cultivated cells were regularly controlled for cell growth and the absence of mycoplasmas.
Viruses
In brief, 1 µg of each of the 8 plasmids containing the cDNA of the virus A/WSN/33 (H1N1) [pHW181-PB2, pHW182-PB1, pHW183-PA, pHW184-HA, pHW185-NP, pHW186-NA, pHW187-M, and pHW188-NS] was transfected into MDCK-293T co-cultured cells, and the virus was rescued at 72 hours post transfection. The transfectant virus was grown and titrated in MDCK cells for further experiments. 16
Purification of bacteriophages
Phage D-10 lysate was concentrated by precipitation in an ultracentrifuge at 30 000 rpm for 2 hours at 4°C, and the pellet was suspended to a final volume of 1 ml. This concentrated phage was purified by a standard cesium chloride density gradient centrifugation procedure to obtain purified phages. 18
Virus infection
For propagation and titration, monolayers of MDCK cells at a concentration of 3 × 106 cells/ml were infected with influenza A/WSN/33 at a multiplicity of infection (MOI) of 0.1. After 1 hour, the inoculum was removed and the cells were washed twice with phosphate-buffered saline (PBS) and supplemented with growth media. AWM-treated influenza A/WSN/33 was infected at different times post treatment. Mock-infected cells at each time point served as controls.
Preparation of 96-well plates for the cell cytotoxicity assay
L929 cells were counted using the Trypan blue (Sigma-Aldrich, India) exclusion method quantified by a TC20 TM automated cell counter (Bio-Rad Laboratories, MLEX, Spain). The cells were plated in 96-well flat bottom plates using a multichannel pipette. Each 96-well plate was partitioned into columns as follows: (1) culture media only, that is, no cells; (2) cells incubated in culture medium alone; (3) test control cells incubated in culture medium containing a cytotoxic ISO recommended product, that is, doxorubicin hydrochloride USP testing specifications (Sigma-Aldrich, India); and (4) test cell cultures incubated in culture media with the suspected toxic AWM samples from given test material. All of the conditions were tested in triplicate wells, and each experiment was repeated 3 times.
Animals used in acute dermal irritation tests and their environmental condition and food
Three female New Zealand white rabbits aged 10 to 12 weeks with body weights of 1.60 kg ± 200 g were used for the acute dermal irritation test, and all 3 rabbits were identified by cage tag and corresponding color body marking. The healthy rabbits selected for study were acclimatized to standard laboratory conditions for 1 week in the experimental room under veterinary examination. After acclimatization and veterinary examination, 3 female rabbits were randomly selected.
Animals were kept in air conditioned rooms with 10 to 15 air changes per hour, a temperature of 20°C to 30°C, a relative humidity of 40% to 60% and an illumination cycle set to 12 hours artificial fluorescent light and 12 hours dark. Animals were housed individually in stainless steel cages provided with stainless steel mesh bottles and facilities for food and water bottles. Pelleted feed was supplied by Pranav Agro Industries Ltd., B7/6 Ramesh Nagar, Delhi, and community tap water was passed through an “Aqua Guard on line water filter” and kept in glass bottles. Food and water was provided ad libitum.
Preparation of animals
The animals were prepared 24 hours prior to application of the test product. The furs from the dorsal area of the trunks were removed with electric clippers to expose an area measuring approximately 6 cm2 of the body surface area of the animal. Care was taken such that abrasion penetrated the stratum corneum only and not the dermis.
Application of test compound
Then, 0.5 ml of the test compound (AWM condensed to water) was applied to a small area (approximately 6 cm2) of intact skin. Each site of application was covered with impervious dressing, which was secured in position with adhesive tape. The treated animals were then housed individually, and a plastic collar was placed around their necks to prevent access by the animal to the patch and resultant ingestion of the test product. After patch removal, the dressing and unabsorbed test product were removed and the site of application was cleaned with lukewarm water. Subsequently, the site of application was observed for skin reaction if any. The intact skin site of each animal was observed for signs of erythema and edema, and the responses were scored following Draize’s method at 1, 24, 48, and 72 hours after application. In addition to the observation of irritation, all local toxic effects, such as defatting of the skin, and any systemic adverse effects were fully described and recorded.
Cytotoxicity test
L929 cell cultures were washed 3 times with the corresponding culture media without 10% FBS. For each washing, 200 μl of culture medium was added to each well, and then the plate was inverted with slight vigorous shaking to discard the culture medium from each well. After the 3 washes, charged/ionized water sprayed through the AMC device was applied to the cell cultures in each well of the corresponding negative group and test group columns. The plates were transferred to an incubator under standard conditions for 30 or 60 minutes. After these exposures, the plates were washed as previously described and then incubated for 24 and 72 hours in culture medium (200 μl per well). At 1 and 3 hours after initiation of the incubation period, each culture was stained with Trypan blue and imaged by phase contrast microscopy (Nikon Eclipse TS100, Nikon España, and Barcelona, Spain). These times were selected for the early detection of cytotoxicity, which was manifested by the loss of cell membrane integrity and penetration of Trypan blue. At the end of each 24- and 72-hour incubation period, the viability of each cell culture was measured using the 3-(4,5-dimethylthiazol-2-Yl)−2,5-diphenyltetrazolium bromide (MTT) cytotoxicity assay. After 72 hours of incubation (the end of the incubation period (t = 72 hours)), the cells were studied under an inverted microscope at 100× magnification. Thereafter, the biological end point was determined by staining with crystal violet supernatant medium. Furthermore, the cells were washed twice with freshly prepared PBS, and MTT solution was added to the plate at a final concentration of 5 mg/ml and incubated for 4 hours in the dark at 37°C. After incubation, the medium was removed and the cells were suspended in DMSO (200 μl). The absorbance of the formazan reduction product was calculated by measuring the optical density at 570 nm using a reference wavelength of 650 nm in an ELISA reader (Bio-Rad, Hercules, CA, USA).
Hemagglutinin assay
Hemagglutinin (HA) assay was used to determine the titer and assess the viability of the influenza viruses before and after treatment following the standard methodology. 19 In brief, 4 ml of fresh chicken blood was taken and washed 3 times with PBS by centrifugation at 800 rpm. PBS was carefully aspirated, which left behind the precipitated blood cells. One milliliter of blood cells was diluted in 9 ml of PBS, and this 10% blood solution was used as a stock, which can be stored at 4°C for up to 1 week. From the stock, 1% of blood was used for the HA assay. This assay was performed in a 96-well round bottom plate, in which 50 µl of PBS was added to each well. Then 50 µl of viral soup was added to the respective first wells and mixed by pipetting and then transferred to the next well, thus generating a 2-fold serial dilution in the successive wells. Subsequently, 50 µl of 1% blood was added to all the wells and kept for 30 to 60 minutes. A cloudy appearance in the well indicated the presence of virus, and button formation indicated the absence of virus.
Plaque assay of vibriophage
Plaque assay was performed to assess the titer of vibriophage D10 following the methods described by Chakrabarti et al. 18 In brief, serial fold phage dilutions mixed with the standard propagating strain were added to molten soft agar (0.8%) and poured onto a nutrient agar plate. After the agar solidified, the plates were incubated at 37°C. The appearance of plaques was monitored, and plaque counts were taken after overnight incubation.
Treatment of vibriophage D-10 with AWM
High-titer pure phage D-10 was serially diluted and treated with the AWM. The interaction of phages with the AWM was studied at different concentrations of bacteriophages. Small drops (approximately 10 µl) of phage dilutions were spotted in sterile Petri plates, and the drops in 3 different Petri plates were exposed to the AWM for 5, 15, and 30 minutes. After the treatment, the drops were accumulated and assayed to determine the effect of the AWM on vibriophage D-10. As a control, phage D-10 was exposed to plain water mist without any treatment.
Treatment of influenza A virus with AWM
Influenza A virus A/WSN/33 with an HA titer of 210 was taken from the stock and used for the experiment. Small drops of virus were spotted in 35 mm tissue culture dishes, and these 3 dishes were placed on the platform of the AMC device. The spots of the virus in the dishes were treated with the AWM for 5, 15, and 30 minutes. As a control, influenza A/WSN/33 virus was spotted and exposed to plain water mist without any treatment. After the treatment, the dishes were removed from the device, spots accumulated, and the treated and control virus samples were assayed for viral titer by HA assay as mentioned above. To determine the viability of the treated virus samples, respective samples were used to infect MDCK cell lines as per the standard methodology. After 24 hours of infection, the cytopathic effect (CPE) was examined under a microscope. Soup from all the respective experimental infections was collected at 36 hours post infection and assayed for the presence of influenza virus by HA assay.
Results and Discussion
Biocompatibility study
Skin reaction
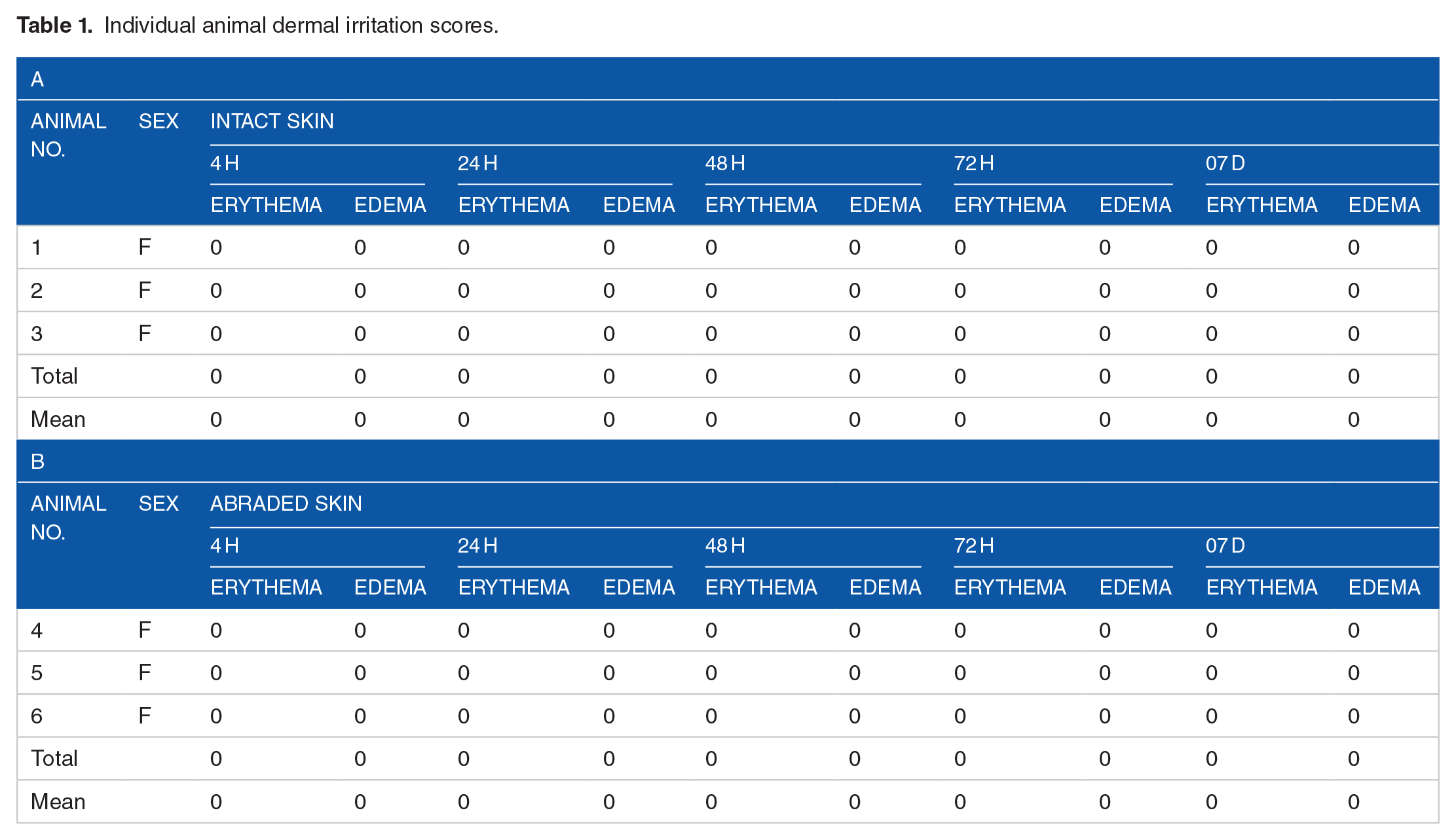
The results obtained from the present study reveal that the test sample (AWM) applied to shaven back skin of female rabbits in the amount of 0.5 ml produced no edema or erythema in intact or abraded skin after 24 hours of application. This result indicates that the charged/ionized water mist produced by the Airlens Minus Corona system is a nonirritant. These responses were graded according to Draize’s scoring method, as shown in Table 1, where a score of 0.00 means nonirritant.
Individual animal dermal irritation scores.
Clinical signs
The test sample (AWM) applied on the shaven back skin of rabbits at a dose level of 0.5 ml did not produce any clinical signs of toxicity throughout the examination period of 7 days, as described in Table 2.
Clinical signs in animals treated with AWM.

Abbreviation: N, no clinical sign.
Cytotoxic effects on L929 cell lines
L929 cells treated with the test sample (AWM) and incubated for 72 hours did not show any signs of cytotoxicity, as observed and interpreted by 3 independent observers. Discrete inter cytoplasmic granules were observed, no cell lysis was observed, and no reduction in cell growth was observed. The cells looked very similar to the untreated group, as shown in Figure 2. Our inference reports a grade 0 reactivity for the test material or AWM. As described in Tables 3 and 4, the test material was sprayed through an AMC device, and the AWM was found to be non-cytotoxic to the L-929 cell line.
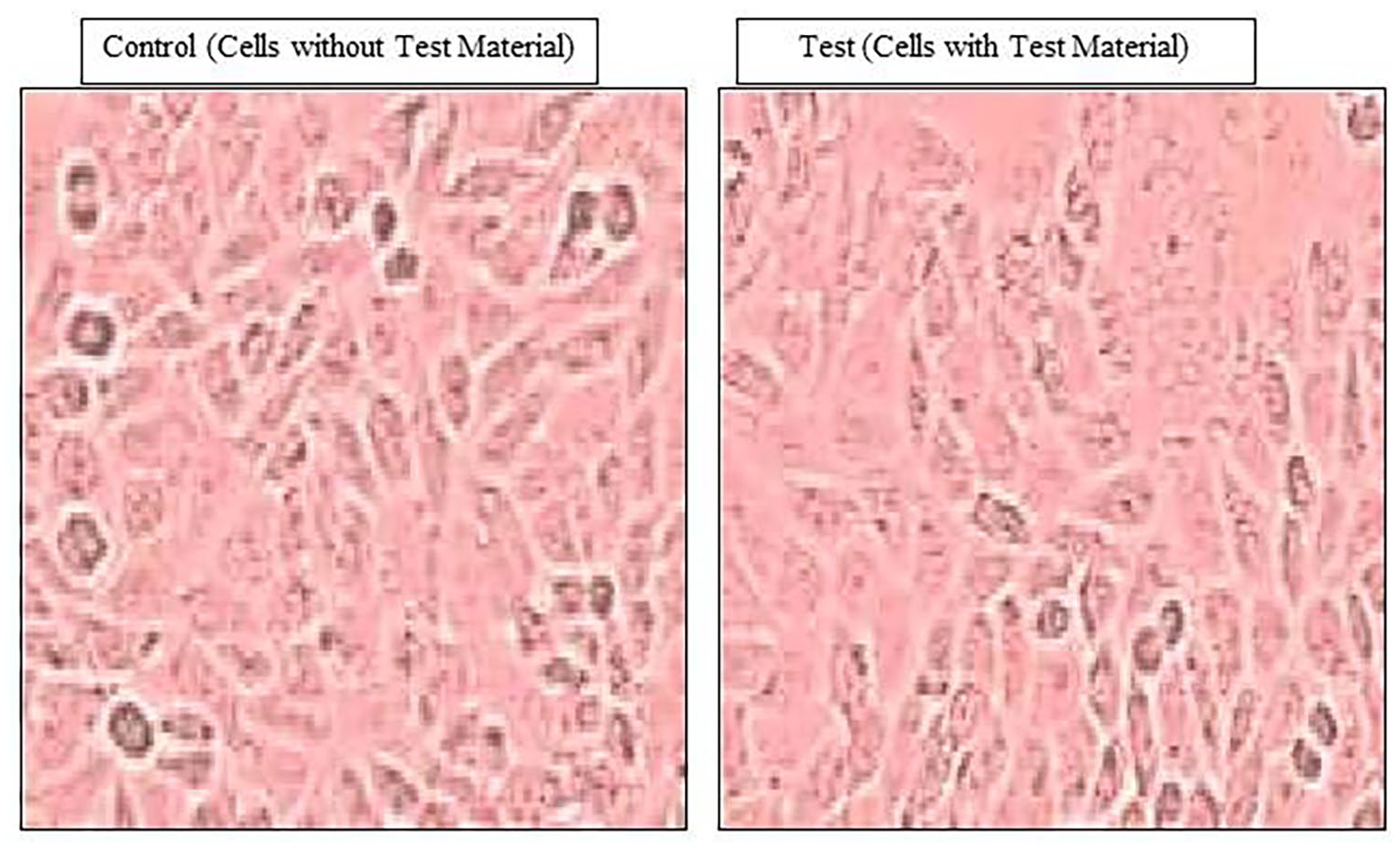
Representative microscopical images of cell viability in control and test material (AWM) treated mouse fibroblast (L929) cell lines.
Growth inhibition reactivity grades for direct contact test.

Cell viability test.
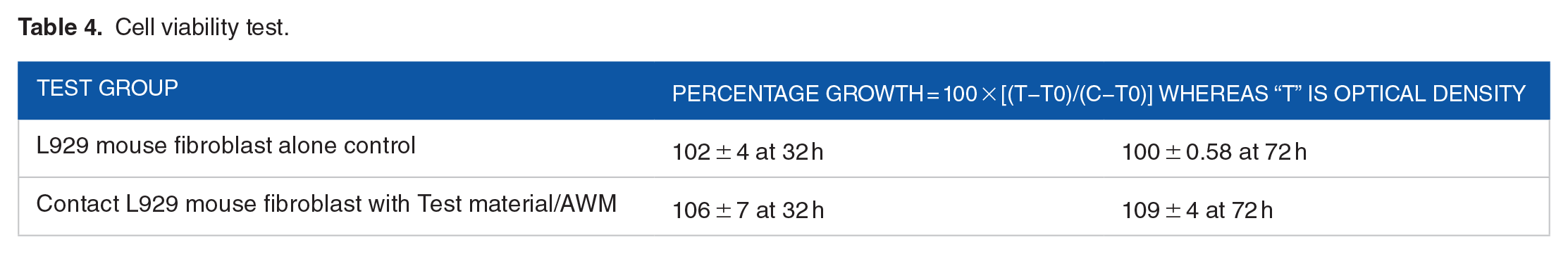
Viral inactivation study: Effect of the AWM on vibrio phage
The AWM was directly applied to vibriophage D-10 to determine the effect of the AWM on bacteriophage particles. The AWM was found to be effective in inactivating the vibriophage. Figure 3 shows the effect of the AWM on different concentrations (3 × 106 pfu/ml and 3 × 105 pfu/ml) of vibriophage D-10. The bacteriophage samples exposed to the AWM were assayed for the presence of phages by plaque assay. The results indicated almost 70% inactivation of vibriophages after treatment for 15 minutes, followed by >90% inactivation of vibriophages at 30 minutes post exposure. Exposure of phage D-10 to the AWM beyond 30 minutes did not show any further inactivation (Figure 3).
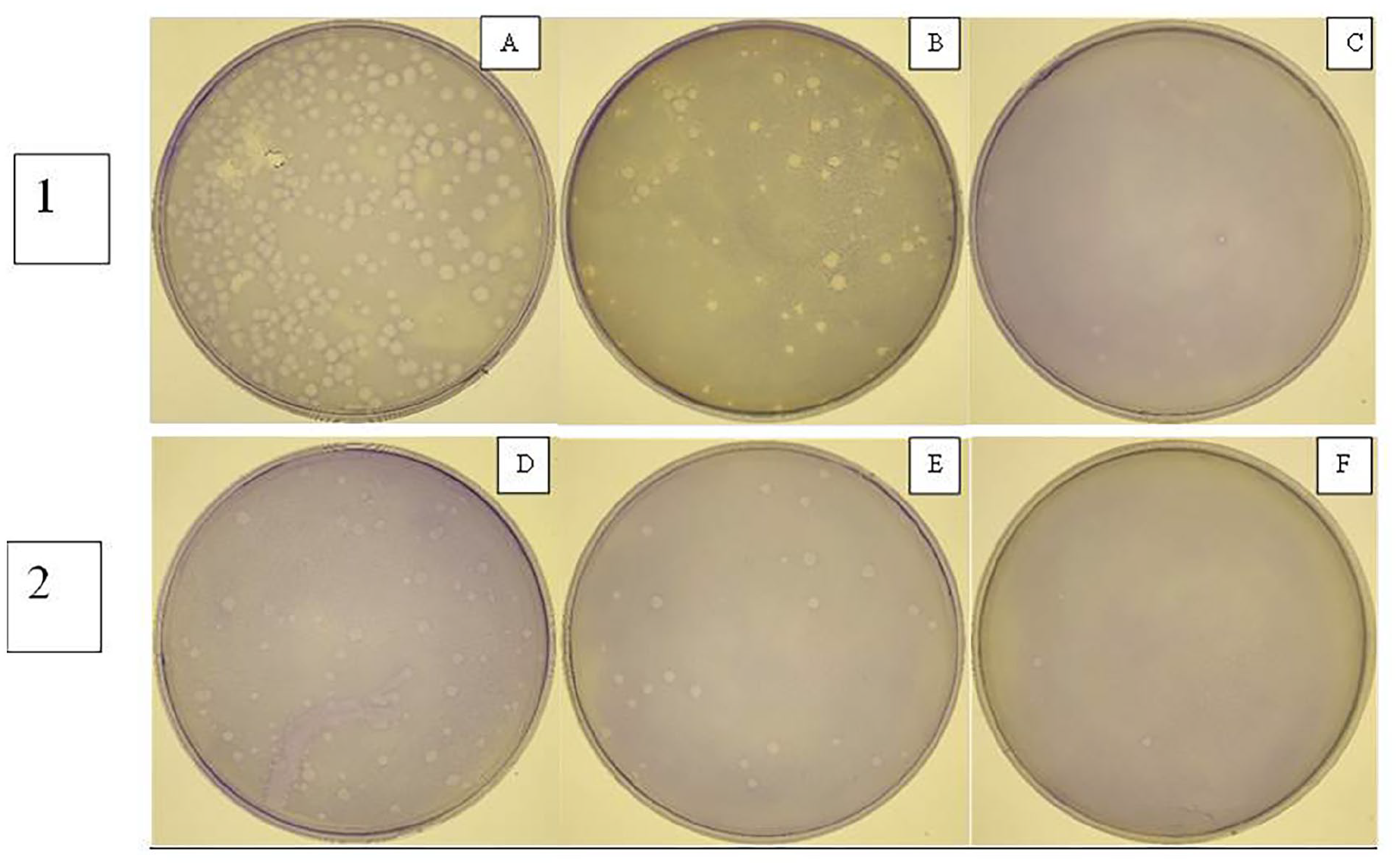
Effect of AWM on bacteriophages. Bacteriophage D-10 was serially diluted and exposed to AWM. Panel 1: Plate A shows number of plaques in control treated phages, plate B shows number of plaques at 15 minutes post exposure and plate C shows number of plaques at 30 minutes post exposure. (Stock phage was diluted to 3 × 106 pfu/ml for this experiment). Panel 2: Plate D shows number of plaques in control treated phages, plate E shows number of plaques at 15 minutes post exposure and plate F shows number of plaques at 30 minutes post exposure. (Stock phage was diluted to 3 × 105 pfu/ml for this experiment).
Effect of the AWM on influenza viruses
Total inactivation of the influenza virus was observed after treatment with the AWM as determined by the hemagglutination (HA) assay. Panel 1 of Figure 4 shows the effect of exposure of influenza A H1N1 (A/WSN/33) virus to the AWM. After exposure of the virus for 5, 15, and 30 minutes to AWM, HA assay was performed, and the results were analyzed with respect to the control virus, which was exposed to plain water mist without any treatment. In the HA assay, the control virus showed no inactivation, but exposure of influenza virus to the AWM for 5 minutes caused almost complete inactivation of the influenza virus, resulting in button formation in the HA assay. For further confirmation of the HA assay results, the AWM-treated and control-treated virus samples were used to infect MDCK cell lines, and the cells were observed for the appearance of CPE at different time points post infection. No CPE was observed in the AWM-treated virus samples, although infection was obvious with the control-treated virus, which was evident from the appearance of considerable CPE (Figure 5).

HA assay of H1N1 influenza virus after exposure to AWM. Panel 1: Influenza A H1N1 was exposed to AWM and HA assay was performed at for different time post exposure. Row A, Control untreated stock influenza virus; Row B, Control treated influenza virus: Row C, AWM treated influenza virus for 15 minutes; Row D, AWM treated influenza virus for 30 minutes. Panel 2: 15 minutes exposed viruses were infected in MDCK cell lines and at 36 hours post infection soup was collected to evaluate infectivity of the AWM exposed virus by HA assay. Untreated stock virus was also infected as a control. Row E, Control untreated influenza virus; Row F, Control treated influenza virus: Row G, AWM treated influenza virus for 15 minutes; Row H, AWM treated influenza virus for 30 minutes.
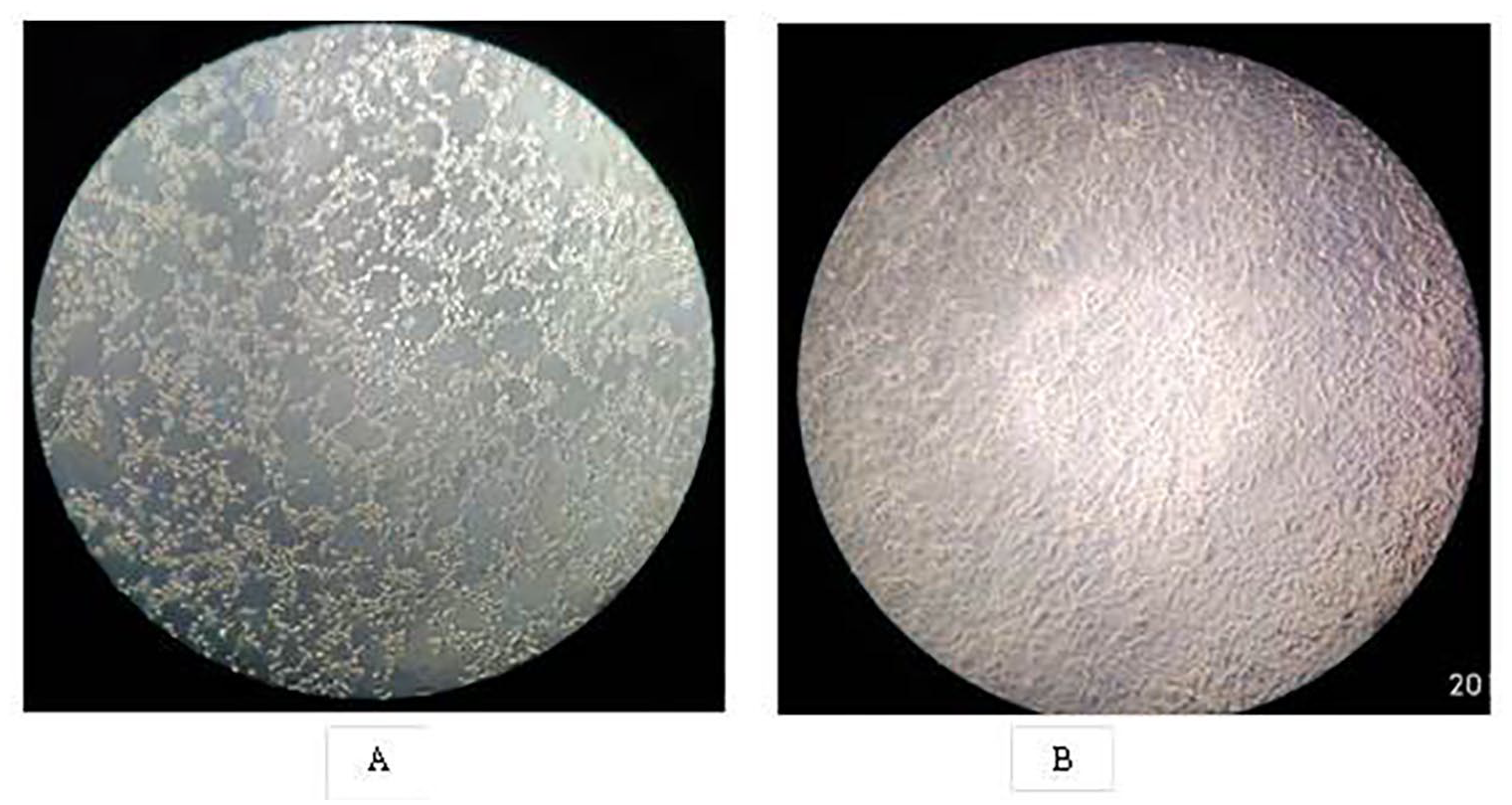
Cytopathic effect in influenza virus infected MDCK cell lines. Influenza A H1N1 virus exposed to AWM was tested for infectivity in MDCK cell line. (A) Control influenza virus infection. (B) Cytopathic effect of AWM treated virus.
To check the titer of influenza virus in the cell lines which was infected with the AWM-treated and plain water treated mist viruses, HA assay was performed on the virus-infected experimental and control soup. Control virus showed HA titer of 210, although the AWM-treated soup showed absence of any virus, indicating no infection with the AWM-treated samples (Figure 4 Panel 2). This experiment showed that treatment of influenza virus with the AWM, inactivated influenza A virus particles within 5 minutes of treatment.
Almost the same results were obtained for the viral inactivation study performed in a 100 cubic foot chamber. Small drops of Influenza A virus in tissue culture plates was exposed in a 100 cubic foot chamber pretreated with the AWM for 30 minutes. In the 100 cubic feet chamber experiments, 30 minutes was required for the complete inactivation of the influenza virus compared with 5 minutes after direct exposure via the dispersion of mist in the chamber.
Discussion
The COVID-19 pandemic has led to extreme fear worldwide because of the significant health risks to patients, healthcare workers and communities. The degree of impact of this unprecedented viral attack on human health is unpredictable. Thus, it is important to limit the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or any other viruses in the future. Corona virus is highly transmissible and can spread through air as well as by surface contamination. To curb the spread of coronavirus from one human host to another, there is an urgent need to develop a safe and effective disinfection technique. Ideally, such a technique should keep air and surfaces disinfected and should be safe for application in the presence of humans. The development of safe disinfection techniques can reduce viral transmission in indoor environments, such as hospitals, offices, and schools.
Under the pretext of the ongoing pandemic, the AMC system has been developed as a new, safe and effective disinfection method that can inactivate viruses within a given indoor environment. It can be designed to disinfect large spaces and keep them continuously disinfected by using water and electricity. Both of these basic components are easily available and do not depend on logistic challenges posed by inconvenient conditions like lockdowns. Moreover, this technology is inexpensive and safe for environment.
DI water with a small amount of dissolved common salt was electrolyzed to produce water containing ROS. Electrolysis of water has been explored in the literature to increase the amount of ROS in water. 15 In this study, a small amount of common salt was added to facilitate the electrolysis process and to make the process practically scalable. The electrolyzed water is then converted into micro droplets of water using ultrasonic waves. Water droplets are mixed with air to generate water mist, which passes through a high voltage (1-20 kV) electric field. These charged droplets passing through a high voltage electrostatic field can acquire a charge due to the effect of the electric field. The accumulation of charge by liquid droplets by the application of a strong electric field has been well studied in the field of electrospraying. 20 In this process, a strong electric field is applied to a capillary, and the liquid coming out of the capillary accumulates a charge at the liquid air interface. In this study, water droplets mixed in air are passed through a strong electric field to acquire a charge. Under a high voltage electric field, the acquisition of charge causes the formation of ROS within the water droplets, as previously studied.13,14 Here, the AWM has been found to inactivate viruses. Thus, a water-based disinfection technique was developed in this study that can be practically applied to large indoor spaces.
This technique is apparently harmless because it does not use harsh chemicals. The biocompatibility of this technique has been studied to prove this point. Moreover, no harmful substances were produced. Ozone can be produced by passing air through a high voltage electric field.21,22 Ozone is harmful to human health 23 and may compromise the safety of this instrument. Thus, the production of ozone was noted in this study. The 8-hour average concentration of ozone was found to be less than 5 ppb, which is well within the prescribed limit.21,22 The effectiveness of this technique to combat viruses was proven in our study. We have demonstrated that this method can inactivate influenza viruses and even inactivate bacteriophages. The ability of bacteriophages to survive under unfavorable conditions is highly diversified, with phage particles capable of surviving in nature for a longer period of time, even in host-free conditions.24-26 Therefore, a technique that is efficient in inactivating bacteriophage particles can inactivate other types of viruses with high efficiency.
The measurement of ROS within water droplets or electrolyzed water was not performed in this study, and ROS generation was referenced from previous works available in the literature. The antimicrobial activity and mechanism of action of ROS have been well demonstrated,12,27-29 and these species are major components within water droplets subjected to a electric field. The goal of this study was to demonstrate that the AMC instrument is safe and effective for the inactivation of viruses and can be applied for the disinfection of large spaces. This instrument can also be useful for combating the COVID-19 pandemic by curbing the transmission of coronavirus. Further studies will be carried out to characterize the produced AWM. Ions may transfer from air to water droplets. 30 However, water condensed from the AWM has shown similar efficacy in the inactivation of influenza virus as the AWM, thus demonstrating that the water has been converted to a disinfectant that inactivates viruses.
The newly developed technique and AMC device have potential antiviral effects by inactivating considerable amounts of virus particles. Based on the encouraging results obtained by the AWM on influenza virus inactivation, this device may be used as a disinfectant tool in the current scenario of the COVID-19 pandemic.
Footnotes
Acknowledgements
The authors are thankful to Dr Shashi Ranjan, Director-R&D of Persapien Innovations, New Delhi for providing the instrument, Airlens Minus Corona, to run the experiments in the laboratory. The authors are also thankful to Mr Debayan Saha, Director-Technology of Persapien Innovations, New Delhi for providing the technical details of the instrument.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by intramural institutional support of ICMR-NICED.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
A.K.C. conceived the idea and supervised the entire work. D.N.T. and S.B. performed the experiments. A.K.C., D.N.T., S.B. and S.D. analyzed the dataset. All authors contributed in preparation of the manuscript.
Statistics
All the experiments shown here were done in triplicate, including animal experiments and experiments with Influenza virus (HA assay) and vibriophages. Based on the inhibition of vibriophage after AWM treatment at different time point one way anova was performed using GraphPad prism (version 5, San Diego, CA, USA). From the above procedure a P value of .0001 was obtained at 5% level of significance which is less than P value .05 (P < .05) and is significant.
Ethics Approval
The animal experiments were performed under the requirements of the OECD Guideline No. 404. Acute Dermal Irritation/corrosion, Section-4: Health Effects (Adopted: 28th July, 2015) in compliance with the principles of current Good Laboratory Practices. (G.L.P.) at Committee for the Purpose of Control and Supervision of Experiments on Animals (
Consent for Publication
All authors read and approved the manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
