Abstract
Background:
Chronic hepatitis B (CHB) infection and nonalcoholic fatty liver disease (NAFLD) are liver diseases which may lead to hepatocellular carcinoma (HCC) formation. Both disease entities have been attributed independently to increase risk of HCC development. While concomitant hepatic steatosis in patients with CHB are becoming more frequent in view of increasing NAFLD prevalence, there is no conclusive evidence linking presence of hepatic steatosis and increased HCC risk in patients with CHB infection. This study explores the association of hepatic steatosis among CHB-infected individuals in HCC development.
Methods:
This is a retrospective study on a cohort of patients with CHB who underwent liver biopsy between January 2000 and December 2014. They were stratified according to presence and severity of histologically proven hepatic steatosis and subsequently followed up to evaluate the association between hepatic steatosis and HCC development.
Results:
Among 289 patients with a median follow-up of 111.1 months, hepatic steatosis was present in 185 patients (64.0%). In all, 27 patients developed HCC on follow-up and 21 of them had hepatic steatosis. Univariate Cox analysis showed that age (hazard ratio [HR] = 1.08, 95% CI = 1.042-1.12), type 2 diabetes mellitus (T2DM) (HR = 4.00, 95% CI = 1.622-9.863), and Ishak score (HR = 1.221, 95% CI = 1.014-1.472) were associated with HCC development, whereas multivariate Cox analysis demonstrated that age and T2DM (HR = 2.69, 95% CI = 1.072-6.759) were significant risk factors for development of HCC.
Conclusions:
Concurrent hepatic steatosis in patients with CHB infection is not a risk factor for hepatocellular carcinoma formation.
Introduction
Hepatitis B is estimated to have infected more than 240 million people worldwide.1-3 The seroprevalence of hepatitis B in Singapore is 3.6% for adults aged 18 to 79 years old. 4 Chronic hepatitis B (CHB) is associated with increased risk of cirrhosis and hepatocellular carcinoma (HCC) formation regardless of the status of chronic infection. 5 In a global study of cancer incidence and mortality, statistics showed that HCC is the second leading cause of death related to malignancy worldwide in 2012. 6
Nonalcoholic fatty liver disease (NAFLD) is becoming one of the most common liver diseases worldwide and is estimated to affect about a quarter of the population in Asia. 7 Due to the high prevalence of NAFLD and metabolic syndrome, metabolic disorders were shown in recent data to contribute numerically more to the burden of HCC formation in the United States. 8 However, except for Japan, CHB is still the leading cause for HCC in Asia-Pacific region. 9
The association/interaction of CHB and hepatic steatosis remains to be clarified. 10 A meta-analysis performed on 4100 patients showed that there were no associations between hepatitis B infection and hepatic steatosis. 11 In a recent study in Korea, hepatitis B surface antigen (HBsAg) positivity was found to be associated with lower risk of developing NAFLD. 12
Currently, there are conflicting data regarding risk of development of HCC in patients with concurrent CHB infection and hepatic steatosis. In this study, we performed a retrospective cohort analysis on CHB-infected patients without any significant alcohol intake history who underwent liver biopsy. The risk of concurrent histologically proven hepatic steatosis and hepatitis B infection in causing HCC on long follow-up is determined.
Materials and methods
Study design
This is a retrospective cohort study performed from January 2000 to December 2014 at Singapore General Hospital, Singapore. Patients above 21 years old and who were diagnosed to have CHB and received a liver biopsy were recruited and followed up on that duration. Inclusion criteria were the following: (1) age >21 years old; (2) diagnosis of CHB with positive serology test for serum HBsAg for at least 6 months 13 ; (3) underwent a liver biopsy for diagnostic purpose, assessment of disease severity or ruling out concomitant liver diseases. Exclusion criteria includes (1) testing positive for antibody against hepatitis C virus (anti-HCV) or antibody against human immunodeficiency virus (HIV); (2) patients with concomitant autoimmune disease, primary biliary cirrhosis, primary sclerosing cholangitis, Wilson disease, or hemochromatosis; (3) patients with HCC diagnosed at baseline visit; (4) patients with significant alcohol intake history (defined as alcohol intake of ⩾20 g per day in men and ⩾10 g per day in women); and (5) patients on long-term hepatic steatosis inducing medications (ie, systemic steroids and methotrexate). The study was approved by the Institutional Review Board of Singhealth Services, Singapore, and informed consents were waived.
Clinical evaluation and histology assessment of cohort population
A retrospective review of inpatient case notes and clinic notes were conducted on the selected patients who underwent liver biopsy and fulfilled the inclusion criteria. Clinical and biochemical data were obtained and recorded. Data collection included patient demographics, clinical parameters, and laboratory values. Type 2 diabetes mellitus (T2DM) was defined as per American Diabetes Association 14 criteria. Liver biopsies were reviewed again by liver histopathologists (T.K-H.L., W.Q.L., W.K.W., R.A.) for hepatic steatosis (defined as presence of >5% steatosis), Non-Alcoholic Steatohepatitis Activity Score (NAS), 15 modified Hepatic Activity Index (HAI) score, 16 and Ishak fibrosis stage. 16
The clinical characteristics were analyzed and stratified by presence (group 1) or absence (group 2) of hepatic steatosis. Patients were followed up and underwent abdominal ultrasound for development of HCC till June 1, 2017. The timing of the follow up was 6 to 12 monthly and it was based on clinical judgment of managing hepatologists and supported by existing guidelines. Hepatocellular carcinoma was confirmed based on contrast-enhanced cross-sectional imaging.
Statistical analysis/methods
Results were analyzed using SPSS version 23.0 (IBM Corp, Armonk, NY, USA). Continuous variables were expressed as mean ± SD. Categorical variables were expressed as actual numbers and their percentages.
Student t test was performed to compare age, hepatitis B virus DNA, and alanine aminotransferase between patients with hepatic steatosis and those without, whereas Fisher exact test was performed to compare the distribution of sex and cirrhosis between these 2 groups of patients.
Time to HCC development was defined from biopsy to the diagnosis of HCC, or last follow-up for censored cases. Cox regression was performed to evaluate the effect of potential factors on the development of HCC.
Results
A total of 289 patients were identified from the system records which fulfilled the criteria for the study. Patients’ baseline characteristics are listed in Table 1. Compared with patients without hepatic steatosis, patients with hepatic steatosis were older (43.2 ± 13.8 years vs 46.4 ± 10.7 years, P = .045) with lower viremia (7.1 ± 1.8 log copies/mL vs 6.7 ± 1.9 log copies/mL, P = .038). Hepatic steatosis was present in the liver biopsy of 185 patients (64.0%). There were 30 patients with cirrhosis at baseline liver biopsies, with equal distribution between both groups: nonhepatic steatosis and hepatic steatosis (10.6% vs 10.3%, P = 1.0).
Baseline characteristics comparison between patients with and without hepatic steatosis.
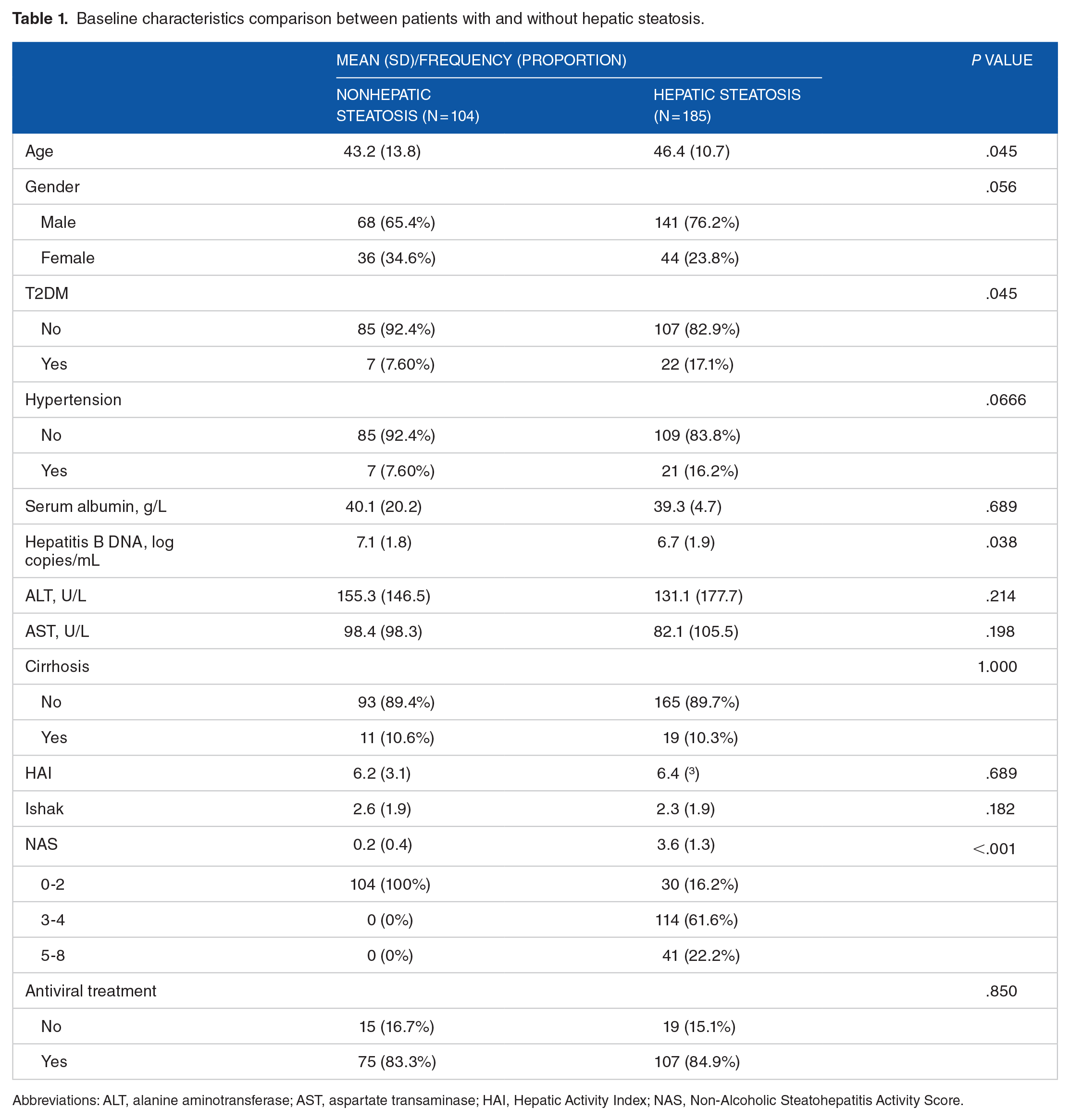
Abbreviations: ALT, alanine aminotransferase; AST, aspartate transaminase; HAI, Hepatic Activity Index; NAS, Non-Alcoholic Steatohepatitis Activity Score.
The median follow-up for HCC development was 111.1 months (range: 0.99-191.1 months). In total, 10 patients died due to non-liver-related cause of death during the follow-up; 21 patients (11.4%) with hepatic steatosis and 6 patients (5.8%) without hepatic steatosis developed HCC. Table 2 shows the list of patients who developed HCC.
Patients who developed hepatocellular carcinoma.
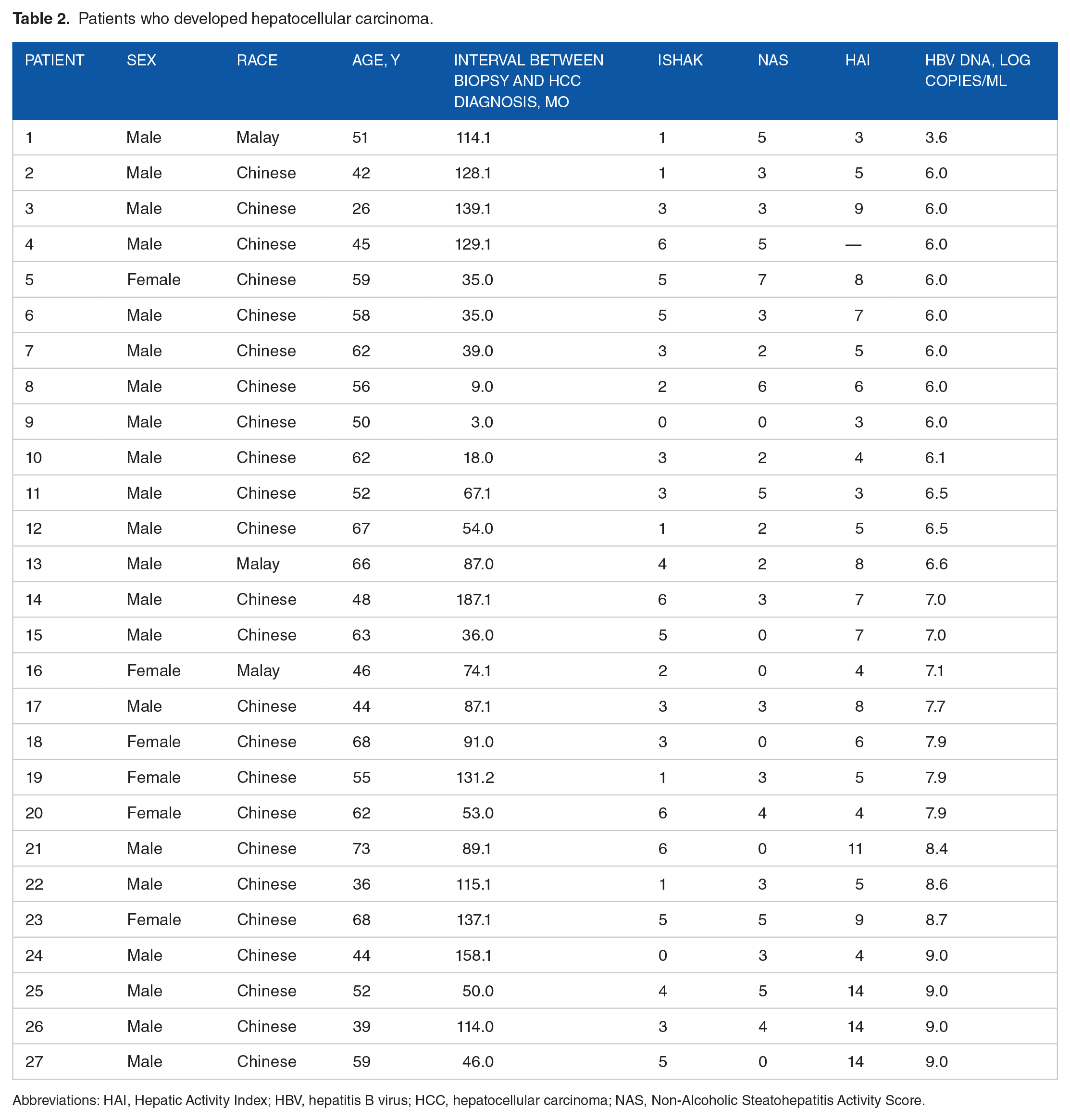
Abbreviations: HAI, Hepatic Activity Index; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; NAS, Non-Alcoholic Steatohepatitis Activity Score.
Univariable Cox regression showed that older patients (hazard ratio [HR] = 1.08, 95% confidence interval [CI] = 1.042-1.12), patients with higher Ishak score (HR = 1.221, 95% CI = 1.014-1.472), patients with hypertension (HR = 4.397, 95% CI = 1.649-11.725) and those with T2DM (HR = 4.00, 95% CI = 1.622-9.863) had a higher chance to develop HCC (Table 3). Multivariate Cox analysis showed similar findings for age and T2DM (Table 4).
Univariable analysis of time to HCC diagnosis.
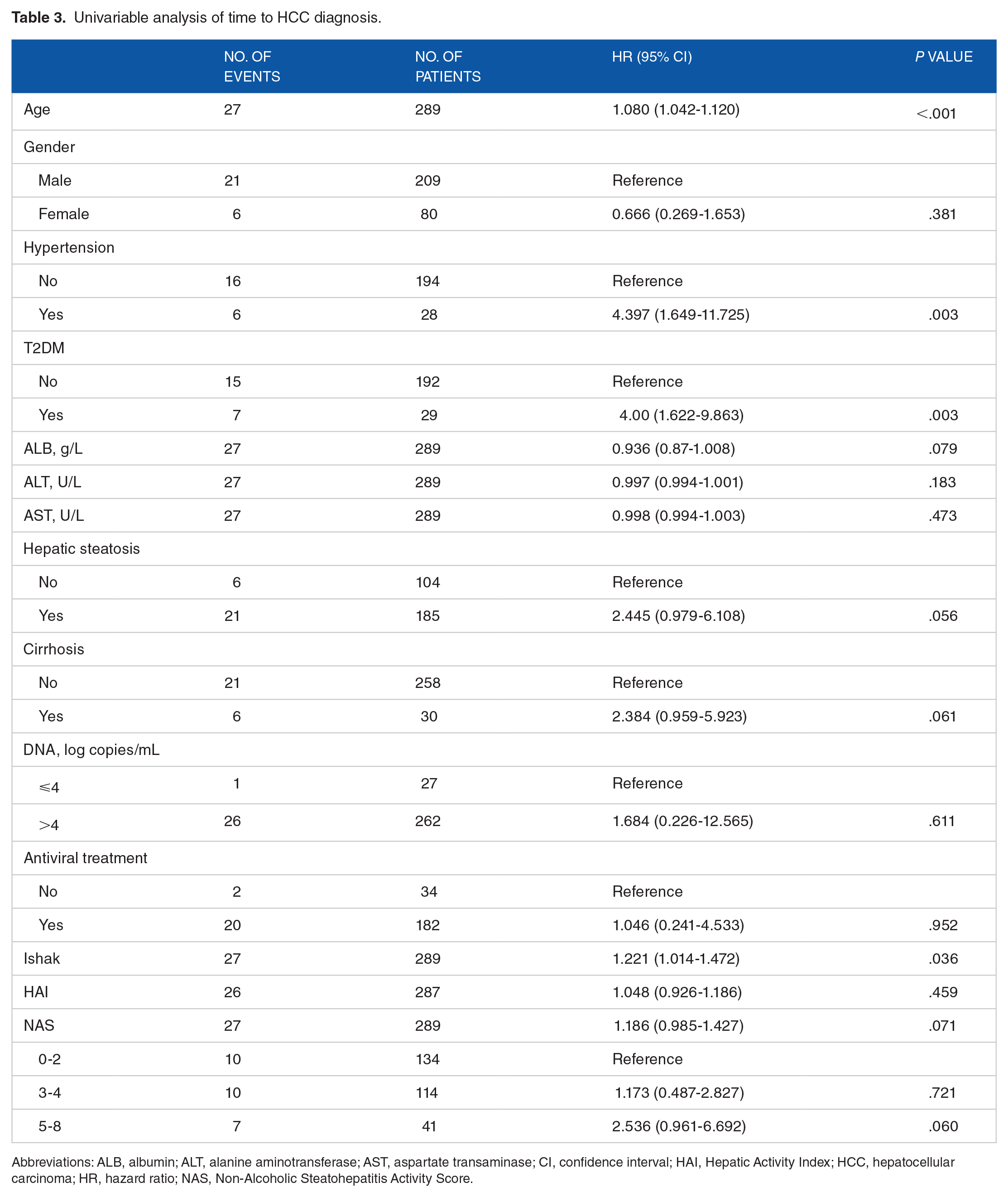
Abbreviations: ALB, albumin; ALT, alanine aminotransferase; AST, aspartate transaminase; CI, confidence interval; HAI, Hepatic Activity Index; HCC, hepatocellular carcinoma; HR, hazard ratio; NAS, Non-Alcoholic Steatohepatitis Activity Score.
Multivariable analysis of time to HCC development.

Abbreviations: CI, confidence interval; HR, hazard ratio; HCC, hepatocellular carcinoma.
Concurrent hepatic steatosis was not a significant risk factor for the development of HCC in the cohort (P = .056, HR = 2.445, 95% CI = 0.979-6.108). Both univariable and multivariable Cox regression for hepatic steatosis was not significant. Similarly, higher NAS score at baseline biopsy was also not a significant risk factor for HCC development (P = .071, HR = 1.186, 95% CI = 0.985-1.427).
Discussion
In our cohort of 289 patients with CHB with liver biopsy, we found that age and presence of T2DM are independent risk factors for developing HCC. This has been studied earlier as risk factor for HCC development as elucidated by other large cohort studies.17-20 Although CHB and T2DM are known to be independent risk factors for HCC development, there are conflicting results in cohort studies on their association with HCC development.
In one study, Wang et al 17 demonstrated in his cohort of 696 CHB-infected patients that T2DM was found to be not a significant risk factor for HCC development (HR = 1.3; 95% CI = 0.3-5.6, P > .05). In a larger and longer follow-up study by Chen et al, 20 T2DM was found to have an increased risk for development of HCC in CHB infection (RR = 2.27; 95% CI = 1.10-4.66). However, in both of these studies, hepatic steatosis was not a risk factor explored for hepatocarcinogenesis.
Recently, Chen et al 21 studied oleic acid–induced hepatic steatosis cells in vitro and found that cell steatosis does promote proliferation and migration of HCC cells. However, clinical results in follow-up patients regarding HCC development and CHB do not show similar data. In our study, we also demonstrated that hepatic steatosis is not a significant risk factor for development of HCC (P = .056). Moreover, increasing NAS score also does not contribute to increasing HCC development (P = .071). A similar result was seen in a study in the Netherlands by Brouwer et al, 22 which was performed on 531 treatment naïve CHB patients who had liver biopsy performed and had a median follow-up of 121.2 months. In their cohort, no significant association of hepatic steatosis and HCC development was seen (HR = 2.2, 95% CI = 0.7-6.5, P = .153), although 34% of their patients have significant alcohol intake history. Lee et al 23 studied 321 CHB-infected patients in Korea with biopsy proven hepatic steatosis and found a 3-fold increased risk of HCC development (HR = 3, 95% CI = 1.12-8.05) over a median follow-up of 63.6 months (range: 34.8-99.6). However, after adjusting for metabolic factors via inverse probability weighting, hepatic steatosis is not a significant risk factor (P = .47), whereas age (P = .02) and diabetes mellitus (P = .03) are independent risk factors associated with HCC development.
Chan et al 24 performed a similar study as well in Hong Kong for a cohort of 270 CHB-infected patients. In their study which was also based on histologically proven fatty liver, hepatic steatosis was associated with a significant increased risk for HCC development. (HR = 7.3, 95% CI = 1.52-34.76). However, their median follow-up time was 79.9 months (range: 17.2-107.0) which was shorter compared with both our study and the study by Brouwer et al. As the incidence of HCC increases over time for CHB infection, a longer follow-up would potentially see more patients with HCC being diagnosed. The study by Lee et al 23 demonstrated similar result as ours but on a shorter follow-up duration showed that current clinical evidence of HCC development among hepatic steatosis with concurrent CHB infection seems to be lacking and do not reflect those found in in vitro study.
Being a retrospective study, our study has its limitations. Most data on demographics and risk factors were captured at the point of liver biopsy and this includes T2DM. Patient’s control of T2DM and duration of the disease were not captured in the system as well. Besides, not all patients who had liver biopsy performed were investigated for T2DM, thus reducing the number of patients we are able to analyze. In our cohort, the number of patients who were found to be cirrhotic on liver biopsy was small. Although our study did not show hepatic steatosis as a risk factor for HCC development, we also do not have any data on whether regression of hepatic steatosis could possibly downplay its significance to HCC development. Hepatic steatosis was present in 64.0% of our population, this is higher than expected as patients with fatty liver will more likely get liver biopsy done to confirm the predominant disease. 25
Further analysis may be possible to prove liver cirrhosis as a risk factor for HCC development if we were to use follow-up imaging to diagnose cirrhosis rather than being dependent on index liver biopsy for diagnosis of cirrhosis. Moreover, the number of cirrhotic cases on index biopsy was small (about 10% of the cohort) and hence could not have a statistical impact as risk factor for HCC. Nevertheless, our study represents a considerable cohort with long duration of follow-up data to be able to sufficiently test our hypothesis. Furthermore, the disease phenotypes were defined by liver histology, which remains the gold standard of assessment.
Conclusions
Our study showed that hepatic steatosis was not a significant risk factor for the development of HCC in a cohort of CHB infection. However, T2DM increases the risk for the development of HCC in this cohort by 2.7-fold.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conception and design: CTL, GBBG, RK;
Administrative support: All authors;
Data analysis and interpretation: HL, CTL, RK;
Liver histopathology review: TK-HL, WQL, WKW, RA;
Manuscript writing: CTL, RK;
Final approval of manuscript: All authors; Research Guidance: WCC.
