Abstract
Development of new antibiotics is always needed in the fight against growing threat from multiple drug–resistant bacteria, such as resistant Gram-negative (G−) Escherichia coli and Klebsiella pneumoniae. While the development of broad-spectrum antibiotics has attracted great attention, careful administration of these antibiotics is important to avoid adverse effects, like Clostridium difficile infection (CDI). The use of broad-spectrum antibiotics, for example, quinolones, can increase the risk of CDI by eradicating the protective bacteria in intestine and encouraging C difficile spore germination. Many common intestine bacteria are G− or anaerobic, including Enterococcus faecalis, Bacteroides fragilis, and E coli. Hence, it may be advantageous in certain therapeutic practices to employ selective antimicrobials. For instance, Gram-positive (G+) methicillin-resistant Staphylococcus aureus (MRSA) that can cause life-threatening sepsis can be controlled with the use of selective antibiotic, vancomycin. Nevertheless, its effectiveness has been limited with the emerging of vancomycin-resistant Staphylococcus aureus (VRSA). A recent report on antimicrobial cationic anthraquinone analogs (CAAs) that show tunable activity and selectivity may provide new hope in the search for selective antimicrobials. In particular, the lead CAA displays prominent activity against MRSA while manifesting low activity against E coli and low cytotoxicity toward normal mammalian cells.
Continuous incidents reported worldwide regarding the bacterial pathogens that are resistant to single or multiple drugs have prompted the call for re-devoting effort into the discovery of new antibiotic. 1 As highlighted in the news, the emergence of multiple drug–resistant (MDR) Gram-negative (G−) Escherichia coli and Klebsiella pneumonia equipped with New Delhi metallo-β-lactamase 1 (NDM-1) that makes bacteria resistant to various antibiotics has attracted great attention for the development of new broad-spectrum antibiotics. 2 Broad-spectrum antibiotics have the advantage of exerting antibacterial activity against both G− and Gram-positive (G+) bacteria, while selective antibiotics may have activity against only G+ or specific strains of bacteria. Vancomycin, for example, is a well-known selective, or narrow-spectrum, antibiotic with effectiveness only toward G+ bacteria, such as Staphylococcus aureus but not G− bacteria.
While the development of broad-spectrum antibiotics has attracted great attention, careful administration of these antibiotics is important to avoid adverse effects, like Clostridium difficile infection (CDI). The use of broad-spectrum antibiotics, for example, quinolones, clindamycin, and cephalosporins, can increase the risk of CDI by eradicating the protective bacteria in intestine and encouraging C difficile spore germination.3,4 Many common intestine bacteria are either G− or anaerobic, including Enterococcus faecalis, Bacteroides fragilis, and E coli. Hence, it may be advantageous in certain therapeutic practices to employ selective antimicrobials. For instance, Gram-positive (G+) methicillin-resistant Staphylococcus aureus (MRSA) that can cause life-threatening sepsis can be controlled with the use of selective antibiotic, vancomycin. Nevertheless, its effectiveness has been limited with the emerging of vancomycin-resistant Staphylococcus aureus (VRSA). 5 Even the newer selective antibiotics, linezolid and daptomycin, have encountered the problem of bacterial resistance.6,7 In short, there is a significant need in the development of new selective antibiotics against formidable bacterial pathogens, like MRSA, while minimizing the risk of disrupting human gut flora.
We have recently reported that latest version of antimicrobial cationic anthraquinone analogs (CAAs) that show tunable activity and selectivity may provide new hope in the search for selective antimicrobials against serious human pathogens, like MRSA or VRSA. 8 In particular, the lead CAA displays prominent activity against MRSA while manifesting low activity against E coli and low cytotoxicity toward normal mammalian cells. The development of CAAs resides on 2 essential guidelines in drug development: (1) low cost of production and (2) accessibility to diverse structural variations with biologically relevant moiety. The cost of production is seldom the focus of early-stage drug development, especially for the laboratories in academia. Nevertheless, a recently Food and Drug Administration (FDA)-approved antibiotic, fidaxomicin, a new class of narrow-spectrum macrocyclic antibiotic, has not been able to compete with vancomycin or other generic drugs in the market due to its higher price. 9
Accordingly, we decide to employ 1,4-naphthoquinone and “Click” chemistry to meet these 2 guidelines; 1,4-naphthoquinone, a redox active scaffold, is a common core in many bioactive molecules.
10
It can react with azides via a [2 + 3] cycloaddition, a Click reaction to enable the introduction of diverse structural moieties readily and lower the cost of production. The inception of CAAs began with [2 + 3] cycloaddition of 1,4-naphthoquinone and glycosyl azides (
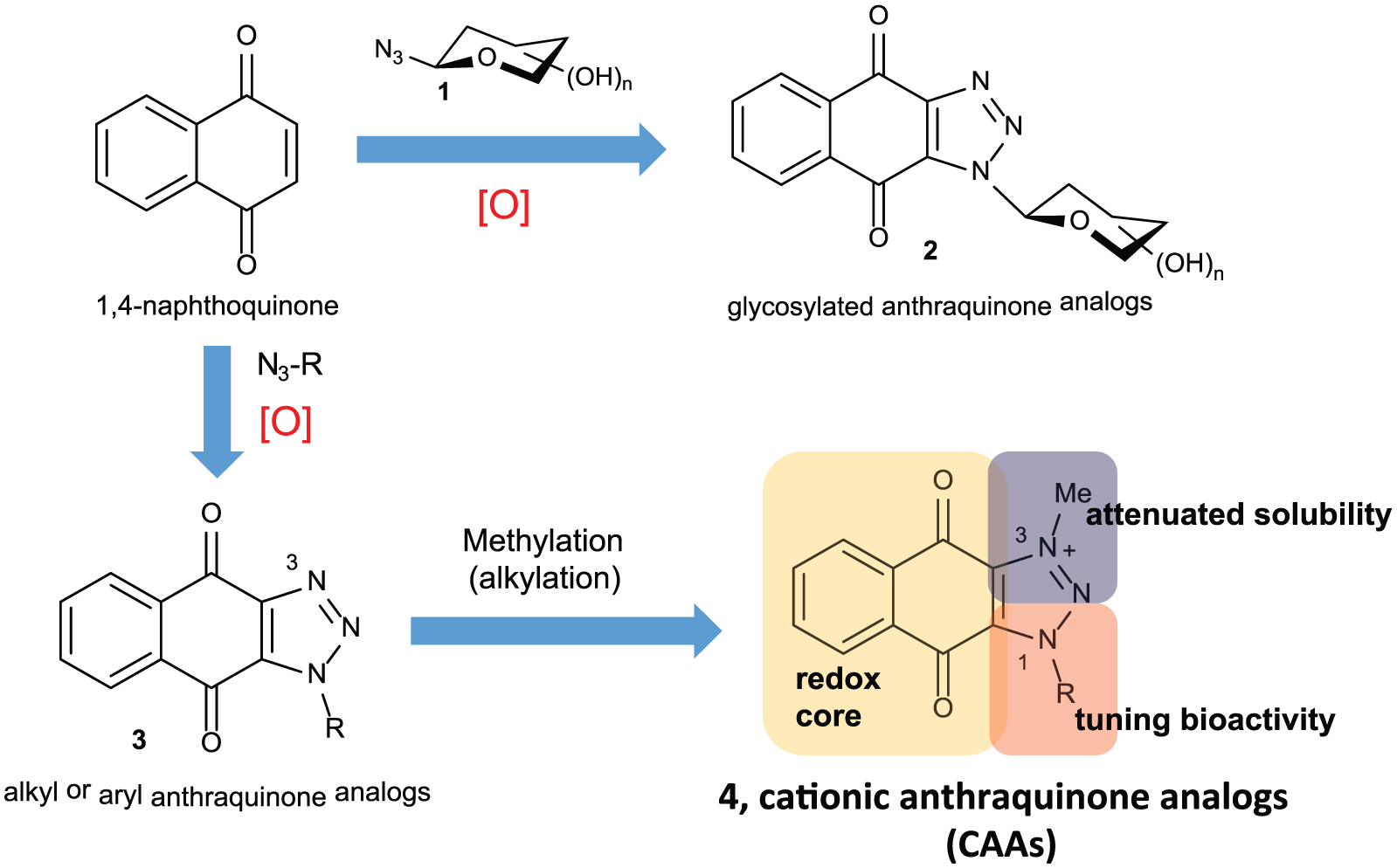
Development of CAAs. CAAs indicate cationic anthraquinone analogs.
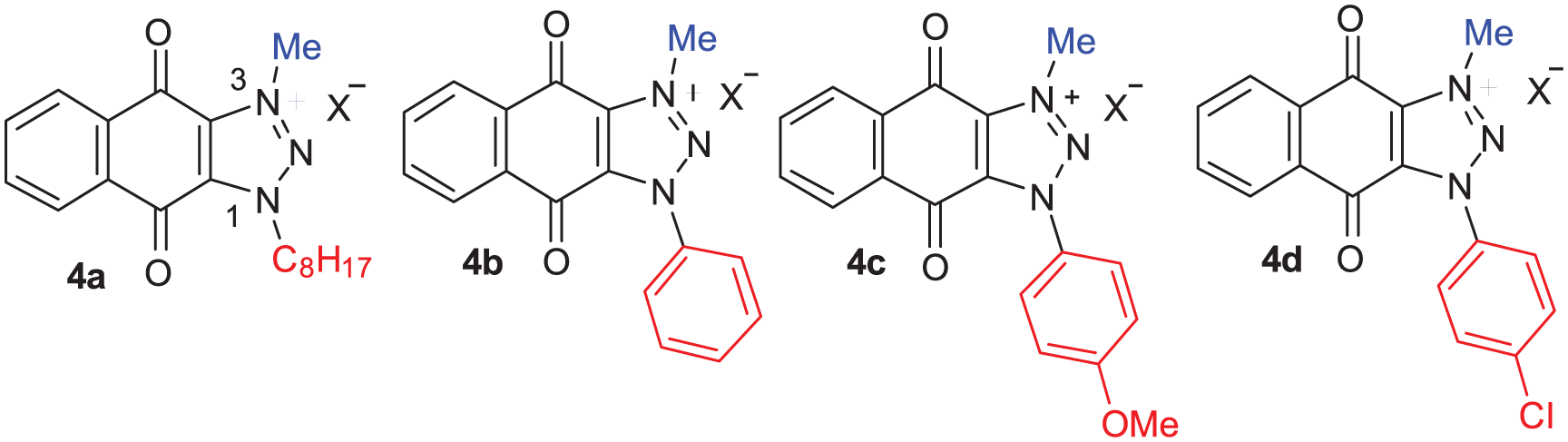
Following the structure-activity relationship (SAR) study of CAAs, the analog with n-octyl group at N-1 (
Structure and activity of CAAs a .
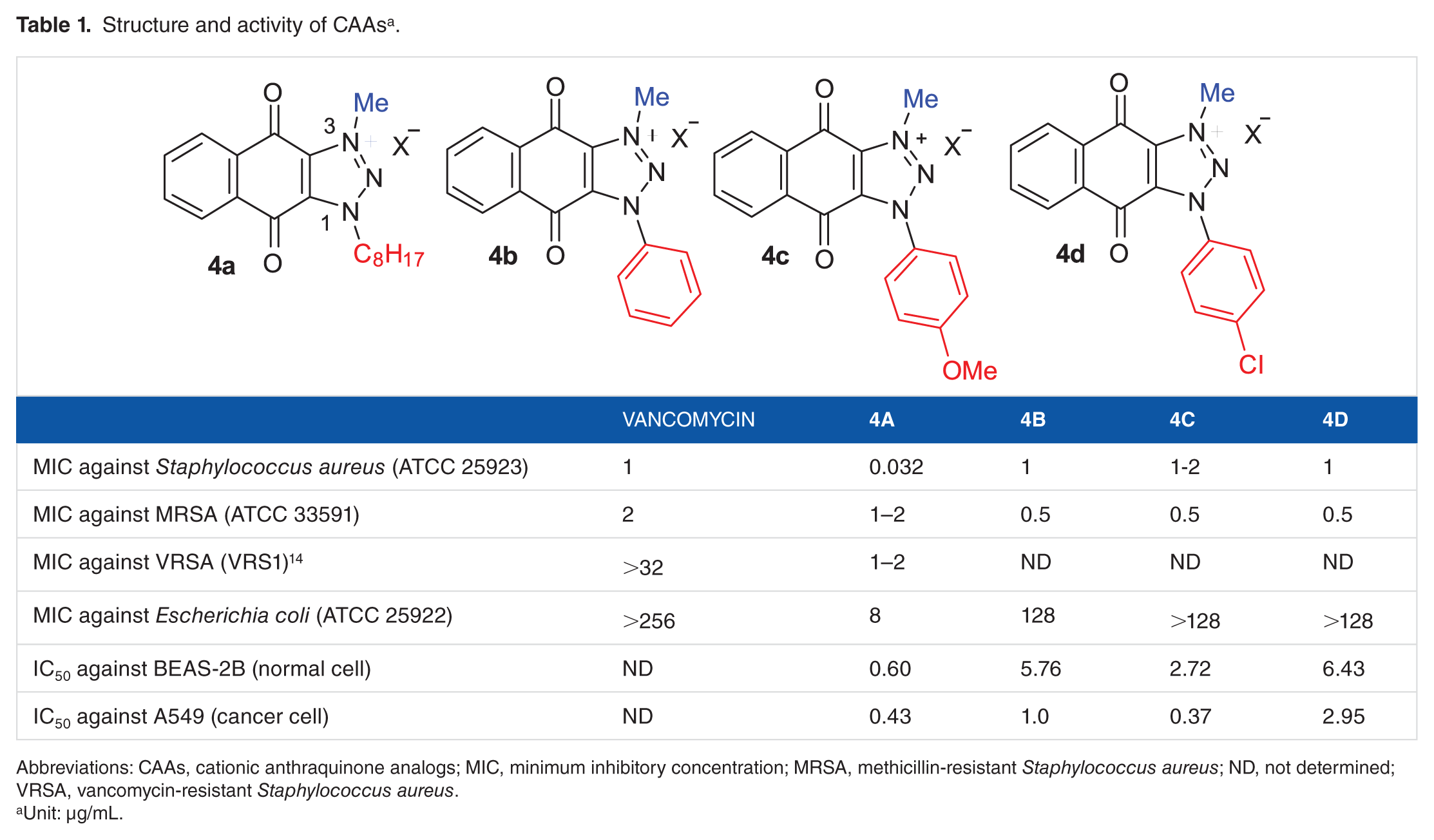
Abbreviations: CAAs, cationic anthraquinone analogs; MIC, minimum inhibitory concentration; MRSA, methicillin-resistant Staphylococcus aureus; ND, not determined; VRSA, vancomycin-resistant Staphylococcus aureus.
Unit: µg/mL.
The revealed SAR led to the synthesis of
Structure and activity of CAAs a .
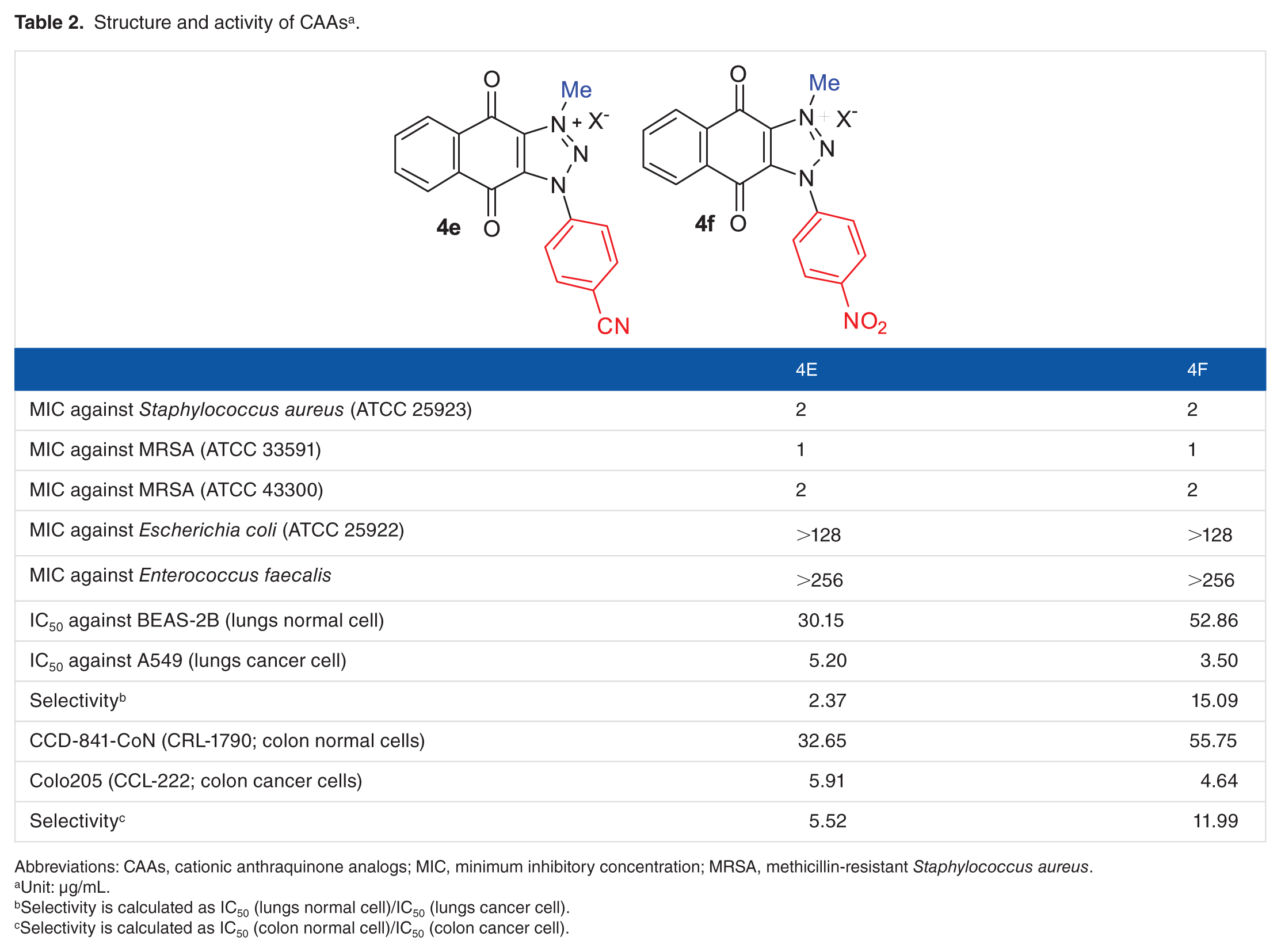
Abbreviations: CAAs, cationic anthraquinone analogs; MIC, minimum inhibitory concentration; MRSA, methicillin-resistant Staphylococcus aureus.
Unit: µg/mL.
Selectivity is calculated as IC50 (lungs normal cell)/IC50 (lungs cancer cell).
Selectivity is calculated as IC50 (colon normal cell)/IC50 (colon cancer cell).
In conclusion, the development of narrow-spectrum antibiotics is important for certain therapeutic applications albeit not receiving as much attention as the development of broad-spectrum antibiotics. Through years of investigation, we have identified 2 lead compounds, which can be prepared with simple chemistry, as the potential selective or narrow-spectrum antibiotics. Such narrow-spectrum antibiotics have the potential of controlling the infections caused by MRSA while not increasing the risk factor of CDI.
Footnotes
Acknowledgements
The authors acknowledge the support from NSF Award CHE-1429195 for 500 MHz Bruker NMR. We thank the support from Department of Chemistry & Biochemistry, Utah State University. The authors also thank the assistance from the Division of Microbiology and Infectious Diseases (DMID) at NIAID, NIH, for testing against various strains of MRSA and VRSA by a contracted laboratory.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
YPS performed the experiments and assisted in manuscript preparation. CWTC designed the experiments and instructed the data analysis.