Abstract
Introduction:
Hereditary angioedema (HAE) is an autosomal dominant disease caused by deficiency of the plasma protein C1 inhibitor (C1-INH). Patients classically present with recurrent localized subcutaneous or submucosal edema lasting for 2 to 5 days, severe abdominal pain, or acute airway obstruction which can be fatal.
Case presentations:
We highlight 2 patients with acute airway compromise secondary to HAE who were successfully treated with plasma-derived C1-INH concentrates.
Conclusions:
The timely administration of plasma-derived C1-INH concentrates for the acute treatment of HAE has been proven to be effective in both patients in aborting an airway complication. A high index of suspicion is required for the early diagnosis and treatment of this potentially fatal condition.
Introduction
Hereditary angioedema (HAE) is an autosomal dominant disease caused by deficiency of the plasma protein C1 inhibitor (C1-INH).
C1 inhibitor deficiency causes unchecked activation of the classic complement pathway and other biochemical systems, including the bradykinin system. Patients classically present with recurrent localized subcutaneous or submucosal edema lasting for 2 to 5 days, severe abdominal pain, or acute airway obstruction which can be fatal. 1
In this report, we highlight 2 patients with acute airway compromise secondary to HAE who were successfully treated with plasma-derived C1-INH concentrates.
Case 1
A 74-year-old Chinese man presented to the emergency department after sustaining an accidental fall at home. He was a known case of HAE. His diagnosis was made after he was screened following the sudden demise of his daughter from cardiorespiratory collapse secondary to HAE.
He had manifested symptoms since he was 30 years old. He had been previously hospitalized for laryngeal edema post-dental treatment 12 years ago and subsequently for facial, lip, and oropharyngeal edema 5 years ago. Apart from this, he also reported at least yearly attacks of milder angioedema that were self-resolving which did not require medical attention. There was a strong family history of HAE as shown in Figure 1, with 2 of his daughters and his grandson having this diagnosis. He had undetectable levels of complement 4 (C4), but had declined further qualitative and quantitative evaluation of C1-INH for cost reasons. He also declined prophylactic treatment with Danazol.
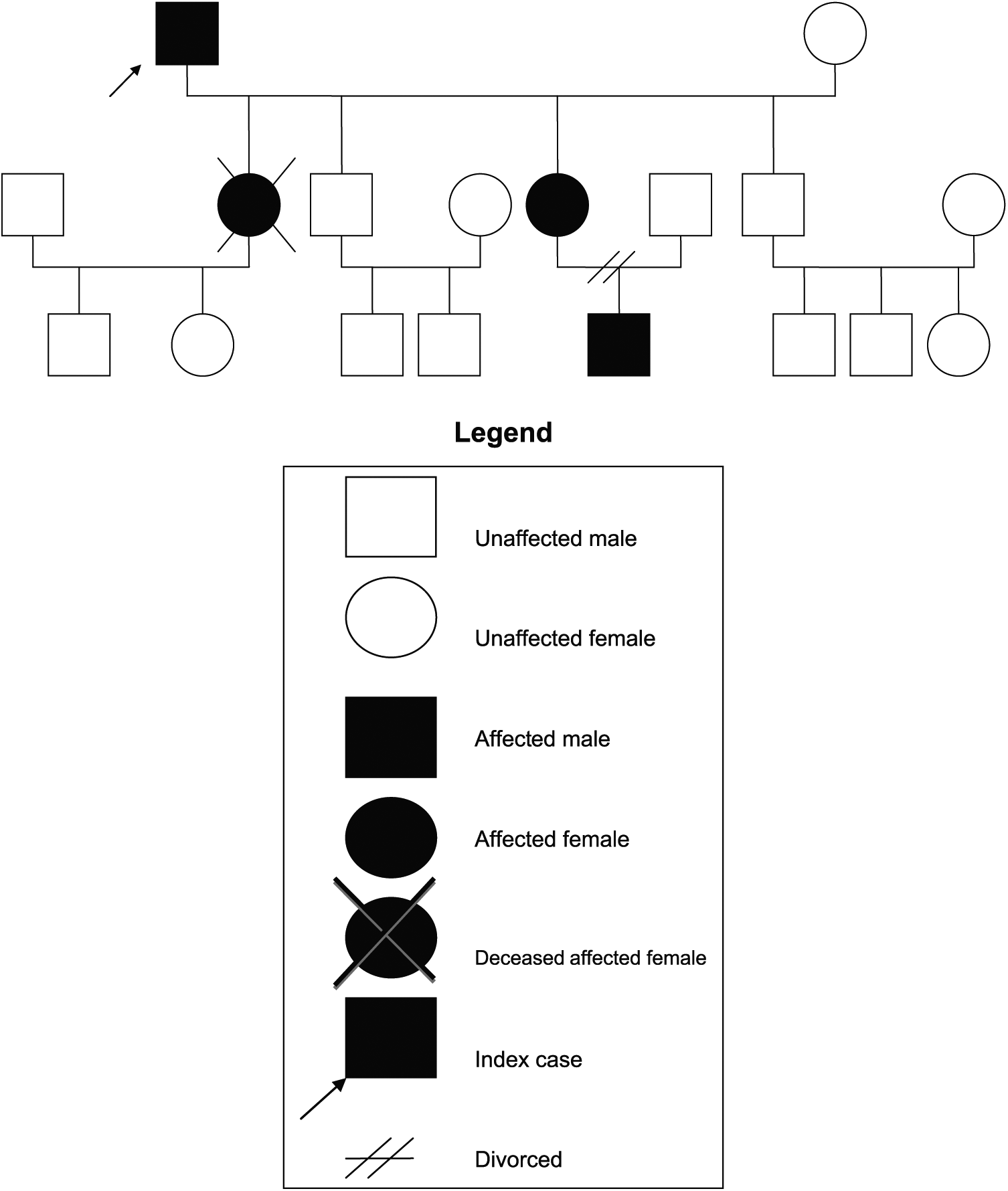
Family tree of affected members with hereditary angioedema (HAE) for case 1.
For the current admission, he did not have oropharyngeal or laryngeal edema on nasoendoscopy examination at initial presentation. However, after 12 hours, the patient complained of increased swelling over the upper lip and neck with dysphagia and a change in his voice quality. A repeat nasoendoscopy examination subsequently showed diffuse oropharyngeal and laryngeal edema.
With an impending upper airway obstruction staring at us, the decision was made for a trial of plasma-derived C1-INH concentrate (Cinryze) with a plan for awake fiberoptic intubation or emergency tracheostomy if he did not improve clinically.
He received a total of 2000 units of Cinryze with gradual resolution of the facial, oropharyngeal, and laryngeal edema on serial nasoendoscopy examinations. By the following day, he was well enough to be discharged.
Over the next 3 years, he had another 2 admissions for acute laryngeal edema secondary to HAE attacks precipitated by dental extraction and upper respiratory tract infection. He received 1000 units of Berinert, plasma-derived C1-INH concentrates on both occasions with complete resolution of his symptoms.
Case 2
A 61-year-old Indian lady presented to the emergency department with acute bilateral periorbital and facial edema after taking Serratiopeptidase (Danzen). A day prior to hospital admission, she noted mild neck swelling with pain. She did not have complaints of voice change, dysphagia, or dyspnea at presentation.
She initially presented with recurrent abdominal pain, vomiting, and diarrhea secondary to gut angioedema in 2010. She was started on Danazol prophylaxis but has been poorly compliant with recurrent flares of HAE. She has no known family history of HAE as she has not been in contact with her other 18 siblings. She had undetectable levels of C4 and low/normal levels of functional C1-INH antigen (43%) in May 2013.
For the current admission, she was initially monitored in general ward but she subsequently complained of dyspnea and was noted to be agitated. Nasoendoscopy examination showed oropharyngeal and laryngeal edema. This is her first admission for acute HAE flare with impending upper airway obstruction.
She was subsequently transferred to the High Dependency Unit for airway monitoring. She received a total of 1000 units of Berinert with gradual resolution of the oropharyngeal and laryngeal edema by the following day. She was educated regarding Danazol compliance.
Since then, the evaluation and treatment of her angioedema have been limited by her financial constraints as well as repeated defaults at the outpatient clinics. Nonetheless, she has always responded to treatment with plasma-derived C1-INH concentrate as per a typical patient with HAE.
Discussion
Hereditary angioedema is an autosomal dominant disease caused by a mutation of the SERPING1 gene, which codes for C1-INH. Its prevalence is uncertain but is estimated to be approximately 1 case per 50 000 persons, with no known differences between ethnic groups. 2 De novo mutations of SERPING1 occur in 20% to 25% of cases.
C1 inhibitor is a serine protease inhibitor whose function is inhibition of complement proteases and contact system proteases (plasma kallikrein and coagulation factor XIIa). Studies have shown that bradykinin is the primary mediator of angioedema in HAE. C1 inhibitor deficiency or dysfunction results in unchecked activation of plasma kallikrein and generation of bradykinin from cleavage of high-molecular weight kininogen.
Three types of HAE have been described. Type 1 (HAE-1) is associated with a quantitative decrease in C1-INH. Type 2 HAE (HAE-2) is associated with normal or high levels of C1-INH which is dysfunctional. 3 HAE-3 is associated with normal C1-INH levels and function.
The disease typically presents with angioedema without urticaria and may be associated with recurrent abdominal pain or upper airway swelling. Symptoms begin in childhood (often as early as 2 or 3 years of age), worsen around puberty, and persist throughout life, with unpredictable severity. Untreated patients have attacks every 7 to 14 days on average, with the frequency ranging from virtually never to every 3 days. 4
Some studies have suggested that minor trauma, stress, infections, dental and other surgery, and estrogens (including pregnancy, oral contraceptives, and hormone replacement therapy) are frequent precipitants of HAE, but attacks can occur without an apparent trigger in many cases. 5
Available treatments for acute attacks of HAE include C1-INH concentrate, kallikrein inhibitors, and bradykinin receptor antagonists. C1 inhibitor concentrates replace the deficient protein and inhibit kallikrein and factor XIIa. C1 inhibitor concentrates may be derived from human plasma (eg, Cinryze and Berinert) or recombinant (Ruconest). Ecallantide is a potent kallikrein inhibitor, which acts by blocking the binding site of kallikrein and inhibiting the cleavage of high-molecular weight kininogen to bradykinin. Bradykinin induces angioedema via activation of bradykinin B2 receptors; its action is blocked by the B2 receptor antagonist, Icatibant.
Berinert is a plasma-derived C1-INH concentrate approved by the United States Food & Drug Administration (US FDA) for the treatment of acute abdominal, facial, or laryngeal attacks of HAE in adult and adolescent patients. Its efficacy has been demonstrated in a randomized clinical trial conducted by Craig et al 6 on 125 patients with acute HAE attacks, where the median time to onset of relief was significantly shorter with Berinert as compared with placebo.
Cinryze, also a plasma-derived C1-INH concentrate, has been approved by the US FDA in 2008 for the routine prophylaxis of HAE in adult and adolescent patients. Similarly, Cinryze has been approved by the European Medicines Agency (EMA) in 2017 for the treatment of acute attacks as well.
In a study by Bork et al, 193 episodes of laryngeal edema in patients with acute HAE were treated with Berinert. They noted that the resolution of symptoms begins as early as 30 to 60 min after administration, and the duration of the upper airway obstruction is substantially reduced with treatment. 7 The authors also mentioned that prior to the diagnosis of HAE or the availability of the medication, these patients would have required acute airway intervention such as tracheostomy.
In 2 randomized trials by Zuraw et al, 8 Cinryze has been shown to shorten the duration of acute attacks and, when used for prophylaxis, reduced the frequency of acute attacks. Patients with laryngeal angioedema were excluded from this study.
In an open-label study to evaluate the efficacy and safety of repeat use of Cinryze for the short-term treatment of HAE attacks, out of 84 patients who were treated for laryngeal attacks, 60% and 77% achieved unequivocal relief within 1 and 4 h, respectively, after start of treatment. Unequivocal relief was defined as at least 3 consecutive post-treatment reports of improvement every 15 minutes. No patient required intubation after administration of Cinryze. 9
Laryngeal edema, the most severe site of attack, is estimated to occur in 50% of HAE patients at least once in their lives. If left untreated, this can result in fatal asphyxiation. This is a major cause of death in patients with HAE, with a mortality rate ranging from 14% to 33% in affected patients. 10 Thus, when laryngeal edema occurs, the most important aspect of management rests in early, aggressive support of the airway. The decision for “watchful waiting” in such patients will often lead to significant morbidity and mortality because laryngeal edema can progress rapidly and potentially require a surgical airway.
For the first patient, he was fortunate that Cinryze had been made available in the hospital after a series of 3 cases involving his family members who presented with acute laryngeal edema secondary to HAE leading to cardiorespiratory collapse.
He has a 17-year-old grandson who was diagnosed with HAE since the age of 14 years old. He has a history of recurrent scrotal and limb swelling, abdominal pain due to gut angioedema, made worse through his non-compliance with Danazol prophylaxis. He presented 1 year ago with acute laryngeal edema requiring emergency tracheostomy.
His daughter (who is the mother of the 17-year-old) also presented 4 years ago with acute laryngeal edema requiring tracheostomy. A less fortunate second daughter also collapsed at home secondary to acute laryngeal edema before she was brought to the hospital. She subsequently died from hypoxic ischemic encephalopathy. Since then, the entire family has been screened for HAE as shown in Figure 1.
For the second case, she presented similarly with her first episode of acute laryngeal edema secondary to poor compliance to Danazol. Having witnessed the efficacy of the plasma-derived C1-INH concentrate in the first case, she was also managed accordingly with Berinert and close airway monitoring. Despite the high cost of the medication, the treatment is cost-effective as acute airway procedures have been avoided in these patients.
In both cases, we have demonstrated that the plasma-derived C1-INH concentrates are effective in the treatment of acute laryngeal edema secondary to HAE. However, it needs to be given early in the course of the attack to allow time for the patient to recover. Patients who are extremely symptomatic or unstable should have their airway secured immediately. Patients who are more stable can be monitored clinically. However, caution is warranted with serial flexible nasoendoscopy examinations for airway assessment as that too can precipitate a flare up in airway edema leading to a compromised airway. Treating HAE patients circumspectly as potential emergent airway obstruction is advocated, and “prophylactic” semi-elective intubation would be preferable to a “crash” tracheostomy.
Conclusions
The timely administration of plasma-derived C1-INH concentrates for the acute treatment of HAE has been proven to be effective in both patients in aborting an airway complication. A high index of suspicion is required for the early diagnosis and treatment of this potentially fatal condition.
Footnotes
Acknowledgements
This work was accepted for poster presentation at the 16th Association of Southeast Asian Nations (ASEAN) Otorhinolaryngology, Head & Neck Surgery (ORL-HNS) Congress 2015.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ECH conceived the concept. WZEF wrote the first draft of the manuscript and SCT contributed to the writing of the manuscript. All authors jointly developed the format and made critical revisions to the paper. All authors reviewed and approved the final manuscript.
