Abstract
As the world continues to respond to the coronavirus pandemic (COVID-19), there is a larger hidden threat of antimicrobial resistance (AMR) lurking behind. AMR remains worrisome in that the pathogens causing resistant infections to thrive in hospitals and medical facilities, putting all patients at risk, irrespective of the severity of their medical conditions, further compounding the management of COVID-19. This study aims to provide overview of early findings on COVID-19 and AMR as well as to provide recommendations and lesson learned toward improving antimicrobial stewardship. We conducted a rapid narrative review of published articles by searching PubMed and Google Scholar on COVID-19 and Antimicrobial Resistance with predetermined keywords. Secondary bacterial infections play crucial roles in mortality and morbidity associated with COVID-19. Research has shown that a minority of COVID-19 patients need antibiotics to treat secondary bacterial infections. Current evidence reiterates the need not to give antibiotic therapy or prophylaxis to patients with mild COVID-19 or to patients with suspected or confirmed moderate COVID-19 illness unless it is indicated. The pandemic has also brought to the fore the deficiencies in health systems around the world. This comes with a lot of lessons, one of which is that despite the advances in medicine; we remain incredibly vulnerable to infections with limited or no standard therapies. This is worth thinking in the context of AMR, as the resistant pathogens are evolving and leading us to the era of untreatable infections. There is a necessity for continuous research into understanding and controlling infectious agents, as well as the development of newer functional antimicrobials and the need to strengthen the antimicrobial stewardship programs.
Background
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first identified in Wuhan, China and human-to-human transmission led to widespread of the virus to other areas of Hubei Province.1,2 Subsequently, the disease spread across the country and then to other nations across the globe. 2 The 2019 novel coronavirus (2019-nCoV) was classified as SARS-CoV-2 by the Coronavirus Study Group of the International Committee on Taxonomy of Viruses. 3 On 11 February 2020, the World Health Organization 4 announced the name of the disease to be coronavirus disease 2019 (COVID-19).
As the world continues to respond to COVID-19, there is a larger hidden threat of antimicrobial resistance (AMR) lurking behind, one that is already killing hundreds of thousands of people globally (about 700 000 deaths annually). Widespread and unnecessary use of antibiotics, among other causes, have facilitated the emergence and spread of resistant pathogens.5-7 AMR remains worrisome in that the pathogens causing resistant infections thrive in hospitals and medical facilities, putting all patients at risk, irrespective of the severity of their medical conditions. 8 This will further confound the management of COVID-19.9,10
Unfortunately, this hidden threat of AMR has not received the same global attention as the COVID-19 pandemic. It would not be incorrect to also call AMR a hidden global pandemic. An AMR commissioned review estimates that annually by 2050, AMR could lead to 10 million deaths globally, with more people dying from drug-resistant infections than from cancer for example. 11 COVID-19 has posed challenges in all aspect of healthcare in resource-poor settings including management of chronic diseases and non-COVID-19 acute bacterial infection. 12 Healthcare systems have triggered the implementation of infection prevention policies through appropriate isolation of infected COVID-19 cases in an attempt to control the spread of the virus. 13 The pandemic has necessitated an unfamiliar response to controlling the spread of infection and to protect the most vulnerable. 14 It has also brought to focus the critical need for the immediate development of accurate diagnostic methods, vaccines, and antiviral treatments to reduce morbidity and mortality among vulnerable population.15,16 Whilst the impact of COVID-19 on AMR remains yet to be fully understood, this narrative review aims to provide overview of early findings on COVID-19 and AMR, including antibiotic use, and to provide recommendations and lesson learned toward improving antimicrobial stewardship.
Method
We conducted a rapid narrative review of published articles on COVID-19 and Antimicrobial Resistance with no date restriction placed on search. PubMed and Google Scholar were searched with the following key terms: “COVID-19,” “SARS-CoV2,” “2019-nCOV,” “Coronavirus Disease,” “Pandemic,” “Antibiotics,” “Antimicrobial Resistance,” “Antibiotic Resistance,” and only papers written in English Language were reviewed.
Antimicrobial Resistance: A Hidden Threat Lurking Behind COVID-19 Pandemic
The rapid spread of COVID-19 globally pushed biopharmaceutical industries to respond by emergency development of vaccines and other therapeutic tools including diagnostics.17,18These advances offer hope to a new paradigm in the discovery of tools required to combat the new global threat. However, there is a bigger threat of AMR lurking behind the pandemic which could implicate treatment and management of COVID-19 and other infectious diseases, leading to preventable loss of lives.9,10,14,19 The emergence of COVID-19 has signaled concern on the impact of this new threat on AMR and antimicrobial stewardship. 14 Whilst AMR is a growing health issue globally,5-7 the negative impact weighs most heavily on Africa and other low- and middle-income countries (LMICs) where there are weak antibiotic supply chain policies, poor regulatory framework, and structures such as poor diagnostics favoring prescribing practices based on observation rather than laboratory results. Africa is also estimated to have a larger burden of deaths due to AMR. 11
Since the beginning of the COVID-19 outbreak and management of cases 20 , some countries have been monitoring and reporting on antibiotic resistance with the aim to preserve most of the gains that have been recorded in the last decade in terms of antimicrobial resistance. The data these countries have provided reveals that a worrying number of bacterial infections are increasingly resistant.21,22,23 Antimicrobial stewardship is important so as not to come out of this global pandemic to be faced by an increase in the need for effective antibiotics. 14 To effectively treat infections, there is a need to preserve health gains made in the past and ensure a secure future through proper monitoring and care. 22 While only a few studies on co-administration of antimicrobial agents and antivirals exists to understand whether coinfection affects disease progression19,24 the need for antimicrobial stewardship programs to step up cannot be overemphasized and this is because experimental therapies for the treatment of SARS-CoV-2 are being explored, for example, hydroxychloroquine and azithromycin. 25
Antimicrobial therapy has a role in the treatment of suspected or confirmed bacterial or fungal respiratory co-infection.18,25 This may be empiric or targeted in patients presenting to hospitals or for the management of nosocomial infections acquired during admission to hospitals, such as hospital-acquired pneumonia or ventilator-associated pneumonia.18,25 In terms of antimicrobial prescribing bacterial/fungal co-infection of the respiratory tract; some patients presenting to hospital with SARS-CoV-2 infection have a clinical phenotype that is not dissimilar from atypical bacterial pneumonia 18 making it difficult to distinguish SARS-CoV-2 infection from co-infection with hospital-acquired and ventilator-associated pneumonia in hospital inpatients.18,25,26 Only a minority of these patients may require antibiotic therapy 19 and this may result in the misuse of drugs including antimicrobials. 14 Some studies revealed the widespread use of antimicrobial therapies as part of the package of clinical care for hospitalized COVID-19 patients in some countries.10,14,18,21 While this suggests that the use of antibiotics or antivirals in hospitalized COVID-19 patients with secondary bacterial infection is appropriate, 19 the possibility of antibiotic prescribing in a large number of patients without established secondary infection is markedly increased, 21 thereby leading to an increase in AMR through driving selection of multidrug resistant (MDR) organisms. 22
A retrospective cohort analysis of 191 patients from 2 local hospitals in Wuhan, China reveals that 181 (95%) patients received antibiotics and 41 (21%) received antiviral drugs. 18 Another cohort analysis of 37 hospitalized patients in 3 local hospitals in Wuhan shows that 36 patients (97.3%) received antiviral treatment, 29 patients (78.5%) received probiotics, 28 patients (75.7%) were treated with traditional Chinese medicines, and 22 patients (59.5%) received antibiotics. 27 The limited data on COVID-19 and superinfection has made it difficult to anticipate the potential impact of the pandemic on antimicrobial stewardship programs and long-term rates of AMR.14,28,29 However, it is plausible to expect secondary bacterial and fungal infection due to immune down-regulation associated with COVID-19 among hospitalized patients.27,28,29,30 A study also highlighted the need for prospective study to analyze superinfection in COVID-19 patients in order to develop antibiotic stewardship strategies and inform rational antimicrobial treatment. 29 Therefore, the potential of COVID-19 to increase AMR must be critically appraised.28,29
African countries are not exempted from the impact of COVID-19 on health security. 13 The increase in the rate of COVID-19 morbidity could further augment AMR on the continent. It is important to consider the deleterious effect of COVID-19 on AMR arising from improper or suboptimal antimicrobial prescribing. 14 Although bacterial and fungal co-infections in COVID-19 patients is evidently low, there is a substantially high prescription of broad-spectrum antimicrobial agents. 28 Antimicrobials occupy temporary roles in the care of patients with COVID-19 10 ; firstly as one of the potential drug candidates being explored as possible direct therapies for SARS-CoV-2 25 and secondly as empiric or targeted drug therapy of bacterial infections secondary to COVID-19 or associated with clinical care. 14 The pandemic has disrupted routine infection control practices and attention is now focused on the diagnosis of SARS-CoV-2 rather than culture identification of resistant bacteria and isolation of patients with MDR organisms. 10 The risk of exposure of healthcare personnel to SARS-CoV-2 aerosolized droplets may also limit diagnostic testing procedures for superinfection in hospitalized patients. 20 It has been suggested that the increased demand on personal protective equipment for COVID-19 patients coupled with increased pressure on limited health resources may exhaust the needed resources in the prevention and management of other infectious diseases. 10 This can have a significant impact on capital and human resources required for infection prevention and control, leading to elongation in healthcare management and extended antimicrobial drug use due to postponement of non-emergency surgical procedures. 10
Secondary Bacterial Infections, COVID-19 Pandemic, and Past Pandemics
Secondary bacterial infections play a crucial role in mortality and morbidity associated with several respiratory viral infections.31,32 Bacterial superinfections often occur during outbreaks of viral infections and often leads to poor outcomes and fatal clinical complications.28,33 Classical examples are the 1918 and 2009 swine flu pandemics both caused by HINI influenza virus, 1957 influenza pandemic caused by H2N2 influenza virus, 1968 Hong Kong flu caused by H3N2 influenza virus, and the 2002 Severe Acute Respiratory Syndrome Coronavirus, 2009 Middle East Respiratory Syndrome Outbreak, and most recently the COVID-19 pandemic.31-33 Over the past 2 decades, viral respiratory tract infections have contributed significantly to mortality in both developed and developing countries, and in most cases, deaths do not result from direct viral damage alone but instead most cases of deaths are associated with secondary bacterial infections. 34
Table 1 shows common respiratory viral-bacterial coinfections.
Common respiratory viral-bacterial coinfections.
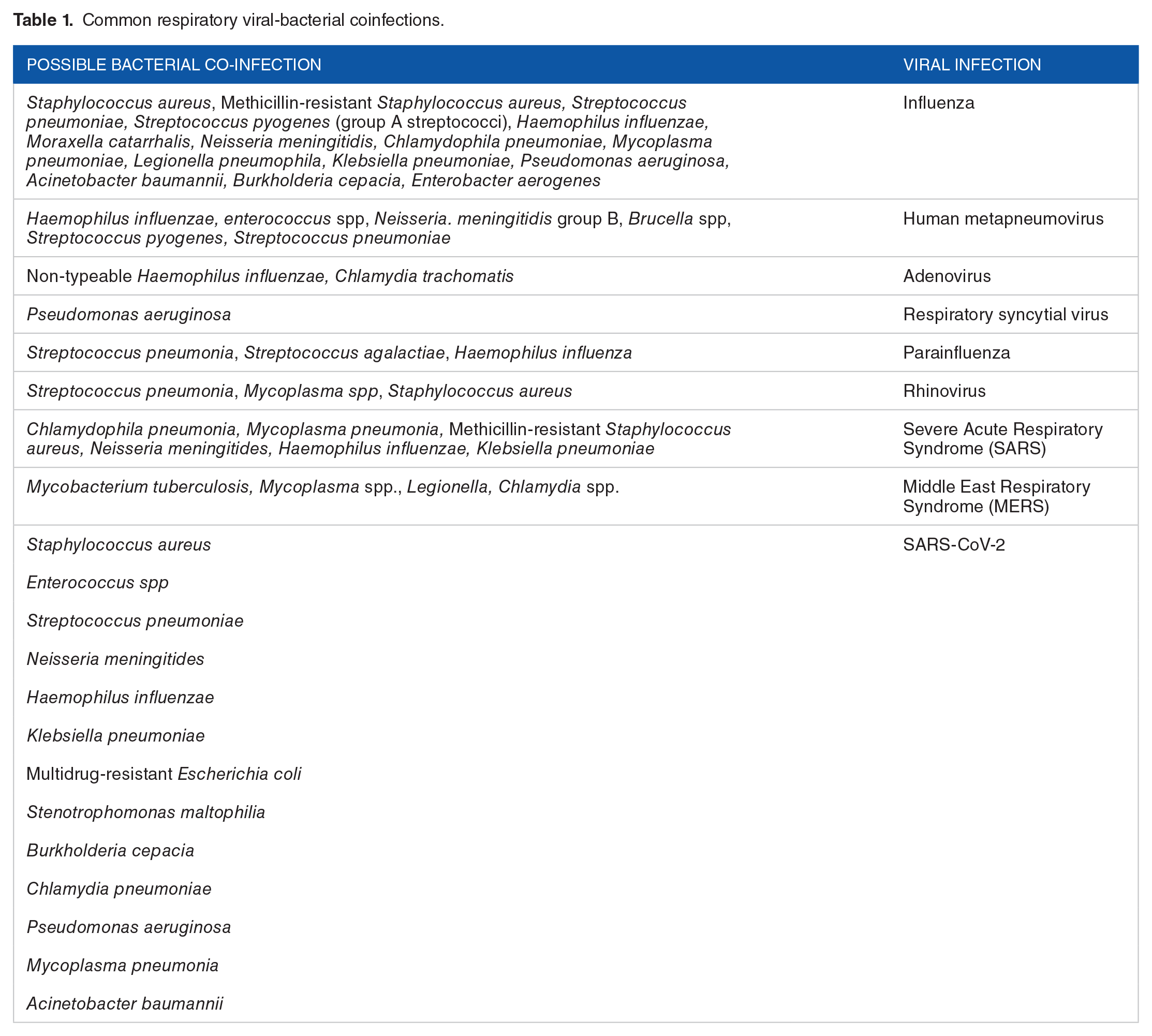
Despite the importance of these superinfections in large outbreaks of respiratory viral infections, they are often understated and understudied. 31 During the influenza pandemic in 1918, there was an estimated death toll of 40 to 50 million, many of which were as a result of pneumonia caused by Streptococcus pneumonia. 31 This implies that secondary bacterial super-infections were highly associated with severe disease. 35 In the 2009 HINI influenza pandemic, 1 in 4 cases of the severe presentation was complicated by a secondary bacterial infection and associated with significant mortality, between 29% and 55% were linked to secondary infections. 36 The disease burden of the present COVID-19 pandemic is greater than both Middle East Respiratory Syndrome and Severe Acute Respiratory Syndrome combined 37 with over 12 million cases and over 500 000 deaths as of 10th of July 2020. A study has shown a correlation between COVID-19 and secondary bacterial infections, with up to 15% of the cases being complicated by secondary bacterial infections and deaths occurring in more than half of these cases. 18
However, the specific mechanism by which a virus can cause the development of secondary bacterial infections is not clearly understood. 35 Some of the possible mechanisms for the bacterial coinfection with viral respiratory infections were highlighted in Table 2. Most recently, studies have investigated the outcome of secondary bacterial infections due to COVID-19.35,38 The epidemiology and pathophysiology of these infections in COVID-19 patients are not clear. Nevertheless, it is known that after a viral infection, the host defense against bacteria in the respiratory tract can be destroyed by mechanical or immunological mechanisms—the impairment of host epithelial cells or through the harmful effect of the virus on the immune system. 35 In a recent study carried out in China, 32 the prevalence of secondary bacterial infections in the Intensive Care Unit was 13.9% (5/36). The incidence of these infections was associated with mechanical ventilation, which disrupted normal airway barriers and aided the entry of opportunistic pathogens. 32
Some possible mechanism of action of viral-bacterial coinfections. 35 .

In Wuhan, China, the clinical features of patients infected with COVID-19 was also studied by Huang et al. 1 Out of 41 individuals infected with COVID-19, all patients had pneumonia and were treated with antibiotics. Complications mostly observed were respiratory distress syndrome (29.0%), acute cardiac injury (12.0%), and secondary infection (10.0%). Moreover, 32.0% of patients were admitted to the intensive care unit. 1 Another study carried out on patients over 60 years old with COVID-19 showed that bacterial infection (42.8%), hypertransaminasemia (28.7%), and acute respiratory distress syndrome (21.0%) were the most common complications observed. 39 Similarly, Chen et al 30 carried out a retrospective study in a cohort of 799 patients. The study revealed that sepsis and acute respiratory distress syndrome occurred in all 113 deceased patients and the most common complications observed in COVID-19 positive patients. Acinetobacter baumannii and Klebsiella pneumoniae were among the pathogens identified in their cultures. 30 Furthermore, a retrospective study of 918 COVID-19 patients conducted in Wuhan, China also showed that 7.1% of hospitalized patients had a fungal or bacterial co-infection with pneumonia (32.3%), bacteremia (24.6%), and urinary tract infections (21.5%) observed to have the highest incidence. 40 Another review of published studies on hospitalized COVID-19 patients identified that while 72% (1450/2010) of patients received antibiotics, only 8% (62/806) demonstrated superimposed bacterial or fungal co-infections. 28
Antibiotics, while not affecting SARS-CoV-2 itself, are used in the treatment of most serious cases of the disease, as empirical treatment for the disease or to prevent or treat secondary bacterial infections. 41 However, it is noteworthy to mention that any surge in the use of antibiotics could potentially contribute to the rising menace of AMR. 41 In fact, many of the commonly identified pathogens in secondary bacterial infections including Staphylococcus aureus, Streptococcus pneumoniae, Neisseria meningitides, Haemophilus influenzae, Klebsiella pneumoniae, and members of the genus Proteus, Enterobacter, and Citrobacter spp. are found in hospitals. 33 This effectively makes many cases of secondary bacterial infections nosocomial. 33 Recent studies have also shown that certain infections experienced by patients with COVID-19 are caused by drug-resistant organisms, such as multidrug-resistant Escherichia coli, Enterococcus, Chlamydia pneumoniae, Klebsiella pneumonia, Pseudomonas aeruginosa, Mycoplasma pneumonia, and extended-spectrum beta-lactamase.39,40,42 Indeed, Fu et al 32 noted that there was a high likelihood that broad-spectrum antibiotics administered previously altered mucosal flora and selected specific resistant bacterial organisms such as carbapenem-resistant B cepacia and S. maltophilia. Some bacteria implicated in coinfection with COVID-1930,32,33,39,40,42 are listed in Table 1.
In the absence of sufficient vaccine coverage for the disease, secondary bacterial infections will continue to play important roles in this current pandemic, therefore adequate attention needs to be paid to it, to curb unnecessary use of antibiotics. 33 Therefore, targeted antimicrobial therapy might play a significant role in the treatment of confirmed or suspected secondary bacterial infections in COVID-19 patients. 35 This further highlights the importance of continuous research and development of new antibiotics as the antibiotic pipeline is drying up and we need new antibiotics to help fight current diseases and emerging ones. In addition, effective infection prevention and control practices will significantly assist in reducing the risk of bacterial infections caused by multidrug-resistant pathogens in COVID-19 patients. 10
Promoting Antimicrobial Stewardship Amid the COVID-19 Pandemic
The impact of COVID-19 on AMR has not been critically examined globally.10,22 This could spell danger on many medical procedures and services as a result of compromise arising from impending AMR. This emphasizes the need for antimicrobial stewardship as COVID-19 may be successfully defeated with the advent of effective medications or development of vaccine.43,56 In the face of the pandemic and due to the absence of specific therapeutic agents for the treatment of COVID-19, antibacterial therapy is being employed in different ways to combat the disease.44,45 For instance, the antimicrobial agents—Azithromycin and Hydroxychloroquine combination was one of the very first therapies employed for COVID-19 management. 46 However, there is limited data on efficacy and potential adverse effects of this combination on health outcomes. 47 A substantial proportion of COVID-19 patients present with fever and cough. 48 Notwithstanding the viral origin of COVID-19 and inadequate testing capacity, a standard response by physicians is to initiate treatment with antibiotics since cough, fever, and radiological infiltrates are key components of bacterial community-acquired pneumonia which requires antibiotic therapy. 48 In COVID-19 patients, antibiotics are utilized in the treatment of reported or suspected secondary bacterial infections. 49 The reason for antibiotic therapy for patients with COVID-19 is not far-fetched as it appears to be centered on previous experiences of bacterial superinfection of influenza. 21
The specific occurrence of bacterial superinfection in COVID-19 is particularly unclear; although there are anecdotal records of reported bacterial superinfections, the occurrence seems to be relatively lower than in serious influenza.14,50 Guidelines on the use of antibiotics in patients with suspected or confirmed COVID-19 differ by region, with certain guidelines likely to be targeted to the common antibacterial therapy for community acquired pneumonia used in that region.6,51 For instance Piperacillin-tazobactam is used in North-American countries, fluoroquinolones in Turkey, and carbapenems-fluoroquinolone combinations in Italy. 6 This vacillating use of antibiotics is further worsened by the fact that health professionals involved in the treatment of COVID-19 patients have a significantly heavy workload and exhibit elevated levels of stress and might therefore not be able to maximize their roles in the review of medication therapies and modulation of clinical practice recommendations 52 which can pose a barrier to antimicrobial stewardship. Unless antimicrobial usage is properly addressed in the course of the COVID-19 pandemic, prolonged unnecessary antibiotic use may bring enormous pressure on the increasingly stressed antimicrobial stewardship programs. 21
To successfully combat AMR, it is important to emphasize the rational prescribing of antibiotics as a part of antimicrobial stewardship (Figure 1). 10 Antimicrobial stewardship programs can support COVID-19 response while preventing impending antimicrobial resistance due to issues associated with poor prescribing practices 53 and continuous engagement of infection subspecialties is required for antimicrobial stewardship response in COVID-19 patients. 10 As such, clinicians including infectious disease physicians, nurses, clinical pharmacists, and other healthcare professionals should be involved in promoting antimicrobial stewardship. Hospitals with functional antimicrobial stewardship systems should continue to identify problems with antimicrobial prescribing even when not easily detectable in their absence. They should also be involved in the development of guidance for their facilities.54,55

Potential antimicrobial stewardship activities focused on COVID-19.
Advocacy for continued stewardship and outlining measures to limit misuse of antibiotic during the pandemic is necessary.29,56,57,58 Primarily, it is proposed that antibiotics be reserved for patients with most severe presentations (eg, high oxygen demand and progressive respiratory failure). It has been suggested that various guidelines must focus on maintenance of good infection control, antimicrobial stewardship, and robust surveillance for antimicrobial usage and resistance.59,60,61,62 Also, microbiological test should be obtained before antibiotics use and when bacterial superinfection probability is low, antibiotic use be evaluated and stopped. Moreover, a switch to oral from parenteral antibiotics should be considered as soon as it is tolerable. Routine azithromycin use due to anecdotal evidence should be dissuaded, antibiotics be used for minimum duration possible and prophylactic antibiotics use be shunned. 21 Procalcitonin measures can also be leveraged as biomarkers to reduce antibiotics use. 28
It is increasingly important to learn now from the current pandemic and prepare to prevent the resultant impact of AMR. The spread of COVID-19 across countries coincides with the critical time for antimicrobial development. There is a need to look into the post-COVID-19 era when antimicrobial resistance will remain a challenge to be overcome.10,22 The pandemic has signaled shortcomings in health systems globally including preparedness and control of drug resistant organisms. 10 However, it has provided valuable insights into various opportunities of antimicrobial stewardship programs in emerging disease outbreak. 53
There is limited evidence on effective and achievable antimicrobial stewardship strategies in many resource-poor settings due to numerous barriers facing implementation. 11 The unpredictable situation of the pandemic has necessitated adaptability of antimicrobial stewardship in the unstable climate. It is expected that COVID-19 will necessitate antimicrobial therapy in a significantly high number of patients. 19 Consequently, antimicrobial stewardship should focus on stringent approaches in reserving antibiotic therapy to patients in which serious bacterial infection is suspected/confirmed. Empirical antimicrobial therapy should be initiated while considering the source of infection acquisition and local pattern of AMR. 46 There is a need to contextualize evidence-based strategies for antimicrobial prescribing and antimicrobial stewardship in order to attain optimal treatment outcomes and avoid unintended consequences of antimicrobial prescription. 49 Evidence also reveals that AMR is under-investigated and under-reported in many limited resource settings due to limited or unavailability of diagnostic tests and lack of microbiology technical resources. 11 These have led to the inability to test clinical isolates for antimicrobial susceptibility, which has resulted in excessive use and misuse of antibiotics, increasing the phenomenon of AMR. 11
Lesson Learned and Recommendations
Table 3 shows some of our recommendations.
Recommendations.

The COVID-19 pandemic has been associated with great involvement of governments’ collaboration with health care teams globally to successfully tackle this major threat to public health. 57 In addition, attention is now focused on preparedness as opposed to response via improved infection prevention and control strategies. This has also further drawn attention to the possible risk of antimicrobial resistance and the need for antimicrobial stewardship engagement. 53 The incidence of COVID-19 in LMICs and other countries of the world have brought to fore the gaps in the healthcare systems and limited epidemic preparedness for outbreaks. This signals the need to consider more efforts in terms of promoting epidemic preparedness globally. 13 While it is still inconclusive whether microbicides from soaps, sanitizers, and disinfectants may lead to antimicrobial resistance, 58 targeted hygiene presents a possible avenue for improving antimicrobial stewardship as it could further prevent transmission of other infections, usually requiring antibiotics management.59,60
Potential stewardship interventions to support reduced antimicrobial prescribing during the COVID-19 pandemic urgently require consideration.54,62 It is noteworthy that overly restrictive stewardship may limit the uptake of new antimicrobials in favor of cheaper, less effective alternatives. To this end, it is important to also focus on encouraging flexibility where necessary. 29 Traditional markers used to support antimicrobial prescribing decisions, such as vital signs, blood tests like white cell count and C-reactive protein, and imaging tend to be abnormal in SARS-CoV-2 infection. This makes the decision making surrounding the requirement for empirical antimicrobial prescription and use challenging. 18 Procalcitonin has, however, been demonstrated to differentiate between bacterial and viral infection and supports early cessation of antibiotics in confirmed bacterial infection with no effect on patient mortality.18,46 This could prove to be an important tool to support reduced antimicrobial use. The pandemic, which has led to high mortality since it was first discovered has now more than ever left clinicians faced with the dilemma to prescribe antibiotics or not in-patient management. 63 Whilst focusing attention on mitigation of the threat of COVID-19 is of utmost significance, it is imperative that medical, public health, policy, and political communities keep sight on curbing AMR and improving antimicrobial stewardship principles amidst dwindling new antibiotics development.10,64
Conclusion
COVID-19 pandemic has further brought to fore the deficiencies in health systems around the world. This comes with a lot of lessons, one of which is that despite the advances in medicine; we remain incredibly vulnerable to infections with limited or no standard therapies. This is worth thinking of in the context of AMR, as the resistant pathogens are evolving and leading us to the era of untreatable infections. There is a necessity for continuous research into understanding and controlling infectious agents, as well as the development of newer functional antimicrobials. Investing in strengthening health systems and preparedness for pandemics and other infectious disease outbreaks is one of the best ways to contain AMR. The current pandemic comes with an important lesson on the need for adequate, strengthened, and efficient surveillance and reporting systems. This is also useful for effective AMR control. Understanding the extent and the landscape of AMR is essential for an effective fight against it. Nevertheless, there will be a period after COVID-19, so we should not lose sight of the challenges that would persist and that this pandemic might potentially exacerbate.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Yusuff Adebayo Adebisi conceptualized the study. Aishat Jumoke Alaran, Melody Okereke, Gabriel Ilerioluwa Oke, Oladunni Abimbola Amos, Omotayo Carolyn Olaoye, Iyiola Oladunjoye, and Azeez Yusuff Olanrewaju conducted the review and analysis under the guidance of Yusuff Adebayo Adebisi and Don Eliseo Lucero-Prisno III. All authors wrote the first draft of the paper. Yusuff Adebayo Adebisi, Nelson Ashinedu Ukor, and Don Eliseo Lucero-Prisno III suggested important improvements to the paper. All authors read and approved the final manuscript.
