Abstract
From the aerial parts of Ruta chalepensis L., grown in Jordan, two furanocoumarins (bergapten and chalepensin), one flavonoid glycoside (rutin) as well as several minor compounds have been isolated. The structural elucidation of these compounds was established based on spectral data (UV, IR, MS, 1 H-NMR and 13 C-NMR). In Jordan, R. chalepensis is recommended for the treatment of rheumatism, mental disorders and menstrual problems. Fresh and dried leaves are used as flavoring agent in food and beverages. Antiplatelet activities of the crude methanolic and ethylacetate extracts in addition to the three isolated major compounds were measured by the aggrometric method according to Beretz and Casenave. Optical aggregometer connected to dual channel recorder was used for measuring aggregation. Both, ethylacetate and methanol extracts inhibited ADP- induced platelet aggregation (ADP-IA) of human blood. However, only ethylacetate extract was able to induce 50% inhibition of collagen-induced platelet aggregation (Co-IA) platelet rich plasma. Bergapten was more active against ADP-IA compared to chalepensin while the latter was more active against Co-IA compared to bergapten.
Introduction
Ruta chalepensis is widely distributed in the Mediterranean and the Middle East region. The aerial part of this plant is used in traditional herbal medicine in many countries for treatment of a wide variety of diseases. Orally, it is used as analgesic, antipyretic, anti-inflammatory, in menstrual problems, antispasmodic, anthelmintic and abortifacient (Ciganda and Laborde, 2003), relief of rheumatic pain and mental disorders (Ageel et al. 1989; Iauk et al. 2004; Oran et al. 1998). Topically, it is used as hair tonic, insect repellent and for snake-bite (Al-Khalil, 1995). In Jordan, R. chalepensis; commonly known as Fijin or Sathab; is used in traditional herbal medicine and it is also used as a flavouring agent in food and beverages (Abu-Hamdah, 2001).
Pharmacological studies carried out with different extracts of Ruta species reported the anti-inflammatory properties (Atta and Al-Kofahi, 1998), antipyretic (Al- Said et al. 1990), antifertility activity (Ulubelen et al. 1994), antifungal (Ali-Shtayeh et al. 1999) and antibacterial effect (El Sayed et al. 2000). Contact dermatitis and photoxicity characterized by erythema, itching and burning of the skin is a common toxic effect, which is usually associated with all Ruta species (Gijon et al. 1995; Heskel et al. 1983). Mutagenicity of this plant has been found to be mediated by furoquinoline alkaloids and furocoumarins upon prolonged exposure to sunshine (Chimmer and Kuhne, 1990; Paulini et al. 1987).
Phytochemical investigations of R. chalepensis reported isolation of furanocoumarins, acridone and quinoline alkaloids, terpenoids, saponins and tannins (El Sayed et al. 2000; El Tawil et al. 1980; Gray and Watermann, 1978; Husain and Devy, 1998; Ulubelen et al. 1988). However, published literature did not report anti-platelet activity of R. chalepensis herb or its constituents. Hence, the present work was designed to investigate the ability of methanolic and ethylacetate extracts as well as the isolated furocoumarins and flavonoid from the aerial parts of R. chalepensis to inhibit ADP- and collagen induced platelet aggregation of human blood.
Materials and Methods
Plant Material
Dried R. chalepensis herb, grown in Jordan, was purchased from a local herbal outlet in Amman. Taxonomic identity was established by comparison with herbarium sample and microscopic key elements using descriptive botany (Abu-Hamdah et al. 2005) and was confirmed by Professor S. Oran, Department of Biological Sciences, Faculty of Science, University of Jordan. A voucher specimen was deposited in the Department of Pharmaceutical Sciences, Faculty of Pharmacy, University of Jordan.
Extraction Procedures
Six kilograms of powdered plant material of the purchased plant was extracted at room temperature by maceration in 96% ethanol for four weeks. Concentration of the aqueous ethanolic extract under reduced pressure provided 700 gm of crude extract. TLC analysis of the extract detected all previously reported compounds in addition to the detection of flavonoids.
500 gm of the dried crude extract were chromatographed on silica gel column (230–400 mesh) using petroleum ether (Pet. Ether), CHCl3, ethylacetate (EtOAc), acetone (Me2CO), butanol (BuOH), methanol (MeOH) and water as eluents. Butanol fraction was chromatographed on silica gel column (70–230 mesh) and was eluted with petroleum ether followed by gradient elution with Pet. Ether: CHCl3 and CHCl3: MeOH. Collected fractions were analysed by TLC in ether: EtOAc (70:30) and toluene: EtOAc: HCO2H (5:4:1). Examination of the TLC plates under UV 365 nm revealed that Pet.ether: CHCl3 and CHCl3: MeOH fractions exhibited strong blue and green fluorescence. Both fractions were subjected to repeated recrystalization in MeOH to isolate two white needle like crystalline compounds. Isolated compounds (1) and (2) were soluble in methanolic NaOH producing yellow coloured solution intensified upon heating.
Acetone and methanol fractions from the crude extract silica gel column were pooled together, based on their similarities after TLC analysis, chromatographed on silica gel column (70–230 mesh) and eluted with a gradient BuOH:MeOH. Flavonoids were detected in collected fractions when analysed by TLC plates after spraying with NP/PEG and FeCl3 reagents. Flavonoid-rich fractions were chromatographed on silica gel column and eluted with CHCl3: MeOH and MeOH: H2O. A yellow crystalline substance (3) was purified by repeated recrystalization under low temperature. All isolated compounds were analysed by UV, IR, EIMS, 1 H-NMR and 13 C-NMR for structural elucidation.
Platelet Aggregation
Ethyl acetate, methanolic crude extract fractions and the three major isolated compounds were tested for their ability to inhibit ADP- and collagen-induced platelet aggregation in vitro. Platelet-rich plasma (PRP) and platelet-poor plasma (PPP) were obtained from male volunteers’ venous blood by free flow prepared according to the procedure reported by Williamson et al. (1981). Acetyl salicylic acid (Aspegic®) solution of 13.9 mg/ml was used as 100% platelet aggregation inhibitor standard. ADP 10 µM and collagen 1 µg/ml solutions (Helena Laboratories, U.S.A) were used as standard 100% platelet aggregation inducers. Test samples were prepared in HPLC-grade methanol and further diluted with TRIS buffer to adjust to the isotonic pH value. 450 µl PRP were incubated at 37°C for 4 minutes stirred in a cuvette prior to the addition of the platelet inducer and/or the extract or compound under investigation. Platelet aggregation was recorded over 8 minutes period measured by a turbidimetric dual channel aggregometer and coagulation tracer (APACT 2) as described by Beretz and Casenave (1991). The change in absorbance was recorded as PRP was stirred with the test reagents. Light transmitting capacity of plasma measured in percent was used to detect aggregation (Craig and Stitzel, 1994). Percentage of aggregation was calculated in reference to 100% aggregation induced by ADP or collagen while percentage inhibition of platelets aggregation was calculated in reference to 100% inhibition induced by Aspegic®. Final concentration of methanol was never >0.5% in all tested solutions.
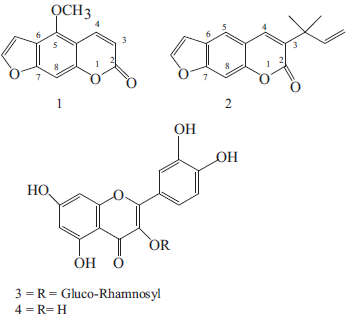
Chemical structures of the isolated compounds from the aerial parts of R. chalepensis.
Results
UV fluorescent compounds (1) & (2) purified by repeated recrystalization were soluble in ethanolic NaOH producing intense yellow color upon heating which indicated the opening of the lactone ring as reported by Murray (Murray et al. 1982) is consistent with coumarins. Spectral data of 1D 1 H- and 13 C-NMR, 2D-HETCOR and HMBC NMR together with EIMS, IR (KBr) and UV were consistent with bergapten and chalepensin respectively (Abilio, 1985; Brooker et al. 1967; Harkar et al. 1984). Assignment of the chemical shifts for both compounds confirmed by 2D HECTOR and HMBC NMR were in accordance with calculated shifts using Internet Chemistry software drawing, Modelling and Information Program Ultraversion 6, 1985–2000 (Cambridge Soft Cooperation).
The isolated flavonoid was analysed by TLC and compared to with rutin reference sample (Fluka, Switzerland) producing same Rf value 0.92 in EtOAc: HCO2H: glacial HOAC: H2O (100:11:11:26) and orange spot when sprayed with NP/PEG and FeCl3. Acid hydrolysis under reflux provided quercetin. Spectral data of both compounds were in full agreement with rutin (3) and quercetin (4) respectively.
Isolated furanocoumarins exhibited significant inhibitory effect on platelet aggregation induced by ADP and collagen as summarized in table 1.
Inhibitory effect of platelet aggregation by R. chalepensis (Rc) fractions and isolated compounds.
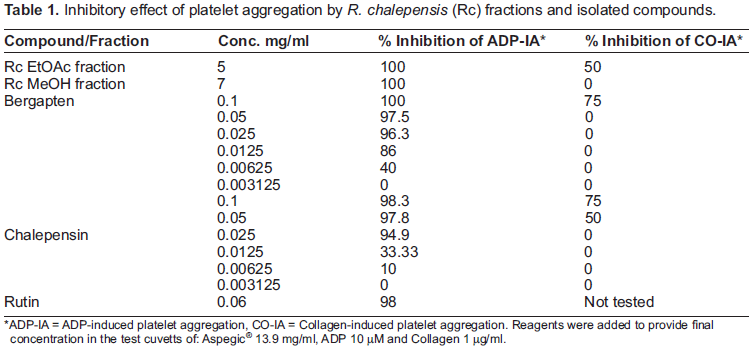
ADP-IA = ADP-induced platelet aggregation, CO-IA = Collagen-induced platelet aggregation. Reagents were added to provide final concentration in the test cuvetts of: Aspegic® 13.9 mg/ml, ADP 10 µM and Collagen 1 µg/ml.
The results are the mean value of 2 experiments carried out in duplicates using different blood samples.
Discussion
Diseases of the cardiovascular system constitute major causes of death throughout the world (Ulrichts et al. 2004). Conditions such as hypertension can lead to other types of diseases, such as stroke, kidney and heart diseases, and need to be treated, usually life-long (Williamson et al. 1996). It is well known that many pathologic conditions of the cardiovascular system is affected by an increase or dysfunction of the blood platelet activity, mainly in arterial thrombi (Golino et al. 2005), since these play a major role in thrombotic disorders (Andrioli et al. 1996, Hernandez Hernandez et al. 1997). Several edible plants and spices have the reputation being used to prevent or at least to decrease the incidence of different vascular diseases based on their ability to prevent platelet aggregation. Garlic, onion, ginger, strawberries and tomatoes are well known and well studied representatives in this aspect (Rahman and Billington, 2000; Goldman et al. 1996; Chrubasik et al. 2005; Dutta Roy et al. 2001). Compounds responsible for the inhibition of platelet aggregation have been determined and reported, such as sulphurous compounds in garlic and onion, gingerol and zingiberine in ginger and phenolics in strawberries (Naemura et al. 2005). Several medicinal plants with diverse active constituents such as alkaloids, flavonoids, tannins and coumarins have shown promising results in inhibition of the platelet aggregation (Afifi and Aburjai, 2005; El Haouari et al. 2006; Mekhfi et al. 2004; Mekhfi et al. 2006; Sheen et al. 1996; Teng et al. 1992). In the present study, the inhibitory effect induced by two isolated coumarin derivatives, namely bergapten and chalepensin appeared to be concentration-dependent in agreement with other similar studies using coumarins (Teng et al. 1992; Hoult and Paya, 1996).
At the highest tested concentration of 100 µg/ml, bergapten was able to completely abolish the aggregation while chalepensin was able to induce 98.3% inhibition. At the lower concentration of 50 µg/ml both compounds were able to induce similar inhibitory effect of 97.5% and 97.8% respectively. Aggregation inhibitory activity was still significant at the lower concentration of 25 µg/ml, 96.3% for bergapten and 94.9% for chalepensin. A dramatic difference in inhibitory action was detected at 12.5 µg/ml of the tested coumarins. Chalepensin's ability to inhibit platelet aggregation was reduced to just 33.3% compared to 86% for bergapten. 40% inhibition of ADP-induced platelet aggregation was recorded for bergapten and 10% for chalepensin when added to PRP at 6.25 µg/ml. Inhibitory effect was completely abolished at the lowest tested concentration of 3.125 µg/ml for both compounds. IC50 for both compounds was calculated, using Sigma Plot version 7 software and found to be 7.07 µg/ml for bergapten and 14.71 µg/ml for chalepensin. This observation suggests that the methoxy group at C5 in bergapten might contribute to the inhibitory effect on ADP-induced platelet aggregation. However, collagen-induced platelet aggregation was less significantly inhibited by the tested furanocoumarins. At the highest tested concentration of 100 µg/ml only 75% inhibition was recorded for both compounds. In addition, furanocoumarins inhibitory effect on the collagen-induced aggregation was one minute delayed compared to instant inhibitory effect on ADP-induced aggregation. At 50 µg/ml chalepensin induced 50% inhibition on collagen-induced aggregation while bergapten was inactive. The presence of the dimethyl allyl side chain at C3 in chalepensin appears to play an inhibitory role on collagen-induced platelet aggregation. Further studies should be carried out to investigate this theory.
Rutin induced 98% inhibition on ADP-induced platelet aggregation at 60 µg/ml which is in accordance with previously reported data in literature about quercetin related flavonoids inhibitory action on washed rabbit platelets (Chung et al. 1993).
Based on the above in vitro test data, we can suspect possible interaction between furanocoumarins and flavonoids-rich herbal preparations when used concurrently with therapeutic anticoagulants. Such interaction might impose a life-threatening condition or at least interfere with concurrent anticoagulant therapy. Further in vitro and in vivo investigations and close patient follow up are recommended.
