Abstract

Introduction
The history of the European Union goes back to the 1950's, when a new approach in economic cooperation started between some states of the continent. As it gradually developed, the four basic freedoms (free movement of persons, goods, services, and capital) assured by the participating countries became the basis of the cooperation. The creation of the European Union in 1992 brought forth a wider sphere of authority than already existed regarding the common economic issues. Some of the provisions related to health care even gained a great publicity, like the introduction of the E111 card or the European Court decision concerning the Simap and Valencia case (as a result of the latter, the member states are bound to regard the time spent on call by doctors in primary health care teams in its entirety as working time)(1). Although matters connected to alternative medicine generally fall within the authority of the member states, policies of the EU do have a certain impact in this area as well. Since a specific legal regulation is absent or incomplete in several countries, an indirect unifying impact of EU activities might emerge and reach a significant level (2)(3).
Alternative medical practices and products are widely used and applied by the citizens of the member states (4). Due to this increasing acceptance some might experience a growing EU activity, like speeches in the parliament, consultations with EU institutions, and advisory panels (2). However promising these manifestations might be at a first glance, all but the directives are not binding for the member states. Up to now, beyond the favoring voices no specific law or significant financial support appeared. Two programs, starting in 2007 might become a turning point in the relation of the EU to alternative medicine. First, the “Programme of Community Action in the Field of Health 2007–2013“ attempts to support mobility between patients and providers of the member states and stresses the assurance of patients’ free choice within health care (5). Second, the Seventh Framework Programme (FP7)–-that will be approved before 2006–-enables the financial support of research in the case of alternative medicine also (6). The legal activity of the EU by means of its provisions or specific policies influences even unintentionally, or indirectly the developmental pathways of alternative medicine. Attempting to highlight some of them, the paper discusses the impact of education, research and migration policies on alternative medicine.
The Legal History of eu and Alternative Medicine
Alternative medicine's first appearance in the case law of the European Court of Justice (EU Court) was with reference to the freedom of movement of persons, and freedom of establishments, wherein the question was raised whether or not to reserve the practice of medicine to allopathic doctors:
[…] in criminal proceedings brought against Marc Gaston Bouchoucha. Mr Bouchoucha, a French national, holds a French State diploma as a masseur-kinesitherapist and a diploma in osteopathy issued on 1 October 1979 by the European School of Osteopathy, Maidstone, Great Britain. He also holds the diploma of “Doctor of Naturopathy” from the London College of Applied Science. However, he holds no diploma, certificate or other qualification entitling him, pursuant to Article L 356-2 of the Code de la santé publique (French Public Health Code), to exercise the profession of doctor. […] In the absence of harmonization at Community level regarding activities which fall solely within the scope of the practice of medicine, Article 52 of the EEC Treaty does not preclude a Member State from restricting an activity ancillary to medicine such as, in particular, osteopathy exclusively to persons holding the qualification of doctor of medicine. (7)
This former decision of the EU Court might be regarded retrospectively as a position taken up against the processes of harmonization in this field, with confirming the authority of the member states for providing practice licenses only for persons with a medical degree. Another important decision made by the EU Court in 2005 suspended the French regulation–-that required permission for the import of homeopathic products from member states–-with reference to unified pharmaceutical market (8).
European Deputy Paul Lannoye's proposal in 1994 that was made for the European Parliament Committee on the Environment, Public Health, and Consumer Protection,–-and unfortunately didn't attained so far–-addressed the issue of alternative medicine (9). In his proposal, Paul Lannoye asked for provisions:
[…] for complementary/alternative medicine within social security systems, the incorporation of complementary/alternative medical systems into the European Pharmacopoeia, an end to prosecutions of non-allopathic practitioners in countries where the practice of medicine is the exclusive domain of allopathic providers, and a pan-European system of recognition and regulation of complementary/alternative medical practitioners along the lines of the British Osteopath and Chiropractor Acts. He also requested a research budget of 10 million Euros per year for five years. At the last moment, the European Parliament cancelled the vote on the proposal.
The European Parliament adopted a resolution about the situation of alternative medicine and called upon the Commission to take measures for accepting alternative medicine after having accomplished the required investigations, and to establish research projects in order to quantify the effectiveness of alternative medicine (10). After eight years, in 2005, the proposal's intention came to be realized. The outcomes of the previously launched research could give answers to the questions posed by the Parliament in many respects. It makes a detailed comparative analysis regarding the jurisdiction, reimbursement and supervision of alternative medicine between member states, and EFTA countries. The work also compares the forms of regulation that has actual validity and also indicates future possibilities. This research was partly funded by the Quality of Life section of the 5th Framework program (11). The Council of Europe has stated that the common European approach to non-conventional medicine based on the principle of patients’ freedom of choice in health care should not be ruled out, and “the Assembly called on the member states to support and speed up the comparative studies and research programmes currently under way in the European Union to disseminate the findings widely“ (12).
The recommendations and the positions taken up by the EU are not binding for the member states, unlike directives that fall within the secondary legislation with a binding force respecting the results. The Court decisions are binding exclusively for the parties to the case. Legislative pieces, called directives only embrace the purpose to be achieved; the manner could be independently chosen by the member states. As a consequence of the basic principle of free movement of goods, the pharmaceutical market became open by establishing the free movement of pharmaceutical products as well. Specific regulations have taken place in 1994 in order to unify the trade of medicinal herbs. Those directives that aimed to assure the European market of homeopathic products came into force in January 1st 1994. The 2004/24/EU recommendation aims to create homogeneous European market for medicinal plants, referring to similar quality requirements in case of the compounds of medicinal plants as in any other registered medical products. However, in the case of medicinal plants the member states might provide a simplified procedure that avoids having to prove effectiveness through trials. On the basis of the simplified procedure products could enter the market, according to the two most important requirements, if these are secure, and their usage in the EU member state started at least 15 years ago. Organizations of alternative medicine reacted with anxiety to the new directives and called forth protests in several states. In the case of supplementary diets (vitamins and minerals) the 2002/46/EC directive is responsible for harmonizing the regulation (which was implemented in 2005). Two appendixes were attached to the directives listing all of the products that could be put into circulation (13).
EU Related Organizations
There are quite a number of organizations of alternative medicine operating within the member states, but most of these have negligible relation with EU institutions. In this respect some of the outstanding organizations must be mentioned. Firstly, The European Forum for Complementary and Alternative Medicine (EFCAM) is an open European forum–-with a head-quarter in Brussels–- for those having some kind of relation with alternative medicine (14). The forum was established in 2004 by 20 different organizations from the alternative medical field. This forum operates within the framework of the European Public Health Alliance (EPHA) that organizes conferences, discussions for non-governmental organizations working in the field of health care (European Parliament Health and Consumer Intergroup). One of their tasks is to provide reports for the European Parliament about the opinions, suggestions that emerged. One of these appointments was held in November 2005 under the title of “Health Challenges and Future Strategy.“ Another important approach runs under the title of “The European Research Initiative for Complementary & Alternative Medicine“ (EURICAM) which was established by 12 experts from the field of alternative medicine who are interested in research beside their general practice. Their main objective was–-soon to be achieved–-to adopt alternative medicine into the 7th Framework Program of the EU. EURICAM has its seat in Vienna, where a conference took place in November 2006 with the aim of defining the objectives and discussing the future agenda (15).
Education and Student Mobility
According to an objective that was set in 2002, the harmonization of educational systems within the EU should reach such a level that would open the door for the free movement of citizens, the acceptance of knowledge and degrees within the states through the assurance to perform work and further studies (16). The envisaged goal of harmonizing the educational training systems includes the harmonization of professional trainings and higher education.
The goal of the EU in relation to professional training is to improve the quality of education, and also to secure the mobility and transparency of professional training systems on a European level (17). In the field of higher education the goal was set to create the European Higher Education Area, which entails–-in accordance with the Declaration of Bologna–-the development of compatibility and mobility within the European systems of higher education. The basis of compatibility is provided by credits that could be collected during the three years period of graduate training (180 credits), and a following two years of master training (120 credits) (18). The harmonization of professional trainings is still in its initial phase that can be contrasted to the practical achievements in higher education. Trainings in alternative medicine differ significantly in the member states (9). The general categorization of professional trainings in the EU, regarding health care, consists of three levels of training: university degree mostly covering the acquisition of theoretical knowledge; two-four year training covering theoretical and practical knowledge; and training which is generally shorter and more or less practical in nature (19)(20). According to the different duration of trainings, different names and titles could be obtained.
The generally used titles and names for alternative medical practices that exist across the EU could cover quite different things regarding the educational background and acquired knowledge of the title's holder. An English herbal therapist most likely has a BSc of Herbal Medicine after a successful completion of a 4 years full time education, while her Hungarian colleague in title finished a two year adult education course. This EU wide divergence might endanger the proper orientation of patients in the system and also impede the appearance of the professional interest of alternative medicine on higher forums. The case is similar in the United States, where training levels could be hardly characterized by transparency. However, there was a proposal for the unification of the requirements and the strengthening of bachelor training (21). Burke's internet based study attempted to give a comprehensive survey about the bachelor training courses in alternative medicine within the U.S.A. However, following this methodology in the EU is not very likely to succeed, because of various languages of the continent. Thus, we used the World Higher Education Database for surveying the master and bachelor courses offered in alternative medicine in the EU. The database is built on the annually refreshed data using the member states “obligatory” reports that should cover all of the accredited courses in higher education. There is very little number of accredited alternative medical courses within the database (it can be reached via the term “paramedical”) (Table 1).
The higher education of CAM in the EU, 2005.
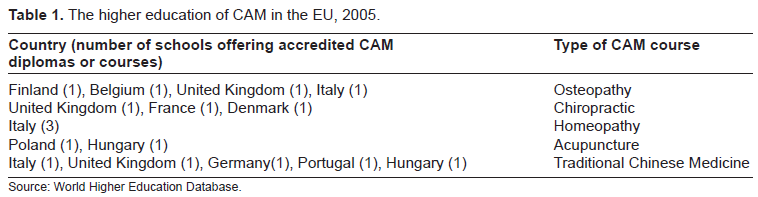
Source: World Higher Education Database.
Although it seems very likely that the collection is deficient, but we could still point to the missing educational background of the profession in the case of university level studies (22). The Bologna process is bound to the master and bachelor training; the assured benefits (Erasmus, Socrates program) could barely reach students of alternative medicine (23). Hopefully the educational policies of the EU will create in the near future a more transparent training system regarding alternative medicine as well.
Mutual Acceptance of Diplomas and Migration
The acceptance of degrees in alternative medicine in a given country basically depends on the local legal context of alternative medicine. If the practice of alternative medicine is bound to a diploma–- qualifies as a regulated profession–-then an employee from another state is also obligated to acquire that diploma, or have her “foreign” training officially recognized as appropriate. Two ways exists for the recognition of diplomas: sectorial or general.
There are seven professions that fall into the sectorial group: medical doctor, dentist, pharmacist, nurse, midwife, veterinary surgeon, and architect. The recognition of these professions is regulated by the sectorial directives of the EU. These directives enable the simple recognition of degrees, because all member states have already harmonized the above mentioned professions and qualifications. It follows from this that–-in the course of the procedure of recognition–-authorities do not examine the training behind the given diploma, but look whether the person satisfies all of the specified requirements of that profession in her native country.
The general system is based on two directives of the Council: 89/48/EC, and 92/51/EC. As a fundamental difference we must mention that the comparison of diplomas is performed by the authorities on an individual basis. They examine what activity is legitimatized by the certificate in the country of origin, and also compare the course of theoretical and practical training with the receiver country's training. If the authorities in the receiver country come to the conclusion that the qualifications are not adequately matching, then they can request: a certification of professional practice, require a period of adjustment, or a successful completion of an aptitude test (24).
The real chance of the mutual acceptance of diplomas is quite low at the present, due to the diversity in training that we have already mentioned. As an example, the bachelor qualification in homeopathy acquired in the U.K. certifies for practice; while in Hungary this activity requires a medical diploma with an additional two years of training in homeopathy (25)(9). There is no mobility between the systems after someone obtained her qualification. An English homoeopathist cannot practice in Hungary, because she is not a doctor; neither can her Hungarian colleague in the U.K., due to her inadequate training in the field of homoeopathy.
It is quite interesting that the member states–-on a European scale–-assured the greatest possible advantages to the employees of health care professions in the field of the mutual acceptance of diplomas. Those working in alternative medicine were left out from these advantages; moreover, they cannot make use of the general system's flexibility whenever they attempt to work in another EU country.
The definite establishment of the sectorial directives and the consensual development of the minimum requirements between the member states are time demanding processes as we have already seen in the case of architects (where the agreement on the minimum requirements preceded by a 17 year long process of negotiation). Thus, it is not very likely that the sectorial directive assuring the simple acceptance of foreign diplomas in the case of either branch of alternative medicine will be framed, as it was proposed by Paul Layonne in 1994, but theoretically cannot be excluded. This necessitates the elaboration of the uniformly accepted basic system of requirements by each profession that could be provided with a legal shape and adopted by the authorities. Some changes will occur in October 2007 in the directive regarding the mutual acceptance of diplomas that will overrule the actually valid sectorial and general rules. According to the expectations the new regulation might bring an easier situation for services that crossing the borders occasionally or temporarily (26).
Unfortunately we have no available statistics regarding the extent, and direction of the mobility within the profession (the Eurostat do not prepare specific statistics concerning the health care sector's mobility). Although we do have data about general mobility that shows 2% of employees work in other member states. Most of the employees that made a request for accepting their qualifications work in the health care sector. The direction of employee's migration within the EU is typically from east to west (27).
The Research Budget Might Increase
The first financial support by the EU given to research in alternative medicine was the COST B4 program launched in 1993 (28). COST is the acronym for Co-operation in Science and Technology and it was launched as a framework for scientific and technical co-operation, allowing coordination of nationally funded research on EU level: “The main objective of the COST Action on ‘Unconventional Medicine’ (UM) was to foster international collaboration in research into the therapeutic significance of UM, it's cost-benefit ratio and its socio-cultural importance as a basis for evaluation of its possible usefulness or risks in the public health“ (29). It was the only program that focused on the systematic evaluation of alternative medical practices. Thus, it seems very likely that getting into the 7th Framework Program of the EU-will be a turning point in supporting research with a special focus on alternative medicine. The framework program consists of 9 “sub-program,” where we can find the research possibilities connected to alternative medicine under the Health section. The probable budget of this section is 6050 million euros between 2007 and 2013 (30). The magnitude of this budget provides a chance for serious work in the field. Although it might seem to be an enormous amount, it is available for and divided to a number of tasks.
According to Edzard Ernst even more is needed, and this amount is just relatively high with comparing it to the NIH's budget for alternative medical research (31). The U.S. Congress established the Office of Alternative Medicine (OAM) in 1992 and the National Center for Complementary and Alternative Medicine (NCCAM) in 1999, which had a yearly budget of 120.4 million $ in 2006 (32).
Among the EU member states there is an outstanding activity in the U.K. regarding the research related to complementary and alternative medicine (CAM). Recently two important investigations were published, which are focusing on the main tasks, objectives and issues concerning the budget that could be assigned for alternative medical research. The report of the House of Lords was published in 2000 that evaluated the main questions regarding the planning of alternative medical research. According to this report, in order to raise this specific field of research to a higher level: “a small number of such centres of excellence, in or linked to medical schools, be established with the support of research funding agencies including the Research Councils, the Department of Health, Higher Education Funding Councils and the charitable sector“ (4). The other important report also stresses the cooperation of research centers and the need for following more rigorous evidence based research projects. The precondition for this is the creation of adequate infrastructure to which state resources and private companies shall also take part (33). The House of Lords report expressed that the most serious impediment to research is the lack of available financial resources. Three resources could be pinpointed that are used by health care research, so could be used for CAM research as well: government, charities, and commercial or industrial resources. According to the report's suggestion the solution for this financial problem would be achievable with raising the central budgets and increasing the number of private companies–-that are engaged in the trade of alternative medical products–-to participate, with a bigger part of their profits in the financial support given to research projects in alternative medicine.
It is reasonable to expect that the “central” budget will increase in the EU following this report's proposals, but the total amount would be still a negligible portion compared to the existing supports given to conventional medicine. Similar disproportionateness was found by Edzard Ernst regarding the support given by medical charities to CAM research, while these topics could not reach 0.5% of the total budget. According to Ernst this is inequitable also for not reflecting the supporter's preferences. Bringing about a change in this disproportionateness would require from the supporters to make their preferences manifest, and also, from the committees to give up their discriminatory practices against CAM topics (31). Although pharmaceutical companies are major participants in research and development projects in health care–-where patent rights and licenses are possible–-these companies have no interest in the field of alternative medicine. The absence of pharmaceutical companies leads not just to the above mentioned problem of research financing, but similarly “responsible” for organizational and marketing problems as well. Pharmaceutical companies well known efficiency in marketing their products could be highlighted by the data that not more than 1% of medical doctors do not apply information of pharmaceutical origin (34).
However, the greatest problem in the field of research seems to get solved by acquiring a stabile budget from the EU in the coming years of 2007– 2013, the main objectives must be well defined in the light of the limits of financial sources. Within the indicated areas of research, the EU urges on the strengthening of basic research (35).
Conclusions
As a consequence of several policies in the field of education, research, and mobility, the European Union certainly has a wide impact on the development of alternative medicine. Except the directives regarding the trading of homeopathic products, medicinal plants and dietary supplements, all other provisions of the EU are lacking a binding force for the member states. However, indirectly other provisions also could have a significant effect on a national level, like the long-term project of the harmonization of education and the creation of the conditions for the employee's mobility within the member states. At the present, professionals of alternative medicine cannot take the opportunity that the European Area of Higher Education seems to offer, since the educational systems of alternative medicine are not suited for the requirements. The number of bachelor or master level trainings should be increased and harmonized in order to be able to access the advantages created by EAHE and the mutual acceptance of diplomas. It is very likely that one of the basic problems regarding the lack of specific and reliable research in the field of alternative medicine will be solved in the long run with the newly emerging 7th Framework Programme. Since these financial sources are limited, it would be compelling to define in an organized manner the main lines and objectives of future research in the field of alternative medicine.
