Abstract
Background:
Premenstrual Dysphoric Disorder (PMDD) is associated with higher suicidality and reduced functioning. Diagnosis and treatment can improve symptoms and quality of life. Poor quality healthcare experiences for people seeking help for PMDD have been documented in other countries, but not in Australia.
Objective:
To quantitatively and qualitatively describe experiences of healthcare for PMDD in Australia, including variations in experience by healthcare provider (HCP) type.
Methods:
We employed mixed methods including an online survey and semi-structured interviews in adults who had sought healthcare in Australia for PMDD symptoms. Quantitative data was analysed using descriptive statistics, chi-square analyses, t-tests, and multilevel logistic modelling. Qualitative data was analysed using reflexive thematic analysis.
Results:
Survey participants (n = 267) had sought help from an average of 5.1 different HCPs in seeking PMDD diagnosis and perceived an average of 51% of HCPs they had seen to have poor PMDD awareness. GPs were the most frequent HCP type seen first (74%) when seeking diagnosis. More than half reported experiencing medical gaslighting (54%) and misdiagnosis (56%). Interview participants (n = 11) reported misdiagnoses such as depression, anxiety, bipolar disorder, and personality disorders. Interview participants reported feeling that their symptoms were minimised by healthcare providers and that their health care concerns were dismissed. Participants were more likely to report poorer experiences with GPs compared to other HCP types. Less than a fifth of the survey sample (19%) had experienced diagnostic methods consistent with DSM-TR recommendations. Diagnosis was described by interview participants as resulting in increased coping, a mindset of self-compassion and reduced self-perceptions of feeling ‘crazy’ or having fundamental personality flaws.
Conclusions:
People in Australia face similar PMDD healthcare barriers to those documented elsewhere. GP training in PMDD screening and clearer patient care pathways with specialist referral are needed to improve healthcare provision and reduce the burden of PMDD.
Keywords
Introduction
Premenstrual dysphoric disorder (PMDD) was first recognised in The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) in 2013 1 and comprises emotional, behavioural and somatic symptoms that present during the luteal phase (7-10 days pre-menstruation) of a menstrual cycle. PMDD prevalence in Australia has not been specified but it is estimated to affect between 1.6% and 7.7% of the population. 2 PMDD is associated with 2.13 higher odds of attempting suicide compared to the general population, 3 disabling levels of psychological distress,4,5 impaired social functioning5,6 and ability to maintain employment/productivity.7-9 PMDD treatment can improve PMDD symptomology10,11 and preliminary evidence suggests that PMDD diagnosis improves quality of life. 4
Studies from the United States, the United Kingdom, Canada and the Netherlands have indicated that PMDD diagnostic experiences are affected by poor perceived PMDD awareness among healthcare providers,9,12,13 diagnostic delay4,13,14 and misdiagnosis.4,13-15 Cross-country survey findings suggest people with PMDD experience average delays of 12 years and visits to six different healthcare professionals (HCPs) between symptom onset and diagnosis. 16 No other studies have quantified the extent of diagnostic delay.
Evidence from those countries further indicate that people seeking help for PMDD have their symptoms dismissed without proper medical evaluation, known as medical gaslighting.4,5,9,13,15,17,18 Funnell et al, 17 the only study to measure frequency of medical-gaslighting experiences among people seeking help for PMDD, found that 45% of people who had sought healthcare for premenstrual symptoms in the United Kingdom felt their symptoms had not been taken seriously at all. In an international sample (n = 2512), Hantsoo et al 1 found that patients were more likely to rate General Practitioners (GPs) as not having believed them when they spoke about their premenstrual symptoms than other HCP types. However, there is currently no evidence from Australia about whether patient experiences of medical gaslighting differ by HCP type.
There is also evidence of low rates of HCPs administering recommended diagnostic methods for PMDD among HCPs.1,19,20 The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition Text Revision (DSM-5-TR) specifies that PMDD diagnosis be made based on prospective daily symptom charting across two menstrual cycles, because retrospective self-report has been found to lack validity in accurate diagnosis.21-23 Hantsoo et al 1 found that 68% of people in a cross-country sample who had visited a HCP for PMDD had never had their daily symptom ratings across two menstrual cycles. Although there is evidence of a disconnect between DSM-TR diagnostic method specifications and clinical practice in some healthcare systems, PMDD diagnostic experiences in the Australian healthcare context have not been researched, and the extent to which DSM-TR diagnostic methods are implemented in Australia is unknown.
Period-tracking apps provide a way for people to use their smart-phones to track physical, psychological, and behavioural patterns associated with the menstrual cycle 24 and may assist people increase their awareness of symptoms associated with menstruation and facilitate conversations with HCPs.25,26 Period tracking apps may have the potential for improving the PMDD diagnostic experience if they have higher levels of user acceptability than other prospective symptom charting tools.
The quality of healthcare provision for PMDD is likely also impacted by a lack of clarity regarding what HCP type one should approach for PMDD diagnosis and treatment because of its intersection with mental and gynaecologic health. 1 People may approach both mental health professionals (eg, psychologists and psychiatrists) as well as health professionals who specialise specifically in gynaecologic/reproductive health issues (eg, gynaecologists and endocrinologists) when seeking help. 1 The Australian health system is organised so that GPs often serve as gatekeepers for access to specialised services 27 and are therefore likely to play a pivotal role in the pipeline of healthcare delivery to people with PMDD. There are currently no clinical guidelines that standardise which HCPs should be accessed for optimal PMDD healthcare delivery or their responsibilities to patients for PMDD healthcare.
Although evidence from healthcare systems in settings outside Australia suggests that PMDD diagnostic experiences for PMDD require improvement in the implementation of diagnostic methods and providing standards of care which meet patients’ needs, no research has been conducted in the Australian health system. This research aimed to describe the lived experience of PMDD diagnosis (methods and usefulness) and other healthcare experiences (HCP PMDD awareness/knowledge, diagnostic delay, misdiagnosis and medical gaslighting) and how they vary by HCP type, for people in Australia.
Methods
An explanatory sequential mixed-methods design was employed to first collect quantitative (Study 1; online survey) and then collect qualitative data (Study 2; semi-structured interviews) for describing the diagnostic experience of PMDD in the Australian health system from the perspective of healthcare consumers. Given the historical psychologisation and trivialisation of women’s symptoms in health settings 28 we privileged the lived experience of health consumers as the site of knowledge production about PMDD diagnostic experience.29-32 Ethics approval for the study was granted by a University Human Research Ethics Committee (QUT Ethics Approval number 7089-HE31).
Participant Recruitment
Participants were recruited via online peer support group pages and social media accounts relating to PMDD on Facebook and Instagram between 11th of November 2023 and 26th of January 2024. A sample size calculation based on a conservative population prevalence of 8% indicated that a sample of N = 384 would be required to obtain survey results representative of females in the Australian population with PMDD with 95% confidence. The first 45 participants of Study 1 were invited to register interest for participation in Study 2 to generate a feasible sample for ‘deep-case-oriented analysis’ of Study 2 data. 33 Eligibility for both studies comprised (1) being over 18 years of age, (2) self-identifying as experiencing PMDD, and (3) having interacted with a healthcare professional in Australia for PMDD symptoms. Study 2 eligibility criteria included having access to Zoom. Participants who registered interest in Study 2 were contacted via email, provided with a participation information sheet, and asked to schedule a time for participation with the researcher when their PMDD symptoms would be absent, to minimise risk of distress. Participant consent for Study 1 (online survey) was obtained via written consent whereas participant consent for Study 2 (semi-structured interviews) was obtained verbally and manually recorded. Participants of both studies were provided with information about the risks and benefits of participation and their option to freely withdraw from participation at any time prior to providing consent.
Measures
Quantitative Data Collection
A bespoke anonymous online survey 34 was constructed using Qualtrics Software (Version, November 2023). The online survey was reviewed by two people from the study target population to collect feedback prior to implementation. Both members of the target population provided the feedback that they were able to easily interpret survey items and items were relevant to their experiences.
Sample Characteristics
Participants were asked socio-demographic questions and the Premenstrual Symptoms Screening Test (PSST) 35 to enable us to describe the sample. The PSST is a 19-item retrospective self-report measure of symptoms reflecting the PMDD DSM-IV diagnostic criteria 36 : 14 assess premenstrual mood, physical and behavioural symptoms and five assess the impact of these symptoms on people’s ability to function in relation to work productivity, relationships, social life and home responsibilities. Items are scored on a 4-point scale rated as ‘not at all’, ‘mild’, ‘moderate’ or ‘severe’. For PMDD diagnosis, at least one of the first four symptoms must be rated as severe, a further four symptoms rated as moderate to severe and one functional impairment domain rated as severe. 35 Although there is evidence that only approximately one third of people who retrospectively report PMDD symptoms have PMDD according to prospective symptom daily tracking,21-23 incorporating prospective reporting of PMDD symptoms into the survey carried risks of high participant attrition as per Henz et al 22 and sampling bias given that higher symptomology has been associated with lower agreement to engage in prospective tracking. 37 Steiner et al 35 established the PSST’s validity for detecting similar prevalence rates of PMDD as those found in large prospective studies. The PSST has been found to have excellent internal consistency (Cronbach’s alpha = .93). 17
Diagnostic Methods
To measure experiences of diagnostic methods across HCP types, participants were asked ‘Have any health care professionals asked you to chart your symptoms daily (across the menstrual cycle) as part of the diagnostic process?’ (‘yes’/’no’). Participants who answered yes were provided with a matrix to indicate which type of HCPs (General Practitioner, Gynaecologist or Obstetrician, Psychiatrist, Psychology or Other – please specify) had asked them to track daily symptoms and for how long each provider type had asked them to track their symptoms (‘for 1 month’/’for 2 months’/’didn’t specify how long’). All participants were also asked: ‘Did any Australian health care professional ask you to complete a survey or checklist about the PMDD symptoms you experience?’ (‘yes’/‘no’/’don’t remember’) and ‘Have you used a tracking app to monitor your PMDD symptoms (e.g. flo, clue etc)?’ (‘yes’/’no’). Participants who responded ‘yes’ to having used a tracking app were then asked, ‘When did you use a period tracking app?’ and to select all responses that applied (‘before diagnosis’; ‘in order to help with diagnosis’; ‘after being diagnosed’.)
Diagnostic Delay
We conceptualised diagnostic delay as both number of years between initial symptom awareness and diagnosis and as number of HCPs visited for help prior to diagnosis. Data from three items were combined: ‘How old were you when you first noticed you were having PMDD symptoms (years)?’; ‘Have you been diagnosed with PMDD from an Australian health care professional? (in other words, has a health care professional told you and/or agreed with you that you experience PMDD?)’ (‘yes/’no’) and ‘How old were you when you were first diagnosed with a health care professional (years)?’.
Healthcare Provider Awareness, Misdiagnosis and Medical Gaslighting
Items assessing experiences of perceived HCP awareness, misdiagnosis and medical gaslighting were tailored according to whether participants had received a diagnosis from a HCP. Participants who had received a PMDD diagnosis were asked to report on their experiences with HCPs prior to diagnosis and those without a diagnosis were asked to report on all of their experiences with HCPs. An item asking how many of each type of HCP had been visited was used to assess the proportion of times each participant encountered a HCP where they (1) lacked awareness (‘How many of these professionals were aware of what PMDD is’), (2) provided a perceived misdiagnosis (‘How many of these professionals did you feel “misdiagnosed you”? That is- provided you with a diagnosis other than PMDD which was not accurate?’) and (3) made them feel medically gaslit (‘How many professionals made you feel like you were making your symptoms up’ or that they were ‘all in your head?’ and ‘How many of these professionals made you feel that you did not have “real” health concern or that your symptoms were normal?’). Items were based on findings in the international literature regarding the type of medical gaslighting people experience when seeking healthcare for PMDD symptoms.4,5,9,13 To support the comparability of the findings, an item from Funnell et al 17 was included to measure medical gaslighting (‘Please think about the first health care professional you sought help for your premenstrual symptoms from. How seriously were your mental health symptoms association with your menstrual cycle taken’). Participants responded to this item on a 4-point scale from ‘Not seriously at all’ to ‘Very seriously’. An open text item was provided for participants to provide further details about their experience of HCP interactions in relation to PMDD symptoms.
Healthcare Provider Type
To measure whether the PMDD diagnosis experience varied by type of HCP visited, participants were also asked: ‘Which type of health care professional did you first seek help for your symptoms from’ (‘General Practitioner’/’Gynaecologist or Obstetrician’/’Psychiatrist’/’Psychologist’/’Other – please specify’). Participants who indicated they had received a diagnosis were asked: ‘Which type of health care professional first diagnosed you with PMDD?’ ((‘General Practitioner’/’Gynaecologist or Obstetrician’/’Psychiatrist’/’Psychologist’/’Other – please specify’).
Usefulness of Diagnosis
Usefulness of receiving a PMDD diagnosis was measured using the item: ‘Was receiving a diagnosis helpful to you?’ (‘yes’/‘no’/‘I’m unsure’) in conjunction with the open text response item: ‘Please explain why you felt this way about receiving a diagnosis’.
Qualitative Data Collection
All semi-structured interviews were conducted by the first author (G.B.) via recorded Zoom sessions between February 13 and March 10, 2024. Interview durations ranged from 22 to 59 minutes (median duration 31 minutes) and were transcribed using Zoom software. The semi-structured interview protocol comprised of open-ended questions designed to elicit richer details pertaining the diagnostic experience for PMDD. The semi-structured interview was refined through role-play practice with three volunteers (one whom met sampling eligibility criteria) prior to data collection.
Data Analyses
Quantitative data were analysed first, followed by the triangulation of the quantitative and qualitative data. Quantitative data 34 were analysed using SPSS (Version 29). Participants were included in the final analysis if they answered questions representing more than 37% progress through survey items (ie, provided data indicating whether they had received a HCP diagnosis of PMDD). Descriptive statistics were used to describe the sample and frequencies of different healthcare experiences. Chi square analysis was used to describe associations between having received a PMDD diagnosis and meeting PSST criteria for PMDD diagnosis and HCP methods of symptom tracking. A t-test was performed to test differences in diagnostic delay between those who became aware of PMDD symptoms before and after DSM recognition of PMDD. Effect sizes were evaluated using Sawilowsky’s effect size rules of thumb. 38 Where descriptive data were reported as percentages, cases of missing data were retained in the denominator (ie, statistics were reported as a percentage of the usable sample). Bivariate associations used listwise exclusion of missing data and statistical significance at P < .05. Six participants reported that more healthcare professionals (HCPs) were aware of PMDD than the number of HCPs they reported having seen. Due to this inconsistency, their data were excluded from the descriptive statistic indicating the median proportion of HCPs visited prior to diagnosis who were perceived to have an awareness of PMDD. One participant reported feeling misdiagnosed by more HCPs than they reported to have visited, this data point was excluded from descriptive statistic indicating media proportion of HCPs participants felt misdiagnosed by. Multilevel logistic regression modelling was performed to test associations between diagnostic experiences and type of HCP visited. Data was re-structured, so each HCP provider visited represented a singular data point and associated dependent variables were coded as binary (eg, 0 = provider did not seem aware of what PMDD is; 1 = provider seemed aware of what PMDD is). Participants that had responses on an outcome variable pertaining to a HCP type but did not indicate how many of that HCP type they had visited were excluded. Like Hantsoo et al, 1 we calculated an ICC (intra class correlation) to assess the extent to which individual differences in response tendencies impacted outcomes (between-person variance) versus the impact that HCP type had on outcome variables (within-person variance). Higher ICCs (closer to 1) indicated that participant specific individual differences had a greater impact on ratings, whereas lower ICCs (closer to 0) indicate that the majority of variance was due to HCP type. The multilevel binary logistic model was conducted using the mixed models function in SPSS (version 29) and consisted of two levels whereby ratings were nested within each participant. The nonindependence of observations for each participant was accounted for by including a random intercept at the participant level. A scaled identity covariance structured was chosen as the best model fit. Statistical significance was determined at P < .05.
Reflexive thematic analysis (reflexive TA) 39 was employed to analyse interview transcripts and was manually conducted by the first author (G.B). The second author (Y.M.) reviewed two interview transcripts independently to support the validity of the findings. We report themes from thematic coding with a deductive orientation (ie, codes reflected pre-existing research questions) which participants endorsed the most often and/or described as carrying significant weight within their experiences. Data saturation was considered reached as no new themes (with a deductive orientation to data analysis) were emerging during analysis of the final interview transcript. To increase reflexive TA transparency, all participant quotes coded as supporting each reported theme are available at Border and Miller. 34
The research questions and subsequent analysis were influenced by the first author’s own personal experience of unknowingly living with PMDD for numerous years. Prior to commencing this research, they already viewed the diagnostic experience as having the capacity to have a profoundly positive impact on quality of life. Throughout the research process, to minimise the influence of lived experience on the objectivity of the data analysis, they critically reflected on how reflexivity was impacting interpretation of the qualitative data. This included engaging in written reflections and discussions with the second author.
Results
Sample Characteristics
Of 296 participants who clicked on the survey link, 12 were excluded because they did not consent to participate, eight were ineligible, and 10 answered an insufficient proportion of the survey for inclusion. The resulting usable sample (N = 267) was estimated to provide representative data for females in the Australian population with PMDD with 94% confidence, falling short of the target for recruitment (N = 384) for 95% confidence due to limitations in project timeline and available resources. Participants included in final analyses were aged 19 to 54 years (Median = 37, interquartile range = 11) and 54.7% (n = 146) indicated experiencing at least one comorbid health condition alongside PMDD. A majority (n = 234; 87.6%) reported having received a PMDD diagnosis from a HCP and had a PSST score that indicated they experienced current PMDD symptomology at or above the threshold for PMDD diagnosis (n = 181; 67.8%; see Table 1). There was no significant association between having met the PSST criteria for a PMDD diagnosis and having received a HCP diagnosis (χ2 (1) = 0.31, P = .58).
Participant Sample Based on PSST Scores.

Calculated for a 2 (diagnosed by HCP) × 2 (met PSST criterion for PMDD) contingency table, (excluding participants with missing data on either variable), there was no significant association between having met the PSST criteria for a PMDD diagnosis and having received a HCP diagnosis, X2 (1, N = 234) = 0.31, P = .58.
Of 45 participants contacted for an interview 13 people responded to the initial email. Of the 13 respondents, two discontinued communications during the interview scheduling process. Interview participants consisted of 11 people, nine of whom indicated they had been diagnosed with PMDD by a HCP.
PMDD Diagnosis Experiences
Diagnostic Methods
Almost half the final sample (n = 120; 44.9%) indicated that a HCP had asked them to track their symptoms for at least some length of time across the menstrual cycle. Having been asked to track symptoms was not associated with having received a diagnosis from a HCP (χ2 = (1) = 2.50, P = .11).
Almost one fifth (n = 50; 18.7%) had been asked by at least one HCP to track their symptoms for 2 months across their menstrual cycle as per DSM-TR guidelines for PMDD diagnosis. Almost one fifth (n = 51; 19.1%) indicated that a HCP had asked them to complete a survey/checklist about their PMDD symptoms and 7.5% (n = 20) reported they did not remember if they had been asked. A total of 109 participants (40.8%) indicated that they had never been asked to fill in a survey/checklist of symptoms nor to track their symptoms across their menstrual cycle by any HCP.
Period-Tracking Apps
Most survey participants (n = 169; 63.3%) indicated that they had used a period tracking app to monitor their PMDD symptoms. Of those, 69.2% (n = 117) indicated having used an app to monitor symptoms pre-diagnosis, 40.8% (n = 69) used a period tracking app to help with diagnosis and 49.1% (n = 83) had used a period tracking app after being diagnosed.
Utility of Diagnostic Experience
The majority of survey participants who had received a diagnosis of PMDD indicated that receiving a diagnosis was helpful to them (n = 186; 79.5%). For 5.6% (n = 13) receiving a diagnosis was not helpful and 9.4% (n = 22) were unsure. Study 2 participants described receiving a diagnosis as resulting in relief that they weren’t ‘crazy’ and that the condition was ‘real’: ‘Yeah, so I was quite relieved that hey that it wasn’t in my head, and that it was actually a real thing’ (Participant 8). The theme of diagnosis resulting in this type of relief was also described in open-text responses to the survey: ‘Gave me clarity that I was not losing my mind, that it was a hormonal issue that needed support, confirmed I wasn’t crazy’. Participants also described the impact of receiving a diagnosis on their belief that they possessed inherent character flaws.
I think it’s fundamental, like there is a certain power in obtaining a diagnosis to know that something is not the fault of your, you know, your mind, your capacity to sort of regulate. . . (Participant 6).
Some described the therapeutic impact of being diagnosed as arising from enabling them to identify when their bodily and mental experience of the world was a result of PMDD and responding with self-compassion.
Essentially, that I think, being of aware of it is also really helpful, even though that’s not really doing anything. . .if I’m going into work and I’m like struggling, going and being like this why I just need to like to accept that I’m not going to be able to do all things that I need to do. And that’s really the only way that I’ve found to cope. (Participant 7).
The therapeutic impact of obtaining a diagnosis also extended to some participants being able to adopt coping strategies such as reminding oneself that the symptoms are transient.
To be able to rationalize the days that I had suicidal ideation and to know not to act on anything. . . To be able to understand. . . This is just your PMDD. You know how this cycle works. . . You just have to hang in to the next minute to the next day (Participant 3).
However, for many participants obtaining a diagnosis was not the final hurdle in their journey to access adequate healthcare for PMDD, detracting from the overall usefulness of diagnosis. Participants described how diagnosis had little impact on their PMDD healthcare needs because they were not offered treatment options: I found [the psychiatrist] was really patronising and dismissive. He said, ‘you know’ (in his diagnosis) ‘Yeah, you know. Very likely have PMDD. . .But never offered any like. . .okay, great. So, what goes next’ (Participant 5) and ‘like I didn’t feel dismissed. I didn’t feel dismissed by the GP. I didn’t feel dismissed by the gynaecologists. I didn’t feel dismissed by the so-called psychiatrist. It was more they didn’t have treatment options’ (Participant 8).
Healthcare Provider Awareness, Diagnostic Delay, Misdiagnoses, and Medical Gaslighting
Awareness and Knowledge
Among participants who specified how many HCPs they had visited (n = 226), the median proportion of HCPs visited prior to diagnosis perceived to have an awareness of PMDD was 50% (interquartile range = 75%). Study 2 participants described challenges associated with finding a PMDD-aware HCP.
I think I called the least 20 to 30 medical centres, just asking if any of their doctors had actually heard of PMDD and not one of them had. (Participant 6).
Some participants described taking an active role in attempting to educate their HCPs about PMDD and the resulting tension they experienced in the healthcare encounter.
. . . there’s some medical professionals that don’t like to have their knowledge questioned and that was the same with the gynaecologist. He didn’t know and instead of being curious and wanting to educate themselves they shut down and push back. Because you know, don’t question my ‘authoritar’ (Participant 10).
Participants also described positive encounters with HCPs because they seemed willing to learn more about PMDD: And the 2 psychologists I’ve had in the last 2 years, neither of them had ever heard of it, but they’ve both researched it since having me as a client. And yeah, they’re really good (Participant 1). Some participants described situations in which they felt that HCP treatment recommendations for PMDD did not align with their understanding of evidence for their efficacy.
She gave me a referral to a gynaecologist who told me that I should count myself lucky this is considered a condition now. Yup. And so we got the Mirena put in, which was the wrong treatment. And then I went on a period of time before finding another GP and doing my own research and getting myself put back on the pill and removing the Mirena (Participant 11).
Diagnostic Delay
Of the participants diagnosed by a healthcare professional (n = 234), the median time elapsed between the onset of symptoms and receiving a diagnosis was 2 years (interquartile range = 6 years), with reported delays ranging from 0 to 32 years. Figure 1 shows the number years of diagnostic delay experienced by participants who became aware of their symptoms before and after PMDD DSM recognition. A bootstrapped independent samples t-test was conducted due to violated assumptions of normality. Participants with a HCP diagnosis who became aware of PMDD symptom onset within the last 11 years (after 2013, when PMDD was included in the DSM) (n = 172), experienced a shorter mean diagnostic delay (2.37 years, SD = 2.46 years) than those who became aware of symptoms before 2013 (12.28 years; SD = 7.05) n = 57). This difference (−9.91, BCa 95% CI −11.95 to −7.83) was significant (t (60.58) = −10.41, P = <.001). The effect size was huge, d = −2.45. As shown in Figure 2, participants with a HCP diagnosis (n = 205) visited a mean of 5.09 (SD = 5.12) HCPs for help with PMDD symptoms prior to diagnosis.

Years of diagnostic delay for participants who became aware of symptoms before (pre-2013) and after (2013 or later) PMDD inclusion in DSM.

Number of healthcare providers visited prior to diagnosis.
Misdiagnoses
More than half (n = 149; 55.8%) of Study 1 participants reported having received a misdiagnosis in relation to their PMDD symptoms. Those who felt they had received a misdiagnosis and specified number of HCP visits (n = 147), reported feeling misdiagnosed by a median of 66.7% of the HCPs they visited (interquartile range = 43.8%). Study 2 participants described being previously misdiagnosed with conditions including depression, anxiety, bipolar disorder, and personality disorders. Some associated the experience of misdiagnosis with not being asked by HCPs about symptoms related to their menstrual cycle when seeking help for their mental health.
So I only really spoke to one GP. . .I would describe my symptoms to him and he had me fill out the depression diagnostic. . . it says: always, frequently sometimes or words to that effect. . .It doesn’t kind of give you the opportunity to say every, you know, it’s cyclical, it depends on the time of the month. . .And then originally he put me on antidepressants and diagnosed me with depression. (Participant 4).
Participants commonly described having been assessed for bipolar disorder. For one participant, this assessment still impacted them when they accessed healthcare: . . .. they tried to diagnose me at one point with bipolar because of the waves that come with PMDD. They tried to diagnose that at one point, and I think some of my files still say, that, that’s what it is but I don’t have the traits of. . . when you look at them clinically, I actually don’t have the mania that comes. You know what I mean, so to speak, with things like bipolar (Participant 3).
Participant responses also suggested that HCPs attributed their PMDD symptoms to comorbid conditions, leading to missed PMDD diagnosis opportunities: They just asked for the symptoms. I do also have endometriosis. So, everyone was just passing it off, as that’s just what it is. They didn’t want to look further. They just told me I was overreacting (Participant 6).
Medical Gaslighting
Almost a third (n = 81; 30.3%) of Study 1 participants indicated that their mental health symptoms associated with PMDD were taken very seriously by the first HCP from whom they sought help; 29.2% (n = 78) indicated their mental health symptoms were not taken seriously at all. More than half (n = 143; 53.6%) indicated that they experienced some form of medical-gaslighting by at least one HCP prior to diagnosis: 45.3% (n = 121) reported being made to feel that they were making their symptoms up/that their symptoms were ‘all in their head’ (from an average of 59.07% (SD = 34%) of the HCPs they visited prior to diagnosis); 49.4% (n = 132)reported being made to feel that they did not have a ‘real’ health concern/their symptoms were normal (from an average of 61.07% (SD = 33%) of the HCPs they visited pre-diagnosis).
Study 2 participants also described interactions with HCPs which made them feel like they didn’t have real healthcare concerns.
. . . the first GP that I went to, and I explained it all. . . He turned around and asked me if I have anybody in my family or friends that I can talk to that’s not my husband, because I just need to talk to some women in my life and that will sort out the problem and he wouldn’t get me a mental healthcare plan. So, I didn’t go back to him (Participant 11).
For another participant, being made to feel like they didn’t have a real healthcare concern resulted in being denied a referral to an Endocrinologist: I have asked GPs for referrals to endocrinologists because I worked out that that’s the specialist that would manage hormones. Yeah, nobody would ever give it to me (Participant 3).
Participants also described interactions with HCPs in which they felt the severity of their symptoms was minimised: it was more the general consensus was always – it’s not that bad. You need to get over it. That kind of thing (Participant 3). Some participants explained the impact of a HCP downplaying the severity of their condition: So, I went in there, and I had read on Doctor Google that if you’re getting migraines with auras you shouldn’t be on the pill. So, I went in there and I spoke to her about it and she said, “stop it immediately” and I said. “You don’t understand I have PMDD. . .I will become suicidal again”. And she’s like “stop it, just stop it, you don’t need it. You’re not using it for contraception. Just stop it”. And then 3 months later, guess what? We are back to square one again. So yeah, I told her that I would become suicidal. I told her that it was PMDD. And it is a serious condition, and she still just disregarded it (Participant 11).
Variations in Experience by Type of Healthcare Provider
GPs were the most common type of HCP first visited for help with PMDD symptoms (73.0%) and 59.8% indicated that a GP first diagnosed them with PMDD (see Table 2). The next most common HCP types that participants reported having received a diagnosis from was a gynaecologist/obstetrician (11.5%) or psychiatrist (11.1%).
Descriptive Statistics for How Diagnosis Experience Differed Depending on Type of HCP Visited.

In total, 232 participants responded to items asking how many HCPs they had visited, by type.
A large portion of the variance in experiences (ie, awareness of PMDD, misdiagnosis, and medical gaslighting) was attributable to participant specific individual differences in outcome tendencies (between-person variance) rather than outcome differences due to HCP type (within-person variance; see Table 3). ICCs ranged from 0.39 to 0.51. The ICC for ‘misdiagnosis’ was 0.51, indicating that a slightly greater portion of variance in people feeling they were misdiagnosed was attributable to participant specific characteristics rather than HCP type.
Contrasts Between HCP Type Based on a Multilevel Binary Logistic Regression with a Random Intercept at Participant Level.
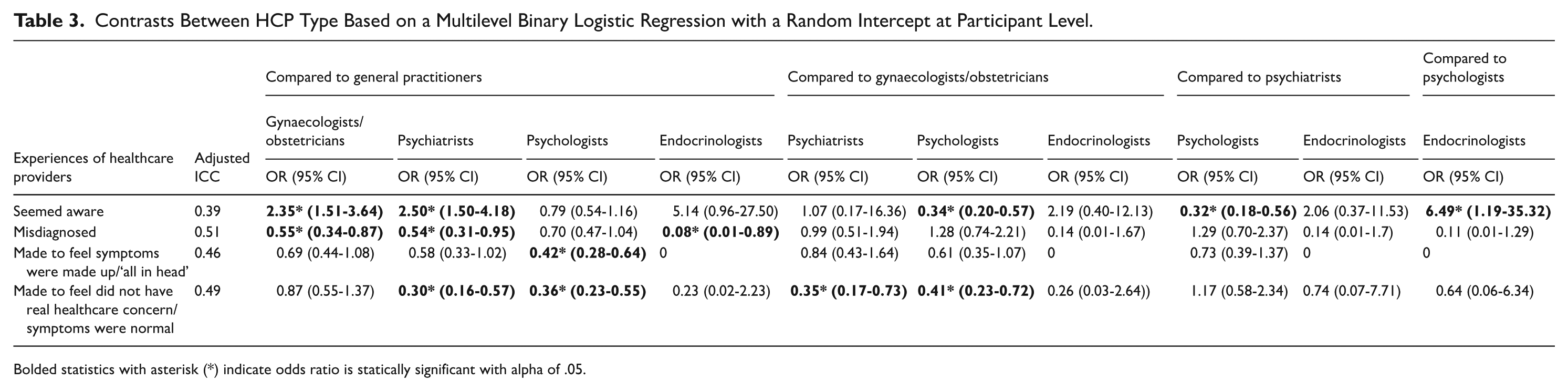
Bolded statistics with asterisk (*) indicate odds ratio is statically significant with alpha of .05.
Awareness by HCP Type
Both psychiatrists (OR = 2.50, 95% CI: 1.50-4.18) and gynaecologists/obstetricians (OR = 2.35, 95% CI: 1.51-3.64]) were rated as significantly more likely to seem aware of PMDD than GPs (see Table 3). Psychologists were rated as significantly less likely to seem aware of PMDD than gynaecologists/obstetricians (OR = 0.34, 95% CI: 0.20-0.57) or psychiatrists (OR = 0.32, 95% CI: 0.18-0.56). Endocrinologists were rated as significantly more likely to have been aware of PMDD than psychologists (OR = 6.49, 95% CI: 1.19-35.32) (see Table 3).
Misdiagnosis by HCP Type
Compared to GPs, all HCP types except psychologists were significantly less likely to be perceived as having misdiagnosed participants (see Table 3). Endocrinologists were the least likely to be reported by patients as having misdiagnosed (OR = 0.08, 95% CI: 0.01-0.89) compared to GPs. Psychiatrists (OR = 0.54, 95% CI: 0.31-0.95) and gynaecologists/obstetricians (0.55, 95% CI: 0.34-0.87) had similarly lower odds of being perceived as having misdiagnosed participants compared to GPs.
Medical-Gaslighting by HCP type
There were no significant differences between most HCP types in the likelihood that they were perceived as having made people feel that their symptoms were made up/‘all in their head’, except psychologists, for whom participants were significantly less likely (OR = 0.42, 95% CI: 0.28-0.64) than for GPs to say they were made to feel this way (see Table 3). Participants were significantly less likely to report that both psychiatrists (OR = 0.30, 95% CI: 0.16-0.57) and psychologists (OR = 0.36, 95% CI: 0.23-0.55) made them feel that they did not have real healthcare concerns/symptoms were normal than GPs (see Table 3). They were also significantly less likely to report that psychiatrists (OR = 0.35, 95% CI: 0.17-0.73) and psychologists (OR = 0.41, 95% CI: 0.23-0.72) made them feel like this than gynaecologists/obstetricians.
Discussion
We aimed to describe the healthcare experiences for people seeking diagnosis and help for PMDD to provide an empirical basis for strengths and areas requiring improvement in healthcare provision for PMDD in Australia. Our findings suggest similar barriers to accessing a PMDD diagnosis as evidenced in the international literature, including difficulty finding HCPs who seem aware of PMDD,9,13 diagnostic delay4,13,16 misdiagnosis4,13,15 and medical gaslighting.4,5,9,13,15 Our finding that the majority of participants with a HCP PMDD diagnosis felt that receiving a diagnosis was useful to them highlights the need for improvements in the healthcare system to mitigate barriers preventing access to diagnosis.
Our findings that the value of diagnosis was associated with reduced self-perceptions of feeling ‘crazy’ or possessing a fundamental personality flaw were consistent with those from Osborn et al. 4 Other research has shown that interactions with HCPs can result in women feeling ‘crazy’ when their healthcare concerns are repeatedly minimised or dismissed. 40 Obtaining a PMDD diagnosis was of inherent therapeutic value to participants because it provided an opportunity to identify when their bodily and mental experience of the world was due to PMDD and enabled them to respond with self-compassion and appraise their symptoms as transient. Habib et al 15 also found that diagnosis enabled people in Canada to better cope with PMDD because they no longer ascribed blame for their symptoms to their self and were better able to recognise the temporal ebbs and flow of PMDD.
We found that many HCPs were perceived as lacking an awareness of PMDD, which may at least partly explain our other findings indicating poor implementation of DSM-TR diagnostic guidelines and instances of misdiagnosis and missed diagnosis. Less than one fifth of survey participants had ever been asked by a healthcare professional to track their symptoms for 2 months across their menstrual cycle as directed in DSM-TR for PMDD diagnosis. Low HCP adherence to recommended diagnostic procedures for PMDD has consistently been found in recent international surveys of both HCPs and patients.1,19,20 A global study indicated that 32% of people who had visited a HCP for PMDD had had their daily symptom ratings across two menstrual cycles assessed by a HCP. 1 Low adherence to DSM-TR diagnostic guidelines may also reflect lack of suitability of the diagnostic method. Clinicians may avoid using prospective daily symptom charting as a diagnostic tool because it is considered too time consuming in clinical settings.19,20 This diagnostic method may also lack suitability for people who suffer symptoms that are significantly impairing and/or experience executive dysfunction. 41 In the Cohen et al 37 study, 53% of people did not agree to rate their symptoms prospectively and they were more symptomatic based on retrospective assessment, suggesting that prospective symptom tracking may be less feasible for people with high symptom impairments. Further development of diagnostic tools for PMDD that consider the feasibility of possible diagnostic methods from the perspective of HCPs and patients may be needed. Our finding that most participants had used a period tracking app to monitor their PMDD symptoms provides some evidence of their acceptability as a monitoring tool from a patient perspective. Further evidence is required to determine their usefulness and validity as a clinical diagnostic tool.
The importance of developing valid PMDD diagnostic methods that enable high HCP uptake is supported by our findings that PMDD symptoms were often attributed to pre-existing comorbid conditions by HCPs. Some participants described never being asked by HCPs about their symptoms in relation to their menstrual cycle. However, PMDD diagnostic tools require more sensitivity than linking symptomology to the luteal phase of the menstrual cycle. Distinguishing between PMDD and Premenstrual Exacerbation (PME, a premenstrual worsening of symptoms of a comorbid condition) may be critical, given preliminary evidence that some PMDD treatments are likely to be ineffective for people experiencing PME. 42 More than half of our participants (54.7%) experienced a comorbid health condition, comparable with findings from a recent cross-country study 43 in which 70% of people sampled with PMDD had a comorbid condition. High prevalence of PMDD comorbidity suggests that distinguishing between PME and PMDD is needed as a standard component of PMDD diagnostic screening.
Our research suggests people in Australia visited an average of five HCPs prior to diagnosis, comparable to findings in a global study that people visited an average of six providers prior to diagnosis. 16 Poor HCP awareness and high levels of medical-gaslighting for people with PMDD symptoms may be contributing to the burden of multiple provider visits prior to diagnosis in the Australian healthcare system. We found that a third of people perceived the first HCP they visited for PMDD symptoms as not taking their symptoms seriously at all. Comparably, Funnell et al 17 found that 45% of people who had sought help for premenstrual symptoms in the UK felt mental health symptoms related to their menstrual cycle had not been taken seriously at all. Incidences of medical gaslighting are likely to increase periods of diagnostic delay which likely places people at risk given the strong association between PMDD and active suicidal ideation. 43
GPs were the most commonly first-visited HCP for help with PMDD symptoms but also rated as the most likely to lack PMDD awareness, misdiagnose, and medically gaslight. In Australia, GPs are gatekeepers for patient access specialised services. 27 Improving healthcare outcomes for people with PMDD may require GPs to be better trained to screen for PMDD and clinical care pathways may also be needed to enable effective GP referral to HCPs that are better equipped to provide PMDD treatment. Endocrinologists were rated positively (characterised by low levels of misdiagnosis, medical gaslighting, and high levels of PMDD awareness) but participants only reported 11 total visits to endocrinologists. This likely reduced the level of power in contrasts between Endocrinologists and other HCPs and may explain many non-significant findings comparing ratings of Endocrinologists and other provider types. Nevertheless, the role of Endocrinologists in clinical care pathways for PMDD should be considered, given their specialty expertise in bio-medical perspectives for PMDD and the impact of hormones on health. Our findings could be used to support the development of clinical guidelines that standardise clinical pathways and responsibilities to patients for PMDD healthcare delivery, which do not currently exist.
Participants in this research were asked to retrospectively recall and characterise visits with HCPs that had occurred, for some participants, over multiple decades. Autobiographical memory is fallible, 44 suggestible 45 and susceptible to hindsight bias. 46 Future research would benefit from assessing the quality of consultations related to PMDD symptoms in real time. Our Study 1 usable sample size (N = 267; estimated to provide representative data with 94% confidence) fell short of our target (N = 384) to provide findings representative of females in the Australian population with PMDD with acceptable confidence. Our research may also have been affected by selection bias associated with our recruitment methods, which targeted people participating in PMDD support communities where the group discourse may have influenced responses. Our sample included a large proportion of people (87.6%) who had received a HCP diagnosis for PMDD. Given the barriers associated with obtaining diagnosis evidenced in our study (eg, medical gaslighting, risk of encountering HCPs who lack awareness, possible misdiagnosis), it is possible that our sample consisted of people who experienced less barriers in the process of receiving a diagnosis compared to the population of people with PMDD who are undiagnosed. Future research pertaining to Australian specific PMDD diagnostic experiences should use larger samples and recruitment methods targeting people who are undiagnosed but likely meet PMDD diagnostic criteria.
The lack of formal pilot testing and reliability and validity of our online survey questionnaire prior to implementation represents another key limitation of this study.
Conclusions
This research plays a critical role in highlighting the unmet needs of people in accessing timely and high-quality healthcare for PMDD in Australia. Our findings suggest that further development of PMDD diagnostic processes and tools is required, and HCPs (particularly GPs) may require further training to improve PMDD awareness and respectful patient communication skills to prevent patients being misdiagnosed and feeling medically gaslit when they present with PMDD symptoms. Standardised clinical care pathways for PMDD should be prioritised to address the existing burden of PMDD and optimise PMDD healthcare in Australia.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr. Leesa Wockner for providing statistical consultation and SPSS software advice.
Author Contributions
GB CRediT = conceptualisation; methodology; formal analysis; investigation; writing (original draft and review and editing). YM CRediT = conceptualisation; methodology; project administration; supervision; validation; visualisation; writing (original draft and review and editing)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
