Abstract
Background:
Exercise-based interventions, particularly dual-task training (DT), have been increasingly recognized as effective strategies for improving cognitive, motor, and functional capacities in individuals with Parkinson’s disease (PD). This study presented a protocol for a randomized controlled trial (RCT) designed to evaluate and compare the effects of land and aquatic-based single-task (ST) and dual-task (DT) training on physical and cognitive outcomes in individuals with PD. The present study aims to describe the protocol of 4 different physical exercise programs, including single-task and dual-task exercises on land and in the aquatic environment, and their potential impact on the cognitive, motor, functional, and quality of life capacities of people with Parkinson’s disease.
Methods:
This randomized controlled trial (RCT) involved individuals with PD who were randomly assigned to 1 of 4 intervention groups: (i) Land Single-Task (LST), (ii) Land Dual-Task (LDT), (iii) Aquatic Single-Task (AST), and (iv) Aquatic Dual-Task (ADT). Participants in each group underwent a 12-week exercise program with standardized volume, frequency, and intensity. The interventions focused on improving cognitive and motor functions, balance, dynamic gait, fear of falling, and quality of life (QoL). A battery of validated assessments was used, including the MoCA, SCOPA-COG, TUG, FTSST, Mini-BEST, DGI, ABC, and PDQ-39, administered at baseline, post-intervention, and at a 12-week follow-up.
Objectives:
This study aimed to investigate whether dual-task training, particularly in the aquatic environment, offered superior benefits over single-task training in improving cognitive, motor, and functional abilities in individuals with PD. Additionally, the study explored the potential of the aquatic environment to provide unique stimuli that enhance neuroplasticity, balance, and overall mobility.
Conclusions:
This protocol outlined a structured approach to evaluating the effectiveness of land and aquatic-based exercise interventions in individuals with PD. The findings from this study will contribute to the development of evidence-based guidelines for exercise prescription in PD, emphasizing the role of aquatic environments in therapeutic interventions.
Introduction
According to the biopsychosocial (BPS) model of health, which underpins the International Classification of Functioning, Disability, and Health (ICF), an individual’s health condition encompasses the domains of body functions (b) and structures (s), activities, and participation (d), denoting the functionality of individuals, along with life contexts that encompass environmental (e) and personal factors. 1 The functionality and contexts of human health can influence and be influenced by people’s capacities, whether cognitive, motor, functional, and also their Quality of Life (QoL). Thus, it is necessary to understand the processes associated with health in the human aging process. 2
With advancing age, various health conditions and/or diseases become more prevalent. Among them is Parkinson’s disease (PD), which significantly increases after the age of 55. In Brazil, there are approximately 220 thousand people with PD, and it is estimated that by 2030 this number will almost triple.3,4 PD is the second most common degenerative disorder of the Central Nervous System (CNS), second only to Alzheimer’s disease (AD). Its prevalence worldwide is around 1% of the population over 60 years old, and its incidence increases by about 10% every 10 years. 5 It occurs about 1.5 times more often in men than in women. 6
PD is characterized as a neurodegenerative condition, in which there is death of dopaminergic neurons in the substantia nigra pars compacta. 7 With the deterioration of these neurons and the consequent decrease in dopamine levels in the basal ganglia, the classic cardinal signs of PD appear, including bradykinesia associated with resting tremor, postural instability, and/or rigidity; non-motor signs including cognition may also be present. Its diagnosis involves various clinical, physical, and functional assessments, as well as multidisciplinary follow-up and treatment, with an emphasis on increasing dopamine concentration in the CNS.1,8
It is known that physical exercise is one of the main resources for reducing motor and non-motor symptoms in people with PD, as its beneficial effects result from increasing levels of neurotrophic factors and stimulating brain plasticity.9,10 Various types of physical exercises have benefits for this population, such as aerobic exercises, gait training, muscle strength training, balance training, aquatic physical exercises (APE), dual-task training (DT), virtual reality, certain types of dances, among others.6,11
One possibility for practicing physical exercises for people with PD is the aquatic environment, as it allows activities to be carried out safely and enjoyably, especially in dynamic and group activities, which can optimize attention to health conditions and extend benefits to these individuals.3,12 Several studies indicate the positive effects of the aquatic environment for people with PD, achieved through the combination of physical and therapeutic exercises with the physical and thermal principles of heated water.9,13
A more effective approach to physical exercise for individuals with Parkinson’s disease may be dual-task training, which progressively and complexly integrates cognitive engagement by simultaneously performing a primary task, with focused attention, alongside a secondary task. People with PD need to train these activities, as they may experience deterioration in cognitive capacity over time. 10 Recent research has found that combining APE with DT promoted improvements in functional mobility, balance, and gait in people with PD, highlighting the aquatic environment as a possibility for stimulating motor and functional skills in these individuals.2,14
Despite the increasing evidence for the benefits of dual-task and aquatic exercises, there remains a critical gap in the literature regarding direct comparisons between dual-task training performed in land-based and aquatic environments. This comparison is essential, as aquatic exercises offer unique physical properties—such as buoyancy, hydrostatic pressure, and viscosity—that may reduce the burden of postural instability and facilitate movement for individuals with PD, potentially enhancing cognitive engagement and motor control.
By integrating motor and cognitive tasks, dual-task exercises are expected to enhance neuroplasticity, improve executive functions, and reduce cognitive decline in individuals with Parkinson’s disease. Additionally, the aquatic environment, with its buoyancy and hydrostatic pressure, provides unique sensory stimuli that promote relaxation, reduce anxiety, and improve confidence, ultimately contributing to better psychosocial outcomes and quality of life.
Understanding these differences could have significant clinical implications for tailoring exercise interventions to the needs of individuals with varying levels of PD severity. Additionally, the study could provide practical applications by identifying cost-effective and accessible interventions that can be implemented in different care settings, improving adherence and outcomes.
The present study aims to describe the protocol of 4 different physical exercise programs, including single-task and dual-task exercises on land and in the aquatic environment, and their potential impact on the cognitive, motor, functional, and quality of life capacities of people with Parkinson’s disease. By identifying the specific benefits of each exercise modality, this study seeks to inform clinical decision-making and guide future research on personalized exercise interventions for PD management.
Method
Trial Design
This is a randomized controlled clinical trial with 5 parallel groups, in which participants are individually randomized to 1 of the 5 groups in a 1:1 allocation ratio. The study is based on the recommendations of the Consolidated Standards of Reporting Trials (CONSORT) and registered on the virtual platform of the Brazilian Clinical Trials Registry - ReBEC (RBR-5yjyr7). This study was approved by the Ethics Committee on Human Research of the Health Sciences Sector of the Federal University of Paraná (UFPR), Curitiba, Paraná (PR), Brazil, no 2 200 372, and follows all determinations established in Resolution of the National Health Council (CNS) 466/2012, the Code of Ethics of the World Medical Association (Declaration of Helsinki), and is aligned with the Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals. All participants signed the Informed Consent Form (ICF) at the time of the first assessment.
Participants
The sample size calculation was performed based on the prevalence of Parkinson’s disease in Curitiba/PR (2%), using a confidence interval of 95% and a margin of error of 5%. Applying the formula n = N.p.q.(Zα/2)²/p.q.(Zα/2)² + (N − 1).E², where:
•
•
•
•
•
Based on this calculation, a minimum of 12 participants per exercise group was determined to be sufficient to ensure statistical power. This approach aligns with similar studies in the literature, which used comparable parameters for sample size determination.
Participants were recruited through a partnership with the Parkinson’s Association of Paraná (APP) and the Ouvidor Pardinho Health Unit (US) in Curitiba, Paraná. After signing the ICF, selected participants were randomized to form groups. The randomization sequence was generated on the “randomizer.org” website. Randomization was performed in blocks, that is, for every group of 25 participants, a randomization process was conducted. A total of 3 randomizations were performed (classes 1-3), totaling 75 included participants. To ensure allocation concealment and minimize selection bias, sealed opaque envelopes were prepared by an independent researcher not involved in the recruitment or intervention procedures. Outcome assessors were blinded to group allocation throughout the study, thus maintaining assessment objectivity.
To minimize potential selection bias and ensure random allocation, all participants who met the inclusion criteria were assessed and randomized, guaranteeing the randomness of group allocation. While the randomization method may not ensure complete homogeneity across groups, the study’s primary goal was to maintain group randomness, as participants were evaluated at 3 different time points during the study to monitor the effects of the interventions. This approach aligns with the CONSORT guidelines and is appropriate for achieving reliable comparisons among groups.
Participants were eligible if they were aged between 50 and 80 years, diagnosed with idiopathic PD according to Movement Disorders Society (MDS) criteria, at stages I to IV on the Hoehn and Yahr scale, did not present recent clinical alterations, had a medical certificate for physical activity (all exercise groups), and had a medical certificate to attend the heated swimming pool (aquatic exercise groups). Exclusion criteria included: not having independent gait, related or not to PD; having another disease that promotes alterations in body balance or level of consciousness; inability to follow verbal and visual commands during activities; missing more than 30% of intervention days; absolute contraindications to attend the heated swimming pool (in the case of aquatic exercise groups); alteration in the daily dosage of Levodopa during the research; altering the routine of physical activities throughout the study; and, finally, not agreeing to the ICF or withdrawing from the research.
Interventions
Four exercise groups were created: (i) Land Single-Task (LST), (ii) Land Dual-Task (LDT), (iii) Aquatic Single-Task (AST), and (iv) Aquatic Dual-Task (ADT). Additionally, there was the control group (CG), which not participated in the intervention and were instructed to maintain their usual activities. The groups underwent 12 weeks of intervention, twice a week, with each intervention lasting an average of 50 minutes, as recommended by ACSM and the most recent European Physiotherapy Guideline for PD.15,16 To ensure the health and safety of participants during all interventions, vital signs were carefully monitored both before and after each activity. Blood pressure was measured using a digital automatic sphygmomanometer (Omron HEM-7124), heart rate and oxygen saturation were monitored with a pulse oximeter (Onyx Vantage 9590), and respiratory rate was assessed manually by counting the number of breaths per minute over a 30-second interval and doubling the value. These measurements ensured the early detection of any abnormalities and allowed for immediate action if necessary.
The aquatic intervention groups, AST and ADT, participated in the therapeutic pool at the Ouvidor Pardinho Health Unit, on Mondays and Wednesdays, with water temperature approximately 33°C and a depth of 1 m and 20 cm. The land-based intervention groups, LST and LDT, participated in training sessions at APP, in the institution’s physiotherapy room, scheduled at times when there is no therapy, which was on Tuesdays and Thursdays.
The single-task exercise groups progressed by increasing the complexity of the motor task (eg, from stable to more unstable, from lower speed to higher speed).10,13 The dual-task groups, in addition to progressing in the complexity of the motor task (which was the primary task for this group), also increased the complexity of the secondary task, which could be either cognitive or motor. For example, motor tasks ranged from holding objects to more complex activities such as passing and throwing; cognitive tasks ranged from naming a person to more complex activities such as progressive and regressive calculations, memorization, etc. 14
The dual-task exercises were included for their ability to engage higher-order brain activity, requiring participants to allocate attention, process information, and perform simultaneous tasks. 6 This type of exercise has been shown to stimulate neuroplasticity and enhance cognitive function, which are particularly beneficial for individuals with Parkinson’s disease who often experience motor-cognitive interference. The rationale for the inclusion of specific exercises was based on their ability to mimic real-life activities and address the deficits in balance, mobility, and cognitive-motor integration commonly observed in PD. 11
This study’s interventions were designed to evaluate how these different environments and task complexities contribute to motor and cognitive improvements, providing a comprehensive comparison of their effects on individuals with PD. 15
Figure 1 shows the study’s Consort flow-diagram and Table 1 shows the periodization of the exercise programs. Appendix 1 provides a complete description of the exercises, and the relationship between the physical exercises, the physical properties of the aquatic environment, the phase of aquatic exercise intervention, and the corresponding outcome with the scale that was trained in the exercise.
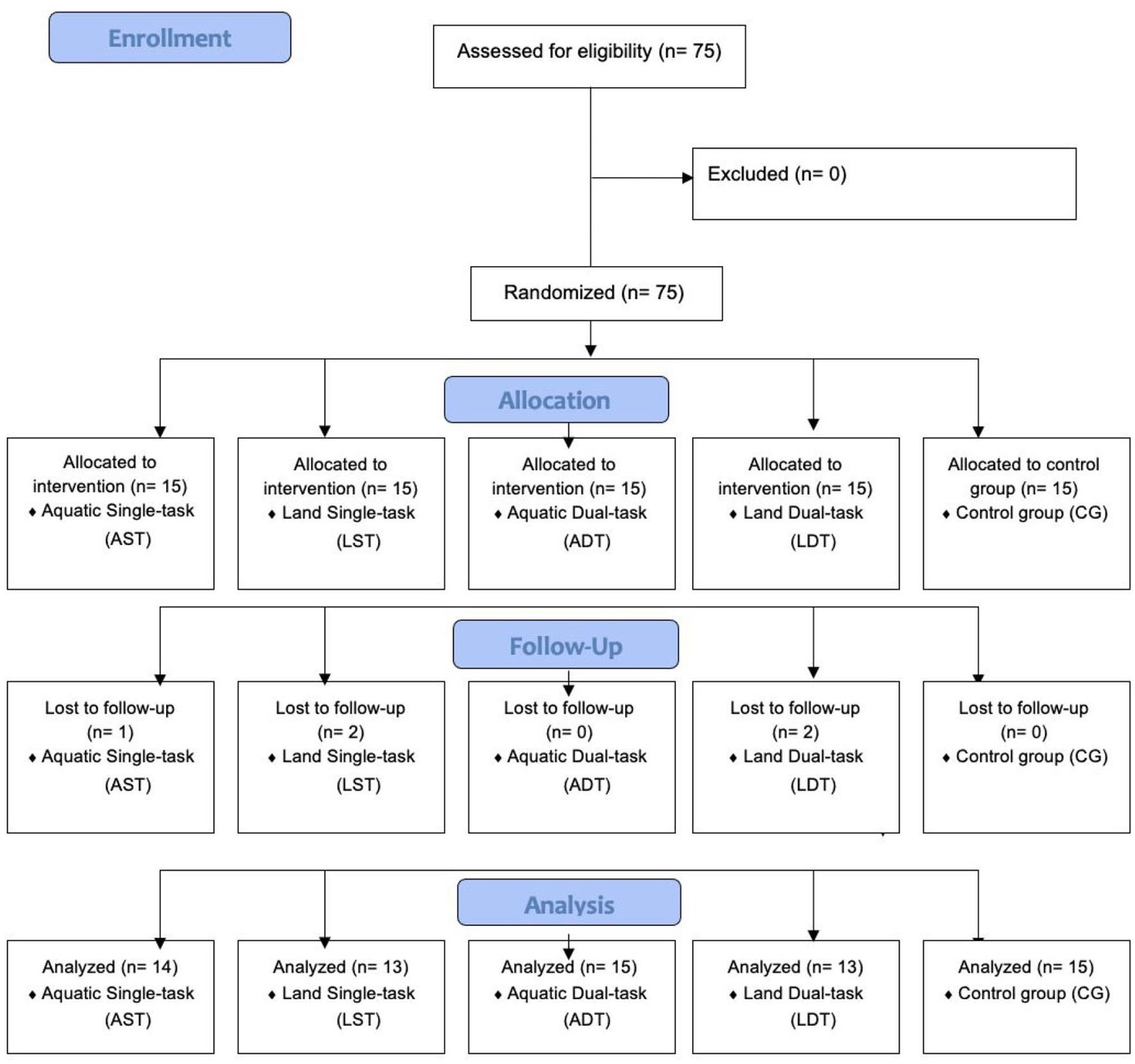
Study’s consort flow-diagram.
Planning of physical exercise programs.

Abbreviations: ADT, Aquatic dual-task; AST, Aquatic single-task; B, Block; CDT, Cognitive dual-task; H, Heating; HDT, Heating dual-task group; LDT, Land dual-task; LST, Land single-task; MDT, Motor dual-task; R, Relaxation.
Measurements
Recruited participants who met the inclusion criteria attended assessment locations (APP and Ouvidor Pardinho) to undergo cognitive, physical, and QOL tests, and sign the ICF. Each individual was assessed over 2 days: on the first day, they were familiarized with team, scales, and functional tests, and completed half of the cognitive tests (approximately 1 hour of assessment on this day); on the second day (the following week), they underwent the remaining cognitive tests, as some questions and activities were similar in some tests (approximately 20 minutes of assessment on this day). 16
All assessments and exercise programs were conducted during the “on” period of Parkinson’s disease medication, as this was when individuals had better motor and cognitive performance. 3
To ensure consistency and reliability of the measurements, healthcare professionals administering these assessments (physiotherapists and undergraduate students in Physical Therapy) underwent standardized training. A pilot study was conducted to evaluate inter-rater reliability using the Intraclass Correlation Coefficient (ICC). The average ICC for the variables analyzed was 0.848, indicating good inter-examiner reliability.
Research participants also responded to an initial questionnaire proposed by the authors with personal information related to Parkinson’s disease. In this assessment, they were asked to bring a report from the neurologist stating the current stage of Parkinson’s disease according to the Hoehn and Yahr scale. This scale describes 5 severity stages of Parkinson’s disease, ranging from 1 to 5, where a higher number reflects greater motor impairment, that is, more advanced Parkinson’s disease. 4
All cognitive, functional, and quality of life assessments employed in this study—such as the MoCA, SCOPA-COG, TUG, FTSST, Mini-BESTest, DGI, ABC, and PDQ-39—are standardized and widely recognized tools for evaluating people with Parkinson’s disease. These instruments were selected based on their clinical relevance, ease of administration, and robust psychometric properties, and are explicitly recommended by the most recent European Physiotherapy Guideline for Parkinson’s Disease. 15 Their inclusion ensures alignment with best practices in the field and facilitates the comparability of results with prior studies.
Cognitive Assessments
For the primary outcome, the Montreal Cognitive Assessment (MoCA) was used to screen cognitive changes, effective in distinguishing between normal cognitive aging and cognitive deficits in older adults. Its administration took around 10 minutes and evaluated 8 cognitive domains: executive function, visuospatial ability, memory, attention, concentration, working memory, language, and orientation. Scores ranged from 0 to 30, with higher scores indicating better cognitive performance. A score below 26 indicated mild cognitive impairment, while a score below 21 indicated dementia in Parkinson’s disease. 17
The Scales for Outcomes in Parkinson’s Disease-Cognition (SCOPA-COG) was used for cognitive screening, including memory, attention, executive functions, and visuospatial function. The assessment lasted approximately 10 minutes, with a maximum score of 43. Scores below 24 indicated mild cognitive impairment, while scores below 10 indicated dementia in Parkinson’s disease. 18
Functional Outcomes and Quality of Life
The Timed Up and Go (TUG) test assessed the time taken by participants to rise from a chair, walk 3 m, turn around, walk back, and sit down. The cutoff for fall risk in Parkinson’s disease was 11.5 seconds. The TUG test was also performed with dual tasks: motor (carrying a plastic cup with water) and cognitive (naming fruits). 19
The Five Times Sit to Stand Test (FTSST) measured the time taken to stand up and sit down 5 times in a chair. A shorter time reflected better functional mobility, with a cutoff of 16 seconds for fall risk in Parkinson’s disease. 20
The Mini-Balance Evaluation Systems Test (MiniBESTest) assessed body balance with 14 items scored from 0 to 2, with a maximum of 28 points. A higher score reflected better balance, with a cutoff of 21 points for fall risk in Parkinson’s disease. 21
The Dynamic Gait Index (DGI) analyzed gait and dynamic posture in different contexts, with 8 tasks scored from 0 to 3, and a maximum score of 24 points. A higher score reflected better gait performance, with a cutoff of 19 points for Parkinson’s disease. 22
The Activities-Specific Balance Confidence (ABC) scale assessed fear of falling through 16 questions about daily activities, with confidence levels ranging from 0% to 100%. The final score was the average percentage, with a cutoff of 76% for Parkinson’s disease. 23
The Unified Parkinson’s Disease Rating Scale (UPDRS) evaluated motor symptoms and daily activities. Sections II and III, most relevant to physical activity, were used. Section II had 13 questions and Section III had 14, both scored from 0 to 4, with higher scores indicating greater impairment. 24
The Parkinson’s Disease Questionnaire (PDQ-39) assessed quality of life with 39 questions across 8 domains. Scores ranged from 0 to 4, with higher scores indicating worse quality of life. 25
Sample Size
The sample calculation was performed using GPower 3.1 software, which determined a minimum sample of 12 individuals per group, assuming an effect size of 0.25 on an F probability distribution, where the value consists of the mean distance between the sample mean and the population mean; a Type I error equivalent to .05; and analysis power equal to .84.
Statistical Analysis
To perform the comparative analysis of the 5 groups, both descriptive and inferential statistical methods were applied. Qualitative variables were presented using absolute and relative frequency distributions. Quantitative variables were presented using measures of central tendency and variation. In the inferential analysis, the normality of the quantitative variables was assessed using the Shapiro-Wilk test.
The following methods were used for evaluation: the longitudinal comparison of quantitative variables (AV1 × AV2 × AV3) was conducted using the Friedman test, following the prerequisites. For comparisons among the 5 groups regarding quantitative variables, repeated measures ANOVA with Tukey’s post-hoc test was applied to control for multiple comparisons. This adjustment ensures that the risk of Type I error is minimized.
Subgroup analyses were also planned to provide additional insights, particularly for different stages of Parkinson’s disease (PD) as classified by the Hoehn and Yahr scale. These subgroup analyses aim to evaluate whether disease severity influences the effects of the interventions on cognitive, motor, and functional outcomes. By incorporating this level of analysis, the study enhances its ability to interpret results across varying levels of disease progression.
An alpha error of 5% was pre-established for the rejection of the null hypothesis. Statistical processing was performed using BioEstat version 5.3 and SPSS version 27.
Discussion
The present study aims to describe and compare 4 different physical exercise programs, including single-task and dual-task exercises, conducted both on land and in aquatic environments, and their potential effects on the cognitive and functional capacities of individuals with Parkinson’s disease (PD). 13
Systematic reviews indicate that both aquatic physical exercise and dual-task training are effective in improving cognitive and functional capacities in individuals with PD. However, the combination of dual-task exercises with aquatic environments still lacks evidence and a more detailed description in prescription, including types of exercises, repetitions, sets, duration, frequency, and training period. 8 Thus, this randomized controlled trial (RCT) established parameters for the prescription and monitoring of these types of physical exercises. This RCT was also developed to test exercise programs that can be reproduced in clinical settings or in other studies because it follows the necessary methodological recommendations for high-quality studies. 3
Critically evaluating these findings against existing evidence, the proposed interventions build upon prior studies that separately explored the benefits of dual-task training and aquatic exercises. Dual-task exercises, in particular, have shown promise in stimulating neuroplasticity and enhancing functional independence. 7 When combined with the physical and thermal properties of water, such as buoyancy, hydrostatic pressure, and viscosity, the interventions may reduce the load on joints, improve balance, and promote safer environments for individuals with PD. These unique characteristics enhance motor performance and allow for more complex cognitive engagement during exercises compared to land-based activities. 2
In terms of practical implications, healthcare professionals can leverage these findings to implement structured dual-task aquatic programs that are adaptable to diverse clinical settings. The scalability of these interventions lies in their simplicity, feasibility, and cost-effectiveness, making them accessible even in resource-limited environments. For instance, group sessions in community pools or rehabilitation centers can maximize social interaction and adherence to treatment while reducing the overall burden on healthcare systems.9,12,13
Additionally, this study underscores the importance of tailoring exercise programs to the individual needs of PD patients, particularly considering disease stage and functional limitations. These insights are essential for healthcare professionals to develop targeted interventions that optimize outcomes.4,26 Moreover, future training workshops for clinicians could focus on integrating the study’s findings into daily practice, promoting evidence-based care for individuals with PD.
This study protocol acknowledges certain limitations that will be important to address during the trial. First, while the aquatic environment offers unique physical properties such as buoyancy and hydrostatic pressure, the potential impact of water temperature on exercise performance will need to be monitored and analyzed. 2 This aspect could influence participant comfort and the outcomes of aquatic exercises. Second, differences in participant motivation between land-based and aquatic environments may emerge during the trial and will be observed and documented to better understand their potential effects on adherence and engagement. 10
Additionally, factors such as medication timing and its interaction with physical performance will be carefully tracked during the study period to assess their influence on variability in results. These considerations will guide future analyses and contribute to a more comprehensive understanding of the intervention effects.1,14
We expect that the results of this RCT will not only identify the most beneficial type of exercise or combination thereof but will also guide clinicians in prescribing interventions to maintain and improve cognitive, motor, and functional capacities in individuals with PD. The broader implications extend to improving quality of life and fostering independence in daily activities, ultimately benefiting both patients and their caregivers.
Conclusion
The AquaDualPark study presents a valuable opportunity to deepen our understanding of the differential impacts of physical exercise protocols on individuals with Parkinson’s disease, particularly by focusing on the unique nuances between land and aquatic environments. The study’s design not only compares single-task and dual-task exercises but also emphasizes the specific guidelines and distinctive benefits associated with terrestrial and aquatic settings. By highlighting these environment-specific strategies, the study aims to identify the most effective approaches for enhancing cognitive, motor, and functional capacities in this population. The rigorous methodology and practical focus of the study ensure that its findings will be both clinically relevant and easily applicable. Although there may be limitations, such as the absence of gold-standard assessments for balance and gait, the emphasis on real-world, reproducible exercise programs enhances its contribution to the field. Ultimately, the results are expected to support more tailored exercise prescriptions, thereby improving management and quality of life for individuals with Parkinson’s disease.
Footnotes
Appendix 1
Acknowledgements
We would like to express our gratitude to the Secretaria de Saúde de Curitiba for providing access to the therapeutic pool at the Ouvidor Pardinho Health Unit, which was essential for the implementation of this study. We also thank the Associação Parkinson Paraná for their long-standing partnership, offering their space and encouraging their members to participate in our research projects.
Authors Contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES), Finance Code 001.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
