Abstract
Introduction:
Alzheimer disease (AD) is the most common type of dementia, affecting 70% of patients with dementia. In Japan, over 5 million people aged 65 years and older had dementia in 2018, and this figure is expected to rise to 25% to 30% of this age group by 2045. In Japan, lecanemab, which was approved in 2023, is expected to be a new treatment for AD. However, lecanemab requires stringent management, including amyloid PET scans and MRI monitoring, necessitating specialized facilities, creating concerns regarding the lack of treatment facilities and poor treatment access.
Methods:
This study assessed spatial accessibility to lecanemab in Hokkaido, Japan, using geographic information system data. Hospitals were categorized into 3 scenarios based on their capacity to meet the treatment criteria. Service area analysis in each scenario evaluated the population coverage within 30-, 60-, and 120-minute travel times. The inverted two-step floating catchment area method was used to calculate the potential high-demand areas index (PHDI) for each hospital.
Results:
Population coverage ranged from 56% to 97%, depending on the scenario and travel time. Coverage for the most feasible scenario (Scenario 1) was 56%, 73.9%, and 88.3% within 30, 60, and 120 minutes, respectively. Northern and southern Hokkaido had the lowest coverage. PHDI analysis identified high-demand areas, with Sapporo facing potential overcapacity issues.
Conclusion:
Lecanemab highlights the need for strategic resource allocation to enhance accessibility and capacity. Establishing additional treatment centers, particularly in areas with poor accessibility and capacity, is crucial to maximize the benefits of treatment for dementia.
Keywords
Introduction
Alzheimer disease (AD) is the most common type of dementia and accounts for approximately 70% of all cases. 1 In Japan, a government survey estimated that, among the population aged 65 years and older, approximately 4 million had mild cognitive impairment (MCI) in 2012 and over 5 million had dementia in 2018.2,3 In 2019, dementia was the most common reason for patients requiring long-term care. 4 Additionally, the prevalence of dementia among those aged 65 years and older in Japan is estimated to reach 25% to 30% by 2045. 5 AD with cognitive and functional impairment creates a significant economic burden for caregivers and the healthcare system. 6 The direct burden on families who are responsible for caregiving and the pressure on the budgets of the insurers who administer long-term care insurance are significant. This economic burden is related to increasing dementia care needs and presents an ongoing challenge for Japan, which is facing a declining birthrate and an aging population. Hence, strategies to reduce the social and economic burden of dementia are needed.
In September 2023, the Japanese government approved lecanemab, a disease modifying therapy with a new approach to AD, following the Food and Drug Administration in the US. In the phase 3 CLARITY AD clinical trial, lecanemab reduced brain amyloid levels and cognitive and functional impairment by 27% after 18 months in individuals with MCI and mild AD. 7 Despite its expected efficacy, treatment with lecanemab requires stricter management before and during administration than conventional therapies. 7 For example, Apolipoprotein E (APOE) genotype testing, amyloid positron emission tomography (PET) or cerebrospinal fluid (CSF) analysis are performed before lecanemab administration to determine adaptation. CSF biomarkers have proven diagnostic accuracy for AD 8 and do not require expensive medical equipment like PET, but samples are unstable after collection and require rapid testing or frozen storage at −80°C.9,10 The potential for a blood test to diagnose dementia is also an area of promising research, although it is still in the development stage. 11 After treatment initiation, patients require periodic magnetic resonance imaging (MRI) to monitor for amyloid-related imaging abnormalities (ARIA), a serious adverse event. Specifically, MRI is needed before the fifth and seventh doses, followed by 6-month intervals thereafter. 12 Additionally, intensive care unit availability is also important for patients with severe ARIA. 12 In short, Lecanemab treatment requires expensive medical equipment and a trained multidisciplinary team including emergency department, knowledgeable MRI reader, clinicians with managing ARIA, 12 only a limited number of hospitals meeting all the requirements. Therefore, appropriate resource allocation is required to make a disease modifying therapy accessible to as many people as possible.
This analysis utilized geographic information system (GIS) data to evaluate spatial access to lecanemab treatment and identify the areas that contribute to improved access. GIS easily captures the address information of facilities and residents as well as road network information, thereby enabling detailed analysis. GIS data is widely used in the health policy context, making it suitable for the purposes of this study. 13 This study proposes improvements to accessibility for disease modifying therapy for AD and suggests that health policymakers and administrators increase access to superior treatments to reduce future social and economic burdens.
Methods
Study design
A cross-sectional study.
Study area
Our study focused on Hokkaido, which has the largest area (83 000 km2) and lowest population density (67 people/km2) in Japan. 14 Hokkaido’s population is 5.2 million, with 1.7 million people aged 65 and over, and an aging rate of 32%. The population is largely concentrated in the urban area of Sapporo (38%), whereas depopulation is progressing in rural areas. 14 Hokkaido faces the challenge of maldistribution of healthcare, and the Hokkaido government has acted to achieve healthcare equity through medical plans. Hokkaido has 21 secondary medical areas (SMAs), which are the basic districts used when planning systems to provide standard medical care. Therefore, we show the SMA levels in Figure 1.
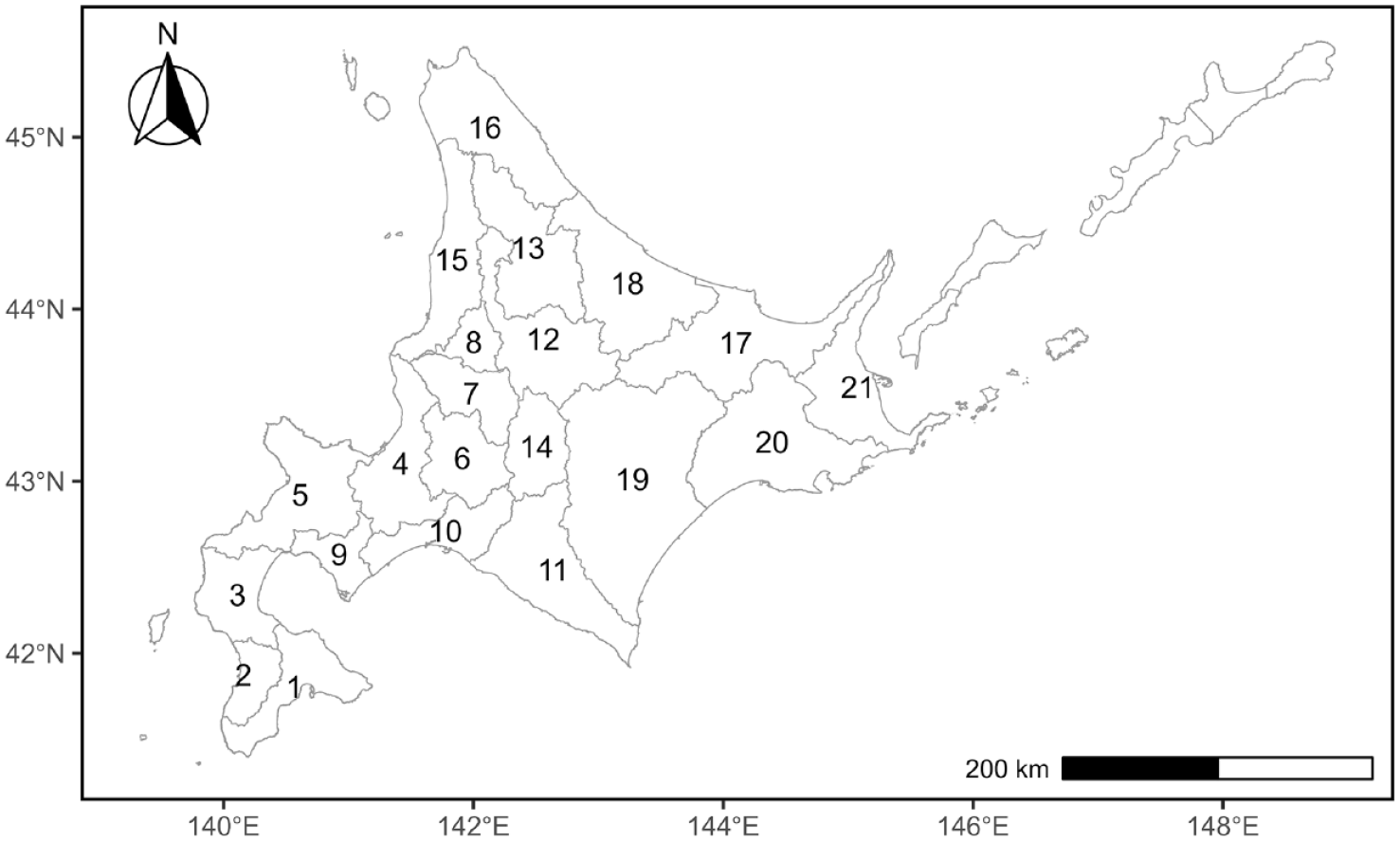
This shows the SMAs in Hokkaido, Japan. 1; Minamioshima, 2; Minamihiyama, 3; Kitaoshimahiyama, 4; Sapporo, 5; Shiribeshi, 6; Minamisorachi, 7; Nakasorachi, 8; Kitasorachi, 9; Nishiiburi, 10; Higashiiburi, 11; Hidaka, 12; Kamikawachubu, 13; Kamikawahokubu, 14; Furano, 15; Rumoi, 16; Soya, 17; Hokumo, 18; Enmon, 19; Tokachi, 20; Kushiro, 21; Nemuro.
Hospitals
We defined hospitals with the capacity for lecanemab as having the following: (1) at least one specialist in dementia from the Japanese Society for Dementia Research (JSDR) on staff, (2) MRIs at 1.5 T or more, (3) amyloid PET scans, or contracting with other facilities to use amyloid PET scans, and (4) a neurosurgery department. In Hokkaido, 9 hospitals met all the criteria, 15 hospitals met criteria (2)–(4), and 19 hospitals met criteria (2) and (3). Scenario analysis was performed based on these criteria. The base scenario (Scenario 1) met all the criteria. Scenario 2 met the criteria except for (1). Thus, the hospital could meet all criteria by assigning a new specialist from the JSDR. Scenario 3 is a hospital with MRI and PET facilities, and thus has equipment but lacks human resources. APOE genetic testing was not included in the hospital’s requirements because it is not covered by insurance in Japan.
Population
The estimated population aged 65 years and older in 2025 was employed in the form of mesh data in the geographical analysis. The projected future population, estimated based on the 2015 census, was publicly available from the Ministry of Land Infrastructure Transport and Tourism. 15 Most patients with dementia are aged 65 years or older 2 ; therefore, this study included a sample population in this age group. This dataset included 1.72 million individuals over the age of 65. The population was close to that reported in the Census 2020, as the population has been declining faster than estimated.
Service area analysis
A service area analysis was conducted to evaluate the spatial accessibility of lecanemab. 16 The analysis defined service areas as 30, 60, and 120 minutes, with each threshold time representing the travel time to the hospital by car. This allowed areas to be identified from where the hospital could be reached within a specified timeframe. To calculate population coverage, the reachable population within each threshold time was divided by the total population in the area. Thus, population coverage by service area analysis was used to estimate the population accessible to lecanemab based on travel time.
Inverted two-step floating catchment area method
The inverted two-step floating catchment area method (I2SFCA) was used to identify hospitals with high demand for lecanemab. The I2SFCA, developed by Wang, 17 calculates the population (ie, number of potential patients) covered by each facility according to the parameters of supply (eg, beds, doctors, medical equipment), demand (eg, population, prevalence of an illness), and travel costs (eg, time, distance, fare). This study called this score as the Potential High-Demand areas Index (PHDI). A positive correlation between PHDI and the number of patients discharged has been demonstrated. 18 This study defined demand as the population aged 65 and over, supply as 1 by all hospitals, and travel cost as the travel time required for residents to access a hospital. Thus, PHDI refers to the number of residents per hospital, indicating the potential number of patients. Finally, we determined the mean PHDI for the SMAs. This represents the mean population aged 65 years and older per hospital in the SMAs. To calculate the PHDI, an origin-destination cost matrix (ODM) was created using GIS. The ODM is a travel time matrix that connects residents to hospitals on a one-to-one basis and is based on travel by car. Thus, remote islands not connected to the hospital by road were excluded.
Data source
We collected hospital information from the Report on the Bed Functions 18 and a specialist in dementia from the JSDR website (March 2024). 19 The geospatial information necessary for the GIS analysis was collected from the Ministry of Land, Infrastructure, Transport and Tourism. 16 Road network data were used with ArcGeo Suite “Douromo 2021” (Esri Japan, Sumitomo Electric, Tokyo, Japan). Descriptive and summary statistics were generated using R version 4.1.1 and R studio.20,21 ArcGIS Pro 3.2 was used for the spatial analysis, such as the service area analysis.
Results
Service area analysis
Hospital service areas were set at 30, 60, and 120 minutes. The population coverage ranged from 56% to 97%. Scenario 3 at 120 minutes provided the highest population coverage (Table 1). For Scenario 1, with high feasibility, 56%, 73.9%, and 88.3% of those aged 65 years and older could access the hospital within 30, 60, and 120 minutes, respectively. Figure 2 shows the distribution of population coverage for the SMAs. The northern and southern areas had low coverage in all scenarios. When assuming a 30- or 60-minute service area, low coverage was noticeable in the north and south.
Population coverage at each scenario, service area in Hokkaido.

( ) is the number of hospitals.

This shows distribution of population coverage for each scenario and service area in the secondary medical areas. A darker color indicates lower coverage, while a lighter color indicates higher coverage: (a) Scenario 1; 30 minutes, (b) Scenario 1; 60 minutes, (c) Scenario 1; 120 minutes, (d) Scenario 2; 30 minutes, (e) Scenario 2; 60 minutes, (f) Scenario 2; 120 minutes, (g) Scenario 3; 30 minutes, (h) Scenario 3; 60 minutes, and (i) Scenario 3; 120 minutes.
We then estimated the population with poor access based on the population coverage (Table 2). For Scenario 1 at 30, 60, and 120 minutes, approximately 0.75, 0.45, 0.20 million people were not covered, respectively. These residents must accept travel costs of at least 30 minutes to access lecanemab. If residents accept travel costs of up to 120 minutes, the population with low access decreases to approximately 200 000. The distribution of the estimated population with poor access is shown in Figure 3. Sapporo and Minamioshima had large populations with poor access in Scenarios 1 (30 minutes) and 2. Therefore, while regional differences in coverage are important from an equity perspective, population is an important factor from an efficiency perspective. According to this analysis, Minamioshima had low population coverage, and a large population with poor access and was thus identified as a priority area for establishing a treatment center.
Estimated population over 65 years old with poor accessibility to lecanemab.


This shows distribution of estimated population over 65 years old with poor accessibility to lecanemab in the secondary medical areas. A darker color indicates large population, while a lighter color indicates small population: (a) Scenario 1; 30 minutes, (b) Scenario 1; 60 minutes, (c) Scenario 1; 120 minutes, (d) Scenario 2; 30 minutes, (e) Scenario 2; 60 minutes, (f) Scenario 2; 120 minutes, (g) Scenario 3; 30 minutes, (h) Scenario 3; 60 minutes, and (i) Scenario 3; 120 minutes.
Potential high-demand areas index by inverted two-step floating catchment area method
Next, we calculated the PHDI for the 3 models. Tables 3 to 5 summarize the PHDI for each scenario. In Scenario 1, 9 hospitals were located in 5 secondary medical areas. The average PHDI for one facility was over 100 000 in Sapporo. In Kamikawa Chubu and Tokachi, PHDI exceeded 100 000 at both 60 and 120 minutes. In Scenario 2, Sapporo’s PHDI was over 100 000 in all cases. However, PHDI did not increase even as the service area expanded from 30 to 120 minutes. In Scenario 3, the PHDI was under 100 000 in almost all cases because the number of hospitals offering lecanemab treatment has increased; however, Minamioshima was an exception, with a PHDI of over 100 000. In the context of this study, the single hospital situated in Minamioshima ensured comprehensive coverage of the accessible population in the surrounding area. The PHDI indicates the population aged 65 years and older covered by each hospital; therefore, the larger the number, the greater the potential number of patients. PHDI distribution is plotted in Figure 4. The darker color and the larger the circle indicate a higher PHDI.
Potential high-demand area index in Scenario 1.
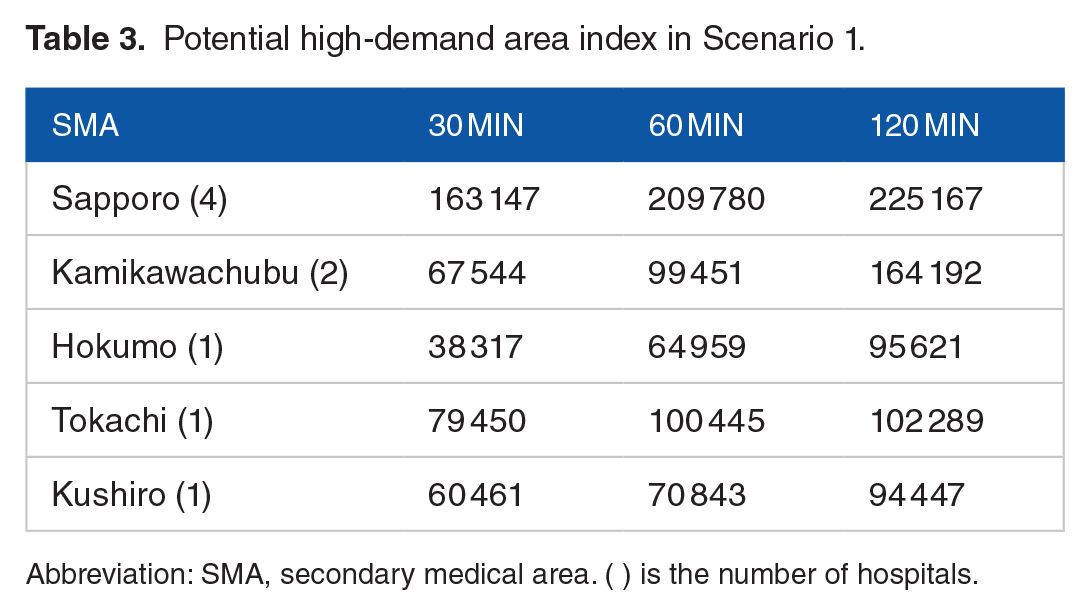
Abbreviation: SMA, secondary medical area. ( ) is the number of hospitals.
Potential high-demand area index in Scenario 2.
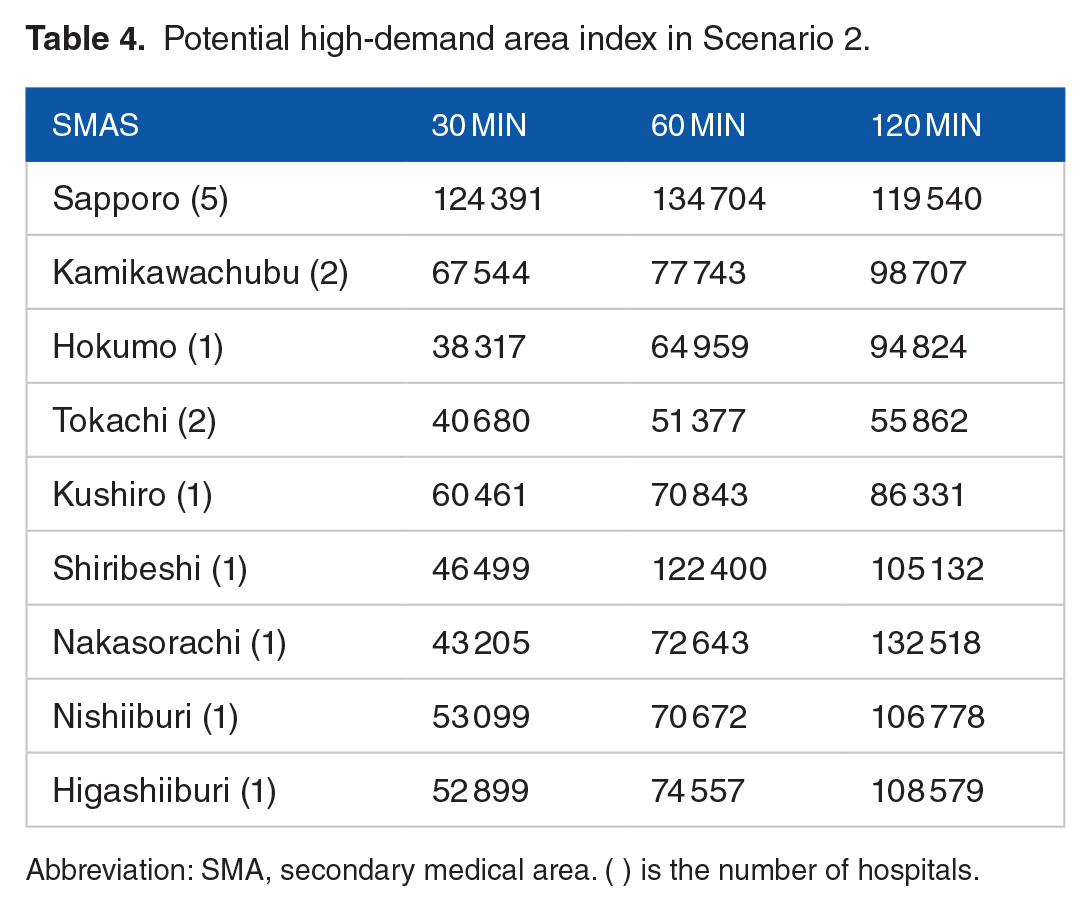
Abbreviation: SMA, secondary medical area. ( ) is the number of hospitals.
Potential high-demand area index in each scenario.
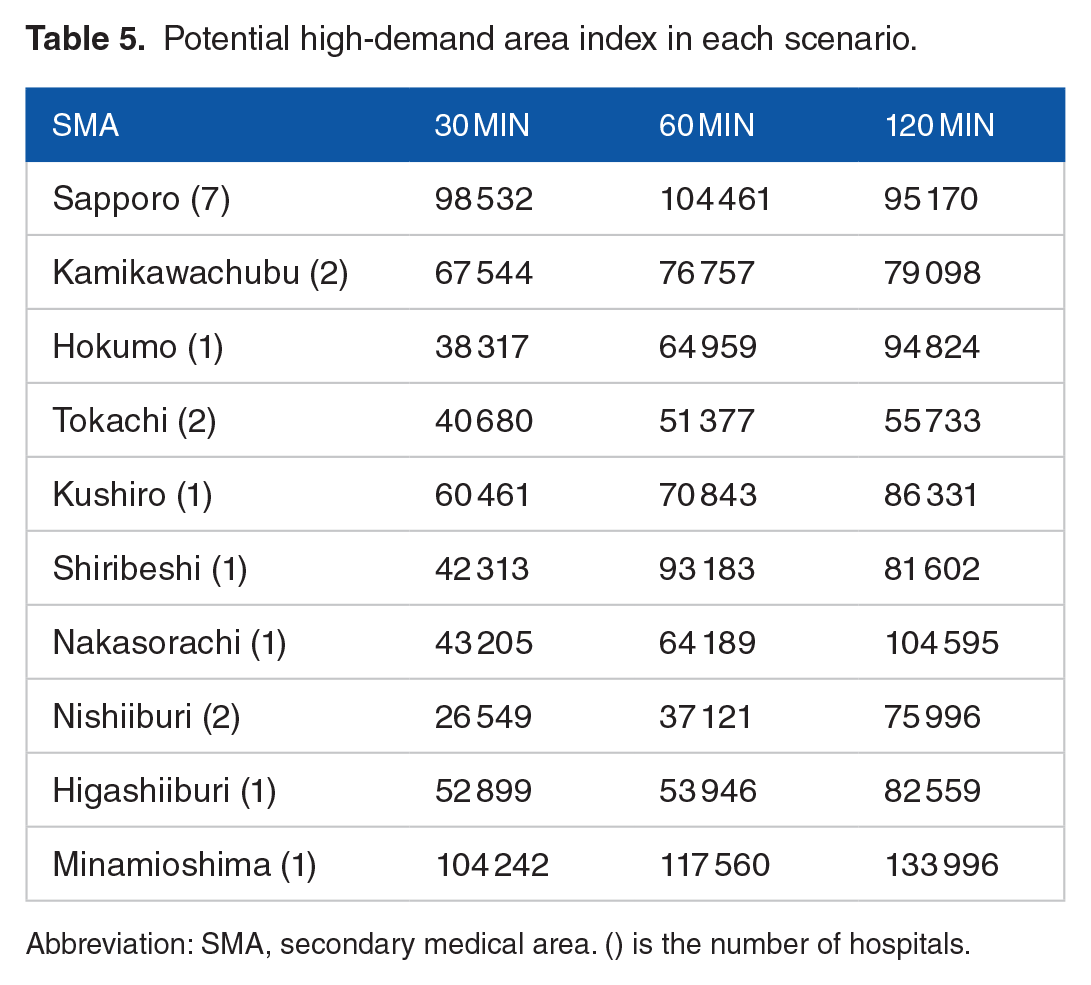
Abbreviation: SMA, secondary medical area. () is the number of hospitals.

This shows distribution of PHDI in the secondary medical areas. The larger the circle and the darker the color, the larger the PHDI. (a) Scenario 1; 30 minutes, (b) Scenario 1; 60 minutes, (c) Scenario 1; 120 minutes, (d) Scenario 2; 30 minutes, (e) Scenario 2; 60 minutes, (f) Scenario 2; 120 minutes, (g) Scenario 3; 30 minutes, (h) Scenario 3; 60 minutes, and (i) Scenario 3; 120 minutes.
Discussion
This study used GIS data to evaluate accessibility to hospitals that can provide lecanemab, for AD. Owing to the need for multiple tests to be performed before and during treatment, the availability of facilities offering lecanemab is limited. Under the medical resource capacity, effective allocation of treatment facilities is expected so that more people can receive lecanemab treatment. Japan has national health insurance and a free-access medical system. Thus, patients in Japan can receive medical treatment at any hospital directly or with a referral letter from their primary care provider. Patients select hospitals based on several factors such as illness severity, family finances, transportation, values, and health literacy. For example, approximately 80% of cancer patients are admitted to a hospital within 45 minutes of their homes to receive cancer treatment. 22 A study of Japanese claims data found that, compared to children and younger adults, older adults were more likely to visit a nearby medical facility. 23 Harada et al 24 revealed that participants with a longer distance from home to the venue (<1, 1-1.99, and 2-2.99 km and ⩾3 km) had a lower rate of participation in cognitive screening. The impact of travel costs is unknown in the context of decision-making regarding dementia treatment. Conventional treatment for early-stage dementia is often continued in neighborhood clinics. However, lecanemab requires a different treatment process that can influence patient decision-making. This study presents 3 different time scenarios. The standard access time was set to 60 minutes or less. However, if travel time is a barrier, it may be closer to the 30-minute scenario, resulting in fewer patients accessing treatment. If interest in dementia and expectations for treatment outweigh travel costs, then the 120-minute scenario would be more feasible. Therefore, by monitoring where patients visit, hospitals that have started offering treatment can identify areas where treatment access is lacking.
We used I2SFCA to identify areas with high treatment demand. In Scenario 1, which met all the requirements for this study, Sapporo had the highest PHDI. If a large proportion of residents accept higher travel costs, the demand for dementia treatment in the Sapporo area may exceed capacity. Fortunately, in terms of the number of CT and MRI owned, Japan has a higher capacity for patients with dementia than other G7 countries. 25 However, not all of them can be devoted to dementia treatment because hospitals equipped with MRI and PET scans do not necessarily have specialists to diagnose dementia. Furthermore, the schedule for lecanemab is one intravenous infusion every 2 weeks. This framework is similar to anticancer treatment, and the 2 treatment types will likely compete for space and staffing in Japan. The standard treatment period for lecanemab is 1.5 years; however, modeling studies have reported an average treatment period of 3.68 years. 6 Therefore, if lecanemab continues under the current protocol, patients will have long wait times and the workloads of doctors and other medical staff will reach their limit. Given the high number of patients with dementia, decentralization of treatment facilities is necessary. Scenario 2, which meets all the criteria except for the dementia specialist, was the most realistic scenario for the next step, as it includes facilities that could become treatment centers by assigning a specialist. At 60 min, this scenario reached 80% population coverage. An appropriate target population coverage has not yet been determined; however, 80% coverage may be achieved by having residents bear the cost of up to 60 minutes of travel. According to the prevalence of MCI (13%, 95% confidence interval 10-16), 10 000 to 16 000 of 100 000 people aged 65 years and older may have MCI. Pittock et al 26 reported that 17.4% of patients with MCI were eligible for lecanemab treatment. Based on this study, out of 10 000 to 16 000 people with MCI, we expect a maximum of 1740 to 2784 patients. Although the actual number of patients receiving treatment is determined by encounter rates, having treatment facilities in locations where travel costs are affordable means that new medications can reach more patients with dementia.
This study identified regions with limited access to lecanemab. In all scenarios, Soya exhibited low population coverage. Remote and rural areas face challenges in securing expensive medical equipment and specialist doctors due to concerns about efficiency. Additionally, Japan’s population is projected to decline, 14 making the expansion of hospitals in these areas an impractical strategy. While establishing a telemedicine model has the potential to address challenges in remote and rural settings, there is currently a lack of evidence supporting its effectiveness. 27 In terms of treatment accessibility, donanemab, 28 which requires less frequent administration compared to lecanemab, holds significant promise. Given the potential of less frequently administered therapies like donanemab to improve access in remote and rural areas, there is hope that these areas may see enhanced treatment availability. Moreover, such therapies could help reduce the burden on medical resources in urban centers. Thus, the development of telemedicine and innovations that reduce the consumption of medical resources will improve accessibility to treatment and benefit not only patients, but also caregivers and administrators.
Limitations
This study has several limitations. First, we conducted a simulation analysis based on open public data. Therefore, verification using real-world data is required. Starting in 2024, several hospitals have initiated lecanemab treatment. The availability of timely claims data allows patient access and regional disparities to be assessed. Second, the definition of treatment center in this study does not fully comply with optimal clinical use guidelines. According to the guidelines for optimal use of Lecanemab in Japan, a specialist from one of the following 4 organizations must be assigned as the person in charge of treatment: Japanese Society of Neurology, The Japan Geriatrics Society, The Japanese Society of Psychiatry & Neurology, The Japan Neurosurgical Society. 29 We confirmed the affiliation of these specialists with 17 out of 19 hospitals employed in this study in March 2024. Furthermore, not all hospitals included in the study scenarios will offer lecanemab treatment because of differences in institutional standards and hospital management strategies. Therefore, to evaluate the accessibility of lecanemab treatment, hospital information should be updated before future analyses are conducted. Third, we did not fully capture collaborations between hospitals. Therefore, if some hospitals try to compensate for the lack of criteria at their institutions by collaborating, our result may underestimate the accessibility of lecanemab. However, we confirmed the opinions of clinicians in this region regarding the presence or absence of collaboration to ensure data reliability. Finally, the transportation mode in this study was automobiles only. Public transportation is well-developed in urban areas but not in rural areas. Therefore, if public transportation is used, the accessibility in rural areas may worsen.
Conclusion
The approval of lecanemab, a disease modifying therapy for AD, has raised expectations in Japan. However, geographical accessibility for this treatment is unclear. This study quantified residents’ accessibility through multiple scenarios and identified areas that require more treatment centers and areas with high patient concentration.
Footnotes
Acknowledgements
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cross-ministerial Strategic Innovation Promotion Program (SIP) on “Integrated Health Care System” Grant Number JPJ012425. The funder had no role in the design of the study; in the collection; analysis, and interpretation of data; in the writing of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: Kazuki O, II, JZ, KS, IY, Katsuhiko O. Data curation: Kazuki O, II. Formal Analysis: Kazuki O. Funding acquisition: Katsuhiko O. Methodology: Kazuki O, Katsuhiko O. Project administration: Kazuki O, II, IY, Katsuhiko O. Resources: Kazuki O, II, JZ, KS, IY, Katsuhiko O. Software: Kazuki O, Katsuhiko O. Supervision: II, KS, IY, Katsuhiko O. Validation: Katsuhiko O. Visualization: Kazuki O, Katsuhiko O. Writing – original draft: Kazuki O. Writing – review & editing: Kazuki O, II, JZ, KS, IY, Katsuhiko O. All the authors have read and approved the final manuscript.
Ethical Considerations
This study was approved by Hokkaido University Faculty of Health Sciences Institutional Review Board as not requiring ethical review.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
Data available on request from authors.
