Abstract
The study objective was to determine whether the discounts provided under the 340B program help address disparities in drug treatment and adverse outcomes among Medicare Fee-For-Service (FFS) beneficiaries initially Medicare-diagnosed with moderate to severe chronic asthma. Using Medicare FFS claims data from 2017 to 2019, we conducted a cross-sectional study that compared risk-adjusted differences in 5 treatment measures and 5 adverse outcomes among beneficiaries treated within 340B and non-340B hospital systems that met the disproportionate share (DSH) criteria and the ownership classification requirement to qualify as a 340B DSH hospital. Our analysis focused on potential disparities that are historically associated with challenges to accessing quality health care. We did not observe fewer disparities in drug treatments or adverse outcomes for beneficiaries with moderate to severe asthma treated at 340B hospital systems compared to non-340B hospital systems. These results raise questions as to whether 340B hospital systems are effectively using discounts to focus on improved access and outcomes for vulnerable beneficiaries.
Main Text
Background
Section 340B of the Public Health Service Act was enacted in 1992 to help “stretch scarce federal resources as far as possible, reaching low-income and/or uninsured patients and providing more comprehensive services” by providing discounts on outpatient drugs to certain safety-net providers known as 340B covered entities (CEs). 1 Since the 340B program was created, it has quickly grown to account for almost 20% of the total value of concessions manufacturers provide for brand-name drugs 2 and 22% of the acute care hospitals in the US. 3 In 2020, purchases made under the program totaled $38 billion, a 27% increase over 2019. 2 In fact, the 340B program is now almost as large as the Medicaid program’s outpatient drug sales. 4 The growth of the 340B program has resulted in scrutiny from critics, some of whom question whether the program is serving its intended purpose. 5
Disparities in health care access and treatment generate an estimated $93B in excess medical expenditures and $42B in lost productivity each year. 6 Many governmental organizations, including the Health Resources Service Administration (HRSA), which oversees the 340B program, and the Centers for Medicare & Medicaid Services (CMS) have included health equity as a part of their strategic goals. In HRSA’s strategic plan, the first goal outlines “actionable steps to achieve health equity and improve public health” and specifies in Objective 1.4 the goal to “incorporate health equity concepts and measures into HRSA programs and policies.” 7 CMS developed an Equity Plan for Medicare that recognized existing disparities in health care access, quality and outcomes among populations CMS serves. 8 The plan presented an approach for achieving health equity that included CMS’ network of quality improvement partners, organizations, health care providers, health plans, and other stakeholders.
One approach to evaluate whether the 340B program is meeting its objectives of reaching vulnerable patients and providing more comprehensive services is to assess how well 340B CEs address known disparities in healthcare. Health disparities, broadly, are “. . .differences and/or gaps in the quality of health and healthcare across racial, ethnic, and socio-economic groups.” 9 Our analysis focuses on 340B disproportionate share hospital (DSH) CEs and Medicare Fee-For-Service (FFS) beneficiaries with initial diagnoses within Medicare of moderate or severe asthma because of the higher prevalence of asthma in vulnerable populations and the observed disparities in health care utilization and outcomes.
There are well documented disparities in asthma treatment and outcomes among vulnerable populations. For example, a 2014 study found that Hispanic patients with asthma had 43% lower odds of receiving a maintenance drug compared to non-Hispanic White patients. 10 A study published in the Annals of Allergy, Asthma and Immunology reported lower controller medication receipt, initiation and use among racial and ethnic minorities with asthma. 11 A separate study that evaluated the relationship between SES correlates, treatment failures and asthma exacerbations found that low income was positively associated with greater risk. 12
Vulnerable populations with asthma also have greater risk of adverse outcomes such as emergency department use and hospitalizations. Data from the 2015 Behavioral Risk Factor Surveillance Survey and the Asthma Call-Back Survey revealed that 32% of Black patients and 23% of Hispanic patients had reported an emergency department visit due to asthma symptoms in the last 12 months, compared to only 14% of White patients who reported the same. 13 Low SES leads to treatment and outcome disparities among asthma patients as well. A systematic review and meta-analysis of 61 asthma-related studies found that lower SES was associated with increased emergency department utilization and hospitalization. 14
Hospital systems participating in the 340B program have an opportunity to reduce health disparities and adverse outcomes if providers use their savings from 340B discounts on eligible covered outpatient drugs to address access barriers and improve the quality of care provided to low-income or uninsured patients (who are more likely to have chronic health conditions). There are no guidelines or requirements as to how 340B hospital systems should use the savings generated from the discounts. Given the intent and mission of the 340B program, many stakeholders believe that the savings from these discounts should decrease treatment or cost barriers experienced by vulnerable patients and their providers, resulting in improvements in the health care of these patients and a corresponding reduction in health disparities. 15 This study provides evidence of whether the 340B program is meeting its objectives for improving access to care for vulnerable beneficiaries with moderate to severe chronic asthma.
The objective of this study was to determine whether the 340B program is associated with fewer or smaller health disparities for asthma-related care from 2017 to 2019 by examining the race/ethnicity, dual Medicare and Medicaid eligibility (dual status), and socioeconomic factors of beneficiaries treated at 340B and non-340B hospital systems. We hypothesize that hospital systems participating in 340B should have smaller disparities in asthma-related drug treatments compared to hospital systems not participating in 340B as the discounts 340B hospital systems received on drugs should help reduce barriers to access, allowing beneficiaries to properly control their asthma. As a result of fewer disparities in drug treatment, we also hypothesize that there will be smaller disparities in adverse outcomes related to asthma for 340B hospital systems compared to hospital systems not participating in 340B. Our work contributes to the literature by measuring disparities in treatment and health outcomes within 340B hospital systems and evaluating whether the drug discounts received by 340B hospital systems lead to lower treatment barriers and fewer adverse outcomes for vulnerable patients with moderate to severe chronic asthma.
Methods
We conducted a retrospective analysis of Medicare beneficiaries treated for moderate to severe asthma within 340B hospital systems and non-340B hospital systems that met the disproportionate share hospital (DSH) and ownership classification criteria to qualify as a 340B DSH hospital. 340B hospital systems, which include owned outpatient sites (known as child sites), were identified using The Office of Pharmacy Affairs Information System (OPAIS) database. The parent hospital of these systems met the 11.75% operating DSH threshold for eligibility and participated in the program at any point between 2017 and 2019. Next, we used Inpatient Prospective Payment System (IPPS) impact files to identify short-term acute hospitals that did not participate in the 340B program during 2017 to 2019 but had an operating DSH percentage that meets the threshold requirement for 340B eligibility and were not classified as for-profit.
The beneficiaries included in this analysis were initially Medicare-diagnosed with moderate to severe chronic asthma treated at the 340B and non-340B hospital systems in our sample. We used Medicare claims (2017-2019) to identify beneficiaries with at least 1 inpatient or 2 outpatient claims within 30 days with the diagnosis of interest, and no asthma-related claims in the 12-months prior to the diagnosis (index date). The ICD-10 diagnosis codes used to identify asthma were J454x and J455x. Beneficiaries were attributed to a 340B or non-340B hospital based on the plurality of non-infusion claims received from hospital outpatient departments.
Our analysis focused on disparities defined by 3 patient characteristics that are generally associated with access challenges to quality healthcare. Those characteristics are race/ethnicity (White vs non-White), dual enrollment status, and socioeconomic status (SES). Beneficiary race/ethnicity and dual enrollment status were obtained from the Master Beneficiary Summary File (MBSF). We used the 2019 Agency for Healthcare Research and Quality (AHRQ) SES index from the Acxiom’s InfoBase®16 and mapped it to beneficiaries using the 5-digit zip code. Beneficiaries were assigned a low SES area if their residence was in the lowest quartile of the metric, and a high SES area if within the highest quartile.
We evaluated 5 drug treatment measures and 5 adverse outcome measures related to asthma occurring within the first 12 months of the diagnosis index date. The treatment measures include number of days to drug therapy initiation from the index date, proportion of beneficiaries receiving maintenance drugs (drugs used to control symptoms of asthma), proportion of beneficiaries receiving novel therapies (biologic therapies used to control symptoms of asthma and approved by the FDA in 2017 or later), proportion of beneficiaries receiving rescue drugs (drugs used for the immediate relief of symptoms), and the proportion of beneficiaries receiving any outpatient drug treatment. Higher quality care is defined as fewer days to therapy start, receipt of maintenance drugs or novel therapy drugs, which implies the beneficiary is managing their asthma, particularly, with newer innovations. Lower quality care is defined as a delay to therapy start or receipt of rescue drugs, which may indicate uncontrolled asthma. Receipt of outpatient drugs reflects the access to outpatient drugs, which the 340B program is designed to provide. The 5 adverse outcome measures include the occurrence of an acute asthma event, all cause emergency department visits, receipt of inhalation treatments, inpatient admissions with asthma as primary reason, and all-cause mortality within 1 year of the diagnosis index date. Visits to the hospital or an outpatient facility for an adverse event indicates that asthma is not being properly managed. The treatment and outcome measures were identified using Medicare FFS claims. Details for how each measure was constructed and the drugs included in the analysis are available in Supplemental Appendix A.
Our study estimated risk-adjusted rates for each outcome measure across 340B and non-340B hospital systems, separately, using a generalized linear model (logit or binomial link). Each model included control variables for age, gender, clinical risk factors, and hospital characteristics. Age and gender were identified using the MBSF, and clinical risk factors were identified in FFS claims using the CMS Hierarchical Condition Category (HCC) indicators from the CMS-HCC model (version 24). Hospital characteristics, which included hospital size (bed size categorized into small, medium, and large hospitals), rural or urban geographic location, and academic medical center affiliation, were identified using the 2019 CMS Provider of Services file. The covariates included in each generalized linear model were selected using a least absolute shrinkage and selection (LASSO) estimator. We estimated a generalized linear model with the selected covariates for each outcome on a reference sample (beneficiaries receiving care at non-340B hospital systems meeting the DSH threshold and ownership requirement), a sample for whom, in the aggregate, the average expected (model-predicted) value equals the average actual value of the outcome. We applied each model to the 340B sample of beneficiaries and created observed and expected rates for all beneficiaries. Finally, we used the delta method to derive estimates of standard errors for each group. We report within-group differences and between-group differences; statistically significant differences were measured based on P-values ⩽.05. The data used in this study was anonymized and therefore this study was exempt from an Institutional Review Board.
Results
In the following section we describe the sample included in this analysis and the results broken out by vulnerable subpopulation.
The sample was comprised of 45 175 beneficiaries attributed to 1275 340B hospital systems and 2638 beneficiaries attributed to 286 non-340B hospital systems. Table 1 shows that approximately 25% of beneficiaries in our sample are non-White, 31% are dual eligible and 36% reside in low-SES areas. More than 70% of the sample is female and the average age is under 70 years old. Hospital system characteristics are presented in Table 2. DSH-eligible 340B hospital systems are more likely to be located in urban areas, to have larger bed-counts, and to be affiliated with a medical school compared to DSH-eligible non-340B hospitals. The difference in number of hospitals included in each sample, combined with the differences in hospital size, contributed to the differences in sample size.
Comparison of beneficiary characteristics.

Source: Medicare Beneficiary Summary File and Medicare FFS Claims, 2017 to 2019.
Abbreviations: HCC, hierarchical condition categories; SES, socioeconomic status.
Comparison of hospital characteristics.
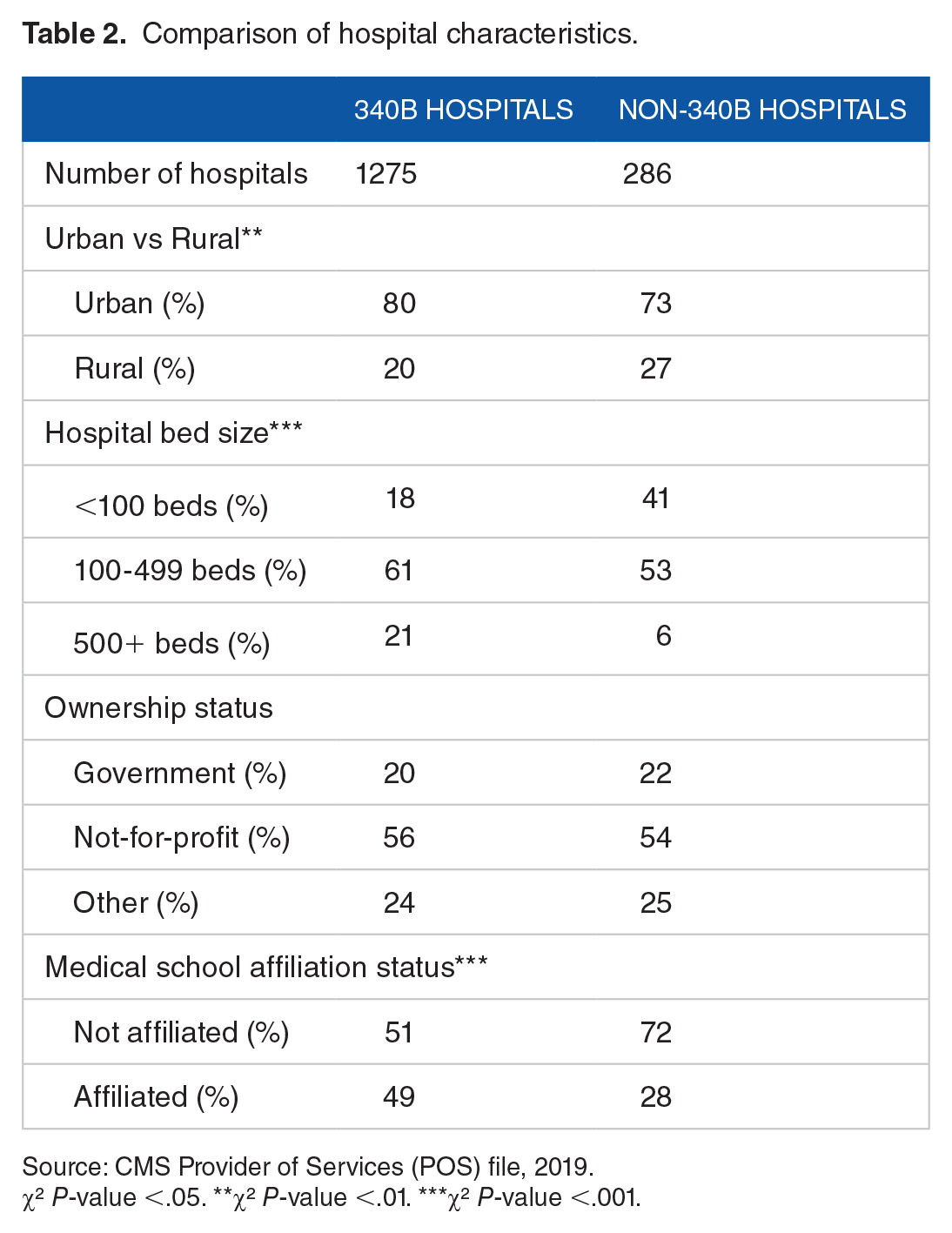
Source: CMS Provider of Services (POS) file, 2019.
χ² P-value <.05. **χ² P-value <.01. ***χ² P-value <.001.
Disparities by race or ethnicity
Our analysis found differences in drug treatment patterns between non-White and White beneficiaries for select outcome measures in 340B hospital systems (Table 3). While there were no differences in the days to drug therapy start or receipt of an outpatient drug, non-White beneficiaries treated at 340B hospital systems were less likely to receive a maintenance drug (8.0% vs 10.8%, P < .001) or a novel therapy drug (17.0% vs 23.3%, P < .001) than White beneficiaries, and more likely to receive a rescue drug (19.1% vs 16.4%, P < .001). A statistically significant difference in treatment by race also occurred at non-340B hospital systems; non-White beneficiaries were less likely to receive a maintenance drug (5.8% vs 9.8%, P < .05).
Risk-adjusted racial disparities for medicare beneficiaries with asthma treated at 340B and non-340B health systems.

Source: Medicare FFS Claims, Enrollment and Acxiom Data, 2017 to 2019.
The outcome measure was analyzed on a subset of our sample limited to beneficiaries who received outpatient drug therapy. Thus, the sample size for these measures is smaller than indicated in the table.
Asterisks represent statistical significance: *P < .05, **P < .01, ***P < .001.
We also observed higher rates of adverse outcomes for non-White beneficiaries compared to White beneficiaries. Non-White beneficiaries had significantly higher rates of acute asthma events (12.5% vs 9.2%, P < .001), ED visits (70.0% vs 59.8%, P < .001), inhalation treatment (37.5% vs 29.8%, P < .001), and inpatient stays (7.7% vs 3.6%, P < .001) than White beneficiaries treated at 340B hospital systems. These patterns were similar for beneficiaries treated at non-340B hospital systems; however, the difference was only statistically different for inpatient stays (8.5% vs 2.7%, P < .01).
Overall, the findings do not show that racial disparities in the quality of drug treatments or adverse outcomes differed between 340B and non-340B hospital systems. However, the magnitude of the disparities observed differed between 340B and non-340B hospital systems for select outcome measures. The rate of ED visits was 10.3% points higher, and the rate of inhalation treatment was 7.7% points higher, for non-White beneficiaries treated at 340B hospital systems, but only 3.0% points and 3.5% points higher, respectively, for non-White beneficiaries treated at non-340B hospital systems.
Disparities by dual eligible status
Disparities in drug treatment for asthma between dual eligible and non-dual eligible beneficiaries were identified for the 5 treatment measures within 340B hospital systems, but in only 1 of 5 measures within non-340B hospital systems (Table 4). Dual eligible beneficiaries treated at 340B hospital systems started drug therapy later (86.8 days vs 77.8 days, P < .01), were less likely to receive a maintenance drug (8.6% vs 10.8%, P < .001) or novel therapy (13.1% vs 26.4%, P < .001), and more likely to receive a rescue drug (22.2% vs 14.8%, P < .001) or any outpatient drug (28.5% vs 23.2%, P < .001). Greater access to any outpatient drug may be a result of the 340B program and a positive outcome, but this result in combination with disparities in a delay to treatment or receipt of specific types of drugs that manage asthma indicates gaps in the care provided. Similar treatment patterns occurred for dual eligible beneficiaries treated at non-340B hospital systems; however only the disparity in the receipt of a novel therapy (10.4% vs 18.8%, P < .05) was statistically significant.
Risk-Adjusted Disparities for Dual and Non-Dual Eligible Medicare Beneficiaries with Asthma Treated at 340B and Non-340B Health Systems.

Source: Medicare FFS Claims, Enrollment and Acxiom Data, 2017 to 2019.
The outcome measure was analyzed on a subset of our sample limited to beneficiaries who received outpatient drug therapy. Thus, the sample size for these measures is smaller than indicated in the table.
Asterisks represent statistical significance: *P < .05, **P < .01, ***P < .001.
Dual eligible beneficiaries with asthma were also more likely to experience adverse outcomes than non-dual eligible beneficiaries at 340B and non-340B hospital systems. Among those treated at 340B hospital systems, dual eligible beneficiaries were more like to have an acute asthma event (11.8% vs 9.2%, P < .001), ED visit (73.4% vs 57.0%, P < .001), inhalation treatment (38.9% vs 28.4%, P < .001), inpatient admission (7.2% vs 3.4%, P < .001), and death (5.2% vs 4.3%, P < .05) than non-dual eligible beneficiaries. Similar differences in adverse outcomes occurred among those treated at non-340B hospital systems, however only differences for ED visits (71.0% vs 55.9%, P < .001), inhalation treatments (35.3% vs 26.6%, P < .05), and inpatient admissions (6.4% vs 3.3%, P < .05) were statistically significant.
There were no statistically significant net differences in the disparities observed among 340B and non-340B hospital systems for dual eligible beneficiaries. Despite the lack of statistical significance, the magnitudes of the disparities were larger for dual eligible beneficiaries treated at 340B hospital systems compared to non-340B hospital systems for 9 of the 10 measures evaluated. The largest net difference occurred for receipt of a novel therapy and inhalation treatment, where there was a relative net difference of 4.8% points and 1.8% points, respectively.
Disparities by socioeconomic status
There is evidence of socioeconomic disparities in drug treatment for asthma at 340B and non-340B hospital systems. Table 5 shows that beneficiaries with low SES treated at 340B hospital systems were less likely to receive a maintenance drug (8.8% vs 10.8%, P < .01) or novel therapy (16.0% vs 25.9%, P < .001), but more likely to receive a rescue drug (20.1% vs 15.1%, P < .001) or any outpatient drug (26.8% vs 23.6%, P < .01) compared to beneficiaries with high SES. Among beneficiaries treated at non-340B hospital systems, differences in drug treatment between those with low and high SES existed for receipt of a maintenance drug (6.0% vs 10.3%, P < .05) and novel therapy (8.4% vs 16.1%, P < .05).
Risk-Adjusted Differences for Low and High SES Medicare Beneficiaries with Asthma Treated at 340B and Non-340B Health Systems.

Source: Medicare FFS Claims, Enrollment and Acxiom Data, 2017 to 2019.
The outcome measure was analyzed on a subset of our sample limited to beneficiaries who received outpatient drug therapy. Thus, the sample size for these measures is smaller than indicated in the table.
Asterisks represent statistical significance: *P < .05, **P < .01, ***P < .001.
Socioeconomic disparities were also present within select adverse outcomes. Among beneficiaries treated at 340B hospital systems, those with low SES were more likely to have an ED visit (67.9% vs 58.2%, P < .001), receive inhalation treatment (36.9% vs 27.6%, P < .001) or have an inpatient admission (6.9% vs 3.8%, P < .001) compared to beneficiaries with high SES. Disparities also exist among beneficiaries treated at non-340B hospital systems, however a statistically significant difference was observed only for inpatient admissions (6.8% vs 2.6%, P < .05).
The net differences in the disparities reported for low SES beneficiaries treated at 340B and non-340B hospital systems were not statistically significant, although the magnitudes in the disparities differed. The magnitude of the disparities was larger at 340B hospital systems for receipt of novel therapy drugs (2.2% point difference). The magnitudes of the disparities were larger for non-340B hospital systems for the receipt of a maintenance drug (2.4% points) and having an inpatient admission (1.0% points).
Discussion
The results indicate that there are risk-adjusted disparities in drug treatment and health outcomes for Medicare FFS beneficiaries with asthma based on race/ethnicity, dual eligibility status, and socioeconomic status. Statistically significant differences across the 10 outcomes were more likely to occur within 340B hospital systems. Furthermore, for metrics in which there were disparities at both 340B and non-340B hospital systems, the magnitude of those disparities were larger at 340B hospital systems than non-340B hospital systems, on average. However, when we compare the disparities in the quality of drug treatments or adverse health between 340B and non-340B hospital systems, we find no statistically different differences.
The disparities observed at 340B and non-340B hospital systems for health outcomes of beneficiaries with asthma were consistent with the literature. Racial minorities and beneficiaries with lower socioeconomic status were less likely to use maintenance drugs and had higher rates of rescue drugs, reflecting higher rates of asthma exacerbations compared to White beneficiaries or those with high socioeconomic status. Racial minorities and beneficiaries with lower socioeconomic status also had higher risk-adjusted rates of ED use and hospitalizations.
Considering that 340B hospital systems receive discounts on drugs, we hypothesized that vulnerable populations would experience fewer barriers to care at 340B hospital systems, receive improved drug treatment from such institutions, and, as a result, experience fewer disparities in the care provided and adverse outcomes. Our results found no statistically significant differences between the disparities observed in 340B and non-340B hospital systems. The drug discounts 340B hospital systems receive had no impact on reducing disparities in drug treatments for asthma, or in the disparities of adverse outcomes related to asthma.
The primary limitation of this analysis was that it only included medications administered at hospitals and outpatient sites and does not include Medicare Part D claims data. As a result, there may be Part D drugs used for asthma treatment or maintenance that are not captured in this analysis. The 340B program does not dictate discount for Part D drugs. Thus, the program’s impact on treatment with Part D drugs is likely limited. However, any treatment with Part D drugs may have an influence on the study’s adverse health outcomes. Our selection of beneficiaries and the drugs examined were identical across subgroups and hospital system types (340B, non-340B). We anticipated that any use of Part D drugs in addition to Part B drugs and their impact on health outcomes will be similar across samples within our analysis.
There are additional limitations. First, this analysis was limited to beneficiaries enrolled in Medicare FFS, but we believe that the results observed between 340B and non-340B hospitals are generalizable across age groups and payer types, particularly non-Medicaid. Second, the models control for beneficiary characteristics, including HCC scores that reflect the previous 12 months of claims, but may not fully reflect the beneficiary’s medical history. Also, this analysis was designed as a cross sectional study, and as such did not analyze changes in disparities in relation to changes in 340B status or changes in disparities within hospital systems over time. As a result, there may be incremental changes in the magnitude of the disparities studied between 2017 and 2019 that were not measured.
These results raise questions as to whether resources allocated to 340B hospital systems are being effectively used to reduce or eliminate treatment barriers and adverse outcomes for vulnerable patients with moderate to severe chronic asthma. In particular, the rate of adverse outcomes for vulnerable patients at 340B hospital systems were generally higher than their rates at non-340B hospital systems. The same is true for treatment with a rescue drug. While the remaining drug treatment outcomes were generally better for vulnerable patients at 340B hospital systems than at non-340B hospital systems, they were also better for non-vulnerable patients.
More days to drug treatment, lower use of maintenance drugs and novel therapies and higher use of rescue drugs among vulnerable subgroups indicate disparities in the access to care and the care provided. We expected that there would be smaller or no disparities at 340B hospital systems compared to non-340B hospital systems, and we find that the disparities were larger or equal to those at non-340B hospital systems.
The current 340B rules allow for participants to obtain discounts on drugs for all non-Medicaid patients (vulnerable and non-vulnerable) but provide no direction for how the savings from the discounts are used. This may lead to misaligned incentives on whether to participate in the program and how to use savings generated from participation. Savings from discounts are not necessarily used to reduce treatment barriers.
Many stakeholders have expressed concern over the evolving implementation of the 340B program, asking specifically whether the program is adequately serving vulnerable beneficiaries, as intended. Despite this concern, there have not been substantial reforms to the 340B program that address accountability. To increase transparency in the 340B program, several Members of Congress have introduced a bill which would permit the Secretary of Health and Human Services to audit 340B hospitals’ records to determine how net income from 340B discounts is used. 17
Policymakers and interested stakeholders continue to explore a wide range of options to strengthen the 340B program and to realign its focus on vulnerable populations. The goals of these policy options are to ensure that vulnerable patients are the focal point of the program by considering guardrails in the use of 340B discounts. Realigning the focus of the 340B program on vulnerable populations is also aligned with HRSA’s strategic plan, and CMS’s broad efforts to improve health equity and eliminate disparities in health care.
Supplemental Material
sj-docx-1-his-10.1177_11786329231169257 – Supplemental material for The 340B Program and Health Disparities Among Medicare Beneficiaries With Chronic Asthma
Supplemental material, sj-docx-1-his-10.1177_11786329231169257 for The 340B Program and Health Disparities Among Medicare Beneficiaries With Chronic Asthma by Amanda S Tripp, Grecia Marrufo, Thomas Kornfield, Melissa Morley, Donald Nichols and Abra Yeh in Health Services Insights
Footnotes
Acknowledgements
The authors would like to thank Ekemini Isaiah, Kolton Gustafson, Megan Peterson, Reed Diskey, Yonatan Mengesha for their research support and Shruthi Donthi and Ed Drozd for programing support.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Genentech, Inc.
Declaration Of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Donald Nichols is employed and Abra Yeh was formerly employed by Genentech, Inc. All other authors declare that there is no conflict of interest.
Author Contributions
TK, DN, AY contributed to the conceptual design; AST, GM, TK, MM and DN contributed to the analysis and interpretation of the data; AST drafted the manuscript; all authors contributed to the manuscript revisions and approved the final manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
