Abstract
Antimicrobial prophylaxis used for surgical procedures remains one of the measures for the prevention of surgical site infections (SSIs). The present study was designed to assess the compliance with clinical practice guideline for antimicrobial prophylaxis in variable surgeries conducted in the intensive care units (ICUs) of a major referral hospital. This cross-sectional investigation was carried out by prospective data collected from October 2017 to March 2018 in the intensive care unit (ICU) of Nemazi hospital in Shiraz. Demographic characteristics, surgery type as well as antibiotic treatment were gathered from medical records and entered in data collection forms. We reviewed compliance and adherence of prophylactic antibiotic administration to the Infectious Diseases Society of America (IDSA) guideline and evaluated the courses of antimicrobial drugs. If an antibiotic administrated for surgical prophylaxis was different from the guideline, the antibiotic was classified as non-guideline-based antibiotics. Most patients participated in this study were male (64.5%). Only 8.75% of the administrated antibiotics chosen for surgical prophylaxis were found to be appropriate antibiotic prescriptions; however, those patients receiving appropriate antibiotics prescribed an inappropriate dosage. In addition, the antibiotics were administrated with inappropriate durations in all cases. Our findings indicated that adherence to the IDSA international guideline seems to be far from ideal in Namazi hospital for antimicrobial prophylaxis, resulting in the unsuitable administration of a wide variety of antibiotics.
Introduction
Surgical site infections (SSIs), based on the Centers for Disease Control and Prevention (CDC), are characterized by infections that occur at or close to the surgical cut up to 30 days following surgery (or up to 1 year after surgery in implant-receiving patients) and influence the incision or deep tissue at the operation site.1,2 The prevalence rates of SSIs were reported to be up to 20%, leading to considerable mortality and morbidity as well as high-cost treatment. SSI prevention is dependent on a mixture of preoperative arrangements, surgical skills, and antibiotic prophylaxis.3,4
In the last decades, a wide variety of investigations have shown that the proper administration of prophylactic antibiotics is effective in decreasing the risk of SSIs. Antibiotic prophylaxis efficiency in the prevention of postoperative lesion infections was demonstrated in a wide variety of investigations.5,6 During last decades, a great number of studies have reported optimal prophylaxis, leading to progress and improvements of guidelines for surgical prophylaxis.7-9 Given the well-established instructions, an appropriate antibiotic/antimicrobial agent should protect patients against SSI-causing pathogens, be established at the appropriate time, and be administered only for 24 h. 10
Despite the accessibility to these surgical prophylaxis guidelines, studies evaluating surgical prophylaxis demonstrated that inappropriate timing of antibiotic administration, inappropriate antibiotic prescribing, and long duration of antibiotic therapy develop complications in surgical prophylaxis.11-13
Overall agreement for the application of prophylactic antibiotic guidelines is widely diverse with low-frequency in the world, ranging from less than 1% in Iran14,15 and Korea 16 to 28%, 33.2%, 36.3%, and 41.1% in Dutch 5 Malaysian, 17 Greek, 18 and French, 19 respectively.
The long-term follow-up of surgical antibiotic prophylaxis (SAP) is critical to ensure the appropriate use of antimicrobial agents.20,21 This will lead to a decrease in the consequences of antibiotic misuse, including increased antibiotic resistance, harmful incidents, and higher costs to the hospital.4,21,22
Therefore, the aim of the present study was to evaluate the quality of clinical practice guidelines for antimicrobial prophylaxis in variable surgeries to the compliance to antibiotic prophylaxis conducted in the intensive care units (ICUs) of a major referral hospital for patients in Fars province, Iran.
Materials and methods
Study design
The present study was established to evaluate antibiotic prophylaxis in patients admitted to the ICU ward for a 6-month period. The study was designed according to the guidelines developed by the American Society of Health-System Pharmacists, Infectious Diseases Society of America (IDSA), Surgical Infection Society, and Society for Healthcare Epidemiology of America.
Ethical approval and setting
The study was conducted in the ICU ward of the largest hospital in Fars province, Iran. The present investigation was carried out in accordance with the Declaration of Helsinki. The protocol of the present study was studied, reviewed, discussed, and approved by the Ethical Review Committee for Research in Human Subjects, the Ministry of Health and Medical Education, and the Ethical Committee of Shiraz University of Medical Sciences. Before the study, written informed consents were obtained from the patients participated in this study.
Data collection
This study was conducted on patients admitted to the hospital between October 2017 and March 2018. Potential participants were excluded from the study if they were unwillingness to participate in the study and/or a history of taking antibiotics before surgery the procedure for reasons other than prophylaxis. Files were reviewed while the patients were in the post-operative period. Data, including demographic information, surgery type, and antibiotic treatment (such as agents, doses, dose intervals, the number of doses, and durations of administration) were collected from medical records and subsequently entered in data collection forms. The criteria for inclusion in the study were based on elective surgical operations.
Assessment of antibiotic prophylaxis
The compliance and adherence of prophylactic antibiotic administration to the IDSA guideline was reviewed. Four features of antimicrobial prophylaxis assessed were as follows:
choice: antibiotic choice for patients
dose
timing of dose
duration of use
The courses of antimicrobial drugs were evaluated. If an antibiotic administrated for surgical prophylaxis was different from the guideline, the antibiotic was classified as non-guideline-based antibiotics. Once a factor related to the antibiotic prescription lacked sufficient data, the case was classified as missing data only in this factor. Table 1 summarizes criteria required for evaluation of adherence.
Summary of Infectious Diseases Society of America guideline recommendations for antimicrobial prophylaxis in adults.

Abbreviations: AP, antimicrobial prophylaxis; GI, gastrointestinal; IV, intravenous.
A Microsoft Excel table was tailored to register type of the surgical procedure used, pharmacotherapy received, antibiotic usage (generic name, doses, dose intervals, and durations of administration).
Statistical analysis
All the data collected in this study were coded and analyzed by SPSS for Windows, version 19.0 (SPSS Inc., Chicago, IL). Frequency and percentages from the data were measured and presented. P values <0.05 were statistically considered significant.
Results
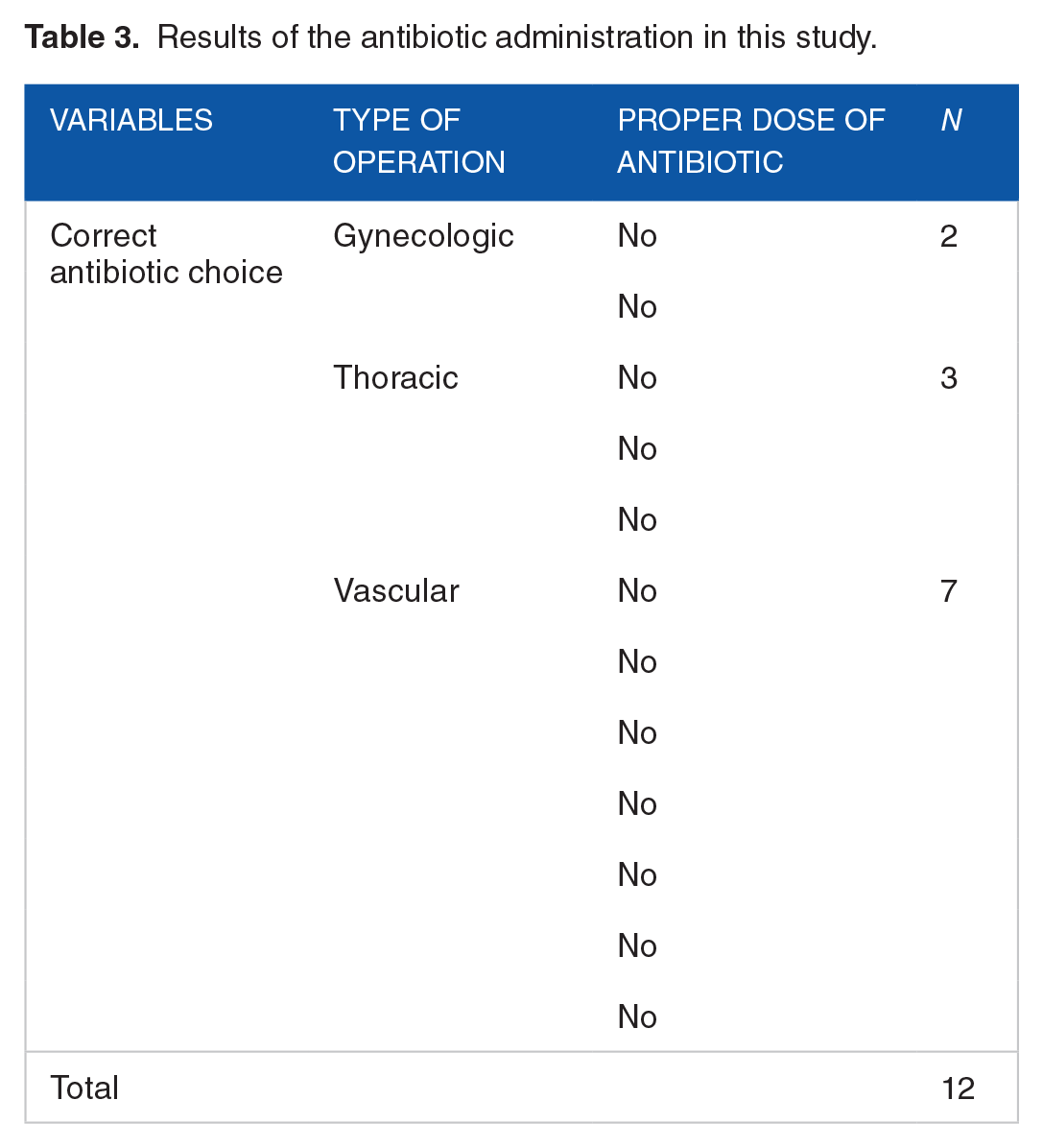
In all, 137 patients were enrolled in this study, in which 92 (64.5%) were males and 45 (35.5%) were females. The age of the participants were from 18 to 92 years (a mean age of 57.74 ± 15.72 years). The most common surgical specialties included gastrointestinal (28.46%), genitourinary (27.7%), vascular (13.1%), orthopedic (8.75%), and neurosurgery (1.45%), respectively. Table 2 represents antibiotic regimens used in operations. Antibiotic prophylaxis, within 2 h pre-operatively, was used in 100% of the cases (137 patients). The selection of antibiotics administered for antibiotic prescription was found to be correct in 8.75% of the cases. Antibiotic administration was performed at the inappropriate dosing in all cases who were administered by correct choice of antibiotic (Table 3). In addition, the antibiotic was administrated at the inappropriate duration of use in all cases, so that the mean of treatment duration was 6.15 days, with a minimum and maximum of 1.1 and 13.6 days, respectively. Most of the patients received the dual therapy (two antibiotics) that was not according to the guideline. This is also worth mentioning that among 137 patients, 26 patients died, 33 patients were discharged, and the rest of the patients were transferred to the wards.
Antibiotic regimens used in operations.

Results of the antibiotic administration in this study.
Discussion
Results from this study showed an extensive discrepancy between Namazi hospital's SSI prophylaxis practice and the protocols offered by international and national guidelines. Our results demonstrated that correct antibiotic selection administered for antibiotic prescription was 8.75%. The antibiotic administration was carried out at an inappropriate dosing in all patients who received the correct choice of antibiotic. Moreover, antibiotic administration was carried out at the inappropriate duration of use in all patients.
Guidelines are used to help clinicians and other practitioners in clinical decision making by defining a range of overall appropriate approaches for the management, diagnosis, or prevention of particular diseases and conditions. 23 An intervention research conducted in a university hospital in the Netherlands demonstrated that the misuse and suboptimal timing of antibiotics used for surgical prophylaxis were approximately 66% and 56% of the procedures, respectively. 24 In the present study, inappropriate timing, inappropriate dosing, and incorrect choice of antibiotics for surgical prophylaxis were observed in 100%, 100%, and 91.25% of the cases, respectively. This non-adherence can be attributed to the lack of paying attention to the guidelines, or inappropriate application of international or national protocols of antibiotic prophylaxis by health care practitioners in Namazi Hospital. Other possible explanations for the non-adherence include physician's concern about increased incidence of complications in under-treated patients in case of “discontinued” use of antibiotics.
Since the past decades, local hospital guidelines were established in a majority of hospitals in the Netherlands for increasing the quality of antibiotic prophylaxis. However, there are critical deficiencies in the adoption of CPGs in practice. Future strategies may circumvent such issues through understanding the forces and variables governing practice as well as applying practice- and community-based methods rather than didactic ones. 25 In a study, in which patients underwent abdominal aortic aneurysm repair, hip replacement, or large bowel resection in 44 hospitals from New York State, Silver et al 26 showed that 14% did not receive antibiotic prophylaxis, and only 63% of those receiving antimicrobials were administered within 2 h before incision. 26 Recent investigations have revealed that inappropriate antibiotic choice, inappropriate timing of antibiotics, and excessive duration of use remain serious problems in surgical prophylaxis.27,28
In a study conducted in Canada, Zoutman et al. investigated the use of antibiotic prophylaxis among patients undergoing surgical repair of a fractured hip using prosthetic material insertion. The authors reported that 70% of the patients failed to pre-operatively receive an antibiotic prophylaxis dose in 2 h; instead, patients received antibiotic prophylaxis either too early or during the procedure. In a study, Zoutman et al 29 reported that 39% of the antibiotic prophylaxis receiving patients did not receive the first dose until the end of the operation.
A wide variety of studies worldwide showed that most countries use adherence to international guidelines in antibiotic prophylaxis. In a study, Gorecki et al 30 demonstrated that 74% of the 211 patients with elective or emergency surgery in a New York private teaching hospital received inappropriate antimicrobial prophylaxis administration according to Surgical Infection Society guidelines. Problems were found to be excessive duration (66%), switch to inappropriate antibiotics (32%), spectrum (31%), and timing (e.g., no pre-operative dose, 22%). 30 In a study conducted in 2006, Van Disseldorp et al 31 investigated the use of pre-operative antibiotic prophylaxis guidelines in León, Nicaragua. They projected that antibiotic selection was discordant with the hospital guidelines in 69% of the cases, doses in 20% of the cases, as well as administration timing and duration in 78% of the cases. In addition, the complete adherence was reached 7% of the patients. 31
In summary, the present study demonstrated that antibiotic misuse and overuse occurs widely in the hospital studied, presumably leading to the emergence of antibiotic-resistant pathogens. The establishment of an antibiotic stewardship program in Namazi Hospital is the best way to ensure the application of antibiotic prescription according to the international guidelines.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance of all patients. This research was supported by Shiraz University of Medical Sciences. A part of the abstract from this manuscript was presented at the 2nd International Congress on Biopharma as a poster presentation with interim findings. The authors would like to announce their appreciation to all contributors who have made the achievement of this study. This work was supported by a research grant (453) from Vice chancellor for research, Birjand University of Medical Sciences.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
Study conception and design: A.V, N.H, F.D, and M.M.
Data collection: M.M
Statistical analysis: A.V and F.D
Interpretation of results: E.A and M.M
Drafting of the manuscript: All authors
Revision of the final manuscript: E.A
All authors approved the final version of the manuscript, including the authorship list.
Compliance with ethics guidelines
The study was conducted in the ICU ward of the largest hospital in Fars’s province of Iran. This investigation was performed in accordance with the Declaration of Helsinki. The protocol of the present study was reviewed, discussed, and approved by the Ethical Review Committee for Research in Human Subjects, the Ministry of Health and Medical Education, and the Ethical Committee of Shiraz University of Medical Sciences. Before the study, written informed consents were obtained from patients participated in this study.
