Abstract
Background:
Schistosoma mansoni remains a significant public health problem in Ethiopia despite over a decade of deworming targeting school-aged children (SACs), often neglecting adults. Prevalence surveys relying on the Kato-Katz (KK) technique may underestimate infection, highlighting the need for updated data using more sensitive diagnostics.
Methods:
A cross-sectional study was conducted from February to June 2023 among 634 SACs and 558 adults in the Amhara Regional State. Data on risk factors were collected via structured questionnaires. S. mansoni infection was assessed using KK and real-time polymerase chain reaction (RT-PCR) on stool, and point-of-care circulating cathodic antigen (POC-CCA) on urine. Egg counts from 2 KK readers were averaged to determine final eggs per gram (epg). Data were analyzed using Statistical Package for the Social Sciences version 21; descriptive statistics and poisson regression model with cluster-robust standard errors were analyzed.
Results:
A total of 452 SACs (71.3%) and 330 adults (59.1%) were tested positive for S. mansoni at least by one diagnostic method. Prevalence in low-, moderate-, and high-transmission settings was 56.9%, 81.5%, and 95.5% among SACs, while it was 50.5%, 55.5% and 96.5% among adults, respectively. KK detected light, moderate and heavy infections among 170 (26.8%), 62 (9.8%) and 29 (4.6%) SACs, respectively, while it was 96 (17.2%), 38 (6.8%) and 3 (0.5%) in adults, respectively. SACs had higher epg than adults (148.0 vs 96.6; P = .001). School-aged children who swam or bathed in surface water had a significantly higher prevalence of S. mansoni infection (prevalence ratio (PR) = 2.679, P = .002). Similarly, SACs who always participated in agricultural activities (PR = 1.266, P = .037) and those who were not aware of schistosomiasis (PR = 1.364, P = .038) had a higher prevalence of infection compared with their counterparts. Among adults, crossing surface water barefoot was significantly associated with a higher prevalence of S. mansoni infection (PR = 1.481, P = 0.018).
Conclusions:
S. mansoni infection remains highly prevalent among both SACs and adults in the study area. In SACs, key risk factors included swimming or bathing in surface water, participation in agricultural activities and lack of awareness about the disease, while in adults, crossing water barefoot increased infection risk. These findings highlight the need to expand deworming programs to adults and implement targeted interventions, including health education and improved water access, to reduce transmission in endemic areas.
Background
Schistosomiasis (SCH) is a neglected tropical disease (NTD) caused by parasitic flatworms of the Schistosoma genus. 1 It is prevalent in communities with limited access to healthcare facilities, potable water and sanitation, making it one of the most widespread NTDs of poverty across sub-Saharan Africa (SSA), the Middle East, Asia, and Latin America. 2 Globally, 70 countries have widespread SCH, putting 700 million people at risk of infection and over 200 million already infected. Despite comprising only 13% of the world’s population, SSA accounts for over 90% of infections and 280 000 deaths annually.1,2
In Ethiopia, 480 of 966 mapped districts are endemic for SCH, affecting 53.3 million people, including 4 million cases. 3 The diversity in the distribution of different Schistosoma species across endemic places is determined by the presence of suitable water bodies, intermediate snail hosts, and environmental, climatic, and physicochemical parameters. 4 Consequently, Ethiopia hosts 2 Schistosoma species: S. mansoni, which is widespread across the country, including the Amhara Region, and S. haematobium, which is limited to the Afar, Somali, Gambella, and Benishangul-Gumuz regions. 5 According to the regional mapping report for 2015 to 2020, S. mansoni was reported in 74 districts out of 154 mapped districts in the Amhara Region. Seven districts (Tegede, Tach Armachiho, Metema, Dewe Harewa, Takusa, Sekota town and Sekota zuria) had ⩾50% S. mansoni prevalence. Moderate and low prevalences were reported in 21 districts each. Twenty-five and 80 districts had <1% and 0% prevalence, respectively. 3 Systematic reviews of previous studies using stool microscopy revealed pooled prevalence between 17.5% and 41.11% in the region.6 -9
Infection occurs when cercariae penetrate the skin of individuals in contaminated water, with activities like swimming, fishing, and washing clothes posing key risks. 10 School-aged children (SACs) are particularly vulnerable, but adults engaged in water-related activities are also at risk.8,10,11
While school-based mass drug administration (MDA) has reduced SAC infections, 3 adults remain untreated, potentially reinfecting treated children. Epidemiological data targeting all at-risk populations in the Amhara Region is inadequate, even though the region is designated as one of the most impacted regions in the country. 3 Moreover, past studies used the less sensitive Kato-Katz (KK) diagnostic method, underestimating the disease burden. 12 Thus, this study aims to determine the prevalence, intensity and associated factors of S. mansoni infection among SACs and adults in the Amhara Region using parasitological, immunological, and molecular diagnostics.
Methods
Study Design
A cross-sectional study was conducted among SACs and adults in the Amhara Regional State, northwest Ethiopia, from February to June 2023.
Study Setting
The Amhara region is divided into 13 zones and 7 city administrations, with the study taking place in 7 zones located in northwest Ethiopia: North Gondar, Central Gondar, West Gondar, South Gondar, East Gojjam, West Gojjam, and Agew Awi Zone (Figure 1). The 7 selected zones are recognized endemic areas for intestinal schistosomiasis, and therefore were prioritized for inclusion in the study. Most of the population resides in rural areas where agriculture is the primary livelihood. The study area is also home to key geographical features, including Lake Tana, the Blue Nile River, and several dams, such as the Koga and Tana Beles dams.

Map of the study area showing selected districts and schools, 2023.
Study Participants
The source population for this study included all SACs and adults living in the districts of northwest Ethiopia. The study populations consisted of SACs attending selected primary schools during the data collection period, and adults residing in selected households. Volunteer SACs (aged 6-14) and adults (aged ⩾18) who had lived in their current residence for at least 6 months prior to data collection were eligible for inclusion. Children and adults who had taken any anthelmintic drugs within 6 months before the data collection were excluded, as were disabled individuals who were unable to respond to research questions or provide stool and urine samples. A single population proportion formula was used to calculate the sample size separately for SAC and adult participants, based on the following assumptions: a 50% prevalence of S. mansoni in northwest Ethiopia, a 5% margin of error (d = 0.05), a 95% confidence level (Zα/2 = 1.96), a design effect of 1.5, and a 10% nonresponse rate. The 50% prevalence was assumed due to the lack of previous epidemiological data using combined parasitological, immunological, and molecular diagnostic tests in northwest Ethiopia or similar study settings. In addition, use of 50% provides the highest sample size.
Hence, the sample size was 635 for SACs and 635 for adults. According to the World Health Organization (WHO) guidelines, districts are classified into 4 endemicity categories based on the prevalence of SCH: nonendemic (prevalence <1%), low-endemic (prevalence 1-9.9%), moderate-endemic (prevalence 10-49.9%) and high-endemic (prevalence ⩾ 50%). 13 In northwest Ethiopia, districts were first grouped according to this classification using the SCH endemicity map (Source: Amhara Regional Health Bureau). One to three districts were randomly selected from each transmission stratum, with the number of districts per stratum determined proportionally based on the total number of districts in that stratum. Nonendemic stratum was excluded from the selection, as data from this area was not relevant to the study’s research questions.
To select SAC participants, 2 primary schools in each district were purposively chosen based on proximity to permanent water bodies (rivers, lakeshores, or irrigation canals). If more than 2 schools in a district met this criterion, 2 were randomly selected using a lottery method. The number of SACs selected from each school was proportionally allocated based on student numbers and classes. Finally, participants from each class were selected by systematic random sampling technique (Figure 2).
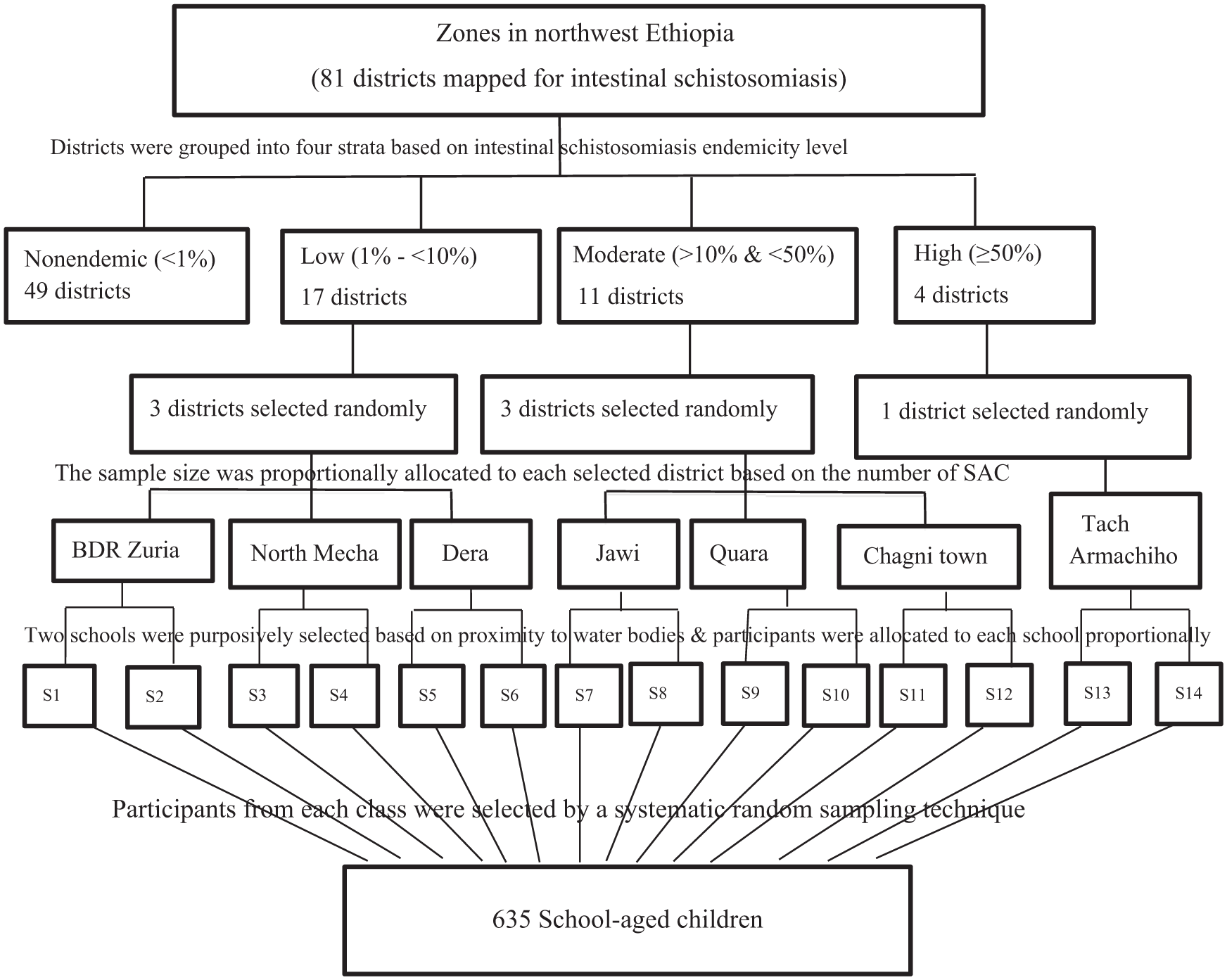
Schematic representation of the sampling procedure for selecting districts, schools and SACs, 2023.
To recruit adult participants (age ⩾ 18), 1 village closest to each selected school was purposively selected. Households in the village were then selected through systematic random sampling, with the number of households matching the number of SACs recruited from the respective school. Household lists were obtained from the corresponding health posts. One adult was randomly selected from each household for participation.
Variables
Schistosoma mansoni infection status (infected/non-infected) was the dependent variable while the independent variables were:
✓ Socio-demographic characteristics of SACs: Age, gender, residence, grade, parent’s marital status family educational status, family occupation, and family size.
✓ Socio-demographic characteristics of adults: Age, gender, residence, marital status, educational status, primary occupation, and family size.
✓ Water, sanitation and hygiene practices: Latrine availability and utilization, sources of water for drinking/bathing/in-house consumption, swimming habit, washing clothes in open water sources, crossing river barefoot, fetching water from open sources, playing near an open water source, participating in irrigation, or fishing practices.
✓ Awareness about intestinal Schistosomiasis
Data Sources
Questionnaire-Based Data
Data were collected through face-to-face interviews conducted by trained health extension workers using the Amharic version of the questionnaire. For SACs, data were collected in the presence of parents or school headmasters. For adults, data were collected through house-to-house visits or at a nearby health post. The questionnaire was designed to collect information on participants’ sociodemographic characteristics, clinical data, and water, sanitation and hygiene (WASH) practices.
Laboratory Diagnostic Approaches
The combined use of KK, point-of-care circulating cathodic antigen (POC-CCA), and polymerase chain reaction (PCR) provides a more comprehensive and accurate assessment of S. mansoni prevalence than any single diagnostic method alone. KK, even though less sensitive, enables standardized comparison with historical data, POC-CCA improves detection of active and light infections in field settings, and PCR offers high analytical sensitivity for confirming low-intensity and subclinical infections. Hence we used a combined diagnostic approach integrating these 3 test methods in order to improve prevalence estimates across transmission settings. 14
Stool and Urine Collection and Examination
After explaining the collection process, participants were given 2 wide-mouthed cups: one for collecting approximately 5 g of stool and the other for 3 mL of random midstream urine. Urine samples were tested for circulating cathodic antigen (CCA) using the POC-CCA test kit (Rapid Medical Diagnostics, Cape Town, South Africa; Batch Number: 220902098). Stool samples were transported at room temperature to nearby health centers for KK processing on the day of collection. The remaining stool samples were transported to Bahir Dar University’s College of Medicine and Health Sciences, Parasitology Laboratory, and stored at −80°C for molecular analysis by real-time polymerase chain reaction (RT-PCR). Stool KK and urine POC-CCA tests were performed according to previously described protocols. 15 Trace POC-CCA results (visible but not clearly pink line on the test line position) were considered as positive. Schistosoma mansoni infection was confirmed when at least 1 of the 3 tests (POC-CCA, KK, RT–PCR) was positive. In this study, participants were classified as infected if positive by at least 1 of the 3 tests (parallel interpretation) to maximize case detection in this post–mass drug administration setting, where infection intensities were expected to be low. However, we clearly acknowledge that parallel interpretation reduces overall specificity, due to the imperfect specificity of tests like POC-CCA.
DNA Extraction and Molecular Detection of S. mansoni From Stool
Schistosoma mansoni egg deoxyribonucleic acid (DNA) was extracted from stool samples using the QIAamp® Fast DNA Stool Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. A 180 to 220 mg stool sample was mixed with 1 mL of InhibitEX buffer and homogenized by vortexing. The suspension was incubated at 95°C for 5 minutes, then vortexed and centrifuged. A 200 µL aliquot of the supernatant was combined with proteinase K and AL buffer, and then heated at 70°C for 10 minutes. After adding 200 µL of absolute ethanol, the mixture was vortexed, and 600 µL was transferred into a QIAamp spin column. The column was centrifuged at 13 200 rpm for 1 minute. The DNA trapped in the column was washed twice with AW1 and AW2 buffers and finally eluted with 200 µL of ATE buffer. The DNA was stored at −80°C for downstream processing.
DNA detection was performed using smcyt748F forward and smcyt847R reverse primers, and the smcyt785T probe targeting the cytochrome c oxidase subunit 1 (cox1) sequence in the S. mansoni mitochondrial genome. The primers and probe sequences, designed to amplify a 99-bp fragment of the target gene, were as follows:
smcyt748F: 5′-CCCTGCCAAATGAAGAGAAAAC-3′
5′-TGGGTGTGGAATTGGTTGAAC-3′
smcyt785 T: 5′-/56-FAM/CCA AAA CCA/ZEN/GAC CCC TCT CAA ATT G/3IABkFQ/-3′
DNA amplification and detection were performed using a QuantStudio 5 RT‒PCR thermocycler (Thermo Fisher Scientific, Life Technologies Holdings Pte Ltd., Singapore) and HID RT‒PCR Analysis Software v1.3. The 25 µL reaction mixture contained 12.5 µL of Hotsplit MasterMix 2X (Biotools), 0.2 µL of 10 µM probe (Integrated DNA Technologies), 0.5 µL of 10 µM forward and reverse primers (Merck Life Science), 1.3 µL of nuclease-free water, and 10 µL of template DNA. The thermal cycling conditions were: initial hold at 95°C for 3 minutes, followed by 40 cycles of 95°C for 15 seconds, 60°C for 30 seconds, and 72°C for 30 seconds.
Positive controls, consisting of DNA from a microscopically confirmed S. mansoni-positive stool sample, were included in each run. A blank containing 25 µL of mastermix and a negative control with 15 µL of mastermix and 10 µL of distilled water were also included. Samples with cycle threshold (CT) values < 38 were considered positive. All samples were tested once, but those positive by POC-CCA or KK but negative by RT‒PCR were retested.
Data Quality Assurance
Data collectors for the questionnaire, stool, and urine samples underwent training prior to the study. A pretest was conducted at Abiot Fana Primary School and the nearby village (West Gojjam, Ethiopia), involving 5% of the sample size (32 SACs and 32 adults). Standard operating procedures and manufacturer instructions were strictly adhered to for all laboratory procedures. The KK smears were prepared in duplicate, and the expiration dates of all reagents and test kits (KK, POC-CCA, DNA extraction, and RT-PCR) were checked before use. The KK smears and POC-CCA tests were independently read by 2 laboratory staff and discordant results were confirmed by a third person. For each sample, the egg counts reported by the 2 readers were averaged, and this mean value was used as the final epg. Positive controls, negative controls, and blanks were included in every RT-PCR assay run.
Data Analysis
Data were entered and analyzed using Statistical Package for the Social Sciences software version 21. The proportion of S. mansoni infection was calculated using descriptive statistics. A Poisson regression model with a log link and cluster-robust standard errors was fitted to estimate prevalence ratios (PRs) and their 95% confidence intervals. Clustering at the school (for SACs) and village (for adults) levels was accounted for by using cluster-robust standard errors, ensuring valid inference in the presence of intra-cluster correlation. Because proportional allocation was used and the probability of selection was similar across clusters, sampling weights were not applied in the analysis. The model assessed associations between S. mansoni infection and selected explanatory variables. Bivariate Poisson regression was first employed to estimate crude PRs for each independent variable. We have assessed multicollinearity using Variance Inflation Factors (VIF), applying a conservative threshold of VIF > 5 to indicate problematic collinearity. To minimize overadjustment, we avoided simultaneous inclusion of variables likely to lie on the same causal pathway. Accordingly, the final multivariable model included only variables that were associated with infection in bivariable analysis (P < .20), were not collinear, were not considered mediators, and had theoretical justification as potential confounders rather than intermediates. 16 Statistical significance was declared at a P-value < .05.
Results
Characteristics of the Study Participants
A total of 634 SACs and 558 adults participated in the study. Attrition among adults was relatively high, with 46 participants unable to provide stool and/or urine samples, 14 households being unavailable after repeated visits, and 16 individuals refusing consent. The mean (± standard deviation) age of the SAC participants was 11.02 (±1.938) years, with ages ranging from 6 to 14 years. The majority (72.9%) of SAC participants were aged 10 to 14 years. Among adult participants, the mean age was 30.91 (±10.524) years, ranging from 18 to 72 years. Three hundred thirty-seven (60.4%) adults were aged 18 to 30 years. Nearly half (49.1%) of the total participants were male, and 677 (56.8%) were rural dwellers (Table 1).
Sociodemographic Characteristics of the Study Participants.

Schistosoma mansoni and Intestinal Helminth Infection by the Kato Katz Technique
A total of 339 (53.5%) SACs and 225 (40.3%) adults tested positive for at least 1 helminth species using KK smear microscopy, with S. mansoni accounting for the highest proportion (41.2% in SACs and 24.6% in adults). Coinfection of S. mansoni with any soil-transmitted helminth (STH) was found in 31 (4.9%) SACs and 30 (5.4%) adults, with the majority of these coinfections involving S. mansoni and hookworms (Table 2). Please note that co-infections are also included within single infections.
Prevalence of S. mansoni and Intestinal Helminth Infection According to the Kato Katz Method.

Prevalence of S. mansoni infection
A total of 452 SACs (71.3%; 95% CI: 67.4-74.6) and 330 adults (59.1%; 95% CI: 54.8-63.2), tested positive for S. mansoni infection at least by 1 of the 3 diagnostic methods. The prevalence by each diagnostic test is presented in Figure 3. The detection rate of KK was the lowest in both SACs and adults (Figure 3).

Prevalence of S. mansoni among SACs and adults by different diagnostic methods, 2023.
The prevalence of S. mansoni in the low-, moderate-, and high-transmission districts were 56.9%, 81.5%, and 95.5%, respectively among SACs. Among adults, the respective prevalence was 50.5%, 55.5% and 96.5% in low, moderate and high transmission settings. District-level analysis revealed that the highest prevalence of S. mansoni was in Tach Armachiho (96.0%), followed by Quara (82.6%) and Jawi (69.9%), with the lowest prevalence found in North Mecha (32.4%; Figure 4).
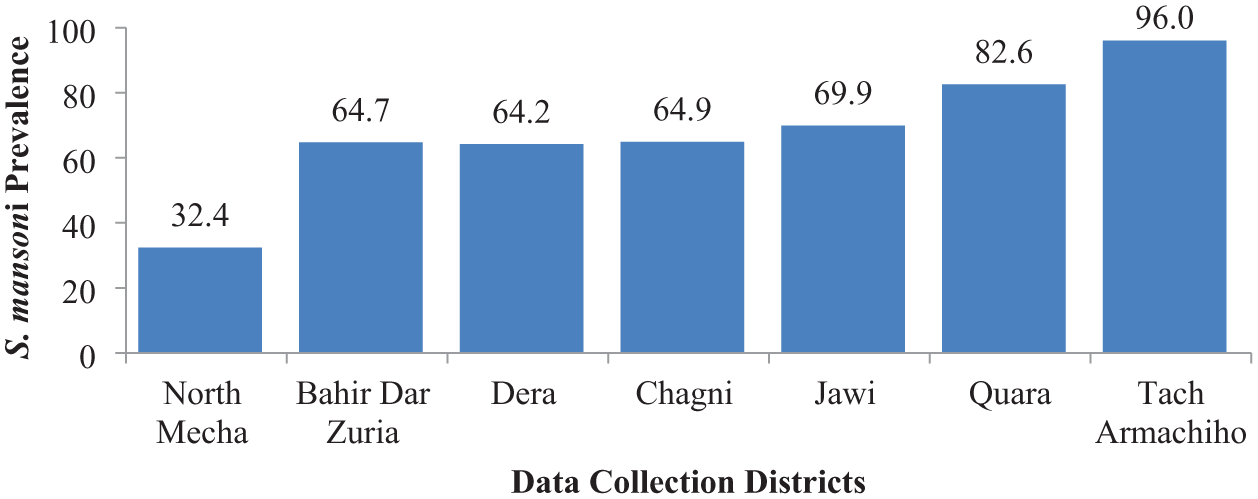
Prevalence of S. mansoni infection in each data collection district, 2023.
School and village-level analysis showed that the prevalence of S. mansoni varied significantly within the same district in low transmission settings (Table 3).
Prevalence of S. mansoni Among School-Aged Children and Adults by Combined Diagnostic Tests in the Respective Villages in the Amhara Regional State, Northwest Ethiopia, 2023 (N = 1192).

Intensity of S. mansoni infection
The arithmetic mean S. mansoni eggs per gram of stool (epg) was 148.0 (95% CI: 127.8-172.1) in SACs and 96.6 (95% CI: 81.6-113.0) in adults. The mean epg differed by study group, with a 51.34 difference between SACs and adults (Supplemental Information S1). Based on microscopy results, 170 (26.8%), 62 (9.8%), and 29 (4.6%) SACs had light (epg < 100), moderate (epg 100-399), and heavy (epg ⩾ 400) intensity infections, respectively. Among adults, 96 (17.2%), 38 (6.8%) and 3 (0.5%) were with light, moderate and heavy intensity infections, respectively. Infection intensity varied by district; of the 32 heavy infections, 21 and 6 were from Tach Armachiho and Quara, respectively.
Associations Between Clinical Signs and Symptoms and S. mansoni Infection
A total of 519 (43.5%) participants reported abdominal pain, while 106 (8.9%) experienced nausea within 2 weeks prior to data collection. Among the reported symptoms, the presence of bloody stool was significantly associated with S. mansoni infection (χ2 = 5.6, P = .018; Table 4).
Clinical Signs and Symptoms Among Study Participants and Their Associations With S. mansoni Infection.

Factors Associated With S. mansoni Infection
School-aged children who reported swimming or bathing in surface water had a significantly higher prevalence of S. mansoni infection compared with those who did not engage in these activities (PR = 2.679; 95% CI: 1.428-5.026, P = .002), indicating that the prevalence of infection was approximately 2.7 times higher among SACs who had a habit of swimming or bathing in surface water. Similarly, SACs who always participated in agricultural activities had a higher prevalence of infection than those who did not participat (PR = 1.266; 95% CI: 1.015-1.580, P = .037), corresponding to a 27% higher prevalence. In addition, SACs who were not aware of schistosomiasis had a significantly higher prevalence of S. mansoni infection compared with those who were aware of the disease (PR = 1.364; 95% CI: 1.017-1.829, P = .038), reflecting a 36% increase in prevalence among unawared children (Table 5).
Poisson Linear Regression of Factors Associated With S. mansoni Infection Among SACs in Northwest Ethiopia, 2023 (N = 634).

Among adults, crossing surface water barefoot was significantly associated with a higher prevalence of S. mansoni infection compared with those who did not cross surface water barefoot (PR = 1.481; 95% CI: 1.069-2.052, P = .018), indicating that the prevalence of infection was approximately 48% higher among adults who crossed surface water barefoot (Table 6).
Poisson Linear Regression of Factors Associated With S. mansoni Infection Among Adults in Northwest Ethiopia, 2023 (N = 558).

Discussion
Ethiopia has adopted the WHO’s goal of eliminating SCH, along with other NTDs, by 2030. 10 This goal is being pursued through MDA, improvements in WASH, vector control, and societal behavior change communications. 3 To achieve this, the country launched its first NTD control program between 2013 and 2015, 17 during which 10 million SACs were treated with praziquantel. The second National NTD Strategic Plan, implemented from 2016 to 2020, resulted in the treatment of 27 million SACs. Currently, Ethiopia is executing the third National NTD Strategic Plan (2021-2025), which aims to eliminate Schistosoma transmission by 2025. 3 Despite significant MDA coverage, the high prevalence of S. mansoni (65.5%) in this study highlights that the disease remains a major public health threat in the study area. However, the reported prevalence should be interpreted as a sensitive estimate of ongoing transmission, rather than a strictly parasitologically confirmed prevalence based solely on egg detection, as we have used a composite result.
The prevalence of S. mansoni infection among SACs based on the KK method was 41.2% (95% CI: 37.2-44.9), which is considered moderate. This rate is similar to a recent finding in Kenya, which reported a prevalence of 43.7%. 18 However, it was higher than earlier studies in different regions of Ethiopia, including Amhara,13,19 -26 Tigray, 27 Southern Nations, Nationalities and Peoples,28,29 Oromia, 30 and Harari. 31 The prevalence was also higher than those reported in other SSA countries.32 -38 On the other hand, these findings were lower than those from studies in other parts of Ethiopia, where higher prevalence rates were reported.39 -41 The observed variations in prevalence can be attributed to differences in the level of endemicity, the duration of MDA, diagnostic methods used, and the age groups included in the studies. For example, including adults in the Tigray study might have contributed to a lower prevalence. 27 In contrast, a study from southwest Ethiopia focused exclusively on a high-endemic area and reported a higher prevalence of 73.8%, 39 whereas our study included SACs from low-, moderate-, and high-endemic districts.
Based on the KK results among SACs, we observed a moderate level of S. mansoni prevalence in North Mecha (12.1%), Dera (24.3%), and Bahir Dar Zuria (31.6%) districts. These districts had previously been classified as low-endemic areas (<10% prevalence) by the Federal Ministry of Health (Supplemental Information S2). The discrepancy may be attributed to our purposive selection of schools located in potential risk areas, whereas the prior endemicity classification was based on data from randomly selected schools. As a result, the focal distribution of the parasite likely contributed to the variation in prevalence. A school-level analysis further supports this, revealing significant variation in S. mansoni prevalence between schools within the same district, particularly in low-endemic districts (Table 3). This finding underscores the importance of subdistrict-level mapping to accurately identify focal transmission areas for targeted interventions.
The higher prevalence of S. mansoni in SACs (71.3%) compared to adults (59.1%) by the combined diagnostic methods may be attributed to both behavioral and biological factors. School-aged children are more likely to come into contact with contaminated freshwater during their play, and they often have lower levels of awareness about hygiene and sanitation. Additionally, their immune systems are still developing, making them more susceptible to infection. 42 However, the fact that 59.1% of adult participants were positive for S. mansoni suggests that current SAC-targeted MDA and other control efforts should be expanded to include adults in endemic areas. Previous studies have similarly reported a significant prevalence of S. mansoni among adults in Ethiopia43 -45 and other SSA countries. 38
The significantly lower detection rate by the KK method compared to the POC-CCA test and RT-PCR highlights the need for urgent revisions to the current diagnostic approaches (Figure 3). The higher prevalence of S. mansoni detected by the POC-CCA test, compared to KK, aligns with findings from previous studies.38,46 -50 The POC-CCA test is more reliable because it detects CCA released by immature worms during the acute phase, even before eggs are excreted in the stool. Additionally, it is less affected by daily fluctuations in CCA excretion in urine.51,52 The POC-CCA test has been shown to be more sensitive than KK, with acceptable specificity.53,54 Therefore, as recommended by the WHO, 55 we suggest that the Federal Ministry of Health revise the current diagnostic methods to incorporate more sensitive and reliable tests.
The distribution and transmission intensity of different Schistosoma species in endemic areas are influenced by the presence of suitable water bodies, intermediate snail hosts, and various environmental, climatic, and physicochemical factors. 4 This results in a focal distribution of the disease, leading to significant variations in prevalence between districts and even between schools or villages located within short distances. Consistent with this, there was a threefold difference in S. mansoni prevalence between North Mecha (32.4%) and Tach Armachiho (96.0%; Figure 4). Similarly, significant differences in S. mansoni prevalence were observed between schools and villages in North Mecha, Dera, and Bahir Dar Zuria districts (Table 3).
The greater mean egg count observed in SACs compared to adults (P = .001) can be attributed to higher exposure to contaminated water, underdeveloped immunity, and a lack of resistance to repeated infections among SACs. 42 This was further supported by the higher prevalence of heavy infections in SACs (4.6%) compared to adults (0.5%). The 2.7% overall prevalence of heavy infection detected by KK microscopy in this study exceeds the national target of reducing heavy infections to <1% by 2025, as set by quantitative PCR. 3 The RT-PCR results also indicate that, of the 670 PCR-positive participants, 375 (56.0%) had moderate or heavy infection (CT ⩽ 30). Notably, heavy infections were disproportionately high in the districts of Quara (6.5%) and Tach Armachiho (12.1%), while the prevalence in other districts ranged from 0% to 1.6%. Similar findings of high heavy infection rates have been reported in previous studies from the Amhara region20,41 and southern Ethiopia.28,39,40 These findings can aid policymakers in designing more targeted MDA modalities based on both infection prevalence and intensity.
Abdominal pain was the most commonly reported symptom, mentioned by 43.5% of the respondents, which aligns with findings from previous study in Ethiopia. 43 Bloody stool within 2 weeks before data collection was significantly associated with S. mansoni infection, a result consistent with a previous study in Ethiopia. 43 Other signs and symptoms reported in this study were not found to be significantly associated with S. mansoni infection. This may be due to the nonspecific nature of these symptoms and the potential coexistence of other intestinal helminths, which exhibit similar clinical manifestations.
In this study, SACs who regularly swam or bathed in freshwater bodies were more likely to be infected with S. mansoni than those who did not. Similar findings have been reported in previous studies conducted in Ethiopia.21 -23,30 However, the habit of swimming or bathing in freshwater was not significantly associated with an increased risk of infection among adults due to multiple multiplicity of contributing factors that were not captured in the analysis.
In general, the continued transmission of S. mansoni in the study area highlights the need to redirect current control and prevention strategies beyond MDA. However, implementing comprehensive solutions, such as providing adequate WASH facilities and large-scale snail control programs, remains challenging due to the country’s limited economic capacity. 3 Consequently, it is essential to introduce context-specific infection prevention strategies. These include boiling, chlorinating, or storing freshwater for at least 24 hours before domestic use,56 -59 as well as vigorously drying the body with a towel immediately after unavoidable contact with freshwater.59,60 Moreover, continuous health education and promotion are crucial, as they empower communities to make informed decisions about their participation in control and prevention activities.3,61 However, data from Ethiopia show that knowledge, attitudes, and practices regarding SCH remain insufficiently developed. 62 Therefore, the implementation of societal behavioral change communication must be reinforced. Additionally, as shown in Table 2, STHs are also prevalent in the study area. Thus, adopting holistic approaches, including integrated MDA and WASH initiatives, will be key to preventing and controlling both S. mansoni and coendemic STHs.
In this study, we assessed the status of S. mansoni infection across different endemic settings in both SACs and adults using a combination of diagnostic techniques. We believe this approach provides a more accurate representation of S. mansoni prevalence in the study area. However, there are some limitations. Use of composite results might have inflated the prevalence as the tests used are not 100% specific. The schools and villages were purposively selected, which may limit the ability to generalize the findings to the broader population. Additionally, factors such as “previous history of participation in MDA” and “distance from home to freshwater bodies” were not analyzed due to recall bias, as the majority of participants responded with “I don’t know.”
Conclusions
Schistosoma mansoni infection remains a substantial public health challenge in the Amhara Regional State, with high prevalence and notable infection intensity across diverse transmission settings. The combined use of KK, POC-CCA, and RT-PCR revealed a far greater true burden than microscopy alone, highlighting the importance of sensitive diagnostic approaches in endemic areas. Behavioral factors—particularly swimming or bathing in surface water, participation in agriculture activities and lack of awareness about the diseases—were strongly associated with infection underscoring ongoing exposure to contaminated water sources. The wide district-level variation in prevalence (32.4%-96.0%) and heavy infection levels reaching 12.1% in some districts indicate that Ethiopia is currently not on track to achieve the WHO 2030 SCH elimination targets. To address persistent transmission, control efforts should expand to include adults and be reinforced through an integrated approach combining regular MDA with vector control, improved WASH services, and sustained health education.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302261437528 – Supplemental material for Prevalence of Schistosoma mansoni Infection and Associated Factors After a Decade of Mass Drug Administration in the Amhara Regional State, Northwest Ethiopia: A Cross-Sectional Study
Supplemental material, sj-docx-1-ehi-10.1177_11786302261437528 for Prevalence of Schistosoma mansoni Infection and Associated Factors After a Decade of Mass Drug Administration in the Amhara Regional State, Northwest Ethiopia: A Cross-Sectional Study by Getaneh Alemu, Endalkachew Nibret, Abaineh Munshea, Melaku Anegagrie, Tadesse Hailu, Animen Ayehu, Tazeb Molla, Girma Zerefaw and Arancha Amor in Environmental Health Insights
Supplemental Material
sj-docx-2-ehi-10.1177_11786302261437528 – Supplemental material for Prevalence of Schistosoma mansoni Infection and Associated Factors After a Decade of Mass Drug Administration in the Amhara Regional State, Northwest Ethiopia: A Cross-Sectional Study
Supplemental material, sj-docx-2-ehi-10.1177_11786302261437528 for Prevalence of Schistosoma mansoni Infection and Associated Factors After a Decade of Mass Drug Administration in the Amhara Regional State, Northwest Ethiopia: A Cross-Sectional Study by Getaneh Alemu, Endalkachew Nibret, Abaineh Munshea, Melaku Anegagrie, Tadesse Hailu, Animen Ayehu, Tazeb Molla, Girma Zerefaw and Arancha Amor in Environmental Health Insights
Footnotes
Acknowledgements
We thank the Mundo Sano Foundation and the Institute of Health for providing the POC-CCA and RT-PCR test kits; Bahir Dar University Science College for granting ethical approval for the study; the Amhara Regional Health Bureau for their support and provision of support letters; the Amhara Public Health Institute for permitting access to the molecular laboratory; and the study participants for their voluntary participation.
List of Abbreviations
CCA – Circulating Cathodic Antigen, DNA – Deoxyribonucleic Acid, epg – Eggs per Gram of Stool, KK – Kato Katz, MDA – Mass Drug Administration, NTD – Neglected Tropical Diseases, POC-CCA – Point of Care Circulating Cathodic Antigen, PRs – Prevalence Ratios, RT-PCR – Real-time Polymerase Chain Reaction, SAC – School-Aged Children, SCH – Schistosomiasis, STHs – Soil Transmitted Helminths, SSA – Sub-Saharan Africa, SD – Standard Deviation, WASH – Water, Sanitation, and Hygiene, WHO – World Health Organization.
Ethical Considerations
Ethical approval was granted by the College of Science Institutional Research Ethics Review Committee, Bahir Dar University (ref: prcsvd/514/2015). Permission was obtained from the Amhara Public Health Institute, and support letters were secured from zonal, district, school, and village authorities.
Consent to Participate
Informed written consent and assent were obtained from parents/caregivers and children, respectively. All participants who tested positive for S. mansoni were treated with praziquantel (40 mg/kg). Those with other intestinal parasitic infections were referred to nearby health facilities for treatment.
Author Contributions
GA conceived and designed the study, participated in data collection, data analysis, and wrote the manuscript. AA, EN, AM, MA, AA, and TH participated in designing the study, data analysis, data interpretation, in editing and reviewing the manuscript. GZ and TM participated in molecular laboratory analysis. All the authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The POC-CCA and PCR reagents and consumables were provided by the Mundo Sano Research Foundation and the Institute of Health, Madrid, Spain, while Bahir Dar University funded the field data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original data for this study is available from the corresponding author.*
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

