Abstract
Beef is considered favorable environments for the growth of bacteria. This study aimed to assess the bacteriological quality of beef and contact surfaces, and to estimate the prevalence of Salmonella in abattoirs and butcher shops in eastern Ethiopia. A cross-sectional study was conducted between June 2024 and January 2025 on 128 beef and 196 contact surface samples. Raw beef (250 g) was purchased from each abattoir and butchery. Beef contact swab samples (100 cm²/contact surfaces) were collected via swab collection kits. Aerobic bacterial, Enterobacteriaceae, and coliform counts as well as Salmonella detection, were analyzed according to ISO standard techniques. In Haramaya town abattoir, the total mean aerobic bacterial count in the meat samples was 7.84 ± 0.60 log CFU/g. A significant proportion of the beef samples examined in Jigjiga, were classified as unacceptable. In Harar abattoir, the mean coliform load of knife swab samples classified as unacceptable (6.10 ± 0.38 log CFU/cm²) was significantly greater than that of surface swab samples. In Dire Dawa city, the mean aerobic bacterial load on cutting board swab samples from butcher shops (9.39 ± 0.91 log CFU/cm²) was significantly higher than that on hand swab samples Among the 324 beef and contact surface samples, 57 were Salmonella positive, with an overall prevalence of 17.6%. Salmonella prevalence was significantly higher (P = .034) in abattoir samples (22.7%) than in butcher samples (13.7%), suggesting that the contamination may mainly originate from the meat source or during flaying, evisceration, and cutting. In conclusion, significant bacterial loads, on beef and contact surface samples, along with the significant prevalence of Salmonella, pose a substantial risk of spoilage and foodborne illness to consumers. We recommend practical interventions such as hygiene regulations and training on sanitation practices to reduce the bacterial load on beef and contact surfaces and safeguard public health along with future detailed research focus on characterizing Salmonella.
Plain Language Summary
Meat can easily grow germs, including Salmonella, because it has lots of nutrients that germs love. This study looked at how clean meat is and if Salmonella is found in meat shops and slaughterhouses in eastern Ethiopia. We tested 324 samples of beef meat and surfaces that touch meat between June 2024 and January 2025. We used standard methods to count the number of germs and to check for Salmonella. What we found that Haramaya (slaughterhouse): Beef meat had a high number of germs that grow with air (7.84 log CFU/g), Enterobacteriaceae (7.58 log CFU/g), and coliform count (6.66 log CFU/g). In Dire Dawa town meat shops, meat was measured to have a high number of Enterobacteriaceae colonies (8.56 log CFU/g). Also, in Jigjiga town meat shops and slaughter houses all the meat samples were not clean enough based on certain standards. The number of germs on cutting boards (9.39 log CFU/g) in Dire Dawa (meat shops was much higher than on workers’ hands (4.90 log CFU/g). We found Salmonella in 18 out of every 100 samples we tested (17.6%). Salmonella was more common in slaughterhouse samples (22.7%) than in meat shop samples (13.7%) with significant difference. Having lots of bacteria, including Salmonella, on beef and surfaces means people could get sick from eating the contaminated beef. We recommend interventions such as hygiene regulations and training on sanitation practices to reduce the germs on beef and contact surfaces and keep public health and more research to figure out what types of Salmonella are in the beef and on the surfaces.
Introduction
Foodborne diseases are associated with the consumption of raw beef and its products. It becomes the most significant food safety threat worldwide. 1 Among the total world burden of food-borne diseases, 70% is from biological hazards. 2 Among several bacterial pathogens that cause food intoxication and spoilage, the genera Salmonella, Campylobacter, Staphylococcus, Escherichia coli, Bacillus, Clostridium, Listeria, Acinetobacter, and Pseudomonas are the most common.3 -5 In general, food-borne pathogens can cause mild gastroenteritis to life-threatening diseases, which are specifically characterized by abdominal discomfort, nausea, vomiting, and various levels of diarrhea. 6 There is high severity, morbidity and mortality of food-borne infections in a targeted population, such as immunocompromised, elderly, and young individuals.
Atlabachew and Mamo 7 evaluated the microbiological quality of raw meat and meat contact surface swab samples that were obtained from a butchery in Debre Berhan town of the Amhara regional state of Ethiopia. In this recent study, the mean bacterial counts for Enterobacteriaceae, total aerobic bacteria, and coliform bacteria in beef samples from butcher shops range from 4.84 to 5.47 log CFU/g. The authors reported mean bacterial loads ranging from 3.96 to 4.17 log CFU/cm2 for total coliforms, Enterobacteriaceae, and total aerobic bacteria, based on swabs taken from contact surfaces. The prevalence rates of Salmonella in beef meat and swab samples in this recent study were 2.50% and 4.16%, respectively, with a pooled prevalence of 7.5%.
In another work by Azage and Kibret, 8 mean counts of aerobic bacteria and total coliform bacteria of 4.53 and 3.97 log10 CFU/gm, respectively, were reported in samples collected from retail butcher shops in Bahir Dar, Ethiopia. The authors reported that 70% of Salmonella isolates were from meat and contact surface swabs. The high risk of contamination of beef by food-borne bacterial pathogens such as Salmonella and the risk of contracting food-borne diseases due to the consumption of contaminated beef and its products are very high. Since the contact surfaces of beef, its products, and beef are highly suitable and favor the growth of microorganisms, 9 the existence of these foodborne pathogens is inevitable in the beef value chain.
Salmonella species are among the most common and leading causative agents of bacterial food-borne diseases. 10 This is because salmonellae are ubiquitous and widespread and can survive several months and years in water, on vegetation, and in soil, allowing them to be easily transmitted to susceptible hosts. 11 Various outbreaks of salmonellosis have been reported in many countries among the food-borne pathogens that contaminate meat, such as in the USA, 12 the Netherlands, 13 Turkey, 14 sub-Saharan Africa, 15 and South Africa. 16 A recent meta-analysis of research findings from South Africa revealed a pooled Salmonella prevalence estimate of 32.3% from samples examined, ranging between 41.9% and 95.9%. 17 In Ethiopia, the pooled Salmonella prevalence was estimated to be 8.34% in contaminated minced beef, 11.86% in mutton and 13.53% in chicken meat collected from markets. 18 A meta-analysis reported that the prevalence of Salmonella in Ethiopia was also estimated to be 5.7% in adults, 8.7% in children with diarrhea. 19 and 9.9% in meat samples. 20 Another limited number of studies conducted in Ethiopia reported the detection and presence of Salmonella in meat, its products, contact surfaces and enabling environments in various proportions.21 -23
There is a high risk of bacterial contamination of beef because the slaughtering, flaying, handling, transporting, and processing of beef and its products are performed in poor hygienic situations. The organisms that are able to contaminate meat mainly emerge from animal hides, gut ingesta, feces, working equipment, personnel, and the environment.24,25 Notably, in Ethiopia, there is a widespread and extended trend and practice of consuming raw beef and its products 18 throughout all regions of the country. Consumers obtain beef from retail butcher shops located mainly in towns and main cities of the country. In turn, abattoirs provide beef to retail butcheries, which is a long process. The consumption of raw beef products or minced raw beef, which is named Kitfo, in Ethiopia is a very common trend. This raw beef and its byproduct eating habits of a significant number of Ethiopians in general and the community in eastern Ethiopia in particular increase the susceptibility of consumers to such types of meat-related bacteria. Bacteriological quality analysis of raw beef is necessary due to the poor handling and hygienic practices observed at abattoirs and butcheries in eastern Ethiopia, which can contribute to the risk of contracting foodborne diseases. The information used to assess the bacteriological quality of beef in the study area is very limited. Thus, the aim of this study was to assess the bacteriological quality of raw beef products and contact surfaces and estimate the prevalence of Salmonella in abattoirs and butcher shops in eastern Ethiopia.
Materials and Methods
Study Area and Sample Collection Sites
The study was carried out in 4 major towns within 3 regional states and 1 administrative city in the eastern part of Ethiopia. For this study, Haramaya town (Oromia Regional State), Jigjiga town (Somali Regional State), Harar (Harari Regional State), and Dire Dawa (Dire Dawa City Administration) were selected (Figure 1). These towns have road access and great production potential for beef farms with abattoirs and butcher shops that provide meat to consumers. The towns were selected purposely on the basis of the high presence and accessibility of abattoirs and butchery shops.
Map of the study areas.
Determination of Sample Size
Chechran’s formula was used to calculate the sample size of the unknown population size, as shown below.
n = sample size
Z = the value on the Z table at the 95% confidence level = 1.96
E = sampling error at 5%
P = maximum variabilities (prevalences) of the population at 8.34% (0.0834) and 12.7% (0.127)
q = 1−p
The sample size calculation for raw beef was based on an expected pooled prevalence estimate of 8.34% Salmonella beef contamination from abattoirs made in different areas of Ethiopia. 18 Therefore, the minimum calculated sample size was 117, which was the 8.34% anticipated prevalence. This was increased by 10%, representing further 11 samples, to increase the statistical power and precision of the sample size estimate, resulting in a total sample size estimation of 128 The beef contact surface sample size was calculated on the basis of a 4.17% previous Salmonella estimate obtained from meat contact surface samples in Addis Ababa butcher shops, Ethiopia. 26 Accordingly, the required sample size is 61. This sample size was increased from 61 to 196 which may enhance the precision of the estimate and reliability of the results, with an overall grand sum of 324 beef and contact surface samples (Table 1).
Summary of Sample Sources and Calculated Sample Sizes.

Study Design and Sample Collection
A cross-sectional study was conducted between June 2024 and January 2025 to evaluate the bacteriological quality of raw beef products and contact surfaces and estimate the prevalence of Salmonella from raw beef and minced beef in abattoirs and butcher shops in eastern Ethiopia. All the required information about all the abattoirs and butchery shops in the towns was obtained from the town’s municipality administration at the study sites. All the abattoirs and butchery houses in the selected towns were visited for sampling. A list of all the sampling sites was prepared using information from the town’s municipality office, and consent to access each site was obtained from the respective owners. Raw beef (250 g) was purchased from each abattoir and butchery within each town using sterile plastic bag (Seward Ltd., UK). The neck, abdomen (flank), thorax (lateral), breast (lateral), and ramp of beef which are sites with the highest rate of contact and contamination was chosen for beef sampling. The swabs (100 cm² or per contact surfaces) of beef contact surfaces (cutting equipment, cutting tables, hand swabs, and from beef processing surfaces) were swabbed and collected aseptically via swab collection kits (Lab M Ltd., Quest Park, UK). A sterile cotton tipped swab fitted with shaft, was first soaked in a 10 mL of Buffered Peptone Water (BPW; Oxoid, Basingstoke, UK) rubbed horizontally, vertically, and then diagonally several times with rotating it to expose all surfaces. Then, the swab was placed into the test tube. All samples were collected aseptically using disposable gloves and were labeled with necessary information including the study site, date of sampling, sample code, source, and sample type. The samples were stored in ice boxes and transported to the College of Veterinary Medicine Research and Molecular Biology Laboratory of Haramaya University in eastern Ethiopia for further analysis within 3 hours of collection.
Determination of the Bacterial Quality of the Beef and Beef Contact Surface Samples
Aerobic Bacterial Counts
The aerobic bacterial count for each sample was analyzed according to the protocol described by the ISO 4833-2 27 standard (horizontal method for the enumeration of microorganisms-Part 2: colony count at 30°C by the surface plating technique). For all beef samples, a 1:10 sample dilution approach was applied. Specifically, 25 g of each beef sample was chopped into small pieces and mixed with 225 mL of sterile buffered peptone water (Oxoid, Basingstoke, UK) in a sterile stomacher bag (Seward Ltd., UK). Then, it was homogenized for 2 minutes at high speed (200-250 rpm) in a stomacher (Nickel Electro LMD, Weston-S-Mare, Somerset, UK). Then, 0.1 mL of the serial dilutions (10−1 to 10−7) of each sample was plated onto pri-dried Plate Count Agar (Oxoid, Basingstoke, UK). The inoculum was spread evenly over the entire surface of the agar using a sterile, bent plastic stick. Plates containing 30 to 300 colonies were counted via a colony counter (UK Sterilin LTD, Staffordshire, UK) after incubation for 24 hours at 30°C to obtain an aerobic bacterial count as a colony forming unit (CFU). The plating procedure was done in duplicate.
Swab samples were obtained from beef contact surfaces using sterile swabs placed in universal bottles or test tubes containing 10 mL of BPW (Oxoid, Basingstoke, UK). For further processing, each swab and its associated BPW were transferred into a sterile plastic stomacher bag with wire closures or into sterile bottles. A 1:10 dilution was then prepared by adding 90 mL of BPW to the sample. The samples were homogenized for 2 minutes using stomacher bag. Following homogenization, 20 mL of the mixture were transferred into a universal bottle which corresponded to a 10−1 dilution and established a lower detection limit of 100 CFU per swab when 1 mL of the mixture was plated. Serial dilutions were prepared as necessary for enumeration. Finally, colony counts were obtained from plates containing colonies between 15 and 150 (or up to 300 colonies), and results were calculated duly.
Enterobacteriaceae Count
For Enterobacteriaceae count, the ISO 21528-2 28 (horizontal methods for the detection and enumeration of Enterobacteriaceae Part 2) method was used. From each separate sample processed as indicated above, like for aerobic bacterial count, 0.1 mL of appropriate dilution for each sample was aseptically spread on pre-dried Violet Red Bile Glucose/Dextrose Agar (Oxoid, Basingstoke, UK) for the enumeration of Enterobacteriaceae.
Coliform Count
ISO 9308-1 29 specifies a method for the enumeration of coliform bacteria. Samples were processed as indicated for aerobic bacterial counts. Then, 1 mL of each appropriate dilution was transferred into sterile plates. Approximately 15 to 20 mL of melted Violet Red Bile Agar at 45°C was poured into the plates. The plates were swirled gently to mix, allowed to solidify, and incubated at 37°C for 24 hours. The total number of colonies (dark red to pink colonies) on the plates (15-150 per plate) was counted. 30 The number of colonies per mL of sample was calculated via the formula CFU/mL of sample = colony count × dilution factor × 10. 31
Enumeration and Detection of Salmonella Species
The horizontal method for the detection of Salmonella species (ISO 6579-1 32 ) was used for detection and enumeration.
Enumeration
The enumeration of Salmonella in beef samples was performed via horizontal enumeration of microorganisms via a plate count technique as described in ISO/TS 6579-2 32 titled “Microbiology of Food and Animal Feeding Stuffs - Horizontal Method for the Detection and enumeration of Salmonella,” Part 2: Enumeration method. Sample preparation was performed according to the procedure described in ISO/TS 6579-2. 32 Samples were processed as indicated in the preceding sections. One milliliter of the suspension was transferred into sterile plates using a sterile pipette for each decimal dilution ranging from 10−1 to 10−10. In between 12 and 15 mL of Xylose Lysine Deoxycholate selective agar (XLD; Oxoid, Basingstoke, UK) was then poured at approximately 44°C to 47°C into each plate within 15 minutes. The inoculum was mixed carefully by rotating the plates and allowing the mixture to solidify on a cool horizontal surface.
The plates were inverted, and they were placed in an incubator adjusted at 30°C for 72 hours. Those plates containing between 30 and 300 colonies were counted and recorded. The results were recorded, and the bacterial load in the original 25 g sample was calculated. The surface swabs of the beef contact samples were diluted 1:10 by adding 90 mL of buffer peptone water. The samples were homogenized for 1 to 3 minutes in a stomacher according to the manufacturer’s instructions. Twenty milliliter (20 mL) of the homogenate was transferred into a universal bottle, which is equivalent to a 10−1 dilution and gives a lower limit of detection of 100 CFU per swab if 1 mL is plated. Further dilutions were performed as needed.
Detection
Salmonella in each sample was detected on the basis of the international standard organization (ISO 6579-1) culture method 32 (horizontal method for the detection of Salmonella spp.). Accordingly, 25 g beef samples were subjected to primary pre-enrichment with 225 mL of BPW (Oxoid, Basingstoke, UK) and incubated for 24 hours at 37°C. This was followed by secondary selective enrichment, in which 0.1 mL of the previously enriched culture was then added to 10 mL of Rappaport–Vassiliadis broth (Oxoid, Basingstoke, UK) and simultaneously added to 1 mL of sample and 10 mL of Mueller Kauffman Tetrathionate Broth (Oxoid, Basingstoke, UK). The mixture was subsequently incubated for 24 hours at 41.5°C (±1°C) and 37°C. Finally, the enriched samples were plated onto 2 Salmonella selective agars. A loopful of culture sample was subsequently streaked onto XLD (Oxoid, Basingstoke, UK; Merck KGaA, Darmstadt, Germany) and BGA (Brilliant Green Agar; Oxoid, Basingstoke, UK) after vortex mixing and incubated for 24 hours at 37°C. After incubation, the plates were examined for the presence of Salmonella colonies that presumably met the description of Salmonella. The presence of red colonies with or without black centers on XLD and pink colonies on BGA plates were considered indicative of suspected Salmonella colonies. Three to five Salmonella colonies were picked carefully and streaked onto nutrient agar (Oxoid, Basingstoke, UK), followed by incubation for 18 to 24 hours at 37°C for further biochemical tests. The following confirmatory tests were performed via biochemical tests according to ISO 6579-1. 33 These tests included Triple Sugar Iron Agar, the Lysine Deoxycholate test, the Urea Agar test, and the IMViC test.
Data Quality Control
During the study, all chemicals, reagents, and media were subjected to a quality control checkup, referring to the standard operation procedure to maintain quality assurance, validate the use of all the reagents and methods, and to verify the reliability and accuracy of the findings. Negative controls and Salmonella reference strain (ATCC 14028) was used as a positive control.
Data Analysis
All data obtained from the laboratory tests were entered into a Microsoft Excel spreadsheet (Microsoft Corporation). After the data were transferred, statistical analysis was performed via IBM SPSS Statistics 25 and STATA 17 software versions. The relationships between predictor variables (raw beef, minced beef, areas; Haramaya, Harar, Jigjiga, and Dire Dawa) and outcome variables (bacterial contamination levels or assessment load and load or colony count of Salmonella) were analyzed via ANOVA and independent samples t-tests. Predictor (independent) variables are those factors that might influence contamination levels whereas the outcome variables refer the results which are measuring to observe the effect of the predictor variables. We used statistical methods such as Analysis of Variance (ANOVA) and independent samples t-tests to analyze the relationship between such predictor and outcome variables which help determine whether differences in contamination levels are statistically significant among beef sample types and geographic locations (the sites where the beef and contact surface samples collected). Bacterial colony counts of Enterobacteriaceae, aerobic bacterial counts, coliform counts, and Salmonella colony counts were converted into logarithmic scales (log10 CFU/g/cm2 of beef and swab samples) for statistical analysis. For bacterial load/count determination, beef contact surfaces with aerobic bacterial counts were considered satisfactory if the colony count was <4 log CFU/g of sample in terms of hygienic conditions, whereas those with counts above 4 log CFU/g of sample were considered unsatisfactory.34,35 This is mainly for susceptible groups including, the young, elderly, and immunocompromised individuals. In terms of Enterobacteriaceae and target pathogen (Salmonella) colony counts, the absence of pathogens per 25 g of sample and beef contact surfaces per 100 cm2 of surface material was considered satisfactory. The presence of pathogens, including Salmonella, per 25 g of sample and contact surfaces per 100 cm2 of surface material was considered unsatisfactory.34,35 Descriptive statistics were used to summarize the data and determine the frequency of the microbial count, mean, and standard deviation of the samples. One-way ANOVA and independent sample t tests were used to compare bacterial counts across the towns, sources, and types of samples.
Results
Total Mean Bacterial Counts of Beef Meat Samples From Abattoirs and Butcher Shops in Towns
The total mean aerobic bacterial count was 7.84 ± 0.60 and 6.10 ± 3.42 log CFU/g in the meat samples collected from abattoirs and butcher shops in Haramaya town, respectively. The mean Enterobacteriaceae counts in the raw beef were 7.58 ± 1.05 and 5.46 ± 3.23 log CFU/g in samples from abattoir and butcher shops, respectively. A mean coliform count of 6.66 ± 1.72 and 4.82 ± 3.12 log CFU/g was found in the beef samples from the abattoir and butcher shops, respectively, within the town. In this town, the mean Salmonella count in the beef samples was 2.60 ± 0.86 log CFU/g at the abattoir and 3.23 ± 0.44 log CFU/g at the butcher shops (Table 2). In Harar town abattoirs and butcher shops, the total mean aerobic bacterial counts were 6.17 ± 2.73 and 6.48 ± 2.25 log CFU/g, respectively. Enterobacteriaceae counts were determined at levels ranging from 0.00 to 7.95 log CFU/g in beef samples, with a mean count of 5.87 ± 2.59 log CFU/g in the abattoir samples (Table 2).
Total Mean Bacterial Count From Meat Samples Collected at Abattoirs and Butcher Shops in the Districts.
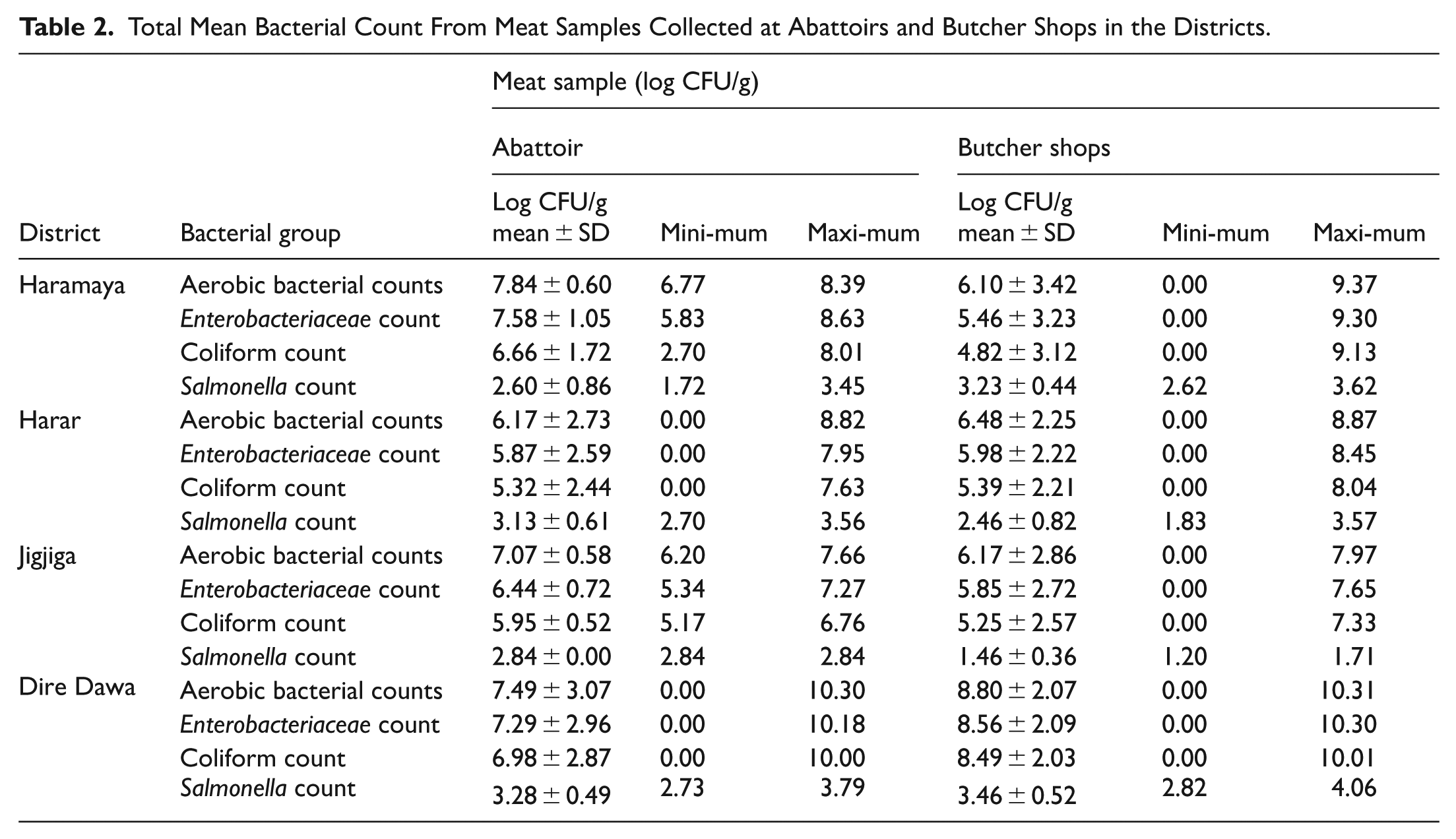
In Jigjiga town, the mean total number of aerobic bacterial colonies was 7.07 ± 0.58 log CFU/g in the samples from the abattoir and 6.17 ± 2.86 log CFU/g from those in butcher shops, respectively. A total mean Enterobacteriaceae count of 6.44 ± 0.72 log CFU/g and 5.85 ± 2.72 log CFU/g was determined from beef samples that were collected from abattoirs and butcher shops, respectively. In Dire Dawa city, a mean aerobic bacterial count of 7.49 ± 3.07 log CFU/g in the abattoir and 8.80 ± 2.07 log CFU/g in the butcher shops was obtained from the beef samples. In addition, the mean Enterobacteriaceae counts were 7.29 ± 2.96 and 8.56 ± 2.09 log CFU/g in the abattoir and butcher shop meat samples, respectively. The average Salmonella count in the beef samples was 3.28 ± 0.49 log CFU/g at the abattoir and 3.46 ± 0.52 log CFU/g at the butcher shops (Table 2).
Total Bacterial Counts of Beef Meat Contact Surface Samples From the Abattoirs and Butcher Shops
The mean aerobic bacterial load in the surface samples from the abattoir in Haramaya town was 6.23 ± 3.06 log CFU/cm2. The mean aerobic bacterial count on meat contact surface samples collected from butcher shops in this town was 6.19 ± 2.67 log CFU/cm². In this town, the average Salmonella load on meat contact surface samples across all butcher shops surveyed was 3.81 ± 0.00 log CFU/cm2 (Table 3). In Harar town, the mean aerobic bacterial counts in beef contact surface samples from abattoirs and butcher shops were determined to be 6.12 ± 1.31 and 5.59 ± 2.51 log CFU/cm2, respectively (Table 3). The Enterobacteriaceae loads in the contact surface samples obtained from the abattoir in this town ranged from 0.00 to 7.36 log CFU/cm2 (Table 3). In Jigjiga town, the mean aerobic bacterial count on beef contact surface samples from the abattoir was 5.85 ± 1.69 log CFU/cm2. However, it was 5.55 ± 2.23 log CFU/cm2 in the butcher shops. In Dire Dawa city, a 7.02 ± 2.51 log CFU/cm2 mean aerobic bacterial count in beef contact surface samples was determined, with a mean aerobic bacterial count of 6.52 ± 3.42 log CFU/cm2 in contact surface samples collected from the butcher shops (Table 3).
Total Mean Bacterial Count From Meat Contact Surface Samples Collected at the Abattoirs and Butcher Shops in the Districts.

Bacterial Load Proportions of Raw Beef Samples Collected From the Abattoirs in the Study Towns With Their Range of Bacterial Counts in Log CFU/g.

Aerobic bacterial counts: <1 (BDL), < 104 (good), 104 – < 106 (acceptable), and ⩾ 106 (unacceptable and potentially dangerous); Enterobacteriaceae and coliform counts: < 102 (good), 102 – 104 (acceptable), and ⩾104 (unacceptable and potentially dangerous).
Proportion or Range of Bacterial Load in Beef Samples From Different Categories
Bacterial Load Categories of Beef Samples Collected From Abattoirs
Among the 9 raw beef samples collected from the Haramaya town abattoir, 88.9% (8 out of 9) had aerobic bacterial counts of >7 log CFU/g, and 11.1% (1 out of 9) had aerobic bacterial counts between 6 and <7 log CFU/g, which were classified as unacceptable. For the Enterobacteriaceae count of the 9 raw beef abattoir samples examined, 22.2% (2/9) had bacterial load counts ranging from 4 to <6 log CFU/g, and 77.8% (7 out of 9) had bacterial loads ⩾7 log CFU/g, which were classified as unacceptable. In terms of the coliform counts, 1 out of 9 (11.1%) had bacterial loads between 1 and <4 log CFU/g, and another 1 out of 9 (11.1%) had bacterial counts between 4 and <7 log CFU/g, and 7 from 9 (77.8%) had bacterial loads >7 log CFU/g. Salmonella was counted in 100% of the Salmonella-positive meat samples that were within the unacceptable range (1 to <4 log CFU/g; Table 4).
In Harar town, of the 14 raw beef samples analyzed, 14.3% (2 out of 14) were classified as acceptable, with aerobic bacterial counts of <10 CFU/g; 7.1% (1 out of 14) were classified as satisfactory (4 to <6 log CFU/g), and 78.6% (11 out of 14) were grouped as unacceptable (6 to <7 and >7 log CFU/g). One out of 14 (14.1%) of the beef samples were classified as acceptable (<10 CFU/g) for both Enterobacteriaceae and coliform counts, and 78.6% (11 out of 14) for Enterobacteriaceae and 50% (7 out of 14) for coliform counts were classified as unacceptable. Salmonella was enumerated in a range between 1 and <4 log CFU/g of Salmonella-positive samples of 2 out of 2 (100%) that were classified as unacceptable (Table 4).
In Jigjiga town, all 9 raw beef samples examined 100% were classified as unacceptable on the basis of aerobic bacterial counts, with bacterial loads ranging from 6 to >7 log CFU/g. Moreover, 100% (9 out of 9) were classified as unacceptable on the basis of Enterobacteriaceae and coliform counts that ranged from 4 to >7 log CFU/g (Table 4). In Dire Dawa city, of the 16 raw meat samples evaluated, 2/16 (12.5%) were grouped as acceptable on the basis of aerobic bacterial and Enterobacteriaceae counts, with bacterial loads below detectable levels (<10 CFU/g), and 14/16 (87.5%) were unacceptable (>7 log CFU/g). One hundred percent (4/4) of the Salmonella-positive meat samples analyzed were classified as unacceptable, with a range of 1 to <4 log CFU/g (Table 4).
Bacterial Load Categories of Beef Samples Collected From Butcher Shops
In Haramaya town butcher shops, 22.2% of the 18 raw beef samples were classified as acceptable based on aerobic bacterial counts, and 77.8% (14 out of 18) were unacceptable (>6 log CFU/g). Based on Enterobacteriaceae counts, 27.8% (5 out of 18) of the beef samples were classified as acceptable, with bacterial load counts <4 log CFU/g, and 72.2% (13 out of 18) were classified as unacceptable, with counts ⩾4 log CFU/g. For the Salmonella count, 100% (4 out of 4) of the beef samples analyzed were classified as unacceptable ranging between 1 and <4 log CFU/g. In Harar, of the 23 raw beef samples explored, 8.7% (2 out of 23) were classified as acceptable on the basis of aerobic bacterial counts (0.00 log CFU/g). Similarly, 17.4% (4 out of 23) were classified as satisfactory with counts between 4 and <6 log CFU/g, and 73.9% (17 out of 23) were unacceptable (⩾6 log CFU/g). Based on Salmonella enumeration, 100% (4 out of 4) of the raw beef samples surveyed were classified as unacceptable with counts between 1 and <4 log CFU/g (Table 5). In Jigjiga, of the 13 raw beef butcher shop samples surveyed, 15.4% (2 out of 13) were classified as acceptable based on aerobic bacterial counts (0.00 log CFU/g), 7.7% (1 out of 13) were classified as satisfactory (4 to <6 log CFU/g), and the remaining 76.9% (10 out of 13) were grouped as unacceptable (Table 5). In Dire Dawa city, aerobic bacterial and Enterobacteriaceae counts in 92.5% (24 out of 26) and 88.5% (23 out of 26), respectively, were classified as unacceptable, and 3.8% (1 out of 26) were classified as satisfactory and acceptable for each based on aerobic bacterial counts (Table 5).
Bacterial Load Proportion/Range of Beef Samples Collected From the Butcher Shops in the Study Districts With Their Range of Bacterial Counts in Log CFU/g.

Aerobic bacterial counts: <1 (BDL), <104 (good), 104 – <106 (acceptable), and ⩾106 (unacceptable and potentially dangerous); Enterobacteriaceae and coliform counts: <102 (good), 102 – 104 (acceptable), and ⩾104 (unacceptable and potentially dangerous).
Total Bacterial Load Comparison of Beef Samples From the Abattoirs and Butcher Shops at the Study Sites
Raw beef samples collected from an abattoir in Haramaya displayed aerobic bacterial counts (7.79 ± 0.62 log CFU/g), which were slightly greater than those of Enterobacteriaceae (7.50 ± 1.09 log CFU/g) and coliforms (6.53 ± 1.78 log CFU/g) (P > .05). The aerobic bacterial count (5.97 ± 3.63 log CFU/g) was higher in the minced beef samples than in the raw beef samples (5.97 ± 3.63 log CFU/g) collected from butcher shops from Haramaya. The mean Salmonella count was insignificantly higher in minced beef samples from butcher shops (3.41 ± 0.30 log CFU/g) than in the beef samples (3.06 ± 0.61 log CFU/g). Minced beef samples collected from butcher shops in Jigiga exhibited a mean aerobic bacterial count (6.63 ± 3.04 log CFU/g), which was higher than that in the beef sample (5.73 ± 2.73 log CFU/g), with no significant difference. Samples of minced beef from butcher shops in Dire Dawa showed a mean aerobic bacterial count (9.19 ± 1.42 log CFU/g), which was higher than that of the beef samples (8.17 ± 2.38 log CFU/g). The mean bacterial loads in abattoir beef samples positive for Salmonella from Haramaya, Harar, Jigjiga, and Dire Dawa were 2.60 ± 0.86, 3.13 ± 0.61, 2.84 ± 0.00, and 3.28 ± 0.49 log CFU/g, respectively, whereas the overall Salmonella prevalence was 17.6% (Table 6).
Mean Bacterial Counts of Raw Beef and Minced Beef Samples Collected From Abattoirs and Butcher Shops at the Study Sites.

Abbreviation: MM: minced meat.
Similar superscript letters in the same column indicate that there are no significant differences among the given mean values. However, different superscript letters indicate significant differences among the means.
Total Bacterial Load Comparison of Contact Surface Samples From the Abattoirs and Butcher Shops at the Study Sites
The mean aerobic bacteria count in the cutting board swab samples from butcher shops located in Haramaya town (7.66 ± 1.25 log CFU/cm²) was higher than that in the other meat contact surface samples. In Harar town abattoir, the mean coliform load of knife swab samples (6.10 ± 0.38 log CFU/cm²) was significantly higher than that of processing surface (PS) swab samples (4.66 ± 4.11 log CFU/cm²) (P < .05). The mean of the Salmonella counts from butcher shop workers’ hand swabs was 3.81 ± 0.00 log CFU/cm². In Harar, the mean aerobic bacterial count of beef processing surface swab samples from butcher shops (6.87 ± 0.75 log CFU/cm²) was higher than that of other meat contact surface swab samples (knife, cutting board, hand). In Jigjiga, the mean aerobic bacterial count (6.85 ± 0.86 log CFU/cm²) and Enterobacteriaceae count (6.15 ± 0.96 log CFU/cm²) were greater in cutting board swab samples collected from the abattoir than in the other contact surface samples. In Dire Dawa city, the mean aerobic bacterial load on cutting board swab samples from butcher shops (9.39 ± 0.91 log CFU/cm²) was significantly higher than that from butcher shop workers’ hand swab samples (4.90 ± 3.08 log CFU/cm²; P = .034). Similarly, in butcher shop samples, the mean Enterobacteriaceae (9.23 ± 0.96 log CFU/cm²) and coliform (9.12 ± 0.97 log CFU/cm²) counts were significantly higher than those in hand swab samples from the study’s butcher shop workers (P < .05; Table 7).
Mean Bacterial Counts of Beef Contact Surface Samples Collected From the Abattoir and Butcher Shops at the Study Sites.

Abbreviations: CB: cutting board; PS: processing surface.
Similar superscript letters in the same column indicate that there are no significant differences among the given mean values. However, different superscript letters indicate significant differences among the means.
Overall Prevalence of Salmonella
Among the 324-beef meat and meat contact surface samples examined for detection of Salmonella, 57 were positive, with an overall prevalence of 17.6% in the study areas. Salmonella prevalence was significantly greater (P = .034) in abattoir samples (22.7%) than in butcher samples (13.7%). Among the study sites, samples taken from Dire Dawa city had the highest prevalence of Salmonella (21.7%), followed by those taken from Jigjiga (17.9%), with no significant variation (P > .05). The prevalence of Salmonella was greater in the minced meat samples obtained from the butcher shops (21.9%) than in the other samples (Table 8).
Prevalence of Salmonella Obtained from Beef and Contact Surfaces in the Beef Value Chain of Abattoirs and Butcher Shops in Eastern Ethiopia.

Abbreviations: CB, cutting board; MM, minced meat; PS, processing surface.
Prevalence is calculated as (number of Salmonella positive/number of samples) multiplied by 100.
Salmonella Prevalence by Town and Sample Type
Salmonella was detected in 17.6% of the total samples, with prevalence varying by sample type and town. In Haramaya town, of the 20 raw beef samples examined, 5 (25%) were Salmonella positive. Among the minced meat samples, 28.6% (2 out of 7) were positive, with no statistically significant difference (P = .210). In Harar, of the 15 knife swabs and 28 meat samples examined, 3 (20%) and 5 (17.9%) were Salmonella positive, respectively. Among the 32 meat and 10 minced meat samples evaluated in Dire Dawa city, 7 (21.8%) and 3 (30%) were positive for Salmonella, respectively (Table 9).
Salmonella Prevalence by Town and Sample Type from Beef and Contact Surface Samples.
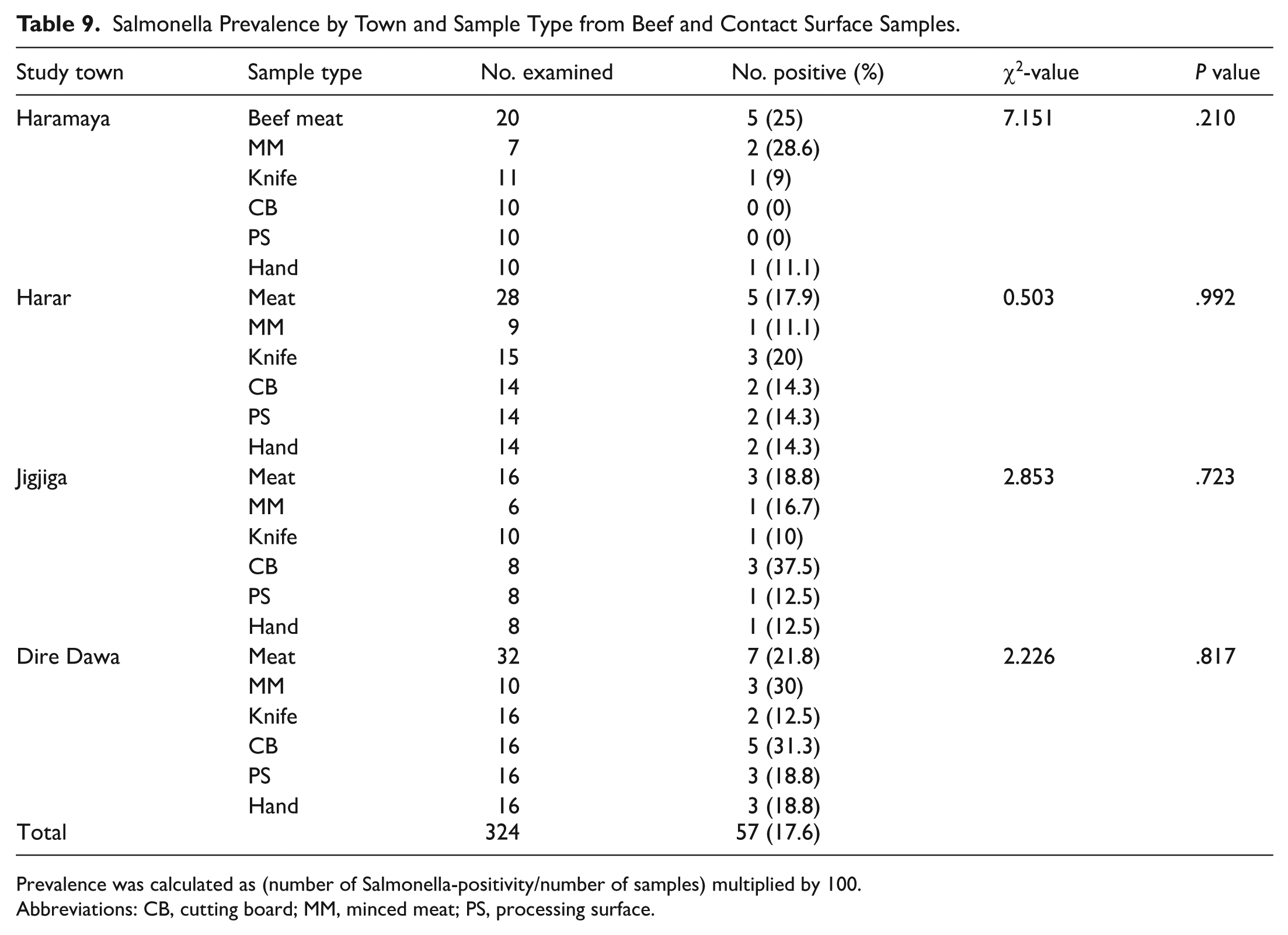
Prevalence was calculated as (number of Salmonella-positivity/number of samples) multiplied by 100.
Abbreviations: CB, cutting board; MM, minced meat; PS, processing surface.
Salmonella Prevalence by Town and Sample Source
A greater proportion of raw beef meat and meat contact surface samples tested in abattoirs were Salmonella positive (22.7%) than those from butcher shop samples (13.7%). Across all towns surveyed, Dire Dawa city, had significantly greater Salmonella prevalence in abattoir samples (31.3%) than in butcher shop samples (13.8%; P = .030), whereas in Haramaya city, a lower Salmonella prevalence was detected in abattoir samples (12%) than in butcher shop samples (13.9%). In Harar town, the prevalence was 21.4% in abattoir samples. Table 10 shows the frequency of Salmonella that was detected to be positive in abattoir and butcher shop samples in the study towns.
Salmonella Prevalence in Beef and Contact Surface Samples From Abattoirs and Butcher Shops Across Towns.

Discussion
This study provides novel insights into the level of mean bacterial load determination and Salmonella contamination on raw beef and meat contact surfaces in abattoirs and butcher shops in selected towns in eastern Ethiopia. The enumeration of aerobic bacterial, Enterobacteriaceae, and coliform counts in raw beef and contact surface samples suggests an inadequate implementation of hygienic practice standard operating protocols in butcher shops and abattoirs. The mean aerobic bacterial count in Haramaya town abattoir was the highest, and that in Harar town abattoir was the lowest, suggesting potential differences in beef processing practices such as handling or evisceration protocols. In Dire Dawa city, a greater mean bacterial count was detected in butcher shop beef samples than in abattoir beef samples, and all other abattoir and butcher shop samples obtained from other towns. The possible factors that contribute to such differences include slaughter hygiene, the quality of the water used, and variations in equipment cleaning protocols among the towns.
The source of the beef in the butcher shops was related to a relatively high bacterial count in the samples. In butcher shops, inadequate or no refrigeration temperatures can potentially increase the growth of bacteria during storage. The ability of warmer temperatures to favor bacterial multiplication and transmission is similar to other findings. 26 Differences in meat handling practices, such as the use of common cutting boards or cutting knives for different types of meat, including offal or internal organs, can increase cross-contamination risk and bacterial counts. In butcher shops, other potential factors include the effectiveness of disinfection and cleaning protocols, such as the cleaning frequency, type of disinfectant and sanitizer used, and time period after slaughter, which may have contributed to the high growth of bacteria. The high mean aerobic bacterial, Enterobacteriaceae and coliform counts observed in the present study are consistent with those reported by Zerabruk et al 26 in Addis Ababa butcheries, who reported total aerobic bacterial, Enterobacteriaceae, and total coliform counts of 6.85 ± 0.83, 6.77 ± 0.69, and 6.84 ± 0.65 log CFU/g, respectively, in minced meat samples, indicating poor personal and working area hygiene and sanitation.
However, in contrast to our findings, Tafesse et al 36 reported relatively low aerobic bacterial, Enterobacteriaceae and coliform counts in raw beef samples from Jigjiga town, potentially because of differences in regulatory slaughtering, such as hide removal, evisceration techniques and hygienic practices, including carcass washing protocols. 37 Our study revealed greater aerobic bacterial, Enterobacteriaceae, and coliform counts in the butcher shops when we compared data from Atlabachew and Mamo, 7 who reported aerobic bacterial, Enterobacteriaceae and coliform counts of 5.47, 4.84, and 4.88 log CFU/g, respectively, from meat, which suggests the importance of adhering to strict standard regulations.
In our study, beef contact surface samples obtained from abattoirs in all towns presented higher mean bacterial counts (aerobic bacterial, Enterobacteriaceae and coliform counts) than did butcher shop samples, possibly suggesting differences in hygienic procedures and/or the effectiveness and frequency of cleaning the premises. Here, the mean aerobic bacterial count on contact surfaces varied highly across the 4 study towns, with Dire Dawa city revealing the highest aerobic bacterial mean count in the abattoir and Jigjiga the lowest. Similarly, higher Enterobacteriaceae counts were detected in contact surface samples collected from butcher shops in Dire Dawa city, with Haramaya having the lowest count, highlighting variations in sanitation, some of which did not use sanitizing or disinfecting agents or hygienic practices. The cross-contamination of shared knives and cutting boards and frequent hand contact with surfaces may have contributed to the high bacterial count on the contact surfaces. Facilities and resource availability for hygiene and cleaning, together with frequent supervision by the concerned authorities, may differ across towns, which impacts bacterial counts on contact surfaces. The training and awareness levels of food handlers in different towns may also contribute to differences in the bacterial load.
Zerabruk et al 26 reported relatively consistent aerobic bacterial, Enterobacteriaceae and coliform counts on contact surface materials, such as 6.31 log CFU/cm2 on a knife and 6.32 log CFU/cm2 on a cutting board in butcher shops. The mean aerobic bacterial, Enterobacteriaceae and coliform counts on contact surfaces obtained in the present study are higher than those reported by Atlabachew and Mamo 7 in meat and contact surface swabs in butcher shops of Debre Berhan, Ethiopia, with mean aerobic, Enterobacteriaceae, and total coliform counts of 4.17, 4.08, and 3.96 log CFU/g, respectively, reflecting potential variations in hygienic practices, sampling techniques, or other factors such as geographical location.
All beef samples collected (100%) from abattoirs in Haramaya and Jigjiga towns considered unacceptable on the basis of aerobic bacterial counts. In Harar and Dire Dawa, the percentage of unacceptable meat samples was greater than 85%. According to Enterobacteriaceae counts, more than 75% of abattoir meat samples in Haramaya, Harar, and Dire Dawa fell into the unacceptable or unsatisfactory category, highlighting a great public health concern. Limitations in sanitation for trained personnel and abattoir workers were observed. The proportion of beef samples that were considered unsatisfactory or unacceptable raises potential food safety risks and suggests the importance of interventions to minimize contamination of beef and its products by bacteria. Compared with samples from butcher shops, a greater proportion of beef samples from abattoirs were categorized as unsatisfactory/unacceptable, potentially suggesting poor sanitation and hygienic practices during slaughter. 38 The higher proportion of unsatisfactory or unacceptable meat samples in many of the towns is most likely attributed to factors such as level of education (training), local control implementation, temperature level, working personnel, handling of animals before slaughter, and slaughter hygiene. 39
The proportion of unacceptable raw meat samples from butcher shops in the towns that sourced meat from the studied abattoirs was significantly greater, reflecting the initial bacterial contamination of the meat. In agreement with the present findings, Zerabruk et al 26 reported that all the meat and minced meat samples collected from the Addis Ababa butcheries were classified as unsatisfactory. The overall significant unacceptable proportion of meat samples obtained in this study is also in accordance with that reported by Kenaw et al 40 in Hawassa city butcher shops, Ethiopia. Zulfakar et al 41 reported a slightly lower proportion of meat acceptability, with possible justification in terms of sampling methods, laboratory processes, and the effectiveness of the intervention differences between slaughter and butchery. Total bacterial loads that were not acceptable and exceeded the standard limit set for aerobic bacterial, Enterobacteriaceae, coliform, and Salmonella counts by the FAO 25 and the European Safety Authority 42 alarm signals to improve overall hygiene practices.
In Dire Dawa city, statistically significant (P < .05) loads of aerobic bacterial, Enterobacteriaceae, and coliform counts were detected on cutting board samples collected from butcher shops, reflecting the need for effective cutting board hygiene to prevent contamination of meat. Significant aerobic bacterial, Enterobacteriaceae, and coliform counts were observed in hand swab samples from abattoir workers in this city compared with those from the other cities, suggesting the importance of employee hygiene and sanitation.
The cutting board and processing surface samples presented the highest potential risk for beef contamination, followed by the knife and hand swab samples. Overall, higher counts of bacteria on equipment surfaces, hand, and processing surface samples that come into contact with the beef in abattoirs and butcher shops highlight the need to follow sanitation enforcement protocols. In support of our observations, the results of Bersisa et al 20 revealed that high bacterial loads contact surface samples (knife and cutting tables), possibly indicating the importance of hand and cutting boards thorough hygiene and frequent cleaning. Ayalew et al 43 also reported higher mean counts of bacteria from knives (6.01 log CFU/cm2) and cutting boards (6.03 log CFU/cm2) from Jigjiga town, Ethiopia. This also potentially suggests inadequate hygiene practices and the temperature situation, which is again supported by Zerabruk et al. 26 The mean total Salmonella count on the cutting board in the current study was slightly lower than in the cutting tables of the study conducted in Addis Ababa, the capital of Ethiopia, by Zerabruk et al. 26 This difference is potentially due to the inadequate hygiene and handling practices maintained at abattoirs and butcher shops.
The overall prevalence of Salmonella across all the beef and contact surface samples was 17.9%, indicating a potential public health concern within the beef value chain in the studied towns. The prevalence of Salmonella varied across the different towns surveyed, with Dire Dawa city exhibiting the highest prevalence (21.7%) and Haramaya town the lowest (13.2%). These findings suggest that local factors, such as sanitation standards or enforcement practices, may play a role in Salmonella contamination across towns. Differences in temperature across towns also favor or retard the multiplication of most bacteria, including Salmonella. This may be due to the increased survival and spread of Enterobacteriaceae, including Salmonella, at relatively warm temperatures. A statistically significant association was observed between Salmonella prevalence in abattoirs and in butcher shop samples (P = .034), indicating that overall beef and contact surface samples contaminated with Salmonella are significant contributors to raw beef and beef product contamination. These findings suggest that the contamination may originate from the meat source or the process of slaughtering the beef in abattoirs, such as flaying, evisceration, and cutting. Detection of Salmonella in the beef and contact surface swab samples suggest both raw beef products and processing surfaces contribute to the overall Salmonella burden in abattoirs and butcher shops.
An increased prevalence of Salmonella was detected in minced beef samples (25%) from Haramaya town compared with other beef and contact surface samples, potentially due to differences in processing methods and contamination during the cutting stage. This is possibly linked to the hygienic practices and sanitary level of the meat handling premises and food handlers. 44 Salmonella was more frequently detected in the cutting board samples collected from Jigjiga (37.5%) and Dire Dawa (31.3%) than in those collected from Harar (14.3%) and Haramaya (0%), potentially suggesting differences in the implementation of hygiene procedures, including frequent washing of working materials and meat-cutting boards. Considerable detection levels of Salmonella in hand swab samples from abattoirs and butcher shop beef handlers reflect workers may also serve as source of contamination.
The total Salmonella prevalence in raw beef and contact surface samples detected in our study (17.9%) is consistent with findings of Zelalem et al, 45 who reported an overall prevalence of 16.7% in butcher shop samples from Addis Ababa and Hawassa cities in Ethiopia. In contrast to our findings, a significantly low prevalence of Salmonella was reported by Tadesse and Gebremedhin 18 (4.53%) in Ethiopia and Gebremedhin et al 46 (5.7%) in Ambo and Holeta and Kore et al 47 (4%) in Hawassa on meat and meat contact surface samples from abattoirs and butcher shops, which suggests possible differences in hygienic practices or sampling approaches. Compared with the current study, a low prevalence of Salmonella was also reported by Zerabruk et al 26 on meat contact surfaces (4.17%) and minced meats (6.25%) from butcher shops, which shows a potential deprivation in hygiene practices through time or other local factors that contribute to Salmonella contamination. Kanko et al 48 reported high Salmonella prevalence (38%) in meat and contact surface samples from butcher shops in Arba Minch town, southern Ethiopia, which is contrary to our relatively low Salmonella prevalence, potentially attributed to differences in techniques of sampling, culture methods, geographical locations or regions, periods of time, and the implementation of more effective prevention and control measures as well as regulatory standards.
Meat handling practices differ (such as the skills of personnel in gut evisceration, carcass examination, and meat handling), hygienic standards in slaughterhouses, and the handling practice during transporting beef can be considered potential factors that have contributed to the contamination of beef by this pathogen. 18 In general, the consumption of raw beef contaminated with Salmonella can lead to food-borne diseases, especially in immunocompromised people. This may be due to a lack of knowledge about the microbial risks associated with processing and handling, the absence or inefficiency of food safety management systems, limited financial resources, lack of awareness among food handlers, and inadequate regulations and regulatory standards.
Significance and Limitations of the Study
Such significant bacterial loads, including Salmonella, on meat and investigated contact surface samples pose a substantial risk of spoilage and foodborne illness to consumers. This study is significant for public health, socio-economic, and scientific reasons. The study significantly contributes in developing foodborne diseases and outbreak control and prevention strategies. It helps manage and monitor the risk of Salmonella including its antimicrobial resistance characteristics as it is a great concern. The bacterial contamination load assessment helps to ensure and improve meat quality and safety with national and international standards. The results help in developing policies and strategies including meat inspection and regulations of hygiene. It provides a guide to the meat inspectors and handlers on the meat related practices such as inspection, slaughtering, handling, and meat selling. The scientific advantage includes building a baseline data and contributes to the wide audience knowledge. The present study did not evaluate the specific cleaning, hygiene, or disinfection practices employed in each facility. This may have limited our ability to assess major factors contributing to the differences in bacterial loads on meat and contact surface samples and limited our ability to detect the most effective and appropriate strategies that help reduce contamination by bacteria, including Salmonella, in meat and meat contact surface facilities. This possibly includes evaluating the effectiveness of different sanitizers, cleaning procedures, or designs of working equipment.
Conclusion
The study demonstrated higher mean counts of aerobic bacterial, Enterobacteriaceae and coliform bacteria in raw beef and contact surface samples from abattoirs and butcher shops in the towns. This implies the presence of potential risk of spoilage that decrease meat shelf-life, leading to a substantial public economic loss and health impact. The significantly higher bacterial counts on the cutting boards in Dire Dawa city, together with the significant proportion of unacceptable meat samples observed in the towns of abattoirs and butcher shops, suggest that poor sanitation of the cutting board is a key factor in bacterial cross contamination of beef meat. Furthermore, the detection of Salmonella in meat and contact surface samples from abattoirs and butcher shops poses a high risk to public health, primarily for vulnerable populations, including young, elderly, and immunocompromised individuals. Although Salmonella was detected in both abattoir and butcher shop samples, the prevalence was significantly higher in abattoirs than in butcher shops, indicating the need for improved sanitation practices at slaughterhouses, such as hide removal, evisceration, and cutting. Our results underscore the importance of enhancing regular surveillance and control measures such as sanitation improvement. We emphasize the need for practical interventions such as hygiene regulations and workers training on sanitation practices, increased cleaning and disinfection practices in butcher shops and abattoirs to reduce the bacterial load on beef and contact surfaces in the beef supply chain and safeguard public health. We further recommend future detailed research focus on characterizing Salmonella strains obtained from beef and contact surface samples.
Footnotes
Acknowledgements
We would like to extend our acknowledgment to the College of Veterinary Medicine of Haramaya University for providing laboratory consumables during the work and allowing us the materials and the laboratory from beginning to end. We are sincere to butcher shops and abattoir workers for their cooperation during sample collection for the study. The study would have been impossible without the genuine cooperation shown by the workers.
Ethical Considerations
The research was reviewed and granted ethical approval by the College of Agriculture and Environmental Sciences _Animal REC (2024/CAES_AREC/2678) on 21 May 2024 in compliance with the Unisa Policy on Research Ethics and the Standard Operating Procedure on Research Ethics Risk Assessment.
Author Contributions
JK and FTT proposed the research idea and designed and outlined the research. JK conducted the sample collection and carried out all the laboratory work, analyzed, and interpreted the data. FTT supervised and assisted in the study. JK prepared the first draft of the manuscript for publication. FTT reviewed the draft manuscript. Both JK and FTT read and approved the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the Research Office of the University of South Africa.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The necessary datasets utilized during the study are available from the corresponding author of the manuscript on reasonable appeal.

