Abstract
Introduction:
Hand hygiene is a cornerstone of infection prevention; however, the water and infrastructure used for handwashing can themselves become sources of microbial contamination. This study assessed the water quality and sanitary conditions of hand hygiene facilities in public and private hospitals in Addis Ababa, Ethiopia.
Methods:
A laboratory-based cross-sectional study was conducted from May to June 2025 in 10 hospitals. Forty samples were collected, including 20 tap water samples and 20 swab samples from tap handles. A sanitary survey evaluated water supply and hand hygiene stations, with onsite physicochemical measurements, and targeted microbial analyses performed. Data were recorded in an Excel spreadsheet and analyzed using SPSS, employing descriptive statistics and Chi-square tests, with significance defined at P < .05.
Results:
The sanitary survey showed that 62.5% of hospitals had continuous running water, with 75% of water systems free from leaks. Handwashing stations were accessible in 55% of facilities, primarily hand-operated (67.5%), followed by elbow-operated (25%) and foot-operated (7.5%). Maintenance deficiencies were noted in 37.5% of stations, with soap available at only 55%. Hand drying materials were present in 12.5% of hospitals, and only 45% of facilities reported regular cleaning of handwashing stations. Physicochemical water quality parameters were generally within acceptable limits. Microbiological analysis revealed high contamination: Escherichia coli was detected in 66.7% of all samples, found on 100% of tap handles compared to 31.6% of water samples (P < .001). Other pathogens included Staphylococcus aureus (55%), Klebsiella (52.5%), Shigella (50%), and Salmonella (50%), all significantly prevalent on swabs than water samples (P < .001), with no significant difference by hospital ownership.
Conclusion:
The study found significant microbial contamination in handwashing infrastructure. To reduce risks, facilities should install non-hand-operated stations, enhance routine cleaning and disinfection, and ensure proper maintenance. Regular water quality monitoring and infection control training are essential.
Keywords
Introduction
Healthcare-associated infections (HCAIs) remain a significant global public health concern, undermining patient safety, prolonging hospital stays, increasing healthcare costs, and contributing to morbidity and mortality worldwide.1 -3 The World Health Organization (WHO) estimates that hundreds of millions of patients are affected annually by infections acquired during healthcare delivery, particularly in low- and middle-income countries (LMICs), where resource constraints exacerbate infection risks and control challenges. 4
In Ethiopia, healthcare-associated infections (HAIs) are a significant concern, with studies suggesting that approximately 10% to 23.7% of hospitalized patients acquire an infection during their stay.5 -7 Among interventions to combat these infections, effective hand hygiene is universally acknowledged as the single most crucial practice to break the chain of infection transmission between healthcare workers and patients, thereby substantially reducing HCAI rates.4,8,9 However, its success depends not only on healthcare workers’ compliance but also on the quality and safety of the water and infrastructure used for handwashing. Factors such as water quality parameters, water volume used, and temperature,10 -18 as well as the concentration of hand hygiene products, contact time, the type of product used, the distance of the hand hygiene supplies from the point of care, and proper handwashing techniques, influence hand hygiene effectiveness.19 -21
Despite extensive efforts to promote alcohol-based hand rubs and improve hand hygiene compliance, the microbiological quality of handwashing water and the condition of hand hygiene facilities have often received inadequate attention. Contaminated water and handwashing surfaces may paradoxically serve as reservoirs and vectors for pathogenic microorganisms, thereby compromising infection prevention efforts.10 -12,15 -18 In particular, hand-operated taps and poorly maintained hand hygiene infrastructure may contribute to cross-contamination, nullifying the benefits of hand hygiene practices. Water quality parameters, such as pH, turbidity, microbial contamination, and presence of enteric pathogens, substantially influence handwashing effectiveness to prevent infection. Water meeting physicochemical standards may harbor opportunistic and enteric bacterial pathogens such as Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Salmonella species, commonly linked to healthcare-associated infections and antimicrobial resistance.15,16,18
In Ethiopia, some studies have documented the presence of pathogenic bacteria, focusing on the drinking water sources, including contamination with fecal bacteria and pathogens like E. coli, S. aureus, and Klebsiella spp., and other.22,23 However, there is a notable lack of evidence specifically addressing water used for hand washing and microbial contamination of tap delivery handles in healthcare settings across both public and private hospitals. 24
Ensuring effective hand hygiene thus requires not only compliance but also regular monitoring and regulation of water quality and hand hygiene infrastructure to meet established safety standards.25 -27 Although drinking water quality is often prioritized, the quality used for personal hygiene, including handwashing, is frequently overlooked, despite its direct impact on infection prevention practices. The contamination of handwashing water with pathogenic microorganisms in healthcare settings threatens hygiene and patient care. Common waterborne pathogens include Klebsiella, E. coli, Salmonella, Shigella, Vibrio cholerae, Shigella, Campylobacter, Legionella, and Helicobacter species. 28 These microbes can survive in water systems and have been linked to healthcare-associated infections (HCAIs), particularly in vulnerable populations such as hospitalized patients with compromised immune systems.
This study focuses explicitly on detecting key pathogens commonly associated with HCAIs: Klebsiella, E. coli, Shigella, Staphylococcus aureus, and Salmonella. It aimed to assess the microbial quality of handwashing water and evaluate the sanitary conditions of hand hygiene facilities in public and private hospitals in Addis Ababa, Ethiopia.
Methods and Materials
The Study Area
The study was conducted in Addis Ababa, which is the capital city of Ethiopia and the seat of the African Union. According to the Ethiopian Ministry of Health, Addis Ababa city has 37 hospitals (13 public, 24 private) and 100 health centers. 29 The city’s water, sanitation, and hygiene infrastructure service delivery shows significant gaps, with 86% of healthcare facilities having basic water, 14% having limited, and 88.4% having limited hand hygiene services. 29 Figure 1 highlights the locations where water and swab samples were collected. 30
Geographic distribution of hospitals where water and swab samples were collected, May 2025.
Study Design and Period
A laboratory-based cross-sectional study, complemented by a sanitary survey and an observational checklist, which was conducted from May 2025 to June 2025 to assess the bacteriological quality of water and handwashing tap handle surfaces in selected hospitals in Addis Ababa.
Sample Size Determination and Sampling Strategy
A stratified purposive sampling technique was employed to ensure representation across hospital ownership types in Addis Ababa. Hospitals were first divided into strata based on ownership (public vs private). Within each stratum, hospitals were purposively selected based on service levels, types, healthcare worker density, and patient flow to capture variation relevant to the study. A total of 10 hospitals, 6 public and 4 private, were chosen based on greater patient volumes and healthcare workers in public hospitals than in private ones, which potentially increases exposure risks.
From these 10 hospitals, a total of 40 samples were collected purposively: 20 water samples and 20 swab samples, taking into account resource constraints, feasibility, and statistical power considerations. In each hospital, sampling areas were systematically chosen from high-contact areas such as outpatient departments, inpatient wards, and emergency units. Sampling procedures were standardized across all facilities to ensure methodological consistency and minimize selection bias.
Sample Collection Procedures
Water Sample Collection
The on-site physicochemical analysis of the water, including pH, total dissolved solids (TDS), temperature, conductivity, and dissolved oxygen (DO), was performed using a digital Oakton PCD 650 multiparameter device. Then, water samples (500 mL) were collected aseptically from handwashing taps in sterile bottles containing 0.2 mL of 3% sodium thiosulfate to neutralize residual disinfectants.
Swab Sample Collection
Surface swabs were taken from tap water delivery handles using standardized sterile swabbing techniques to assess microbial contamination on handwashing tap water handles. 31
For swab samples, 10 mL of tryptic soy broth was added to the sampling tube, and 2 swab sticks were used for sample collection. The swab sticks were placed inside the broth tube for transport to promote the recovery and enrichment of microorganisms from the surfaces. Samples were transported in a cold box at 0°C to 4°C to the microbiology laboratory.32,33
Sanitary Survey Data Collection
A sanitary survey was conducted using a structured observational checklist to assess water supply infrastructure, hand hygiene station accessibility, maintenance, and environmental conditions at each facility. Key parameters included the availability of running water during sampling from the main water source, station cleanliness and functionality, availability of soap and drying materials, presence of leaks or cross-connections, regular cleaning schedules, waste management, and signs of contamination or poor ventilation. This survey aimed to evaluate the operational status and hygiene of handwashing facilities to support infection control measures.
Operational Definitions
In this study, the definitions were based on the WHO/UNICEF Joint Monitoring Programme (2016). Basic water service refers to water that is available from an improved source located on the premises. Limited water service describes situations where water is obtained from an improved source but is either located off premises or is not consistently available on-site. No water service indicates that the facility relies on unprotected water sources or lacks any water supply. For hand hygiene, basic service is defined as the availability of hand hygiene materials, such as soap and water or alcohol-based hand rub, at points of care. Limited hand hygiene service means that hand hygiene facilities are available either at the point of care or the toilet, but not both. No hand hygiene service is when hand hygiene stations are absent or present without soap or water. 34
Sample Analysis
Physicochemical Water Samples Analysis
The water samples were analyzed on-site for pH, TDS, temperature, conductivity, and DO using the Oakton multiparameter probe.
Microbial Analysis
The analysis of water samples and water tap handle surfaces for the presence of microorganisms and bacterial isolation was conducted at the Microbiology Laboratory of Addis Ababa University. All the samples were analyzed within 24 hours of arrival at the microbiology laboratory.
Water Sample Analysis
The assessment included the detection of Escherichia coli, Staphylococcus aureus, Klebsiella spp., Shigella, and Salmonella spp. A membrane filtration (MF) method was employed to enumerate Escherichia coli in water samples. A 100 mL volume of each water sample was passed through a sterile 0.45 µm membrane filter. The filter was then aseptically transferred onto m-ColiBlue24® Broth-saturated pads and incubated at 35°C for 24 hours. The presence of blue colonies indicated Escherichia coli.35 -37
A streak plate method was utilized to detect other target microorganisms from water. Water samples were initially enriched by adding 10 mL of the sample to sterile broth to promote microbial growth. From this enriched broth, 1 mL aliquots were aseptically withdrawn and used to inoculate selective agar plates. The inoculation was performed using the streak plate technique, spreading the inoculum over the agar surface with a sterile loop to obtain isolated colonies.
For Staphylococcus species identification, samples were streaked on mannitol salt agar and incubated at 37°C for 24 hours; yellow fermentation zones indicated Staphylococcus presence. Confirmation of the Staphylococcus aureus was performed using the coagulase test.38,39
Klebsiella detection was performed by inoculating aliquots from selective enrichment broths onto MacConkey agar and incubating at 37°C for 24 hours. After incubation, large, mucoid, lactose-fermenting red colonies with a diffusing red pigment were suspected as Klebsiella. These colonies were further confirmed by biochemical tests; those negative for hydrogen sulfide production and positive for methyl red and Voges-Proskauer tests were identified as Klebsiella.40,41
To detect Salmonella and Shigella, samples underwent a two-step enrichment process: first in buffered peptone water at 37°C for 24 hours, followed by incubation in selenite F broth at 42°C for 48 hours. Enriched cultures were then streaked onto Salmonella-Shigella agar and incubated at 37°C for 18 hours. Suspected colonies were further subjected to biochemical identification tests.35 -38
All biochemical tests were incubated for 18 to 24 hours at 35°C to 37°C and monitored for characteristic color changes indicative of metabolic activity. For the catalase test, a few drops of 3% hydrogen peroxide were applied to bacterial cultures on a glass slide, with the formation of bubbles indicating a positive reaction. Gram staining was also performed to differentiate bacteria based on cell wall properties, with Gram-positive bacteria appearing purple and Gram-negative bacteria appearing pink after counterstaining with safranin. All microbiological procedures adhered strictly to the standards set by the Clinical and Laboratory Standards Institute (CLSI). 42
Swab Sample Analysis
Swab samples were vortexed to dislodge microorganisms from the swab cotton. From this broth, 1 mL aliquots were aseptically taken for subsequent culturing. Each 1 mL aliquot was inoculated onto selective media specific to the target microorganisms using a sterile inoculating loop. The inoculation was performed using the streak plate technique, spreading the inoculum over the agar surface with a sterile loop to obtain isolated colonies.
Inoculated selective agar plates from both water and swab samples, typically 90 mm in diameter Petri dishes, were incubated at 37°C for 24 to 48 hours, depending on the specific growth requirements of the microbial species. After incubation, visual inspection was performed to assess colony morphology, color changes, and growth patterns. Biochemical tests to confirm the bacteria were conducted using the same methods as for the water samples.
Quality Control
Aseptic techniques were strictly maintained throughout the sampling and handling processes. Instrument calibration for onsite physicochemical analyses, including the Oakton multiparameter probe, was performed to ensure accuracy.
Water samples were collected in sterile bottles, transported in an icebox, and immediately refrigerated upon arrival at the laboratory. Bacteriological analyses followed standard operating procedures and laboratory safety guidelines. To verify media sterility, 5% of each batch was incubated overnight and monitored for contamination.
Quality assurance measures, including the use of field blanks and negative controls (laboratory blanks) were implemented to ensure accurate species identification. Samples were inoculated onto nutrient agar plates and incubated at 37°C for 24 hours. Resulting colonies underwent biochemical testing for confirmation. All media were prepared according to the manufacturer’s instructions, with sterility confirmed by overnight incubation at 37°C before use.
Data analysis and Interpretation
Data were coded and analyzed using SPSS version 25.0. Descriptive statistics, including percentages, frequencies, means, ranges, and standard deviation, were calculated and presented in tables, charts, and graphs. The Chi-square test was employed to evaluate associations between bacterial presence (dependent variable) and independent variables (sample types: taps vs swabs; hospital ownership: public and private) at a 95% confidence interval (CI), with statistical significance set at P < .05.
Ethical Considerations
Ethical clearance was obtained from the Addis Ababa University, Ethiopian Institute of Water Resources Ethical Review Committee (Ref: EIWR/25/2024). Institutional support letters were secured, and verbal consent was obtained from participating hospitals. Data confidentiality was maintained by anonymizing records.
Results
Sanitary Survey of Water Supply and Hand Hygiene Stations in Addis Ababa Hospitals
The Water Supply Systems of the Hospitals
The sanitary survey conducted in hospitals across Addis Ababa assessed the condition of water supply systems used for hand hygiene and revealed critical gaps. Only 62.5% of the facilities had running water available during sampling, highlighting a significant limitation in access to a basic requirement for effective infection prevention. While the majority of systems (75%) showed no visible leaks or damage, 12.5% of facilities exhibited potential cross-connections between potable and wastewater drains, posing a contamination risk. Additionally, only 57.5% of the surveyed facilities had adequate water flow, which is essential for maintaining proper hand hygiene (Table 1).
Sanitary Survey of the Water Supply Systems in Addis Ababa Hospitals, May 2025 (N = 40).

Hand Hygiene Stations in Addis Ababa Hospitals
The sanitary survey revealed that 62.5% of the facilities had running water available during sampling, with a notable difference between public (44.0%) and private (93.3%) hospitals. Soap was present at 55% of hand hygiene stations (93.3% in private and 32% in public hospitals). Paper towels or hand dryers for hand drying were available in just 12.5% of hospitals. While 62.5% of hand hygiene stations were clean and well-maintained 44% in public and 93.3% in private facilities, accessibility was limited, with only 55% of stations easily reachable by healthcare workers and patients. Most handwashing stations were hand-operated (67.5%), which may affect ease of use and contamination risk. Regular cleaning and disinfection were reported in only 45% of facilities, with 57.5% having a formal cleaning schedule. These deficiencies may compromise the effectiveness of hand hygiene services and increase the risk of healthcare-associated infections (Table 2).
Sanitary Survey of Hand Hygiene Stations Service and Management in 10 Hospitals in Addis Ababa, Ethiopia, May 2025 (N = 40).

The evaluation of hand hygiene services in hospitals across Addis Ababa revealed significant disparities between public and private hospitals. As shown in Figure 2, only 10% of hospitals provided advanced hand hygiene services, 40% offered basic services, and 42.5% reported limited services. Notably, private hospitals had better service levels than public hospitals, highlighting critical gaps in hand hygiene service provision in public hospitals.
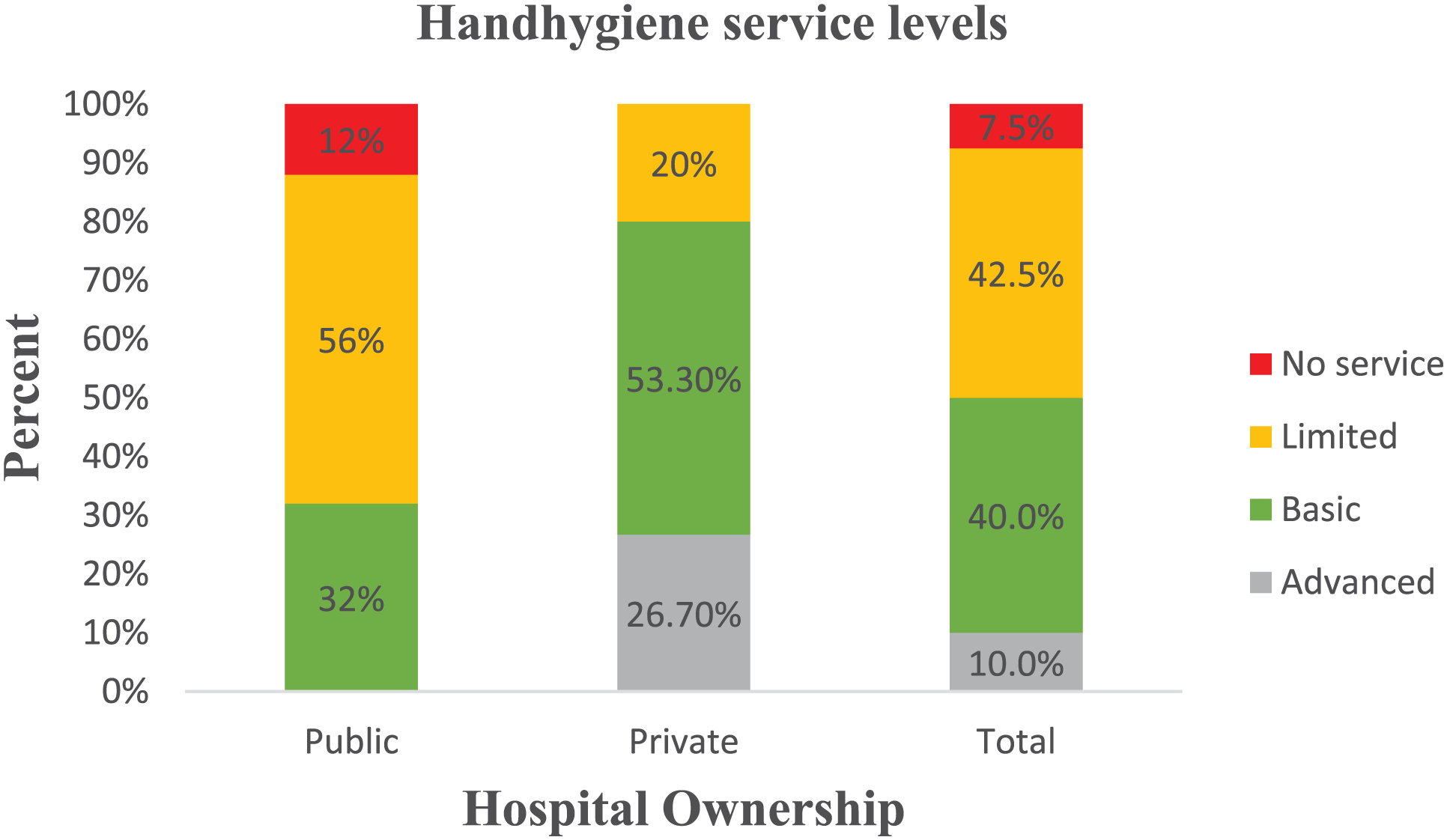
Hand hygiene service level in Addis Ababa hospitals, Addis Ababa, Ethiopia, May 2025.
Physicochemical Quality of Hospitals' Tap Water
The physicochemical quality of tap water across various sampling sites revealed that pH values ranged from 6.3 to 8.3, total dissolved solids (TDS) varied from 92.3 to 269 mg/L, conductivity levels ranged from 163 to 538 µS/cm, and dissolved oxygen (DO) levels ranged from 4.8 to 7.8 mg/L. Table 3 presents the detailed physicochemical parameters of water samples used for hand hygiene at Addis Ababa hospitals.
Summary of Physicochemical Characteristics of Handwashing Tap Water Samples from Hospitals in Addis Ababa, Ethiopia, May 2025.
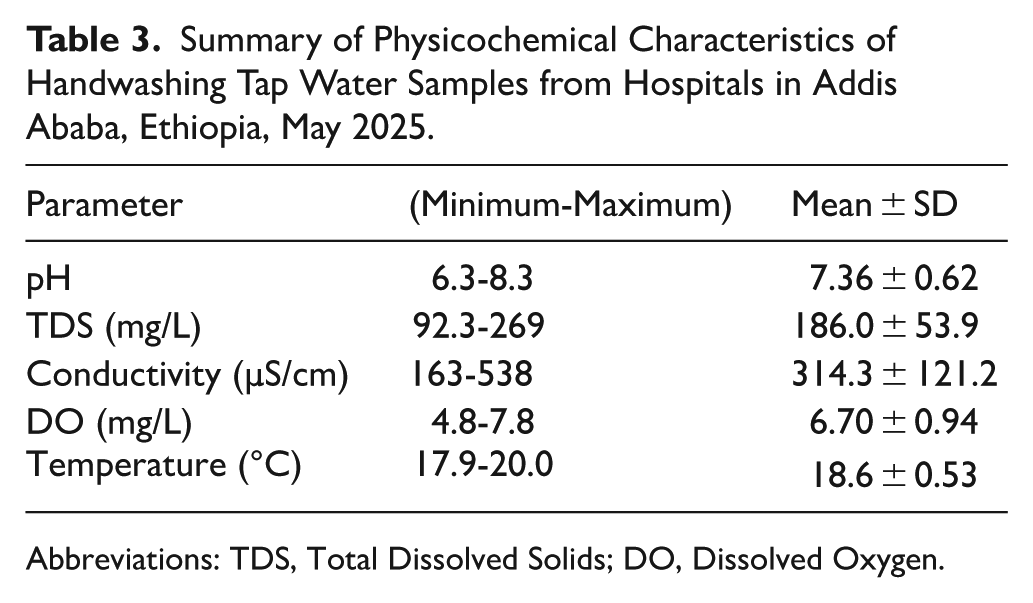
Abbreviations: TDS, Total Dissolved Solids; DO, Dissolved Oxygen.
Microbial Contamination of Water and Handwashing Tap Handles at Addis Ababa Hospitals
Microbial Contamination of Water at Addis Ababa Hospitals
Escherichia coli was the most frequently detected bacterium in water samples, accounting for 31.6% of all positive findings. Klebsiella was detected at a rate of 20%, followed by Shigella at 15% (Table 4).
The Distribution of Microbial Pathogens in the Water Samples Addis Ababa Hospitals, Ethiopia, 2025 (N = 20).

Microbial contamination of handwashing tap handles at Addis Ababa Hospitals
Escherichia coli and Staphylococcus aureus were the most frequently detected bacteria in swab samples, accounting for 100% of all positive findings. Salmonella was detected in 94.7% of samples, followed by Klebsiella and Shigella at 85% (Table 5).
The Distribution of Microbial Pathogens in the Handwashing Tap Handles at Addis Ababa Hospitals, Ethiopia, 2025 (N = 20).

Figure 3 graphically contrasts the detection rates of pathogens from tap swabs and water samples. Notably, bacterial presence in tap swabs is markedly higher than in water, highlighting a possible contamination source in tap handles. Swab positivity rates ranged from 85% to 100% for all bacteria, while water samples ranged from 10% to 31.6%. Staphylococcus aureus and E. coli were the most frequently detected in all swab samples (100%) but were much less common in water (55% and 66.7%, respectively).

The distribution of microbial pathogens in the water and swab samples from handwashing tap handles at Addis Ababa Hospitals, Ethiopia, 2025 (N = 40).
Table 6 presents that all tested pathogens are significantly more prevalent on tap handle swabs compared to water samples (P < .001 for all). For example, while Staphylococcus aureus was detected in 55% of the total sample set, its presence shows a dramatic contrast between tap swabs (100%) and water samples (10%). Similarly, Escherichia coli was present in 65% of total samples, occurring in all swab samples (100%) but only 30% of water samples. Statistical analysis using the chi-square test revealed these differences to be highly significant (P < .001 for all pathogens), indicating a strong association between sample type and bacterial contamination.
Statistical Comparison of Microbial Pathogen Distribution Between Water and Tap Swab Samples in Addis Ababa Hospitals, Ethiopia, 2025 (N = 40).

Abbreviations: χ², Pearson chi-square; df, degrees of freedom, and Fisher’s Exact Test confirmed significance for small counts.
Statistical significance P < .05.
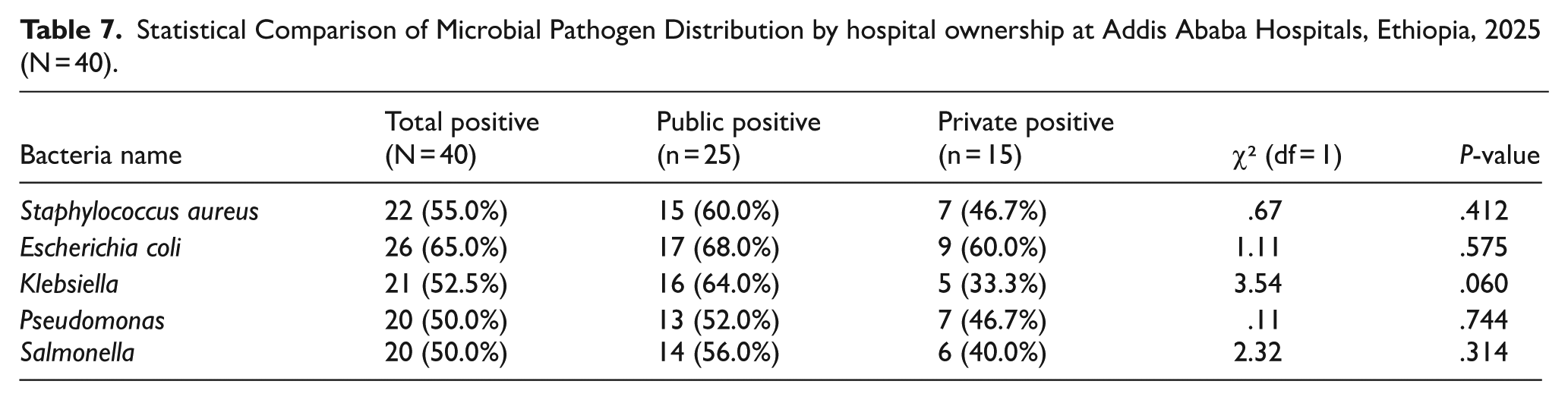
The prevalence of bacteria such as Staphylococcus aureus, Escherichia coli, Klebsiella, Shigella, and Salmonella was slightly higher in public hospitals compared to private ones. However, these differences were not statistically significant, as indicated by chi-square tests (P > .05 for all pathogens), suggesting similar contamination levels across hospital ownership types. Detailed counts, percentages, and statistical results are presented (Table 7).
Statistical Comparison of Microbial Pathogen Distribution by hospital ownership at Addis Ababa Hospitals, Ethiopia, 2025 (N = 40).
Discussions
This study assessed the water quality, sanitary conditions, and presence of pathogenic microbes on handwashing taps in public and private hospitals in Addis Ababa, Ethiopia, which have direct implications for infection prevention and control (IPC). The findings reveal significant gaps that undermine effective hand hygiene practice and increase the risk of healthcare-associated infections (HAIs).
The availability of running water at hand hygiene stations (62.5%) falls short of the WHO’s recommendation for continuous water supply in healthcare settings, which is essential to maintain effective handwashing practices and reduce pathogen transmission in healthcare settings. 41 This finding aligns with previous studies done in Addis Ababa, Ethiopia, showing suboptimal water and hygiene infrastructure in public health care facilities. 29 Although the physicochemical parameters of water samples were within acceptable limits, microbial contamination with key pathogens such as Escherichia coli, Staphylococcus aureus, Klebsiella spp., Shigella spp., and Salmonella spp. was alarmingly high, particularly on handwashing tap handles. The prevalence of pathogens is notably higher on tap handles than in water, raising serious concerns about contamination sources and potential healthcare-associated infection (HAI) risks. This is a critical finding that exposes taps as potential reservoirs facilitating cross-infection. 43 The presence of E. coli, particularly on water handles (100%), points to inadequate sanitation and the potential for fecal-oral transmission within the healthcare environment. This prevalence of microbial contamination on tap handles is supported by the findings from other low- and middle-income countries (LMICs), where hand-operated taps, inadequate cleaning and maintenance, and limited access to hygiene supplies heighten the risk of cross-contamination. 44
Moreover, the sanitary survey further identified that a quarter of water supply systems had visible leaks or damage, posing risks of both water loss and microbial ingress. The lack of cross-connections between potable and non-potable systems is reassuring, as it reduces the risk of gross contamination. Access to hand hygiene stations is generally poor, with only 55% of stations easily reachable and strategically located, which is crucial for promoting frequent handwashing. Additionally, the predominance of hand-operated stations (67.5%) over more hygienic options like elbow and foot may facilitate cross-contamination, as hand contact with taps is a known vector for pathogen transfer. 45 This finding underscores the urgent need for infrastructural upgrades such as elbow- or foot-operated taps that minimize hand contact and reduce microbial spread.
In addition, only 55% of stations had soap available, and a mere 12.5% offered hand drying materials.41,43 Limited soap availability and hand drying facilities have repeatedly been identified as barriers to proper hand hygiene compliance and can directly increase HAIs.46,47
The evaluation of hand hygiene services in Addis Ababa hospitals revealed notable disparities between public and private facilities, with only 10% of hospitals providing advanced service levels, 40% basic, and 42.5% limited services. This finding is consistent with other studies in Ethiopia, where public hospitals frequently face challenges such as limited availability of running water, soap, and functional hand hygiene stations, all of which undermine effective infection prevention efforts.29,43,47 Resource constraints, inadequate maintenance of infrastructure, and inconsistent supply chains within the public sector contribute to these gaps, placing healthcare workers and patients at increased risk of healthcare-associated infections (HCAIs). In contrast, private hospitals, often better resourced and managed, demonstrate higher compliance and superior hand hygiene infrastructure.
Similar trends have been observed in other developing countries. For instance, studies from Nigeria and Kenya show that private healthcare facilities generally outperform public hospitals in hand hygiene infrastructure and compliance.48,49 The WHO/UNICEF Joint Monitoring Programme further highlights that many healthcare facilities in low- and middle-income countries still lack reliable water access, adequate hand hygiene infrastructure, and sufficient supplies, factors that directly affect infection control outcomes. 41 To bridge this gap requires substantial investments in water and hygiene infrastructure, regular maintenance and upgrades, particularly in public hospitals, as well as comprehensive training for healthcare workers in both public and private facilities. These deficits likely contribute to suboptimal hand hygiene compliance and heightened risk of HCAIs.
Notably, although public hospitals exhibited slightly higher microbial prevalence than private ones, the differences were not statistically significant. This suggests that hand hygiene service challenges extend beyond ownership status, pointing to broader issues such as management and enforcement of cleaning, disinfection, and maintenance protocols. Training and awareness raising for healthcare facility management on effective hygiene facility maintenance are needed to address microbial contamination universally across facilities.
The implications for healthcare worker safety and patient outcomes are significant. Contaminated hand hygiene stations elevate the risk of HCAIs, underscoring the critical importance of comprehensive hand hygiene improvement strategies. In Ethiopia, low hand hygiene compliance rates have been linked to persistent gaps in material availability, ongoing education, and effective promotion, despite gains from multimodal campaigns.46,50
Limitations of the Study
This cross-sectional study provides a snapshot of microbial contamination at one time point and does not capture temporal variations. Furthermore, it focused only on selected bacterial pathogens, excluding viruses and fungi that may also affect hand hygiene. Finally, while significant contamination was identified, no direct causal link was identified between contamination levels and clinical infection outcomes in patients or healthcare workers. Future longitudinal studies are needed to explore these relationships and evaluate intervention impacts.
Conclusion
The study revealed significant microbial contamination of handwashing tap handles and handwashing water in Addis Ababa hospitals, highlighting a critical gap in infection prevention and control. The higher prevalence of multiple pathogens, including E. coli, Staphylococcus aureus, Shigella, Salmonella, and Klebsiella, on tap handles compared to the water source significantly elevates the risk of infection transmission within these healthcare settings. Although water quality was generally better than contamination on tap handles, contamination still occurred and should not be overlooked. Addressing this issue requires a multi-faceted approach involving the implementation of enhanced routine cleaning and disinfection protocols, the installation and upgrading of facilities making non-hand-operated hand hygiene facilities, the consistent provision of hand hygiene products, and improved accessibility to hand hygiene stations. These measures, combined with regular monitoring of both water quality and surface contamination of hand hygiene stations, and ongoing staff education and training on hand hygiene and infection prevention and control, are vital to improving patient safety and reducing the incidence of healthcare-associated infections.
Footnotes
Acknowledgements
The authors gratefully acknowledge the support and cooperation of the Ministry of Health, Addis Ababa Health Bureau, and the participating hospitals. We extend our sincere appreciation to the healthcare workers, hospital managers, the Infection Prevention and Control (IPC) focal points, and Addis Ababa University Biotechnology Research Center, who provided valuable assistance during data and sample collection and analysis.
Author Contributions
Abayneh Melaku: Conceptualized and designed the study, developed the methodology, performed data collection and analysis, interpreted the results, and wrote the initial draft of the manuscript. Dr. Tesfaye Admasu: Contributed to the study design, provided technical support for data collection and analysis, assisted with data interpretation, and critically reviewed and edited the manuscript. Prof. Argaw Ambelu: Provided overall guidance and supervision of the study, contributed to the study design, contributed to data interpretation, and critically reviewed and edited the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets analyzed during this study are available from the corresponding author upon reasonable request.
