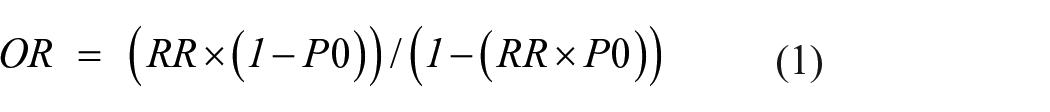Abstract
Paternal lead exposure has emerged as a potential contributor to adverse pregnancy outcomes, yet its impact remains underexplored compared to maternal exposure. This systematic review and meta-analysis synthesize evidence on the association between paternal lead exposure and pregnancy outcomes to inform public health interventions and future research. To evaluate the association between paternal lead exposure and adverse pregnancy outcomes, including spontaneous abortion, low birth weight, preterm birth, small-for-gestational-age, and congenital anomalies. A systematic search of PubMed, Scopus, and Google Scholar was conducted up to August 2024. Observational studies examining paternal lead exposure (⩾15 µg/dL) and its effects on pregnancy outcomes were included. Data synthesis adhered to PRISMA 2020 guidelines, and study quality was assessed using the Newcastle-Ottawa Scale. Meta-analysis was performed using a random-effects model to compute pooled odds ratios (ORs) with 95% confidence intervals (CIs). Eleven studies were included in the systematic review, with 7 contributing to the meta-analysis. The pooled OR for congenital anomalies associated with paternal lead exposure was statistically significant (OR = 2.09, 95% CI: 2.09-3.35; P < .01), indicating a strong association. However, no significant associations were observed for other outcomes: spontaneous abortion (OR = 1.11, 95% CI: 0.75-1.64), low birth weight (OR = 0.98, 95% CI: 0.68-1.39), preterm birth (OR = 1.57, 95% CI: 0.61-4.05), and small-for-gestational-age infants (OR = 0.92, 95% CI: 0.78-1.09). Heterogeneity was low for most outcomes, except for spontaneous abortion (I2 = 39%) and preterm birth (I2 = 52%). This study highlights a significant association between paternal lead exposure and congenital anomalies, emphasizing the need for occupational and environmental regulations targeting lead exposure among men of reproductive age.
Plain Language Summary
Paternal lead may be of reproductive health concern

Keywords
Introduction
Pregnancy outcomes are critical indicators of maternal and child health, significantly affecting the well-being of mothers and their newborns. Adverse pregnancy outcomes contribute substantially to global infant mortality rates. According to United Nations Children Fund (UNICEF), 1 the annual reduction rate in infant mortality decreased from 3.8% between 2000 and 2015 to 2.1% between 2015 and 2022, stalling the progress toward achieving Sustainable Development Goal (SDG) 3. Findings from World Health Organization (WHO) 2 and Center for Disease Control and Prevention (CDC) 3 suggests that the Leading causes of global infant mortality are congenital birth defects, chromosomal abnormalities, low birth weight, and preterm birth. Despite improvements in facility-based births, these leading causes are prevalent especially in low- and middle-income countries. 2 Among various possible factors, studies indicate environmental toxins, particularly parental lead exposure is strongly associated with adverse pregnancy outcomes.4 -7
Considerable progress has been made in reducing lead exposure globally over the past few decades.8,9 Historically, the 1960s established a benchmark of 60 μg/dL as the acceptable limit for human lead exposure, 10 but emerging evidences has shown that lead is harmful even at lower concentrations. Currently, WHO has set the reference limit for blood lead levels at 5 μg/dL, emphasizing that no level of lead exposure is known to be without harmful effects. 11 The impact of lead exposure is significant in countries with historically high industrial activities and in low- and middle-income (LMIC) countries12 -14 where enforcement of environmental health regulations may be inadequate. 14 In the study by Huang et al 15 findings revealed that Asian and south African populations have higher blood lead levels than those in South America, North America and Europe, furthermore, populations in Pakistan, Iran Egypt, and China have BLLs exceeding the WHO recommended alert level of 5 μg/dL. Meanwhile, in countries like the United States and Western Europe, workplace safety regulations have reduced occupational lead exposure, though risks persist in certain industries. 8
Lead exerts its toxic effects on pregnancy by interfering with essential cellular processes and disrupting the body’s normal metabolic functions. 16 According to the World Health Organization (WHO), there is no safe threshold for human lead exposure as lead can cause harm even at low blood lead levels (<5 μg/dL). Exposure to lead generates reactive oxygen species (ROS), leading to oxidative stress that damages DNA, lipids, and proteins, 17 which can trigger inflammatory responses in pregnant women increasing the risk of hypertensive disorders and abnormal placental functioning. Lead also disrupts the hypothalamic-pituitary-gonadal axis, causing hormonal imbalances that can adversely affect pregnancy outcomes. 18
Experimental studies have associated lead exposure in male animal species with outcomes such as reduced implantation rate, alterations/sperm cell damage, suppression of pubertal growth rate, decrease in serum testosterone levels, neurobehavioral changes, and hepatic dysfunction in offsprings.19 -23 These adverse outcomes are linked to lead’s effect on the hypothalamic-pituitary-gonadal axis causing impaired spermatogenesis and lead’s transfer through seminal fluid, also the genetic consequences of sperm DNA fragmentation. 24 Epigenetic changes in sperm DNA induced by lead exposure can alter gene expression patterns in the developing fetus. 16 Additionally, report from some studies indicate that there is a positive correlation between paternal and maternal blood lead level.25,26 Men who work in lead-exposed environments may inadvertently carry lead dust on their clothing or personal items, bringing it into their homes. This “take-home” exposure can indirectly expose their partners to lead increasing the risk of adverse pregnancy outcomes.
Paternal lead exposure commonly occurs through occupational sources such as construction, battery manufacturing, ammunition manufacturing and handling (including military personnel), glass and ceramics production, steelwork and welding, firefighting, nuclear medicine (where exposure to lead-containing radiation shielding and equipment occurs), heavy machinery handling, artisanal mining, smelting, e-waste recycling, renewable energy technology, and 3D printing and additive manufacturing.8,11,14,27 -31 Inhalation, ingestion, and dermal contact with lead-contaminated air, food, water, or occupational materials are potential exposure pathways. 11 These occupational risks, though focused on paternal exposures, do not exclusively affect men, as most jobs in these sectors employ both men and women. Therefore, similar occupational lead exposure risks can also be considered for female workers. While the harmful effects of maternal lead exposure on pregnancy outcomes are well-documented,32 -34 the impact of paternal lead exposure has received comparatively less attention. Although some human studies have explored the relationship between paternal lead exposure and pregnancy outcomes, the findings are inconsistent.5,35 -37
Therefore, this review aims to systematically and through meta-analysis evaluate and synthesize existing evidences on the impact of paternal lead exposure on pregnancy outcomes, examining associations so as to inform future research and public health strategies aimed at mitigating adverse pregnancy outcomes associated with paternal lead exposure.
Materials and Methods
This review protocol was registered with the International Register of Ongoing Systematic Review (PROSPERO) database and the registration number
Search Strategy
Relevant studies were identified by comprehensive search of 3 electronic data bases; PubMed, Google Scholar and Scopus from inception to August 2024 using a combination of key terms linked with and Boolean operators, parenthesis, and quotation marks. These subject headings and Boolean operators were used: (male OR paternal OR father OR men OR man OR semen OR sperm OR testes OR testicular) AND (“maternal outcome” OR “pregnancy outcome” OR Preeclampsia OR Pre-eclampsia OR eclampsia OR “gestational hypertension” OR “gestational diabetes” OR GDM OR perinat* OR miscarriage OR “still birth” OR still-birth OR stillbirth OR “still born” OR “still-born” OR obstetric OR intrauterine OR pregnancy OR preterm OR congenital OR abortion OR birth* OR prenatal OR postnatal OR fetal OR placent* OR congenital OR infant OR newborn OR baby OR gestation* OR embryo* OR “live birth” OR “maternal mortality” OR child OR “maternal death” OR “birth defect” OR “preterm birth”) AND ((Pb AND metal) OR Pb2+ OR “lead metal” OR “lead Pb” OR “heavy metal” OR “lead exposure” OR “exposure to lead” OR “Lead level” OR “Pb exposure” OR lead[ti] ; Supplemental Data). The reference lists of included studies were also searched for additional relevant articles.
Eligibility Criteria
The eligibility criteria were developed based on PECOS framework; Population, Exposure, Comparator, Outcome, and Study designs of interest as stated below.
Population: The population in the studies should include fathers/men.
Exposure: Studies must have assessed exposure to lead (Pb) metal.
Comparator: The studies should compare pregnancy outcomes among fathers with varying levels of lead exposure.
Outcomes: The selected outcomes were based on clinical importance and availability in published studies which included; preterm birth, congenital birth defects, spontaneous abortion, still birth, low birth weight.
Study Design: observational studies; Cohort, case-control and cross-sectional studies published in peer reviewed journals. The included papers were those written in English language
Exclusion Criteria
Review articles, conference papers, case studies and editorials were not considered to ensure reliability of evidence. We also excluded invitro/animal studies and papers whose full text was unavailable or retracted
Study Selection Process
Two independent authors screened the retrieved reports in conformity with the inclusion criteria and went ahead to retrieve and review full-text copies of the studies for eligibility. Discrepancies on selected studies were resolved with dialog and consensus.
Data Extraction Process
Two independent authors extracted relevant data from each paper using Microsoft excel spread sheet, the extracted data included; authors/year of publication, country of study, study design, sample size, method for determination of lead exposure and findings.
Study Quality Assessment
The quality of the studies included in this review was evaluated using the New-Castle Ottawa Quality Assessment Scale (NOS) for case-control study, cohort study 39 and a modified version for cross-sectional study. The NOS contains 3 main categories, selection, comparability, and ascertainment of exposure/outcome. A 9-star rating system was applied for the quality assessment of the cohort and case-control studies. For the cross-sectional studies, the scores rated from 0 to 10. Scores between 7 and 9 indicated high quality, 4 to 6 was considered moderate quality and less than 3 was defined as low quality.
Data Synthesis and Meta-Analysis
Pooled OR Computation
Studies selected for the meta-analysis were those who’s actual or estimate blood Pb-exposure level is up to 15 μg/dL or those whose group categories included a BBL of 15 μg/dL or more. Meta-analysis was only performed for pregnancy outcomes that were reported in more than one of the eligible studies. The meta-analysis was performed with Odds Ratio (OR) values (and its associated 95% CI), as such relevant studies that reported a Risk Ratio (RR) value were converted to their OR equivalent using the equation (1) below as reported by Zhang and Yu. 40 This conversion was performed on the RR and their associated confidence intervals.
where P0 is the baseline risk (ie, the probably of the outcome of interest in the unexposed group
Studies that reported OR and RR values for multiple exposure groups were pooled internally by meta-analysis to obtain a representative estimate that was used for the main meta-analysis. For the meta-analysis, the OR and confidence interval were initially log-transformed in order to obtain the standard error of the treatment estimate (accomplished by dividing the difference between the log-transformed upper limit and log-transformed lower limit by 3.92). 41 The resultant value was subsequently applied in computing the pooled OR estimate using the metagen function from “meta” package 42 of R studio statistical software (version 3.6.1). 43 I 2 values were computed and used to assess the heterogeneity of the pooled estimate. A maximum-likelihood random effect model (DerSimonian-Laird Method) was assumed to minimize the influence of inter-study variation on the pooled OR estimate. 44
Results
Study Selection Process
The systematic review identified a total of 10 882 records from 3 databases: PubMed (n = 5549), Google Scholar (n = 240), and Scopus (n = 5093). After removing 5062 duplicate records, 5820 records were screened for relevance based on titles and abstracts. A total of 5795 records were excluded due to irrelevance to the study question, leaving 25 reports sought for retrieval. Of these, 2 reports could not be retrieved. After assessing the remaining 23 full-text articles for eligibility, 12 were excluded for the following reasons: Experimental/animal studies (n = 3).19,45,46 Outcomes not related to pregnancy (n = 5).47 -51 Others include a review paper, a case report, a conference paper and a paper that did not specifically investigate lead exposure among the heavy metals examined.52 -54
Ultimately, 11 studies met the inclusion criteria and were included in the systematic review, out of the 11 studies, only 7 were included in the meta-analysis. The PRISMA flow diagram outlining this process is presented in Figure 1.

PRISMA flow diagram.
Characteristics of Included Studies
The Characteristics of the included studies are summarized in Table 1. Studies were from North America, 3 from Europe, and 3 from Asia. Most studies were conducted in occupational settings where paternal lead exposure was evaluated using biological markers (eg, blood lead levels [BLL]) or job exposure histories. Sample sizes ranged from small cohorts of 54 participants to large registry-based studies of over 6000 participants. Of the 11 studies included in this study, 4 were cohort studies,6,35,55,56 4 case-control studies,5,37,57,58 and 3 cross-sectional study designs.25,26,59
Eligible studies included in the systematic review and meta-analysis.
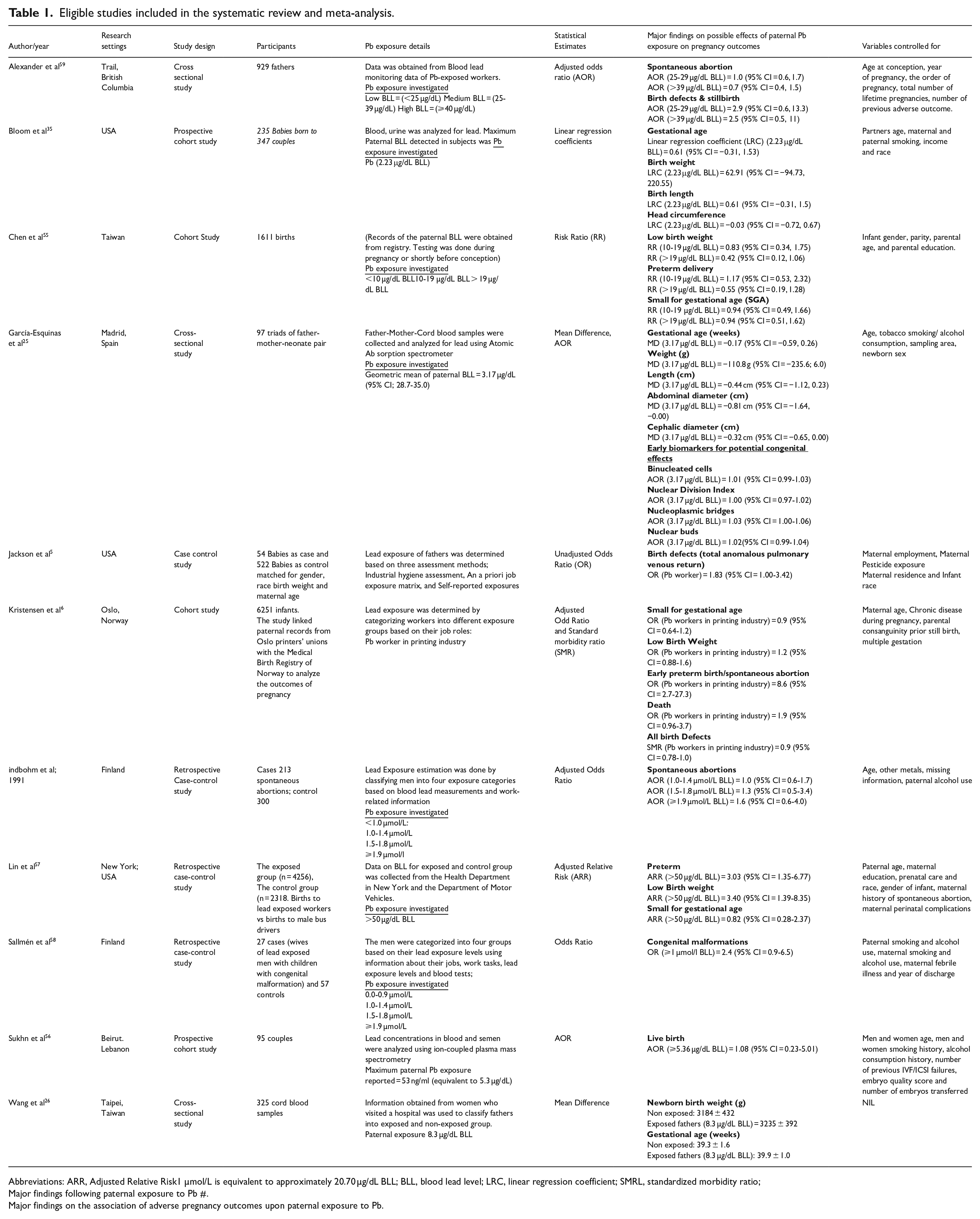
Abbreviations: ARR, Adjusted Relative Risk1 µmol/L is equivalent to approximately 20.70 µg/dL BLL; BLL, blood lead level; LRC, linear regression coefficient; SMRL, standardized morbidity ratio;
Major findings following paternal exposure to Pb #.
Major findings on the association of adverse pregnancy outcomes upon paternal exposure to Pb.
Quality Assessment
Following the assessment of the study quality by 2 independent reviewers using the Newcastle Ottawa scale, 9(82%) of the 11 included studies were classified as high-quality studies, 1(9%) was of moderate quality and 1(9%) was of low quality. Details of the Newcastle-Ottawa gradings are shown in Table 2
Risk of bias assessment (Newcastle-Ottawa Quality Assessment Scale criteria).

*Represents a point awarded for meeting a quality criterion, with a maximum score of 9.
Exposure and Outcome Assessment
Predominantly occupational lead exposure with BLLs categorized by tiers. Each study reported on more than 1 pregnancy outcome. Outcomes reported on includes spontaneous abortion (n = 3), preterm birth (n = 6), low birth weight (LBW; n = 5), small for gestational age (SGA; n = 4), and congenital anomalies (n = 5). See Table 1. The meta-analysis yielded a pooled OR that is mainly influenced by occupational exposure concentrations between 10 and 60 μg/dL.

Forest plots of summary odds ratio (ORs) and 95% confidence intervals (CIs) for the association between paternal lead exposure and spontaneous abortion.
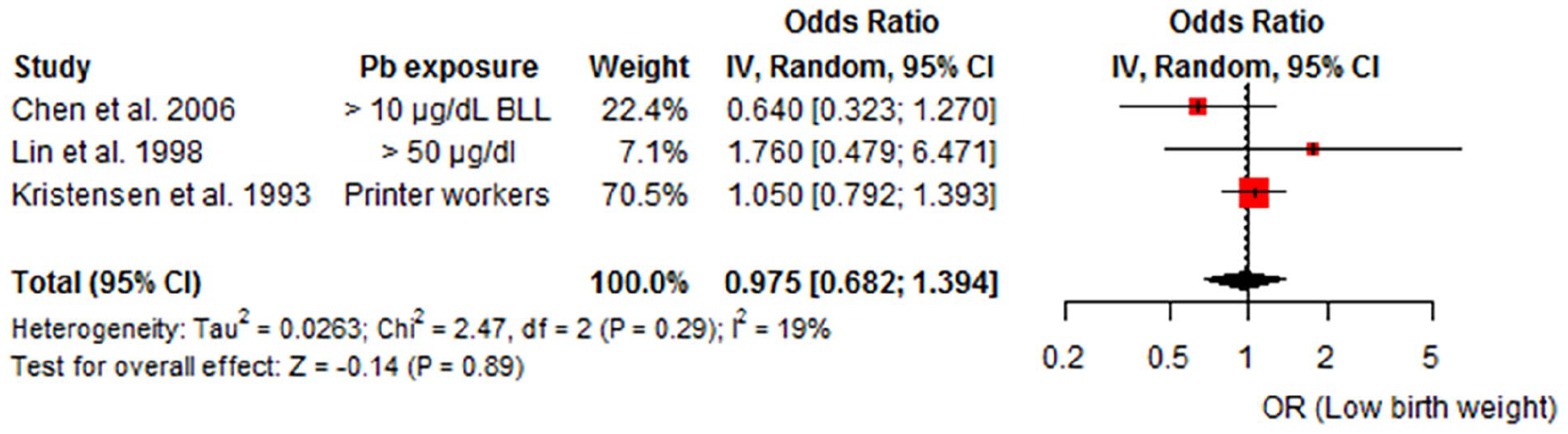
Forest plots of summary odds ratio (ORs) and 95% confidence intervals (CIs) for the association between paternal lead exposure and low birth weight.

Forest plots of summary odds ratio (ORs) and 95% confidence intervals (CIs) for the association between paternal lead exposure and preterm births.

Forest plots of summary odds ratio (ORs) and 95% confidence intervals (CIs) for the association between paternal lead exposure and small for gestational age.

Forest plots of summary odds ratio (ORs) and 95% confidence intervals (CIs) for the association between paternal lead exposure and birth defects.
Discussion
This systematic review and meta-analysis investigated the association between paternal lead exposure and adverse pregnancy outcomes, synthesizing data from 11 studies conducted across North America, Europe, and Asia. Findings from meta-analysis showed that the pooled OR for birth defects (OR = 2.092, 95% CI: 2.092-3.351, P < .01) was statistically significant, suggesting that paternal lead exposure is strongly associated with birth defects/congenital abnormality however, the results for other pregnancy outcomes, which include spontaneous abortion, low birth weight (LBW), preterm birth, and small-for-gestational-age (SGA), were not statistically significant.
The correlation between paternal lead exposure and birth defects signifies a critical pregnancy risk. This finding is consistent with previous studies that suggests that exposure to lead causes sperm DNA damage or epigenetic abnormalities that can lead to impaired fertilization or congenital disorders.60 -64 Conversely, findings from Ali et al 65 found no significant association between paternal work place exposure to lead and congenital malformations. Evidence has shown that Lead causes oxidative damage to sperm’s plasma membrane and DNA by increasing reactive oxygen species (ROS) while depleting the body’s antioxidant defenses.61,66 Lead also disrupts the epigenetic regulation of sperm DNA. It alters patterns of DNA methylation which can interfere with gene expression necessary for proper sperm function and embryonic development.62,64,67 Additionally, Lead exposure has been linked to chromosomal abnormalities and breaks, it also inhibits enzymes involved in DNA repair pathways and causes damage to mitochondrial DNA which compromises sperm integrity and genetic stability. 68 According to La Llave León and Salas Pacheco 69 Lead also impairs spermatogenesis and alters the hypothalamic-pituitary-gonadal (HPG) axis disrupting the release of testosterone and luteinizing hormone which are essential for sperm production and maturation. Findings from research indicate a dose-response relationship between paternal lead exposure and some reproductive outcomes, with higher paternal blood levels associated with an increased risk of adverse outcomes. While some studies highlight complexities and limitations in defining this relationship, the overall body of evidence supports a positive correlation.64,70 Some studies report a threshold effect, beyond which the risk of adverse pregnancy outcomes increases significantly, however this threshold varies across studies. 71 The variability in the sources of paternal lead exposure; (occupational, environmental and lifestyle sources) may have contributed to the challenges in determining a well-defined dose response relationship. Additionally, individual susceptibility to lead toxicity is influenced by nutritional status, genetic predisposition, and epigenetic modifications, which affects lead metabolism and detoxification pathways.71,72 These factors highlight the need for more standardized and extensive longitudinal studies on the impact of paternal lead exposure on pregnancy outcomes.
Other mechanisms of the impact of paternal lead exposure on pregnancy is the indirect maternal transfer of lead through seminal fluid or from household contamination.8,25,26,73,74 Indirect transfer to the mother may contribute to maternal lead burden, potentially leading to hormonal imbalance, irregular menstrual cycles, reduced fertility, and increased risk of miscarriage. 71 During pregnancy, lead can cross the placenta barrier, directly affecting fetal development. 75 It can also interfere with placental function, limiting the transfer of essential nutrients and oxygen to the fetus. 75 This disruption increases the risk for hypertensive disorders of pregnancy (PIH and preeclampsia), which has potentially life-threatening consequences for maternal and fetal health.76 -80 Additionally, maternal lead exposure has been associated with a higher risk of preterm birth and low birth weight,32,81 both of which can contribute to neonatal health challenges and in severe cases, congenital abnormalities and stillbirth.82,83
Findings from the meta-analysis of the other pregnancy outcomes; spontaneous abortion, low birth weight, preterm birth, and small for gestational age suggests that contrary to some previous studies, paternal lead exposure may not be a significant risk factor for these adverse outcomes. Specifically, an epidemiological review by Anttila and Sallmén 52 indicated that paternal exposure to lead or mercury might be associated with the risk of spontaneous abortion, similarly Bellinger 70 in his review article also reported a possible association between paternal lead exposure <30 μg/dL and spontaneous abortion although significant limitations in the quality of the underlying data was acknowledged. Furthermore, Bellinger 70 also stated that increased paternal exposure to lead (blood lead level (BLL) >10% of the Threshold Limit Value (TLV) or >25 μg/dL for at least 5 years) is a risk factor for preterm and low birth weight, while Min et al 84 added that the odds of low birth were significantly higher with a fivefold increase among infants of fathers who were exposed to high levels of lead exposure during the 6 months before conception through the duration of the pregnancy. The inconsistencies in these findings highlights the need for further research to explore the potential mechanisms involved and clarify the effect of cofounding factors that may contribute to pregnancy complications.Differences in study design, population characteristics, exposure assessments, and methodologies contributed to the observed heterogeneity across the included studies. Heterogeneity was however low or absent for some outcomes, including low birth weight (I2 = 19%), small-for-gestational-age infants (I2 = 0%), and birth defects (I2 = 0%), indicating consistent findings among the studies for these outcomes. Moderate heterogeneity was observed for spontaneous abortion (I2 = 39%), and preterm birth demonstrated slightly higher variability (I2 = 52%).The overall quality of included studies was high, with 9 of 11 studies rated as high quality based on the Newcastle-Ottawa Scale. This strengthens confidence in the reliability of the findings. However, the single low-quality study might be a source of bias in the pooled estimate. The major strength of this study is that, to the best of our knowledge the study represents the first systematic review/meta-analysis of the extant literatures/studies on paternal exposure to lead and a range of pregnancy and birth outcomes. Majority of the studies included in this review were high quality studies that encompassed diverse population and controlled for important cofounders. Additionally, the low heterogeneity in most analysis increases the reliability of the pooled results. However, there are some limitations to the study, the relatively small number of studies pooled for the outcomes may have limited statistical power. Secondly, some of the studies estimated paternal lead exposure level by occupational histories/settings and not by actual blood testing. Additionally, majority of the studies were conducted in the 1990s and 2000s, with fewer recent studies assessing the impact of lead exposure on pregnancy outcomes. These limitations may introduce potential bias and limit the generalizability of findings.
Implications for Public Health and Research
The significant association between lead exposure and birth defects underscores the urgent need for stricter occupational and environmental regulations to minimize lead exposure, particularly among reproductive-age individuals. Comprehensive preconception health programs should involve conducting screenings and assessing the risk of lead exposure in both men and women. Furthermore, educational campaigns targeting communities in industrial or mining regions will increase awareness on the harmful effects of lead exposures and promote measures to minimize exposures. Given the nonsignificant findings for other pregnancy outcomes, further research will be necessary to elucidate the impact of lead exposure on pregnancy outcomes, long term, large cohort studies involving diverse populations that includes participants in low- and middle-income countries where lead exposure is more prevalent is crucial to informing tailored interventions.
Conclusion
This study evaluated the association between paternal lead exposure and adverse pregnancy outcomes with focus on birth defects (congenital abnormalities), spontaneous abortion, low birth weight, preterm births and small for gestational age. While there is a potential association between paternal lead exposure and birth defects, the results for other pregnancy outcomes showed no significant association. Enhanced awareness and appropriate public health interventions is essential to mitigating the effects of environmental lead exposure particularly in low- and middle-income countries with limited awareness and lack of strict environmental regulations. There were variations in methodology and quality of the included studies which may have influenced the results and highlights the need for further large cohort investigations with robust and well-designed methodologies.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302251327535 – Supplemental material for Paternal Lead Exposure and Pregnancy Outcomes: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ehi-10.1177_11786302251327535 for Paternal Lead Exposure and Pregnancy Outcomes: A Systematic Review and Meta-Analysis by Kenechi A. Aliche, Francis U. Umeoguaju, Catherine Ikewuchi, Faith C. Diorgu, Opeyemi Ajao, Chiara Frazzoli and Orish E. Orisakwe in Environmental Health Insights
Footnotes
Abbreviations
UNICEF: United Nations Children Fund
WHO: World Health Organization
CDC: Center for Disease Control and Prevention
SDG: Sustainable Development Goals
DNA: Deoxyribonucleic Acid
ROS: Reactive Oxygen Species
PRISMA: Preferred Reporting Items for Systematic Reviews
PECOS: Population, Exposure, Comparator, Outcome, and Study design
Pb: symbol for Lead metal
NOS: Newcastle-Ottawa Scale
OR: Odds Ratio
RR: Relative Risk
CI: Confidence Interval
SGA: Small for Gestational Age
BLL: Blood Lead Level
LBW: Low Birth Weight
HPG: Hypothalamic-Pituitary-Gonadal axis
TLV: Threshold Limit Value
PIH: Pregnancy Induced Hypertension
Statements and Declarations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.