Abstract
Background:
Foodborne diseases pose a significant public health threat, particularly in regions with poor sanitation and food handling practices. These diseases, mainly caused by microbiological hazards like bacteria, fungi, and parasites, affect millions globally. Despite the global burden, the true extent of these hazards remains underestimated, especially in low- and middle-income countries like Ghana. This study aimed to map the available literature on foodborne microbiological hazards in Ghana, providing an overview of the evidence and identifying areas where further research is needed.
Method:
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis Extension for Scoping Reviews. A detailed search was done in PubMed, Scopus, Web of Science, and Google Scholar, and articles were exported to Rayyan for screening. A three-phase screening process was used to identify relevant articles. Data from the included articles were extracted and analysed, with specific information related to food type, specific hazards, sample population, and hazard groups summarised using proportions and tables.
Results:
This review included 72 studies which were published between 2001 and 2023. Eighty-five percent of these studies (85%) reported on bacterial hazards, while 19%, 11%, and 6% reported on fungi, parasites, and mycotoxins, respectively. The most reported bacterial, fungal, and parasitic hazards were Escherichia coli, Aspergillus spp. and Trichuris trichiura, respectively. Aflatoxins were reported in maize, groundnut, and spices, with prevalence ranging from 61% to 100% and at levels exceeding standards set by Ghana Standards Authority and European Food Safety Authority.
Conclusion:
This review highlighted the spectrum of microbiological hazards in foods in Ghana. The hazards identified pose significant public health risks, particularly among vulnerable populations. It is crucial that stricter enforcement of food safety laws and improved food handling practices are implemented in the country, particularly in the informal food sector, to protect consumers.
Introduction
Foodborne diseases, which result from foodborne hazards, pose a significant public health threat, particularly in regions where high prevalence of sanitation lapses and poor food handling practices exist.1,2 These foodborne hazards, typically categorised as biological, chemical, and physical, are extensively distributed globally, and dictate the spectrum of foodborne ailments and conditions. 3 Biological hazards, which include pathogenic organisms (spanning across viruses, bacteria, parasites, and fungi) and their associated toxins, constitute the predominantly implicated hazard in about 97% of foodborne illness outbreaks and cases worldwide.4,5
Globally, millions of foodborne disease cases occur every year, with reports indicating that the burden is comparable to those of major infectious diseases like HIV/AIDS, malaria, and tuberculosis.4,6 According to a recent global estimate by the World Health Organization (WHO), 600 million cases of foodborne illnesses caused by over 30 foodborne hazards are reported each year, resulting in approximately 420,000 deaths.4,7 Notably, Africa bears the highest burden of these foodborne diseases with an estimated 1200-1300 disability-adjusted life years (DALYs) per 100,000 people compared to 35–71 DALYs in other regions. 8 However, the true extent of the health burden associated with foodborne hazards and diseases (such as morbidity and mortality) remains largely unknown, especially in low- and middle-income countries where the burden is frequently underestimated. 8
In Ghana, there is an immense public health risk posed by foodborne diseases. A report by the country’s Ministry of Food and Agriculture and the World Bank noted that on an annual basis, approximately one (1) in every forty (40) individuals (an estimated 420,000 persons) suffers from foodborne illnesses, approximately 65,000 of whom lose their lives. 9 Despite the presence of regulatory bodies in Ghana, there appears to be a problem with adherence to food safety standards within the food industry, particularly in the informal sector, and hence the high risk posed by foodborne hazards. 10
It is crucial to gather and consolidate information on foodborne hazards within a country and identify specific foods that are mostly contaminated by these hazards in order to effectively prioritise food safety policies and allocate resources to areas with the greatest food safety risk. 11 Such studies have been conducted in a number of African countries to aggregate information on microbial contamination in food, enabling evidence-based policies and decision making by various stakeholders.12 -16 In Ghana, previous reviews on the microbial safety of food are limited in scope and are now outdated. Notably, the review conducted by Saba & Gonzalez-Zorn 17 over a decade ago reported on a limited number of microbiological hazards, failing to account for fungi, parasites, and other clinically-significant foodborne bacterial pathogens, such as Listeria monocytogenes, Clostridium perfringens, and Escherichia coli O157:H7. This review provided findings based on a limited number of studies (11 articles), 17 resulting in a restricted perspective of the evolving landscape of food safety research in Ghana. Additionally, the review by Yeleliere et al. 18 was narrative and focused on select cities, and thus, may not provide a comprehensive overview of the food safety situation in the country. Besides, there has been a significant increase in research outputs on the theme over the years. Synthesising the wealth of contemporary knowledge on this topic is, therefore, warranted to yield a more accurate and up-to-date understanding of the microbiological hazards present in foods in Ghana. The objective of this study was to systematically map the available literature on foodborne microbiological hazards in Ghana, providing an overview of the evidence and also identifying areas where further research is needed.
Methodology
This review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR). 19 The primary focus of this review was on peer reviewed-literature; hence no gray literature sources were searched.
Study design and search strategy
Original articles were sourced from different databases, namely PubMed, Web of Science, Scopus, and Google Scholar, to reduce biases.20,21 The literature search for this study was conducted between December 7 and 11, 2023, and March 1 and 5, 2024. The search terms, as presented in Table 1, were used to search the databases and the results were exported into Rayyan for screening. 22 For Google Scholar, only the first 400 results were collected. The search was specifically limited to peer-reviewed articles published in the English language by December 2023. A total of 1708 results were retrieved for screening.
Databases, search terms, and results retrieved.

Eligibility criteria
Original articles were required to meet at least one of these requirements in order to be included in the study: (i) reported specific microbiological hazards in foods in Ghana and (ii) reported on the prevalence and/or microbial load of these hazards in foods in Ghana. Studies that exclusively investigated microbiological hazards that are unrelated to foodborne diseases were excluded from the analysis. Those that reported microbiological hazards solely in non-food items, such as animal faeces, packaging materials, and food processors, were also excluded.
Study selection
A three-phase screening process was used to retrieve articles of interest for the review. In the first screening phase, duplicates were removed manually using the Rayyan Systematic Review (RSR) platform. 22 Titles and abstracts of the retained articles were screened and assessed for eligibility using the RSR platform in the second screening phase. In the final screening phase, the full-text articles were thoroughly read to include relevant articles based on the objectives of the study. A set of criteria was employed to evaluate the quality of full-text articles, encompassing the utilisation of scientifically rigorous methods, appropriate laboratory procedures, and precise reporting of results. Figure 1 below shows the process of article selection.
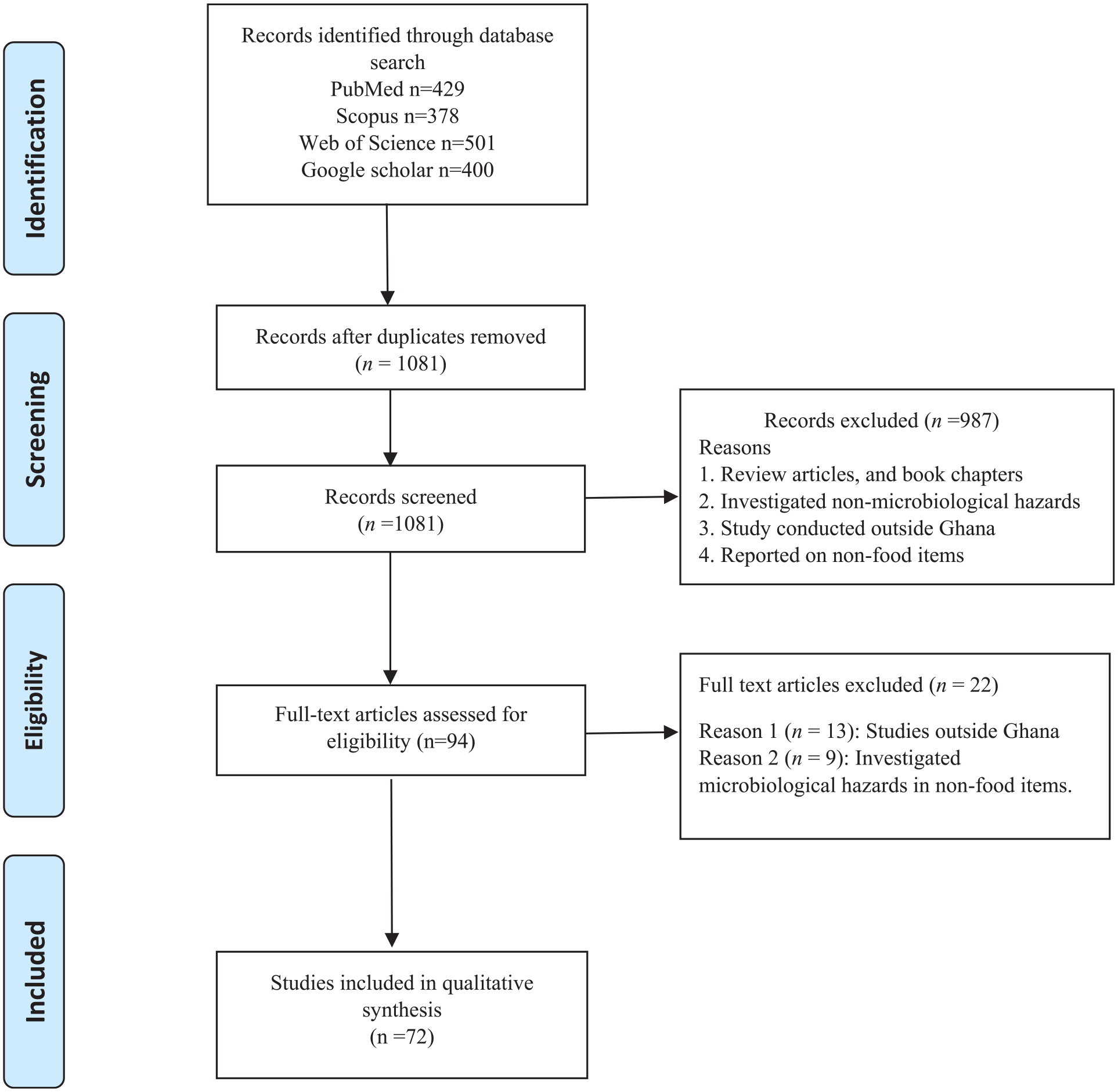
PRISMA flow diagram providing a visual representation of the article selection process of this scoping review.
Data extraction and analysis
Data from each included article was extracted and recorded in an Excel spreadsheet, categorised by author(s), year of publication, study sites, sampling points, food type, specific microbiological hazards, and hazard groups. The extracted data were then analysed and summarised as proportions. A narrative description of the included articles was presented, along with an analysis of the number and geographic distribution of the articles. The results and conclusions obtained were thematically combined and presented in tables and figures, to provide a visual representation of the findings.
Results
Following the completion of all screening processes, a total of 72 articles were selected for inclusion in this scoping review (Table 2).
Included studies in the systematic review of foodborne hazards in Ghana with study sites, sampling points, sample types, food hazards, and hazard group.

Ayoyo—leafy green vegetable (Jute mallow)
Fufu—Pounded boiled cassava, yam and plantain
Hausa koko—Millet pudding
Waakye—Boiled rice and beans
Tuo-zaafi—Stiff porridge made from maize or millet flour.
Solom—Millet beverage
Khebab—Skewered and grilled meat; typically made from lamb, beef or chicken.
Nunu—Fermented yogurt-like beverage
Brukina/Burchina—Local beverage made from ground millet and pasteurised milk.
Wagashie/wagashi/Waagashie—Fermented Curd cheese.
Characteristics of the studies
This review comprised a total of 72 articles of which 82% (59/72) were conducted between 2012 and 2023, and the remaining 18% (13/72) were conducted between 2001 and 2011. In terms of study locations, the majority of the studies (44%, 32/72) were conducted in Accra, which is the capital city of Ghana. The remaining studies were conducted in Kumasi (17%, 12/72), Tamale (15%, 11/72), Bolgatanga (6%, 4/72), Cape Coast (6%, 4/72), Koforidua (3%, 2/72), Ho (3%, 2/72), and Yeji (1%, 1/72). Four (4) studies, representing 6%, were conducted across several regions in Ghana. Most of the studies (85%, 61/72) reported on bacterial hazards; 19% (14/72) reported on fungi; 11% (8/72) investigated parasites. Four articles (6%, 4/72) reported on mycotoxins, specifically aflatoxin and fumonisins. It is worth noting that the total count of articles reporting on bacteria, fungi, parasites, and mycotoxin exceeded the number of articles included in the review as certain articles addressed multiple hazards concurrently.
Studies included in this review assessed microbiological hazards in different food samples (Table 2), including vegetable and fruit samples (carrot, onion, tomato, roselle leaves, jute leaves, African spinach, African eggplant, cabbage, cucumber, lettuce, green bell pepper, pawpaw, mango, sugarcane, pineapple, and watermelon), cereal and legumes and their products (maize, boiled rice, “solom”—a local millet beverage, tiger nuts, and groundnut paste), dairy products (raw milk, boiled milk, yogurt, “nunu”—a local milk drink, and “woagashie”—a local cheese made from cow’s milk), meat (beef, goat meat, and grilled beef sausage), chicken, raw and smoked fish (Sardinella aurita), spices (cloves, negro pepper, cumin, calabash nutmeg), “fufu”—a local delicacy prepared from boiled cassava, yam, and/or plantain, “tuo-zaafi”—a local staple prepared from maize or millet flour, and hibiscus tea (“bissap/sobolo”).
Foodborne microbiological hazards
Bacteria
Of the studies reviewed, 85% (61/72) reported on bacterial hazards in various foods.23 -83 A total of 31 different genera of bacteria were isolated from both ready-to-eat (RTE) foods and raw foods, as shown in Figure 2. The most common bacterium reported was Escherichia coli (46/72, 64%). It was most detected in RTE vegetable salads, cabbage, lettuce, raw beef, raw chicken, raw goat meat, “Hausa koko”, “khebab,” as well as fresh and smoked fishes, with individual prevalence ranging from 2.1% to 100%.23 -41,44,46,48,50 -55,57,60-62,64 -67,69,74,75,77 -83 The highest prevalence (100%) was recorded in lettuce and cabbage samples from a vegetable farm in Kumasi. 26 Staphylococcus spp. was the second most isolated bacterium, reported by 44% (32/72) of the studies23,25,27 -29,31,33,35 -39,41,44,48,49,52,54,55,57,62,65 -67,70,71,74,75,79 -82; 19 of these 32 studies specifically reported on Staphylococcus aureus, with individual prevalence ranging from 4% to 97%.23,25,27 -29,31,33,41,48,57,65 -67,70,71,74,75,79,82 It was most detected in raw beef, chicken, and grilled beef sausage. The highest prevalence (97%) was recorded in “tuo-zaafi” sampled from Tamale. 25 Salmonella spp. was reported by 32% (23/72)23 -25,28,29,31,35,36,38,44,50,51,53,58,60,66,74,76,78,80 -83 from foods such as raw beef, chicken, chevon, salad, mixed spices, and locally produced fruit juices. The prevalence of Salmonella spp. ranged from 1% to 73%. The highest prevalence was recorded in RTE vegetable salad. Klebsiella spp. was reported by 28% (20/72) of the studies included,27,31,33,34,40,44,49,50,54,55,57,63,65,68,70,71,75,80 -82 with prevalence ranging from 0.4% 50 to 63%. 34 Most commonly isolated species were Klebsiella pneumoniae and Klebsiella oxytoca. Bacillus spp. was reported by 24% (17/72) of the studies.24,28,31,33,37,38,55,57,61,62,65 -68,73,75,79 It was most common in vegetable salad and raw beef, with individual prevalence ranging from 10% to 93%. Also, Enterobacter spp. was reported by 22% (16/72) of the studies,31,33,34,40,49,50,54,55,57,61,63,67,68,70,71,80 with prevalence ranging from 1% to 71%. Proteus spp. was also reported by 19% (14/72) of the studies, with prevalence ranging from 1% to 38%.27,33,34,37,38,40,49,50,54,55,62,68,70,71 Proteus mirabilis and Proteus vulgaris were the most isolated Proteus species.

Percentage frequency of bacterial isolates found in all food samples in this review.
Other bacteria isolated were Streptococcus spp. (17%, 12/72), with prevalence between 3% and 11%,28,33,35 -38,40,44,46,65,67,81 Pseudomonas spp. (17%, 12/72), with prevalence between 1% and 14%,27,31,33,37,38,50,54,57,65,67,68,70 Shigella spp. (14%, 10/72), with prevalence between 1% and 77%,23 -25,28,40,50,53,68,78,83 Enterococcus spp. (13%, 9/72), with prevalence between 6% and 16%,33,35,40,43,53,54,56,67,76 Citrobacter spp. (11%, 8/72), with prevalence between 0% and 6%,33,50,54,57,63,68,70,71 and Aeromonas spp. (7%, 5/72), with prevalence between 2% and 21%.27,31,50,54,57
Also recorded in this study were Acinetobacter spp. (6%, 4/72),50,54,63,71 Serratia spp. (6%, 4/72),34,50,54,80 Clostridium spp. (4%, 3/72), Yersinia spp. (4%, 3/72),31,50,55 Vibrio spp. (3%, 2/72),50,54 and Campylobacter spp. (3%, 2/72).47,78 Erwinia spp. 34 Micrococcus spp., 62 Lactobacillus spp., Lactococcus spp., Leuconostoc spp., 40 Corynebacterium, 46 and Mycobacterium spp. 55 were reported by one study each. Rahnella spp., Providencia spp., Kluyvera spp., Pasteurella spp., and Stenotrophomonas spp. were all reported by Baah et al. 50
Fungi
In all, 14/72 articles reported on various fungal species, as detailed in Figure 3. These fungi were mostly isolated from RTE vegetable salads, smoked fishes (tuna/mackerel) “solom,” “fufu,” hibiscus tea, “nunu,” raw egg, beef, and chicken.28,34,40,46,61,62,65,66,69,80 -82,84,85 The most isolated fungal species was Aspergillus spp. (14%, 10/72),28,34,46,62,65,66,80 -82,85 followed by Penicillium spp. (10%, 7/22),28,34,66,80 -82,85 and Fusarium spp. (8%,6/72).62,66,80,81,84,85 Other fungal species identified were Rhizopus spp. (7%, 5/72),34,62,66,82,85 Rhodotorula spp. (7%, 5/72),28,66,80,81,85 Mucor spp. (7%, 5/72),37,46,62,66,85 Candida spp. (4%, 3/72),40,80,81 Cladosporium spp. (4%, 3/72),82,84,85 Saccharomyces spp. (3%, 2/72),40,62 and Eurotium spp. (3%, 2/72).62,84 Carvulania spp. 28 Geotrichum spp., Paecilomyces spp., 34 Zygosaccharomyces spp., and Yarrowia spp. 40 were each reported by one study.

Percentage frequency of fungal isolates found in all food samples in this review.
Fungal counts reported by Aboagye et al. 28 for “sobolo” and “asaanaa” ranged between 2.29–4.86 log10 CFU/ml and 2.098–4.23 log10 CFU/ml, respectively. Toxigenic fungal species, including those from the Aspergillus, Fusarium, and Penicillium genera, were isolated from these local drinks. 28 The study by Boampong et al. 53 , recorded a high fungal count of 3.73 log CFU/g in watermelon. The count of fungal colonies obtained from vegetable sauce, fried chicken, mixed salad, fried rice, and goat light soup, investigated by Darko et al. 84 exceeded the acceptable levels set by the WHO. These food samples reported fungal loads between 2.2 log CFU/g and 4.0 log CFU/g, indicating that these foods could potentially transmit these fungi to unsuspecting consumers. 84 Also, Kortei et al. 85 reported the fungal counts in “solom” (a millet beverage). The counts observed ranged from 1.68 ± 0.8 to 4.11 ± 0.9 log10 CFU/ml. The study by Nkekesi et al. 66 reported fungal contamination in street-vended grilled beef sausage in Ho. The total fungal counts varied from 0.0 to 9.83 × 103 CFU/g. The species identified in the samples included Aspergillus spp. and Rhizopus spp.
Parasites
Of the studies included in this review, 11% (8/72) reported on the identification of various parasites as contaminants in various food samples, as illustrated in Figure 4.49,53,56,86,87,88,89,90 These parasites were isolated from vegetable salads, tiger nuts, fresh vegetables such as cabbage, lettuce, carrot, tomato, cucumber, green pepper, green onions, and fresh cut fruits such as pawpaw and pineapple. The most isolated parasite was Trichuris trichiura (8%, 6/72)53,56,86,87,88,89; detected mostly in lettuce, cabbage, and green onions.87,88,89

Percentage frequency of parasitic organisms found in all food samples in this review.
Other parasites isolated include Cryptosporidium spp. (7%, 5/72),49,53,56,89,90 Giardia lamblia (7%, 5/72),53,56,86,87,89 Entamoeba histolytica (7%, 5/72),53,56,86,87,89 Entamoeba coli (7%, 5/72),53,56,86,88,89 hookworm (7%, 5/72),53,56,87-89 Cyclospora cayetanensis (6%, 4/72),49,53,56,89 Ascaris spp. (6%, 4/72),53,56,87,88 Isospora belli (6%, 4/72),53,56,87,89 Enterobius vermicularis (4%, 3/72),53,56,87,89 Stronglyoides stercoralis (3%, 2/72),87,88 Taenia spp. (1%, 1/72), 87 Moniezia spp. (1%, 1/72), 86 Ancylostoma duodenale, 49 Faciolopsis buski, 89 Balantidium coli, and Fasciola spp. 90 The highest contamination rates were detected on vegetables such as spring onion (97%), 90 lettuce (76.2%) 88 , and cabbage (66.7%). 88
Mycotoxins
Among the 72 studies analysed, four (6%, 4/72) investigated the presence of mycotoxins in various food samples.91 -94 These mycotoxins were detected in maize, groundnut paste, and spices. Aflatoxins and fumonisin were both detected in maize samples investigated by Danso et al. 91 from the Ashanti Region. The mean levels of aflatoxin detected on-field in the study (7.2–14.2 ppb) were all below the limit of 15 ppb set by the Ghana Standards Authority (GSA). 91 However, the levels detected in heaping and post-drying stages (16.6–24.9 ppb) were above the threshold. Out of 153 individual samples tested for aflatoxin, 29 samples were above the threshold. 91 Fumonisin levels detected ranged between 0.7 and 1.9 ppm, well below the threshold. 91
Out of a total of 180 maize samples tested by Kortei et al. 92 , 131 tested positive for aflatoxin and 103 tested positive for ochratoxins. Out of the 180 samples analysed for total aflatoxins, 127 (70.50%) exceeded the limit set by the European Food Safety Authority (EFSA). The levels of aflatoxin in these samples ranged from 4.27 to 441.02 µg/kg. Similarly, 116 samples (64.44%) surpassed the limit set by the GSA, with concentrations ranging from 10.18 to 441.02 µg/kg. 92 Regarding ochratoxin A (OTA), 94 samples (52.22%) exceeded the tolerable limit established by the EFSA. The OTA concentrations in these samples ranged from 4.00 to 97.51 µg/kg. Additionally, 89 samples (49.44%) surpassed the limit set by the GSA, with OTA levels ranging from 3.30 to 97.51 µg/kg. 92
Kortei et al. 93 investigated the presence of aflatoxins in groundnut and groundnut paste. The study revealed that out of the 80 samples examined, 49 (61.25%) tested positive for Aflatoxin B1, with concentrations ranging from 0.38 ± 0.04 µg/kg to 230.21 ± 22.14 µg/kg. The same proportion of samples tested positive for total aflatoxins, with levels ranging from 0.38 ± 0.02 µg/kg to 270.51 ± 23.14 µg/kg. The limits set by the GSA and the EFSA were used as benchmarks for AFB1 and total aflatoxins (AF total) (5 and 10 µg/kg for GSA, and 2 and 4 µg/kg for EFSA). A total of 33 samples (41.25%) exceeded the limits for both AFB1 and total aflatoxins. 93
Discussion
The findings of this review present an encouraging trend of research activities related to microbial food safety in Ghana. The majority of the studies (82%, 59 out of 72) were conducted between 2012 and 2023, while 18% (13 out of 72) were carried out between 2001 and 2011. This indicates an upward trajectory in microbial food safety research in Ghana. This contradicts earlier reports by Saba & Gonzalez-Zorn, 17 which had characterised microbial food safety research in Ghana as “abysmal.” Several of these studies were carried out in the Greater Accra, Ashanti, and Northern Regions, suggesting a limited geographical scope. Approximately, 78% of the total studies were conducted in these regions, with a particular emphasis on their capital cities (Accra, Kumasi, and Tamale). This finding aligns with reports by Botha et al. 95 and Saba & Gonzalez-Zorn 17 , which identified these regions as having a high number of food safety research activities in Ghana compared to the other regions in the country. This could be attributed to a higher occurrence of food contamination in these regions. 95 Paudyal et al. 16 highlighted that food safety studies in Africa often exhibit a limited capacity, focusing on specific geographic areas within the countries under investigation, similar to the observation in this study.
The review reported various microbiological hazards, with the majority (85%) of the included studies reporting on bacterial hazards. Among these hazards, the most frequently isolated bacterial pathogens were Escherichia coli, Staphylococcus spp., Salmonella spp., and Klebsiella spp. These findings align with those of a similar review conducted by Makinde et al. 96 in 2020, which investigated the microbiological safety of RTE foods in low and middle-income countries. Specifically, E. coli, Klebsiella spp., and Salmonella spp. were the most commonly reported bacterial pathogens in that study. Additionally, findings from the meta-analysis of Paudyal et al. 16 that focused on selected African countries also identified Escherichia coli, Staphylococcus spp., and Salmonella spp. as the most studied pathogens in food. The presence of these pathogens in food poses a potential risk, rendering it unsafe for human consumption and posing a clear threat to consumer health.16,96 These pathogens are responsible for diarrhoea, particularly among children under five years of age. 4 A report by Osei-Tutu & Anto 97 identified cholera, typhoid fever, shigellosis, and viral hepatitis as the four most clinically diagnosed foodborne diseases reported at a hospital in Accra, Ghana. All the pathogens responsible for these diseases were identified in foods in this study, except viral hepatitis.
In this review, Escherichia coli was found to be the most identified bacterial pathogen from both RTE and raw foods in Ghana. Notably, Shiga toxin-producing strains of Escherichia coli (E. coli 0157:H7) were identified in vegetable salads, 31 groundnut, 62 raw milk, boiled milk, “brukina,” “nunu,” and raw “wagashie” samples, 74 with overall prevalence ranging from 1.1% to 21%. In line with this finding, studies from other countries98 -101 have reported the presence of E. coli 0157:H7 in RTE food samples despite zero-tolerance policy against it (Todd, 2004). Wang et al. 102 reported a study which investigated an outbreak of E. coli 0157:H7 linked to spinach consumption in 26 states in the USA and Canada. Among 199 cases reported, three deaths occurred, while 16% developed acute renal failure and 51% required hospitalisation. The presence of this pathogen in RTE food poses a significant public health risk due to the severe illnesses it can cause, such as haemorrhagic or non-haemorrhagic diarrhoea, haemorrhagic colitis, haemolytic uremic syndrome, and thrombotic thrombocytopenic purpura.103,104
Other bacteria reported from RTE foods in this study included Klebsiella spp., Shigella spp., Salmonella spp., Pseudomonas spp., Bacillus spp., Streptococcus spp., and Staphylococcus spp.23 -25,28,30,39,41,49,53,54,57,60,62,65 -67,70,74. In this review, it was found that locally prepared drinks such as “asaanaa” and “sobolo” contained these harmful microorganisms, with many exceeding the acceptable limits set for RTE foods.28,65 Jimma et al. 60 reported the presence of E. coli and Salmonella spp. in locally processed fruit juices, with microbial loads above the acceptable limits and found no traces of these organisms in industrially processed fruit juices. This finding is consistent with that of a study by Sultana et al. 105 from Bangladesh which found bacterial contamination in locally processed juices to be above tolerable limits, while industrially processed fruit juices had negligible counts. This finding suggests that locally prepared drinks are more susceptible to microbial contamination than industrially processed drinks, which could be attributed to good sanitation practices, addition of preservatives, and the use of automated and aseptic juice processers in the industries.60,105
Fruits and vegetables were identified in this review as common reservoirs of several microbial pathogens, predominantly, E. coli, Salmonella spp., Shigella spp., Enterococci spp., Citrobacter spp., Staphylococcus spp., and Klebsiella spp. in Ghana.53,56,59,70 The review revealed that fresh-cut RTE fruits and vegetables are prone to contamination by various microorganisms, both pathogenic and non-pathogenic, due to the processes involved in their preparation and handling before and during sale.53,70 Contrary to this finding, 10,070 samples of fresh-cut fruits and vegetables analysed for bacterial pathogens in Canada reported zero incidence of Escherichia coli, Salmonella spp., Shigella spp., and Campylobacter spp. 106 Proper handling and storage throughout the entire process of harvesting, processing, preparation, storage, and retail display are crucial to ensuring the safety of RTE fresh-cut fruits and vegetables for consumers. 107
Also in this review, several bacteria were identified in raw beef and raw chicken samples.29,31,33,36,37,44,47,50,64,71,78,79,81,82 The most commonly isolated bacteria from raw beef and chicken samples were E. coli, S. aureus, Klebsiella spp., Salmonella spp., Klebsiella spp., and Bacillus spp. This finding agrees with those of studies conducted by Mpundu et al. 108 and Madoroba et al. 109 in which E. coli, Staphylococcus aureus, Salmonella spp., and Bacillus spp. emerged as major contaminants in raw chicken and meat samples from Zambia and South Africa. The occurrence of these bacteria may be attributed to unhygienic practices both at slaughterhouses and retail shops as well as the water used in washing the meat before retail. 109 Also, these bacterial contaminations may be from the soil as most post-slaughter processes in abattoirs are done on the floor. According to Bhandare et al. 110 it has been emphasised that a large number of abattoir and retail beef workers in developing countries lack proper training on, and awareness about, hygienic practices that could help reduce bacterial contamination in beef products. 111 Retailers must prioritise strict adherence to hygiene conditions, proper handling practices, and appropriate storage methods for meat products. 111 Moreover, it is essential to implement continuous monitoring of bacteriological profiles and loads at abattoirs and sales points to ensure the safety of these products.
The parasitic organisms reported in this review, Trichuris trichiura, Cryptosporidium spp. Giardia lamblia, Entamoeba histolytica, Entamoeba coli, Hookworm, Cyclospora cayetanensis, Ascaris spp., Isospora belli, Enterobius vermicularis. Stronglyoides stercoralis, Taenia spp., Moniezia spp., Ancyclostoma duodenale, Faciolopsis buski, Balantidium coli, and Fasciola spp., have been documented in various regions worldwide, as evidenced by studies conducted by Mohamed et al., 112 Eraky et al., 113 Said et al., 114 and Chau et al. 115 In this review, these parasites were isolated from RTE salads, fruits, and vegetables.49,53,56,86-90 Among the identified parasites, Trichuris trichiura was the most prevalent. This parasite is the known cause of trichuriasis—a condition characterised by inflammation of the colon, diarrhoea, bloody stools, weight loss, anaemia particularly in children.116,117 A review conducted by Karshima 2018, reported these parasites (Taenia spp., Ancylostoma duodenale, Ascaris lumbricoides, Enterobius vermicularis, Balantidium coli, Entamoeba coli, Strongyloides stercoralis, and Trichuris trichiura) as major parasitic contaminants in fruits and vegetables in Nigeria as reported in this study. This finding emphasises the public health concern associated with the potential acquisition of parasitic infections through the consumption of contaminated fruits and vegetables. These contaminations may be due to water used for irrigation and washing of fruits and vegetables and also poor hygienic practices by food handlers.87,88,118
Various fungal species were reported in a number of studies in this review, predominantly in RTE foods such as boiled rice, vegetable salad, “fufu,” “solom,” “sobolo,” grilled beef sausage, “nunu,” groundnut, smoked fish (Mackerel, Tuna, and Sardine), cabbage, lettuce, and scallions. The analysis revealed that salads and vegetable dishes were the most heavily contaminated food items with fungi. The most reported fungal species in these foods were Aspergillus spp., Fusarium spp., and Penicillium spp. This finding agrees with that of a study by Izah et al. 119 which reported Aspergillus spp., Fusarium spp., Mucor spp., and Penicillium spp. in RTE foods in Nigeria. According to Hashem, 120 these fungi are known to produce mycotoxins such as aflatoxins and ochratoxins (produced by Aspergillus spp.), moniliformin and fumonisins (produced by Fusarium spp.), and citrinin and cyclopiazonic acid (produced by Penicillium spp.).119 -122 These mycotoxins could cause liver diseases, among other health complications. 120
Some studies in this review identified the presence of some mycotoxins, specifically aflatoxins, fumonisin, and ochratoxin in maize, groundnuts, groundnut paste, and spices sold in local markets throughout Ghana. The findings revealed that a significant proportion of maize, groundnut, and spice samples, had aflatoxin levels that exceeded the established thresholds set by the GSA and the EFSA for total aflatoxins.91 -94 Aflatoxins could have adverse effects on human health, including teratogenic, carcinogenic, hepatotoxic, and mutagenic outcomes, even when consumed in small quantities.123,124 In Ghana, maize and groundnut blend is a very popular complementary food for children. 125 A report by Ismail et al. 126 indicated that aflatoxin exposure during pregnancy and infancy, whether in utero or via breast milk, infant formula milk, and infant foods, has been associated with various health concerns, such as hindered growth and development, weakened immune system, and impaired liver function, particularly in African and Asian nations due to high exposure rates. This is major public health concern in Ghana due to a high rate of consumption of maize and maize products. Aflatoxin contamination in food also has economic implications. According Kortei et al. 92 elevated levels of aflatoxins in food samples could potentially lead to rejection during export, as they do not meet the required safety standards. It is important that good agricultural practices and good hygiene practices are implemented to prevent the formation of aflatoxins in food during storage. 92 The various hazards identified in this review have been reported to cause severe health complications, particularly in vulnerable populations, such as immunocompromised individuals, children, the elderly, and pregnant women. 127
Conclusion
This review has provided an overview of the spectrum of several microbiological hazards in foods in Ghana that are of importance to public health. It has revealed that several foods, such as vegetable salads, fruits and vegetables, “khebab”, meat (beef, goat chicken) and milk, commonly have high levels of microbial contaminations. The widespread contamination of food with such hazards (e.g., Escherichia coli, 0157:H7, Staphylococcus spp., Klebsiella spp., Salmonella spp., Shigella spp., Aspergillus spp., Fusarium spp., Penicillium spp., coupled with their toxins, and other parasitic contaminants), as documented in this review, could pose significant public health concerns, particularly for vulnerable populations. Consuming such foods could lead to illnesses, and in severe cases, death. It is essential that regulatory authorities rigorously enforce existing food safety laws and take proactive measures to ensure compliance particularly in the informal food sector. It is also important that food vendors and handlers adhere to safety standards, maintain proper personal hygiene, and adequately prepare food to protect the safety of consumers. Moreover, food safety research in Ghana must extend beyond the capital cities of Greater Accra, Ashanti, and Northern Regions to encompass a wider range of geographical areas. This is crucial for a comprehensive understanding of the unique food safety challenges that exist in other areas throughout the country. Although no articles in the review investigated foodborne viruses, viral hepatitis was reported as a common foodborne illness in a Ghanaian hospital. 97 It is important that research is conducted in this area to better understand the impact of foodborne viruses on foodborne disease.
Limitation
This scoping review is subject to some limitations due to the heterogeneity of included studies. Despite using a standardised data extraction template, the studies included varied in design, methodology, food types, microbial hazards, and data reporting, making direct comparisons and analysis challenging. Additionally, the review primarily focused on published articles indexed in PubMed, Web of Science, Scopus, and Google Scholar, potentially excluding relevant grey literature and articles available in hard copy.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Institutes of Health, USA, through the “Application of Data Science to Build Research Capacity in Zoonoses and Food-Borne Infections in West Africa (DS-ZOOFOOD) Training Programme” hosted at the Department of Medical Microbiology, University of Ghana Medical School (Grant Number: UE5TW012566-01). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contribution
Conceptualisation, ESD; methodology, WKA, FCNK, and ESD; validation, FCNK, PBT-Q, and ESD; formal analysis, WKA, FCNK, PBT-Q, and ESD; resources, ESD; data curation, WKA, FCNK, PBT-Q, and ESD; writing—original draft preparation, WKA, FCNK, and ESD; writing—review and editing, WKA, FCNK, PBT-Q, and ESD; visualisation, WKA, FCNK, PBT-Q, and ESD; supervision, PBT-Q and ESD; funding acquisition, ESD.
