Abstract
Along with expanding urbanization and industrialization, environmental pollution which negatively affects the surroundings, has been rising quickly. As a result, induces heavy metal contamination which poses a serious threat to living organisms of aquatic and soil ecosystems. Therefore, they are a need to ameliorate the effects cost by cost pollution on the environment. In this review, we explore methods employed to mitigate the effects caused by heavy metals on the environment. Many techniques employed to manage environmental pollution are tedious and very costly, necessitating the use of alternative management strategies to resolve this challenge. In this concept, bioremediation is viewed as a future technique, due to its environmental friendliness and cost-effective measures aligned with sustainable or climate-smart agriculture to manage contaminants in the environment. The technique involves the use of living entities such as bacteria, fungi, and plants to deteriorate toxic substances from the rhizosphere. Currently, bioremediation is thought to be the most practical, dependable, environmentally benign, and long-lasting solution. Although bioremediation involves different techniques, they are still a need to find the most efficient method for removing toxic substances from the environment. This review focuses on the origins of heavy metal pollution, delves into cost-effective and green technological approaches for eliminating heavy metal pollutants from the environment, and discusses the impact of these pollutants on human health.
Keywords
Introduction
Globally, the Industrial Revolution played a significant impact on the development of the economies of many different countries because it changed an economy that was predominately based on agriculture and handicrafts into one that was dominated by industry and machine manufacturing.1,2
In India, the Industrial Revolution had a pivotal role in the economic rise of developing countries. In the case of India, the Industrial Revolution commenced post-1850 and notably bolstered the rural economy. 3 However, this period of scientific and technological development had concomitantly brought pros and cons in the long run. As a result, it led to unprecedented outcomes due to human activities which were first ignored until the publication of Silent Spring by Rachel Carson on September 27, 1962. 4 The Silent Spring unveiled the mystery behind the use of synthetic chemical inputs and its negative impact on the environment. 5 Their effects were environmental pollution that was formally categorized as anthropogenic activities,6 -9 resulting from the dumping of industrial, home trash, and synthetic agricultural inputs. Therefore, to sustain the environment, alternative methods need to be employed to mitigate the effects induced by synthetic chemicals on the environment.
The anthropogenic activities, primarily emanating from the agricultural, industrial, and urbanization side, are currently releasing contamination to the rhizosphere or atmosphere which includes accumulation of heavy metals and other toxic fumigant chemicals that pose an environmental threat. 10 Heavy metal contamination is considered as one of the most critical environmental issues that reduce crop productivity and directly or indirectly jeopardizes the survival of almost all types of living entities on the planet. 11 The toxic metals absorbed by plants result in chemical residues on marketable produce causing mutagenic reactions which result in cancer in human beings. 12 Because wildlife depends on plants, they are also affected by heavy metal pollution, which disturbs the balance of mother nature and reduces biodiversity. 13 On the other side, pesticides used in plant protection also kill or affect the reproductive potential of untargeted organisms like beneficial nematodes, insects, birds and earthworms. 14 The prevention of heavy metal infiltration into terrestrial, atmospheric, and aquatic habitats as well as the remediation of damaged land are therefore imperative.
Heavy metals are a distinct group of metals that possess comparatively high densities, atomic numbers, and atomic weights within the periodic table.15,16 Typically, heavy metals are non-biodegradable and persist in the environment for several decades. 17 Heavy metals such as mercury (Hg), cadmium (Cd), lead (Pb), chromium (Cr), and arsenic (As) are considered to pose a significant threat to untargeted living entities due to their toxicity character, even at low concentrations. 18 As a result, bioremediation is viewed as a future technique to ameliorate the effects caused by pollution on the environment due to anthropogenic activities. This technique is suitable for remediating contaminants and it is eco-friendly. 19 Bioremediation involves the use of living entities such as bacteria: Acinetobacter sp., 20 Alcaligenes odorans, 21 Bacillus subtilis, 22 Corynebacterium propinquum, 23 Microbacterium sp., 24 Pseudomonas sp., P. putida, P. aeruginosa, 25 and Ralstonia sp. 26 to deteriorate toxic substances from the rhizosphere as well as the atmosphere. 27 It also employs the use of plants, technically known as green biotechnology where Brassica juncea, 28 Helianthus annuus, 29 Pteris vittate, 30 Salix viminalis, 31 and Solanum lycopersicum 32 plants were employed and shown the ability to extract or reduce heavy metals from the soil. This review summarizes a variety of bioremediation techniques, with a focus on their efficacy in thoroughly eradicating heavy metal pollution from the environment. It does so by doing a thorough analysis of the current literature.
The Principal Sources of Pollution
Heavy metals are released into the environment from various sources including mining, urbanization, chemical industry, sewage plants, pesticide plants, biomedical and unsafe agricultural practices (Figure 1) The United Nations Environment Program (UNEP/GPA) and the Global Plan of Action (GPA) recognize electronic waste (e-waste) which includes devices like mobile phones, tablets, computers, and smartwatches as a major threat to the environment and human well-being. This is primarily due to the presence of heavy metals like Hg, Cd, and Pb in electronic devices, which can pose serious risks to both the environment and human health if not properly disposed of UNEP/GPA 33 and Tchounwou et al. 34 The pollution levels of these heavy metals are influenced by industrial activities, geographic locations, regulatory oversight, and diverse sources. 35 For instance, Hg primarily emanates from coal combustion, electric/light bulb, wood preservatives, leather tanning, ointments, thermometers, adhesives and paints. 36 Cd often originates from industries like battery manufacturing, paint pigments, pesticides, galvanized pipes, plastics, polyvinyl and copper refineries. 37 Pb, an extremely toxic metal, commonly originates from substances like Pb-based paints, gasoline and mobile batteries. 38 Cr is emitted from a variety of industrial activities, including petroleum refining, electroplating, leather tanning, textile manufacturing, and pulp processing. 39 As, a naturally occurring element in the Earth’s crust, is released into the environment through a variety of human activities, including mining, agricultural practices, automobile exhaust and industrial dust, wood preservatives, and dyes. 40

A schematic diagram illustrating the origins of heavy metal pollution.
Soil plays a vital role in supporting terrestrial ecosystems and their biodiversity. Heavy metals are prevalent pollutants within the soil environment, and their presence can adversely affect microorganisms, plants and animals. The European Environment Agency (EEA) has set limit values for soil pollutant levels of various heavy metals, including Hg (0.20 ppm), Cd (0.44 ppm), Pb (0.48 ppm), Cr (0.20 ppm), and As (0.11 ppm).41,42 According to World Health Organization (WHO) guidelines, the acceptable levels of heavy metal pollutants in drinking water are as stated: Hg—0.001 ppm, Cd—0.005 ppm, Pb—0.05 ppm, Cr—0.05 ppm, and As—0.05 ppm. 43 The Food and Agriculture Organization (FAO) of the United Nations (UN) and the WHO set maximum limits for the consumption of heavy metals, as higher levels can cause health problems. The permissible limits for heavy metals consumption through vegetables are as follows: Hg—0.05 mg/kg for all vegetables; Cd—0.2 mg/kg for leafy vegetables, 0.3 mg/kg for root vegetables, and 0.1 mg/kg for other vegetables; Pb—0.15 mg/kg for all vegetables; Cr—0.1 mg/kg for all vegetables, and As—0.1 mg/kg for all vegetables.44 -46
Managing Pollution
Several techniques are employed to decontaminate the environment from these pollutants and avert the entry of toxic metals into the environment. Nevertheless, these methods tend to be costly and exhibit suboptimal efficacy.47,48 The increasing concerns surrounding environmental contamination have initiated the development of suitable technologies to assess the presence and mobility of metals in soil, water, and wastewater (Figure 2). Private and government institutions face a technical challenge to removing contaminants from the environment. Phytoremediation has emerged as a popular and economical plant-based technology for effectively addressing environmental issues. The process entails utilizing plants to extract and remove elemental pollutants or lower their bioavailability in soil or water. 49 In modern science, this technology is widely accepted due to its eco-friendliness, affordability, and high effectiveness. 50 Phytoremediation takes advantage of the unique and selective uptake capabilities of plant root systems, coupled with the translocation, bioaccumulation, and contaminant degradation abilities of the entire plant body. 51 Both aquatic and terrestrial plant species have been harnessed to eliminate pollutants from the environment. 52
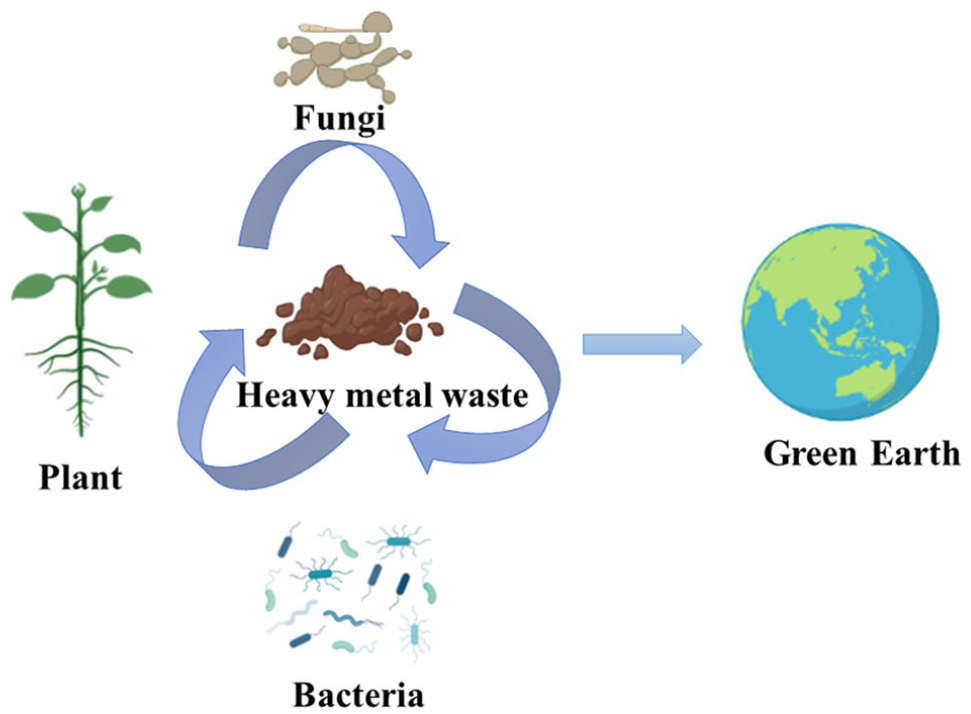
An illustrative diagram elucidating bioremediation, highlighting the crucial roles of plants, bacteria, and fungi (Created with BioRender.com).
More than 400 species have been identified as metal accumulators of Hg, Cd, Pb, Cr, As, and various radionuclides from contaminated soils (Tables 1–4). Arabidopsis sp. is well known for its metal tolerance and hyperaccumulation of Zn. 53 Aquatic plant species such as Azolla pinnata, Ceratophyllum demersum, Eichhornia crassipes, Lemna minor, Myriophyllum spicatum, Nasturtium officinale, Pistia stratiotes, Potamogeton pectinatus, Phragmites, Salvinia herzogii, Salvinia minima, Spirodela intermedia, Scirpus spp., and Typha latifolia, are of particular importance due to their high contaminant removal capacity.54 -58
Heavy metal accumulation in plants.

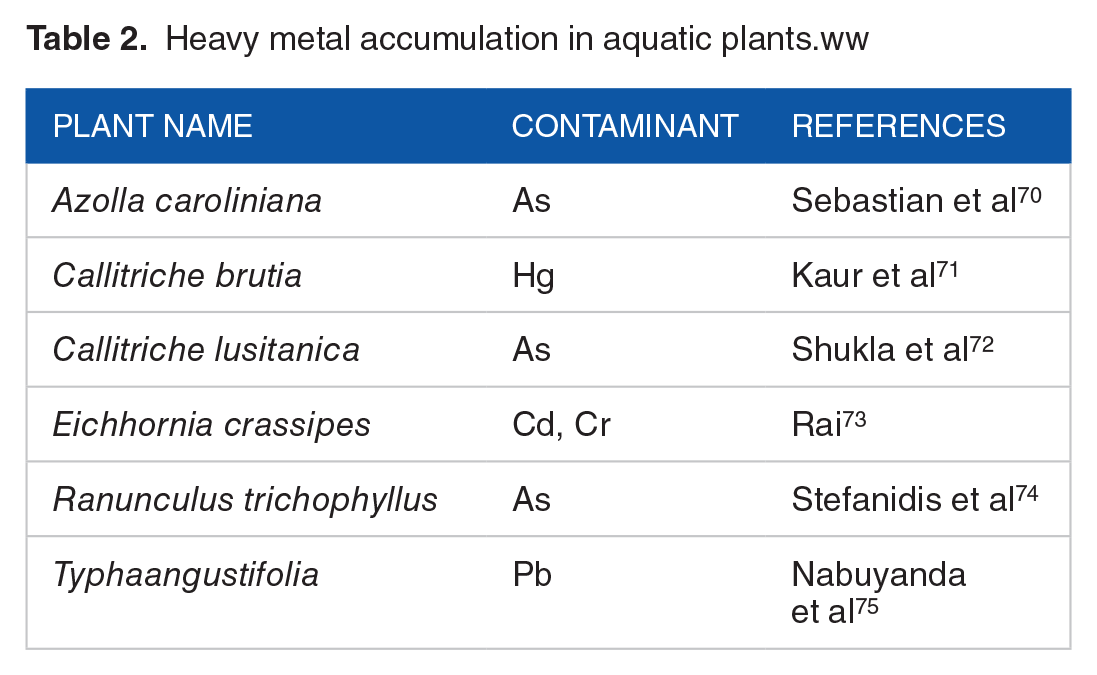
Heavy metal accumulation in aquatic plants.
Heavy metal accumulation in genetically modified plants.

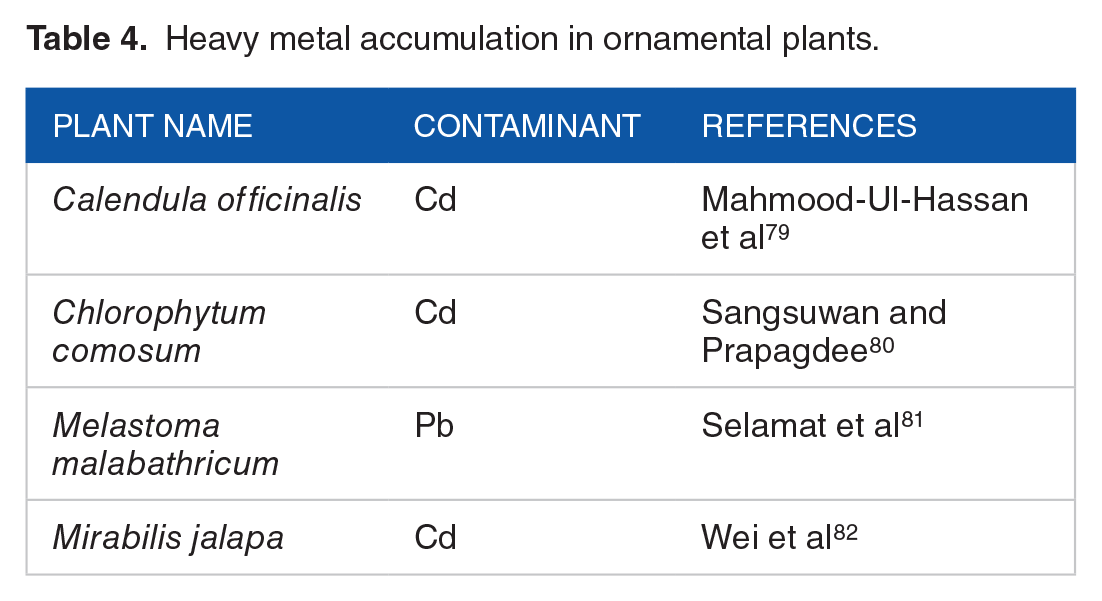
Heavy metal accumulation in ornamental plants.
Mechanism of phytoremediation
Phytoremediation encompasses several processes, including phytoextraction, phytoaccumulation, phytovolatilization, phytostabilization, and phytotransformation (Figure 3). 49 Phytoextraction is a technique that involves the absorption of organic and inorganic pollutants through the roots and stems. Besides, some particular plant species, like Brassica juncea, Cassia alata, Celosia argentea, Kummerowia striata, Helianthus annuus, Momordica charantia, Nicotiana tabacum, Salix mucronata, Salix viminalis, Solanum lycopersicum, Solanum melongena, Swietenia macrophylla, Pteris vittata, and Vigna unguiculata, have the potential to be used as suitable plant selections to enhance the phytoextraction process.83 -86 In phytostabilization, in this process, plants accumulate and immobilize heavy metals by binding with biomolecules. 87 Miscanthus giganteus, Avena sativa, and Sinapis alba can also help to stabilize heavy metal compounds in the soil. 88 There are several processes by which plants can reduce contaminants.

A schematic diagram depicting the underlying mechanisms of phytoremediation processes (Created with BioRender.com).
Phytoextraction
Phytoextraction, also called phytoaccumulation, involves the accumulation of heavy metals from earthland. In this method, the uptake and translocation of contaminants by plants root into the aerial portions of plants and deposited into vacuoles. The mechanism during the accumulation process is used to absorb and precipitate the toxic metals by metal-phytochelatin complex before translocating into the shoot, leaf and stem parts of the plant. The hyperaccumulator species accumulate a higher concentration of heavy metals.89,90
Rhizofiltration
Rhizofiltration involves the elimination of heavy metals using plant roots. Though it is comparable to phytoextraction, in this process, plants remove contaminants from wastewater or groundwater rather than soil. In this process, plant roots assimilate or adsorb pollutants from wastewater, groundwater, or surface water. Generally, aquatic plant species are employed to eliminate pollutants through rhizofiltration. Rhizofiltration is effective for removing Cd, Pb, and Cr, which are primarily accumulated in the roots. Sunflower, tobacco, and spinach exhibit promising potential in removing Pb from water. 91
Phytostabilization
Plant roots can limit the movement of heavy metals by phytostabilization, a process that reduces toxic effects. This process involves the capture of contaminants on the root surface using transport proteins or secondary metabolites. Furthermore, the process involves the breakdown of complex organic molecules into simpler ones by coupling them with protein, amino acid, and sugar derivatives. Black nightshade, sunflower, and cowpea are among the plant species that employ phytostabilization mechanisms. 92
Phytovolatilization
This process entails the uptake of contaminants by plants from the soil and their conversion into less toxic volatile compounds that are released into the atmosphere. The volatile compounds are primarily released from aerial plant parts such as stems and leaves. This mechanism is effective when the contaminants are less toxic. 93
Phytodegradation
Phytotransformation, also known as this process, refers to the absorption of contaminants by plants, which are then metabolized or broken down into less toxic compounds and translocated to various plant organs. The organic compounds are then degraded into non-toxic forms inside the plant tissue. 94
Microbial-assisted remediation of heavy metal
Microbial remediation is the process of using living microorganisms such as bacteria, fungi, and archaea to break down and detoxify various chemical and metallic hazardous wastes from the environment. 95 Bioremediation involves the direct application of microorganisms to the polluted site in order to facilitate the degradation of contaminants. Microorganisms are used in a variety of remediation techniques, including bioaugmentation and biostimulation. In bioaugmentation, specific microorganisms are added to a contaminated site to enhance the breakdown of contaminants. In biostimulation, the environmental conditions at the site are modified to promote the growth and activity of naturally occurring microorganisms that can degrade contaminants. Physical and chemical treatments are conventional remediation methods that have drawbacks such as high cost, heavy machinery, logistical glitches, and potential environmental toxicity. 96 In contrast, bioremediation technologies have seen significant growth and development, making it a promising method for treating soil and water contamination (Table 5). Among these methods, bioremediation of oil spills is the most lucrative and environment-friendly technique. 97
Bioremediation of heavy metal by microorganisms.

Fungi are used for the remediation of pollutants in mycoremediation, a type of bioremediation. Fungi play a vital role in cleaning up contaminated sites in both soil and aquatic ecosystems. 111 These microorganisms, which are widely present in nature, can thrive in a diverse range of environmental conditions. These microorganisms survive in extreme conditions and produce some extracellular ligninolytic enzymes like peroxidase and laccases. These enzymes help fungi to transform pollutants into non-toxic forms. Pollutants can be adsorbed by extracellular enzymes. 112 Diverse fungal species such as Aspergillus sp., Bjerkandera adusta, Coriolus versicolor, Cryptococcus sp. Hirschioporus laricinus, Inonotus hispidus, Mucor sp., Penicillium sp., Phanerochaete chrysosporium, Phlebia tremellosa, Phanerochaete chrysosporium, Pleurotus sp., and Trametes versicolor, have been reported for bioremediation.113,114
Role of genetical engineering microbes in bioremediation
The potential of microbes for bioremediation is vast but unexploited. Genetically engineered organisms are the best way to enhance bioremediation activity.115,116 Further research is required to formulate advanced bioremediation techniques in engineering that can effectively eliminate the complex mixtures of pollutants found at various sites. Several microbes use the contaminants as an energy source through their metabolic processes. Bacteria and fungi in the environment help to degrade or detoxify harmful substances. Modern science relies on biotechnology to facilitate the development of genetically modified organisms (GMOs), which can be instrumental in comparing them with their wild-type variant. GMOs possess the necessary protein machinery, which they utilize to uptake and regulate heavy metals through the implementation of gene regulatory elements such as promoters, binders, and terminators. These organisms produce a heavy metal binding protein that protects from toxicity by strongly binding to heavy metals (Figure 4). Mesorhizobium huakuii strain B3, produces phytochelatin protein which accumulates Cd as reported by Sriprang et al 117 . Bae et al 118 reported that P. putida 06909 produced metal-binding peptide (MBP) EC-20 that has a high affinity for Cd. Al Hasin et al 119 found that Methylococcus capsulatus can remediate Cr (VI). Wagner-Döbler 120 demonstrated that recombinant bacteria allow detoxifying Hg2+ to the non-toxic form of Hg0 through mercury reductase and subsequent release of Hg. The mechanism for detoxification of heavy metals is controlled by the mer operon gene that regulates transcription levels at both positive and negative. P. fluorescens HK44 was applied for large-scale field-based remediation of pollutants. 121 Patel et al 122 reported that the recombinant Caulobacter crescentus strain JS4022/p723-6H was able to eliminate Cd.
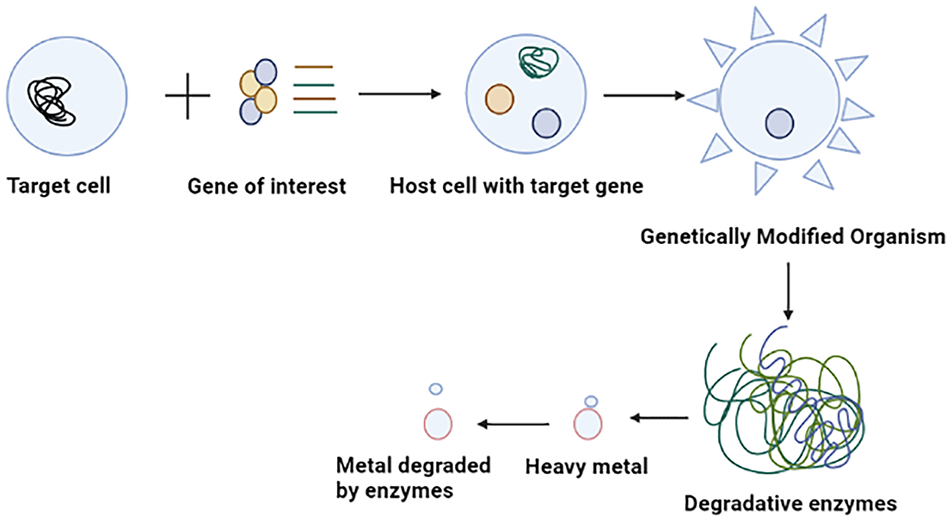
Illustrating the process of heavy metal degradation using genetically modified organisms (GMOs) (Created with BioRender.com).
According to several researchers, certain microbes can remove heavy metals from their environment by either accumulating them or developing a tolerance toward them. There are several microorganisms, including Acinetobacter sp., Alcaligenes odorans, Aspergillus niger (fungus), Aspergillus versicolor, Bacillus subtilis, Corynebacterium propinquum, Fomitopsis pinicola, Microbacterium sp, Pseudomonas sp., P. putida, P. aeruginosa, Ralstonia sp., and Streptomyces, that play a role in removing heavy metals.123 -126
Effect of Heavy Metal on Human Health
Certain edible crops can accumulate heavy metals, even in very small amounts. When these heavy metals enter our food chain, they disrupt the food pyramid and pose a threat to human health by causing cancer and liver diseases. Vegetables such as brinjal, gourd, spinach, coriander, tomato and pumpkin are particularly susceptible to heavy metal uptake by their roots, which can then be transported to the edible portions of the plant.127,128 As a result, consuming these vegetables that contain heavy metals can be extremely hazardous to human health. Alexander et al 129 carried out research involving vegetables cultivated in soil contaminated with heavy metals. Significant variations were observed among the vegetables in terms of the levels of metal accumulation. For Cd, lettuce exhibited a higher accumulation (8.6 mg/kg dry matter) compared to spinach (5.8 mg/kg dry matter), onion (3.6 mg/kg dry matter), carrot (2.0 mg/kg dry matter), pea (0.29 mg/kg dry matter), and French bean (0.07 mg/kg dry matter). Remarkably, lettuce recorded the highest concentration of Pb, nearly double that of onions, which held the second-highest average value. The sequence was as follows: lettuce (14.6 mg/kg dry matter) > onion (7.5 mg/kg dry matter) > carrot (5.8 mg/kg dry matter) > spinach (1.8 mg/kg dry matter) > pea (0.78 mg/kg dry matter) > French bean (0.34 mg/kg dry matter). A study conducted by Zhu et al, 63 revealed that the concentration of heavy metals in the edible parts of vegetables varied, with leafy vegetables having the highest amounts, followed by stalk vegetables, root vegetables, and solanaceous vegetables, and then legume vegetables and melon vegetables. Previous reports have also suggested that edible crops grown in industrial areas such as coal mines and petrochemical plants tend to contain higher levels of heavy metals. 130 Human exposure to heavy metals primarily occurs through the consumption of edible crops, which accounts for 90% of the exposure. The remaining 10% is attributed to the inhalation of polluted air particles as reported by Khan et al. 131
Excessive levels of heavy metals have the potential to pose harm to the body. They have the capacity to inflict damage on various organs such as the brain, muscles, nerves, liver, kidneys, and heart (Figure 5). Previous studies have specified that heavy metals can impair different organs within the human body, as illustrated in Table 6. The European Protection Agency (EPA) has reported that prolonged exposure to heavy metals can result in severe cancer. Research conducted by the WHO has shown that higher exposure to heavy metals puts 10% of women at risk of infertility.132,133

Illustrating the health implications of exposure to heavy metals on human well-being (Created with BioRender.com).
Effects of heavy metal contamination on human well-being.

Hg, a highly toxic metal found in air, water, and soil, is considered to be highly carcinogenic by the EPA. Hg exposure can result in various health problems, including Alzheimer’s disease, lung damage, and skin issues such as the common ailment. 151 Acrodynia is a common skin ailment in which skin color becomes pink. 152 Similarly, Cd is a highly toxic metal that causes bone damage and acute exposure can lead to renal dysfunction, while prolonged exposure to high levels of Cd can result in lung damage. Heavy metals such as these can also induce DNA damage, cause chromosome aberrations, and alter DNA replication and transcription.153 -155 Exposure to Cr over a long period can result in the formation of ulcers. Human activities have resulted in the contamination of the environment with heavy metals, which can have adverse effects on human health. Excessive uptake of heavy metals poses a significant threat to human health. The entering of heavy metals into the human body can initiate cancer by the production of reactive oxygen species (ROS) which mainly disrupts DNA molecules. Heavy metals can cause damage to specific organs within the human body. In an animal model of acute toxicity, Wister rats exposed to 1 mg/kg of Hg caused alterations in their kidneys. A study reported that oral exposure to Hg in rats resulted in diarrhea. Additionally, scientists found that guinea pigs exposed to 0.1 to 0.4 M of Pb increased serum endothelial and serum total protein levels, along with lung infection. 156 Male adult rats exposed continuously to Pb (0.4%) exhibited a significant reduction in white blood cell count, as reported by Mugahi et al. 157 Furthermore, the administration of Pb (10 mg/kg) was observed to increase the levels of lactate dehydrogenase and acid phosphatase in rats. 158 In rats, Patlolla et al 159 demonstrated that the administration of 10 mg/kg of Cr increased the levels of ROS and malondialdehyde in the liver and kidney. High doses of Cr(VI) caused the immune system to reduce, resulting in the development of allergic contact dermatitis.160,161 Fay et al 162 investigated Cd toxicity (0.6 mg/kg for 12 weeks) in the renal cortex of rats and found that Cd exposure significantly increased the volume of urine while decreasing the excretion of protein in urine. Cd toxicity can cause osteoporosis and bone fracture by increasing the dynamin-related protein, as demonstrated by Ma et al. 163 A close relationship between osteoporosis and high intake of Cd was also proven by Pouillot et al. 164
Conclusion
Recent technological advances have made bioremediation a more effective tool. This method is distinct and effective because it does not rely on chemicals or complex machinery. In the current study, bioremediation was shown to be a potential technique for resolving or reducing the negative effects of environmental contamination. Since it uses living entities to manage pollution, it cannot worsen the problem of heavy metal buildup or ozone depletion and is thought to be both environmentally friendly and economically effective, making it applicable to both emerging and developed nations globally. The results of the toxicity evaluation indicated that heavy metals constitute a substantial threat to living entities that are not specifically targeted. Therefore, funding ongoing research and innovation in bioremediation technologies is crucial for solving the 21st century’s expanding environmental issues.
Footnotes
Acknowledgements
Department of Biotechnology, The University of Burdwan, Burdwan, India, and the University of Limpopo’s Department of Plant Production, Soil Science and Agricultural Engineering, Green Biotechnologies Research Centre of Excellence, Private Bag X1106, Sovenga, 0727, South Africa, are gratefully acknowledged by the authors. Authors are sincerely acknowledged Tshepo S. Mashela, the University of Limpopo’s Department of Plant Production, Soil Science and Agricultural Engineering, Green Biotechnologies Research Centre of Excellence, Private Bag X1106, Sovenga, 0727, South Africa, for his invaluable assistance in manuscript preparation.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
CRediT Authorship Contribution Statement
Data Availability
The manuscript contains all the necessary data to support the findings of this study.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
