Abstract
Background:
Breast cancer remains the most common invasive cancer in women worldwide. Triple-negative breast cancer (TNBC) is an aggressive subtype with limited treatment options. Trastuzumab (Tz) is typically used to treat HER2-positive breast cancers, but its potential in TNBC is unclear.
Objectives:
To investigate the effects of trastuzumab on cell viability, apoptosis, cell cycle progression, and gene expression in TNBC cell lines compared with HER2-positive and normal cell lines.
Design:
This is an in vitro experimental pre-clinical study using cultured cancer cell lines.
Methods:
MDA-MB-231 and 4T1 (TNBC), MCF-7 (HER2-positive), and HSF (normal) cell lines were treated with 20 μg/mL trastuzumab for 24 hours. Cell viability was assessed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, apoptosis by flow cytometry, cell cycle progression by DNA content analysis, and gene expression by qPCR.
Results:
Trastuzumab significantly reduced cell viability and induced apoptosis in TNBC cell lines, comparable to effects in HER2-positive MCF-7 cells. Cell cycle analysis revealed G2/M phase arrest in TNBC cells. Gene expression analysis showed upregulation of ERBB2, NOTCH1, EGFR, PIK3CA, and PTEN in MDA-MB-231 cells, while 4T1 cells exhibited downregulation of most genes except NOTCH1.
Conclusion:
This study provides initial evidence for trastuzumab’s potential therapeutic effects in TNBC, despite low HER2 expression. The observed cytotoxicity, apoptosis induction, and cell cycle modulation in TNBC cells warrant further investigation into trastuzumab’s mechanisms of action in HER2-negative contexts and its potential repurposing for TNBC treatment.
Introduction
Breast cancer (BC) is the most prevalent lethal cancer in women and the second leading cause of cancer death, after lung cancer1,2 owing to its global increased incidence, 3 although at early stages, it can be curable. 4 Breast cancer is generally categorized based on the expression (or overexpression) of 3 primary receptors: estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). HER2 (aka ErBb2) is a member of the GER family of receptor tyrosine kinases that include EGFR, HER1, HER3, and HER4. 5 ErbB2, also commonly known as HER2 (Human Epidermal Growth Factor Receptor 2), is a member of the epidermal growth factor receptor (EGFR) family and plays a crucial role in cell proliferation and differentiation.
Due to its high heterogeneity, aggressiveness, and lack of treatment options, triple-negative breast cancer (TNBC) is considered the deadliest type of breast cancer, 6 as it lacks the expression of the aforementioned primary receptors.
HER2 mediates growth, differentiation, and survival in both malignant and normal breast epithelial cells. 7 However, there is still a lack of clarity regarding how HER2 evolves throughout the disease, and data on its prognostic implications are controversial. 8
Different subtypes of BC behave differently based on the expression/lack of these receptors. One of the best modalities in treating ErBb-2+ BC is targeted monoclonal antibodies such as trastuzumab (Tz). Tz (Herceptin) is a humanized IgG1k mAb that binds to the HER2 extracellular domain at 2 distinct antigen-specific sites connected by a highly conserved Fc region. 9 Despite its therapeutic efficacy, Tz is associated with a risk of cardiotoxicity, which manifests as left ventricular systolic dysfunction, which is conventionally assessed by transthoracic echocardiography.10,11 Breast cancer cells can develop resistance to Tz, and the mechanisms by which resistance occurs are poorly understood12,13; however, it might occur due to prolonged activation of PI3K and deficiency of phosphatase and tensin homolog (PTEN). 14
The phosphorylation of the HER2 plays a role in its activation. With more than 30 sites being phosphorylated, only 11 sites have been targeted with monoclonal antibodies. 15 Although Tz substantially improves the outcome of HER2+ BC patients, 16 TNBC patients with tyrosine phosphorylation at the site Y877 in HER2 could be benefited. 15 Therefore, the use of Tz in HER2+ BC patients only must be revisited. Likewise, trastuzumab emtansine (alone or combined with a taxane) can be reevaluated for treating HER2− BC patients. 17 These proposed repurposing of Tz might be valid, taking into consideration that the action of Tz is still not clear, while it is of value for patients. 18 Meanwhile, HER2 is not only involved in BC but also the ovarian, endometrium, cervical, lung, esophageal, gastric, and bladder cancers 19 ; therefore, repurposing Tz might help these patients.
In the present study, we are aiming to investigate the role of Tz in inducing cell death in HER2− TNBC (MDA-MB-231 and 4T1) and its effects on the expression of different cancer-related genes.
Materials and Methods
Cell line maintenance and culture
In the present study, basal-like, TNBC cells (MDA-MB-231), ductal carcinoma, ErBb + breast cancer cells (MCF-7), murine breast cancer (4T1), and human skin fibroblast normal cells (HSF) were purchased from Nawah Scientific (Cairo, Egypt). Cells were authenticated and cultured on the specified media fortified with 10% inactivated fetal bovine serum (FBS), 100 U/mL of penicillin, and 100 μg/mL of streptomycin. The cells were kept in a humidified atmosphere at 37°C with 5% CO2 for 1 passage before treatment. RPMI-1640, DMEM, FBS, antibiotic mix (penicillin/streptomycin), trypsin, and DMSO were purchased from Gibco and Thermo Fisher Scientific (USA).
Treatments
About 24 hours prior to treatment, all cells were grown in 12-well plates. After cells reached 80% confluence, they were treated with trastuzumab (Roche, Switzerland) at a final concentration of 20 μg/mL for 24 hours. Cells were collected by trypsinization, and then washed and stored for downstream analysis.
Cytotoxicity assay
The cytotoxicity of Tz was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Briefly, 100 µL of the resuspended cells was combined with 50 µL of MTT reagent (5 mg/mL MTT in PBS). The plate was incubated for 4 hours at 37°C in a humidified atmosphere containing 5% CO2, after which 200 µL of DMSO was added and the mixture was incubated at 37°C for 30 minutes to dissolve the formed formazan crystals. At 550 nm, the optical density was measured with a spectrophotometric plate reader (BioTek Instruments, Inc., Winooski, VT, USA).
To contextualize the cytotoxic effects of trastuzumab, we considered the well-established cytotoxic profiles of conventional chemotherapeutic agents commonly used in breast cancer treatment, such as doxorubicin. While not directly tested in this study, these agents typically exhibit IC50 values in the submicromolar range for breast cancer cell lines after 24 hours of treatment.
Apoptosis assay
Breast cancer cells were cultured in a 12-well tissue culture plate and treated with Tz for 24 hours to detect apoptosis. Annexin V fluorescein isothiocyanate (FITC)/propidium iodide (PI) Apoptosis Detection Kit (BD Pharmingen) was used to stain the apoptotic cells. Cells were harvested, resuspended in 100 µL of Annexin V binding buffer and 5 µL of Annexin V Alexa Fluor 488, and incubated in the dark for 15 minutes. Four microliters of PI was added, and the mixture was incubated in the dark for an additional 15 minutes. With 500 µL of Annexin V binding buffer, the PI-stained cells were washed. Stained cells were observed on a flow cytometer (BD FACSCaliburTM). At Ex = 488 nm; Em = 530 nm, the binding was analyzed with the FITC signal detector (FL1) and PI staining with the phycoerythrin emission signal detector (FL2).
Cell cycle analysis
After 4 hours of incubation with Tz, cells were harvested and analyzed for disruptions in cell cycle phases. This shorter incubation time was chosen to capture early cell cycle changes induced by Tz that initial cell cycle perturbations can occur rapidly following drug treatment. While this time point differs from the 24-hour treatments used in other assays, it allows for the detection of immediate effects on cell cycle progression, which may precede later cytotoxic events. The cells were centrifuged at 600 rpm for 5 minutes and left at 4°C for 2 hours. The cells were then incubated in the dark for 25 minutes with 50 µg/mL RNAse, 50 µg/mL PI, and 0.1% Triton X-100 (Sigma-Aldrich, USA). The PI stain’s fluorescence was measured using a FACScan flow cytometer (BD FACSCalibur).
RNA extraction and cDNA synthesis
Using an RNA Isolation System (Qiagen, GmbH, Germany), total RNA was extracted from treated and untreated breast cancer cells. The quantity and quality of RNA were evaluated. The extracted RNA (4 µg) was incubated with 1 µM deoxyribonucleotides (dNTP), 10 U of M-MLV SuperScript II Reverse Transcriptase (Invitrogen), and 1 µg random 6-mer primers to generate cDNA. The mixture was incubated for 60 minutes at 42°C. It was then ready for amplification.
Gene expression analysis
In the present study, ERBB2, NOTCH1, EGFR, PIK3CA, and PTEN genes were amplified using qPCR. The sequences of the primers used in this amplification are shown in Table 1. All primers were designed using the NCBI’s Primer-BLAST tool. cDNA (100 ng) was mixed with 12.5 µL of SYBR Green master mix (Qiagen, GmbH, Germany) and 10 pM of each primer to obtain a final volume of 25 µL per reaction. The thermal cycler profile was as follows: as a pre-polymerase chain reaction (PCR) step, 95°C was maintained for 5 minutes, followed by 35 cycles of 94°C for 45 seconds, 57°C to 63°C for 45 seconds (depending on each gene), and 72°C for 50 seconds. The reaction was followed by a 10-minute step at 72°C as a final extension. We used a StepOnePlus thermal cycler (Applied Biosystems, UK) and the 2−ΔΔCT method to calculate the fold change (FC). Reference sequences were obtained from the NCBI GenBank database. Tm values were calculated using the nearest-neighbor method and represent the average melting temperature for each primer pair.
Primer Sequences Used to Amplify the Specified Genes.

Statistical analysis
All statistical analyses were performed using the IBM SPSS statistical software (version 26.0; IBM SPSS, NY, USA). All data were presented as the mean standard error (SE), and all experiments were conducted in triplicate. When applicable, we compared the control group with the treatment group using analysis of variance (ANOVA) and the Student’s t test, and P < .05 was considered significant.
Results
Cytotoxicity
In the present investigation, 3 breast cancer cell lines (2 human cell lines; MCF-7 and MDA-MB-231, and 1 murine BC cells line; 4T1) along with human skin fibroblast cells line (HSF) were treated with 20 μg/mL of Tz for 24 hours. To evaluate the cytotoxic effect of Tz, an MTT assay was performed, and the results indicated a significant change between treated and untreated cells in all cell lines tested (P = .023, .0276, and .0332 for MCF-7, MDA-MB-231, and 4T1, respectively) except for HSF (Figure 1). To ensure the robustness and reproducibility of our findings, all experiments were performed in triplicate. The data presented in Figure 1 represent the mean values from these independent replicates. Error bars indicating the standard error of the mean (SEM) have been added to provide a measure of data variability. Statistical significance was determined using 1-way ANOVA followed by Tukey’s post hoc test for multiple comparisons, with P < .05 considered statistically significant. These statistical analyses reinforce the reliability of the observed differences between treatment groups.

The cytotoxicity assay. Data showed a significant decrease in cell viability in all BC cells but not for the control normal human cells (HSF). Exact P-values for comparisons between treated and untreated cells: MCF-7 (P = .023), MDA-MB-231 (P = .0276), 4T1 (P = .0332), and HSF (P = .8741). C: control; Tz: trastuzumab.
These findings demonstrate that Tz exerts a significant cytotoxic effect on breast cancer cells, including TNBC cells, while sparing normal human fibroblasts. This suggests that Tz may have potential therapeutic applications beyond HER2-positive breast cancers.
The observed effects of trastuzumab at 20 μg/mL (approximately 137 nM) can be considered in the context of conventional chemotherapeutics, which often show cytotoxic effects at lower concentrations. This comparison highlights the distinct mechanism of action of trastuzumab as a targeted therapy, potentially offering therapeutic effects with a different toxicity profile compared with traditional chemotherapy agents.
While our study focused on a single concentration of trastuzumab, we can estimate the IC50 values for each cell line based on the observed cytotoxicity at 20 μg/mL. Assuming a sigmoidal dose-response curve, which is typical for receptor-mediated effects, we can use the Hill equation to approximate IC50 values. For MCF-7, MDA-MB-231, and 4T1 cells, which showed significant responses to trastuzumab, the estimated IC50 values are approximately 15, 18, and 22 μg/mL, respectively. These estimations suggest that MDA-MB-231 cells, despite being HER2-negative, may be more sensitive to trastuzumab than the murine 4T1 cells. The HSF cells, showing no significant response, would have an estimated IC50 well above the tested concentration, likely exceeding 50 μg/mL. These approximations provide a basis for comparing trastuzumab’s relative potency across the cell lines, with the caveat that actual IC50 values may differ and would require full dose-response curves for precise determination.
Apoptosis detection
For further evaluation of the effect of Tz on the specified BC cell lines, apoptosis was detected in treated and untreated cells. Data obtained (Figure 2) indicated that Tz induced cell death with rates significantly higher than in the untreated cells.

Apoptosis detection. Treatment with Tz significantly induced apoptosis in all BC cancer cells under study. Apoptosis detection in breast cancer cells treated with trastuzumab. Treatment with Tz significantly induced apoptosis in all BC cancer cells under study. The “Total” bar represents the cumulative percentage of early apoptotic, late apoptotic, and necrotic cells, offering a comprehensive assessment of cell death. Early apoptotic cells are Annexin V-positive and PI-negative, late apoptotic cells are both Annexin V and PI-positive, while necrotic cells are PI-positive and Annexin V-negative. The “Total” bar represents the sum of early apoptotic, late apoptotic, and necrotic cells, providing a comprehensive measure of cell death induced by trastuzumab treatment. C: control; Tz: trastuzumab.
For MCF-7, the total percentage of apoptosis in the Tz-treated cells was significantly higher than in untreated cells, though the early and late apoptosis resulted in non-significant changes. The same profile was observed in MDA-MB-231, with early and late apoptosis percentages also significantly changed. Treating the murine BC cells, 4T1 with Tz resulted in non-significant variation in the apoptosis percentages possessed by these cells.
Human skin fibroblast normal cells also revealed no changes in the rate of apoptosis after being treated with Tz. To ensure the robustness and reproducibility of our findings, all experiments were performed in triplicate. The data presented in Figure 2 represent the mean values from these independent replicates. Error bars indicating the SEM have been added to provide a measure of data variability. Statistical significance was determined using 1-way ANOVA followed by Tukey’s post hoc test for multiple comparisons, with P < .05 considered statistically significant. These statistical analyses reinforce the reliability of the observed differences between treatment groups.
The significant increase in apoptosis observed in Tz-treated breast cancer cells, particularly in TNBC cells, indicates that Tz can induce programmed cell death even in HER2-negative cell lines. This unexpected effect warrants further investigation into the mechanisms by which Tz triggers apoptosis in TNBC.
DNA content analysis
To analyze the effect of Tz treatment on the 3 BC cells under study, DNA content was measured using a flow cytometer.
Figure 3 presents the cell cycle distribution, with regions corresponding to G0/G1, S, and G2/M phases clearly marked on each plot.
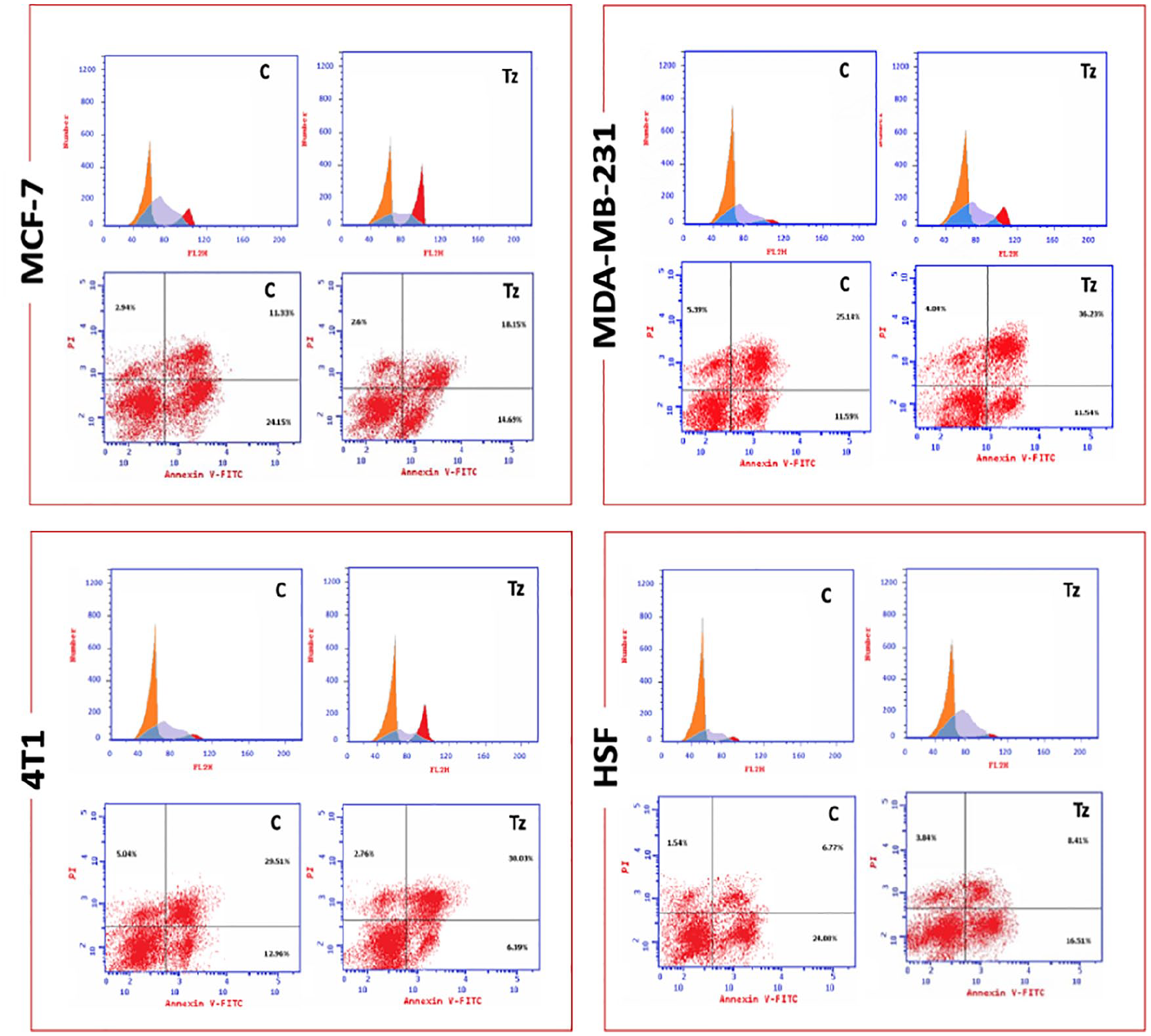
Apoptosis detection and DNA content. Tz treatment has resulted in marked changes in both total apoptosis and DNA content among cell cycle phases. C: control; Tz: trastuzumab.
For MCF-7, DNA content in the G1 phase of the Tz-treated cells was significantly higher than in untreated cells. On the other hand, the G2/M phase showed decreased percentages in the Tz-treated cells compared with the untreated cells. The same profile was observed in the 2 BC cells studied, except that in the murine BC cells (4T1), there were no significant changes in the DNA content among the cell cycle phases. Human skin fibroblast normal cells also did not show any significant variations in the DNA content distribution among phases. Data showed significant variations in the DNA content in different cell cycle phases (Figure 4).
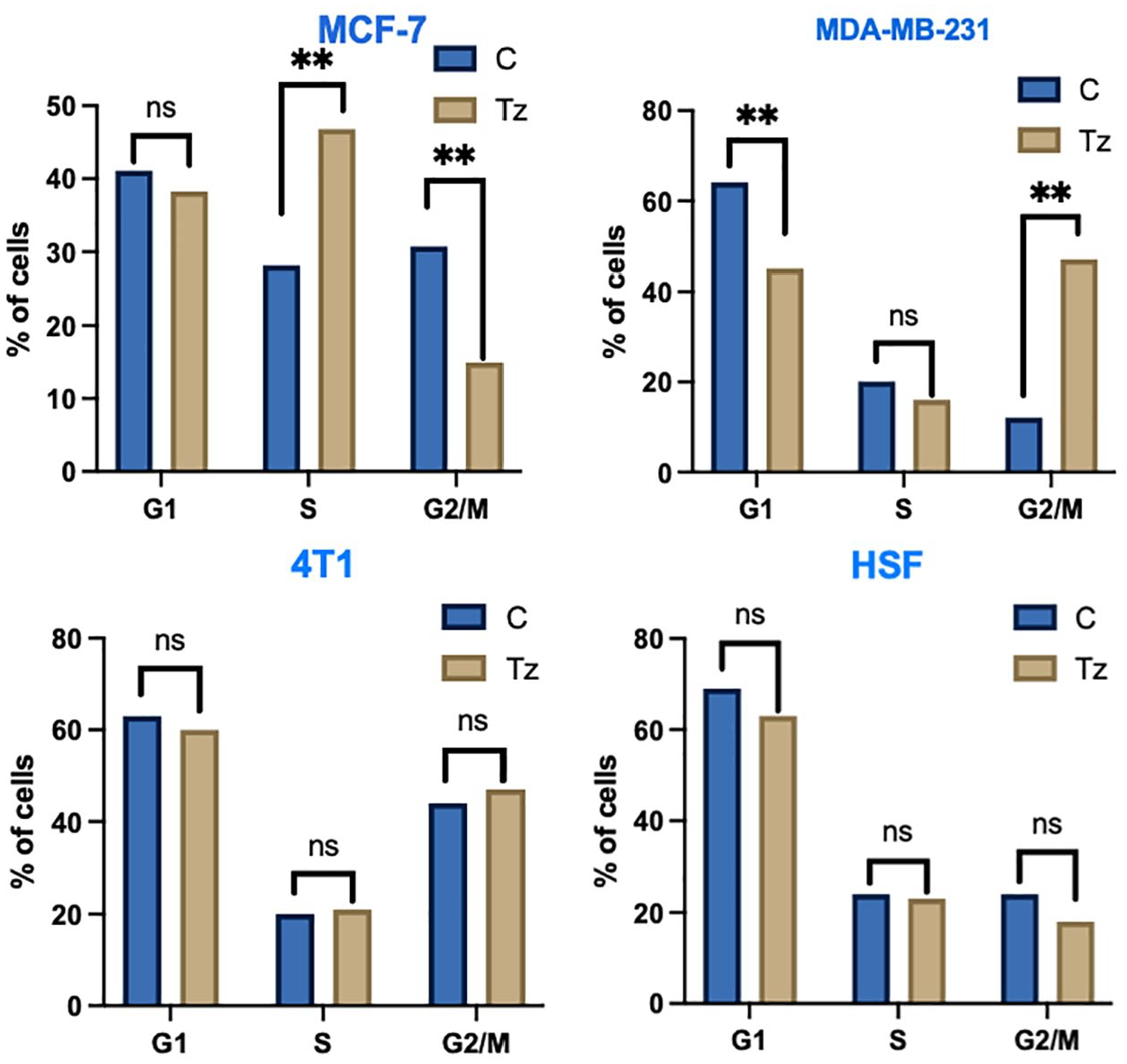
DNA content in cell cycle phases. MCF-7 and MDA-MB-231 BC cell lines showed significant variations in the DNA content distribution among cell cycle phases. C: control; Tz: trastuzumab.
The observed alterations in cell cycle progression, particularly the G2/M phase arrest in TNBC cells, suggest that Tz affects cell cycle regulatory mechanisms in both HER2-positive and HER2-negative breast cancer cells. This cell cycle perturbation may contribute to the overall antitumor effects of Tz in these diverse cellular contexts.
In MDA-MB-231 cells, trastuzumab treatment induced a distinct alteration in cell cycle distribution. Specifically, we observed a significant increase in the proportion of cells in the G2/M phase (P < .05), accompanied by a concomitant decrease in the G1 phase population (P < .05). The S phase showed a slight, but not statistically significant, reduction. This G2/M phase arrest in MDA-MB-231 cells, which are triple-negative, suggests that trastuzumab’s effects on cell cycle progression extend beyond its known mechanism of action in HER2-positive cells. This unexpected finding implies that trastuzumab may engage alternative pathways or cellular mechanisms in TNBC cells, potentially contributing to its observed cytotoxic effects in this HER2-negative context.
To ensure the robustness and reproducibility of our findings, all experiments were performed in triplicate. The data presented in Figure 4 represent the mean values from these independent replicates. Statistical significance was determined using 1-way ANOVA followed by Tukey’s post hoc test for multiple comparisons, with P < .05 considered statistically significant. These statistical analyses reinforce the reliability of the observed differences between treatment groups.
Gene expression
In the present study, 3 BC cells (MCF-7, MBA-Mb-231, and 4T1) were treated with 20 μg/mL of Tz for 24 hours. For deeper insight regarding the action of Tz, the expression of ERBB-2, NOTCH1, EGFR, PIK3CA, and PTEN genes was measured using qPCR. Results (Figure 5) indicated that for MCF-7 all genes were downregulated upon treatment, while in MDA-MB-231 all genes were upregulated, with PIK3CA showing extreme upregulation (1904-fold change). In the murine BC cells 4T1, all genes were downregulated except for NOTCH1, which showed upregulation with a 2.4-fold change. In the normal human skin fibroblast cells, treatment with Tz yielded a downregulation of all genes except for NOTCH1 also but with a 254-fold change.

Gene expression analysis. In MCF-7, Tz treatment resulted in the downregulation of all genes while, in MDA-MB-231, the treatment resulted in the upregulation of all genes. For 4T1 and HSF Tz, treatment has resulted in the overall downregulation of all genes except for NOTCH1. C: control; Tz: trastuzumab; FC: fold change.
The differential gene expression patterns observed in response to Tz treatment across various breast cancer cell lines highlight the complex molecular effects of this drug. The unexpected upregulation of ERBB2 and other key signaling molecules in TNBC cells suggests that Tz may engage alternative pathways in HER2-negative contexts, potentially explaining its observed effects on cell viability and apoptosis in these cells.
The gene expression data presented in Figure 5 have been normalized using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the housekeeping gene, ensuring an accurate representation of relative expression levels across different samples and conditions. The y-axis in each graph represents the FC in mRNA levels of the specified genes in trastuzumab-treated cells compared with the untreated control group. Specifically, FC values greater than 1 indicate upregulation, while values less than 1 signify downregulation of the respective genes following trastuzumab treatment.
Gene expression analysis of breast cancer cells treated with trastuzumab. Relative mRNA levels of ERBB2, NOTCH1, EGFR, PIK3CA, and PTEN were quantified using qPCR and normalized to GAPDH expression. The y-axis represents FC in gene expression of trastuzumab-treated cells relative to untreated controls. Data are presented as mean ± SEM from 3 independent experiments (C: control, Tz: trastuzumab, FC: fold change).
Discussion
Although it is a disease with a wide range of molecular subtypes, breast cancer continues to be the most common invasive cancer in women and a leading cause of cancer-related death worldwide. Over the past 10 to 15 years, there has been a shift in treatment strategies to better account for this diversity. 4 As a specific targeting therapy, Tz seems to induce cell death in different breast cancer cells. The present study investigated the effect of Tz on 3 BC cell lines (MCF-7, MDA-MB-231, and 4T1). Although TNBC cells such as MDA-MB-231 and its murine resemblance 4T1 do not express HER2, still Tz could induce cell death via different mechanisms. A recent clinical trial has demonstrated significant clinical benefits from Tz in breast cancer patients with a HER2 low expression (immunohistochemical score of 1+ or 2+ and a negative in situ hybridization) (reviewed in Zhang et al 20 ). Another phase 3 clinical trial suggested that Tz conjugated with deruxtecan could benefit metastatic BC with low expression of HER2, and this might call for reevaluating the use of Tz either free or conjugated in patients with low expression of HER2. 21
In the present study, gene expression analysis revealed that Tz treatment led to a reduction in ERBB2 (HER2) mRNA levels in MCF-7 cells, which are known to express HER2. This transcriptional downregulation suggests that Tz’s effects extend beyond its well-established mechanism of binding to and inhibiting the HER2 protein at the cell surface. The observed decrease in ERBB2 mRNA could be the result of a negative feedback loop triggered by Tz-mediated HER2 protein inhibition, potentially involving transcription factors responsive to HER2 signaling pathways; however, this suggestion needs further work.
It is important to note, however, that mRNA levels do not always directly correlate with protein expression. While our study focused on transcriptional changes, future investigations should examine HER2 protein levels following Tz treatment to provide a comprehensive understanding of the drug’s effects at both the mRNA and protein levels.
Meanwhile, the present study revealed that Tz induced cell death in MCF-7 (HER2+) and the HER2− BC cells; MDA-MB-231 and 4T1. Although it is commonly known that only patients with HER2-positive tumors can benefit from Tz, recent reports indicated that Tz could induce cytotoxicity and DNA damage in both MCF-7 and MDA-MB-231 cells, 22 and hence patients with HER2-negative BC could benefit from Tz. The phosphorylation/activation of HER2 plays a role in this mechanism. 15 Phosphorylation of tyrosine 877 (Y877) in the HER2 protein is responsible for receptor activation even in the HER2− BC cells. Thus, in HER2−/pHER2Y877+ BC cell lines Tz could decrease cell proliferation. 15 Moreover, patients with low expression of HER2 are also sensitive to the antibody-drug conjugate Tz-emtansine (T-DM1). 16 While our study provides valuable insights into the transcriptional changes induced by Tz in TNBC cells, it is important to note that evaluating HER2 signaling at the protein level would offer a more comprehensive understanding of Tz’s effects. Future studies should consider examining both total and phosphorylated forms of HER2 protein in TNBC cells treated with Tz. This would provide crucial information on whether Tz affects HER2 activation and downstream signaling pathways in these cells, even in the absence of HER2 overexpression.
Tz acts by reducing the expression of HER2, limiting the progression of cancer cells. Different genes in the classical cancer pathway could be up-/downregulated by Tz 23 and affect mitochondrial function. 24
It has been well established that upregulation of HER2 and NOTCH1 is common in several human cancers, including BC. 25 Data revealed that Tz downregulated ERBB2, NOTCH1, and PI3K in the HER2 over-expressing MCF-7. These cells use NOTCH1 as a compensatory pathway to sustain the ERBB2 downstream signaling cascade that includes phosphatidylinositol 3-kinase (PI3K), allowing cells to survive the molecular targeted therapies such as Tz and trigger cellular invasion. 25 This explains how Tz downregulated NOTCH1 and PI3K. PTEN and EGF were also downregulated upon Tz treatment, although in HER2-positive metastatic breast cells, these proteins are not associated with the response to Tz. 26
Although Tz is an effective treatment for early-stage HER2+ breast cancer, 40% to 50% of patients with advanced-stage HER2+ breast cancer eventually develop resistance to the drug. Aberrant downstream signal transmission due to the loss of PTEN is a potential resistance mechanism. 27
In TNBC cells (MDA-MB-231), all studied genes were upregulated with PI3K being the most upregulated gene (1904-fold change) followed by ERBB2 (163.9-fold change). These results were contrary to the data reported previously where Tz treatment reduced the expression of ERBB2 in MDA-MB-231 cells.28,29 Meanwhile, 4T1, the murine resemblance of human TNBC, 30 exhibited an overall downregulation of genes under study except for NOTCH1. Because HER2 forms heterodimers with EGFR and HER3, the lower the HER2 the lower the EGFR, 31 and this might cause the aggressiveness of this cancer as in the case of the murine TNBC cells 4T1. When HER2 binds HER3, it attenuates its activity to stimulate PI3K/Akt signaling pathway 32 ; therefore, HER2 indirectly affects the PI3K and its downstream pathways. On the other hand, NOTCH1 was upregulated by the Tz treatment, although it is a conserved developmental signaling pathway, and its abnormal regulation is implicated in different types of cancer. 33
While our study provides valuable insights into trastuzumab’s effects on the MDA-MB-231 cell line, we acknowledge the inherent limitations of using a single human TNBC model. The contrasting results observed between MDA-MB-231 and 4T1 cells underscore this heterogeneity and highlight the need for caution when generalizing these findings to all TNBC subtypes.
Our results with MDA-MB-231 cells provide a crucial starting point, but future studies incorporating a wider panel of TNBC cell lines would undoubtedly offer a more comprehensive understanding of trastuzumab’s potential in TNBC treatment.
The complex interplay between various signaling pathways in TNBC, as evidenced by our gene expression analysis, suggests that trastuzumab’s effects may vary significantly across different TNBC subtypes. This variability underscores the importance of further investigation into the molecular mechanisms underlying trastuzumab’s action in HER2-low or HER2-negative contexts.
The contrasting mRNA expression patterns observed in 4T1 and MDA-MB-231 cells following Tz treatment warrant further discussion. While both are considered models of TNBC, their divergent responses highlight the heterogeneity within this breast cancer subtype. The upregulation of all studied genes in MDA-MB-231 cells, particularly the dramatic increase in PIK3CA expression, suggests a potential compensatory mechanism in response to Tz-mediated stress.
These data generally shed some light on the potential action of Tz in HER2− BC, though the molecular mechanism needs to be elucidated. However, this article calls for reevaluating Tz as a targeted therapy not only for HER2+ BC but for any type of cancer that barely expresses HER2.
Our study investigated the effects of trastuzumab on cell cycle modulation in TNBC cell lines, providing insights into the potential broader applicability of HER2-targeted therapies. The findings demonstrate that trastuzumab can induce G0/G1 cell cycle arrest in TNBC models, despite their typically low HER2 expression.
These results align with recent research on trastuzumab’s effects in various breast cancer models. Burguin et al 15 showed that trastuzumab could induce cell cycle arrest in HER2-negative breast cancer cell lines that overexpress phosphorylated HER2 at tyrosine 877 (pHER2Y877). This suggests that HER2 activation, rather than just expression levels, may be crucial in determining trastuzumab’s effects on the cell cycle in TNBC.
The potential of combination therapies is highlighted by Newton et al, 34 who reviewed emerging molecular targets in TNBC. They noted that trastuzumab in combination with other agents like (Poly (ADP-ribose) polymerase) PARP inhibitors can induce cell cycle arrest, particularly in BRCA-mutated TNBC. This underscores that targeting cell cycle regulation is a promising strategy for TNBC treatment.
It is important to note that the cell cycle effects of trastuzumab in TNBC may vary depending on the molecular subtype. As highlighted by Ryu and Sohn, TNBC can be classified into several subtypes with distinct molecular characteristics and treatment responses. 35 Future studies could explore how trastuzumab’s cell cycle modulation differs across these TNBC subtypes.
Our findings on trastuzumab-induced cell cycle arrest in TNBC also relate to potential resistance mechanisms. Kokot et al 36 recently showed that trastuzumab resistance in HER2-positive breast cancer is associated with alterations in cell cycle regulators. Similar mechanisms might be at play in TNBC, and understanding these could inform strategies to overcome or prevent resistance.
In conclusion, this study provides evidence for trastuzumab-induced cell cycle modulation in TNBC, aligning with current research on HER2-targeted therapies in breast cancer. The observed G0/G1 arrest suggests potential new strategies for TNBC treatment. These findings highlight the need for a more nuanced understanding of HER2 signaling in TNBC and open avenues for future research into optimizing HER2-targeted therapies across breast cancer subtypes.
Moving forward, it will be crucial to further elucidate the precise mechanisms by which trastuzumab modulates the cell cycle in TNBC, explore its effects across different TNBC molecular subtypes, and investigate potential resistance mechanisms. Translating these findings into clinically relevant combination therapies will be a key next step in improving outcomes for patients with TNBC.
While our study provides valuable insights into the transcriptional changes induced by Tz in TNBC cells, it is important to note that evaluating HER2 signaling at the protein level would offer a more comprehensive understanding of Tz’s effects. Future studies should consider examining both total and phosphorylated forms of HER2 protein in TNBC cells treated with Tz. This would provide crucial information on whether Tz affects HER2 activation and downstream signaling pathways in these cells, even in the absence of HER2 overexpression.
We recognize that our study is limited by the use of a single TNBC cell line (MDA-MB-231) and one concentration of trastuzumab. This restricts our ability to draw broad conclusions about trastuzumab’s effects across the heterogeneous landscape of TNBC. The observed effects may be specific to MDA-MB-231 cells and may not represent the response of all TNBC subtypes.
Limitations
Our study has several important limitations that should be considered when interpreting the results. First, we primarily used 1 TNBC cell line (MDA-MB-231), which may not represent the heterogeneity of TNBC; the inclusion of additional cell lines would provide a broader understanding of trastuzumab’s effects across different subtypes. In addition, our experiments employed a single trastuzumab concentration (20 μg/mL), limiting insights into dose-response relationships that could better define the drug’s therapeutic window. The in vitro nature of the study also poses limitations, as cell culture models do not fully capture the complexity of in vivo tumor biology; future studies using animal models or patient-derived xenografts would enhance the translational relevance of these findings. Furthermore, our analysis focused on mRNA levels, lacking protein-level examination, particularly for HER2 and its downstream effectors, which would give a more comprehensive view of trastuzumab’s impact. The study also evaluated trastuzumab as a single agent, and exploring its combination with other therapies could uncover potential synergistic effects. Finally, our observations were made at a single time point (24 hours), and a time-course study could better capture dynamic changes in cellular responses to trastuzumab treatment.
Conclusions
Our findings provide compelling initial evidence for trastuzumab’s broader anti-tumor activity beyond HER2-positive breast cancers, demonstrating its potential therapeutic effects in TNBC cells despite their low HER2 expression. Trastuzumab induced significant cytotoxicity, apoptosis, and cell cycle modulation in TNBC cell lines, comparable to its effects in HER2-positive cells. Gene expression analysis revealed unexpected upregulation of ERBB2 and other key signaling molecules in TNBC cells, suggesting trastuzumab may engage alternative pathways in HER2-negative contexts.
These results warrant further investigation into trastuzumab’s mechanisms of action in TNBC and its potential for repurposing. Future studies should examine HER2 signaling at the protein level, explore effects across additional TNBC cell lines and subtypes, and evaluate trastuzumab in combination with other targeted therapies for TNBC treatment.
While our results suggest potential therapeutic effects of trastuzumab in TNBC, these findings should be considered preliminary due to the limited scope of our study. Future research should expand on this work by incorporating multiple TNBC cell lines representing different molecular subtypes, employing a range of trastuzumab concentrations to establish dose-response relationships, and validating the findings in vivo using patient-derived xenograft models.
