Abstract
Background:
Historically, hepatitis C virus (HCV) pretreatment evaluation has required multiple visits, frequently resulting in loss to follow-up and a delayed initiation of treatment. New technologies can accelerate this process. We investigated the feasibility of a single-day evaluation program and its impact on evaluation completion, treatment eligibility awareness, and treatment initiation among people who inject drugs (PWIDs).
Methods:
HCV-infected PWID who were unaware if they were eligible for treatment were recruited in a prospective evaluation of an accelerated model of care between 2017 and 2019 and compared to a historical cohort. The patients underwent a medical evaluation, rapid HCV viral load testing, and transient elastography during a single visit, at the end of which they were informed whether they were eligible for treatment. A historical cohort of patients fulfilling the same inclusion criteria and evaluated with the usual standard of care spanning several visits who were examined at the addiction medicine clinic from 2014 to 2016 served as the comparison group.
Results:
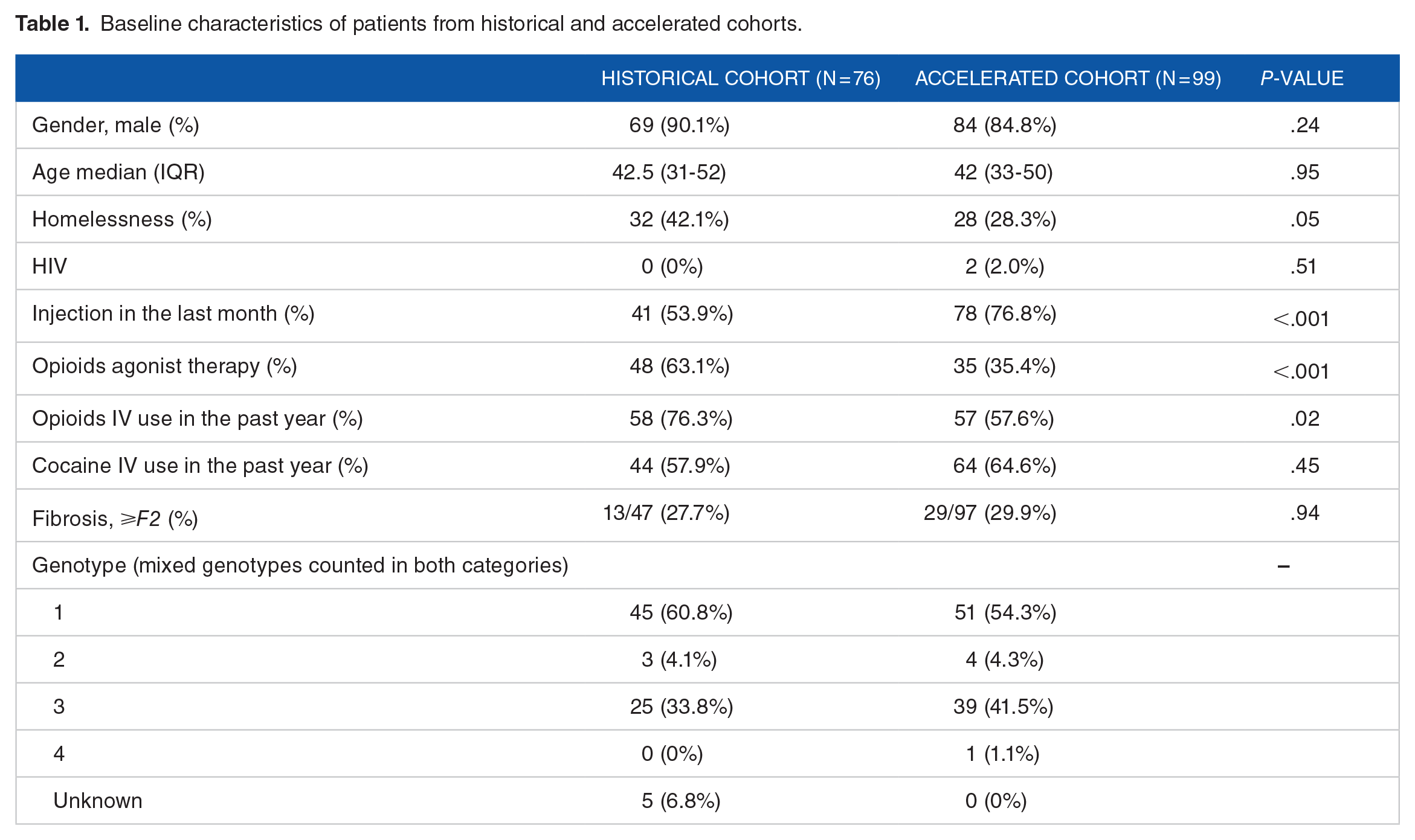
The accelerated and historical cohorts included 99 and 76 patients, respectively. The cohorts did not differ significantly by age and gender, but more patients in the historical cohort were undergoing opioid agonist therapy, while more patients in the accelerated cohort injected drugs in the last month. An accelerated evaluation resulted in a higher rate of evaluation completion (100% vs 67.1%; P < .001). Among those eligible for treatment, the proportion of those initiating treatment was similar between the groups (51/64 (79.7%) vs. 26/37 (70.3%); P = .28). The delay in the initiation of treatment was shorter in the accelerated cohort than in the historical cohort (69 (IQR: 49-106) days vs. 219 (IQR: 141-416) days; P < .001).
Conclusions:
Accelerated evaluation enhanced the awareness of eligibility and reduced the time to initiation among eligible patients.
Trial Registration:
This study is registered on www.clinicaltrials.gov (NCT02755402).
Introduction
Hepatitis C (HCV) treatment has been completely revolutionized by both the development of direct-acting antiviral (DAA) treatments with a 95% sustained virological response (SVR), the generalization of noninvasive hepatic fibrosis assessment methods, and access to treatment through front-line healthcare professionals (primary care providers, nurses, etc.).1-3 To achieve the World Health Organization’s elimination targets, there is a need to treat individuals who are most likely to drive ongoing transmission, such as people who actively inject drugs (PWID), in addition to those with liver disease.4-6 Despite concerns about reinfection risk and nonadherence to HCV treatment, there is some evidence that HCV treatment is effective in this population.1,7-10
HCV infection is typically diagnosed and assessed in several sequential steps, including antibody (anti-HCV) testing followed by confirmation of the presence of an active infection (HCV RNA), determination of the HCV genotype, and evaluation of liver fibrosis. This approach requires multiple visits, and there is a risk of attrition in between visits. 11 There is also a long delay between the diagnosis and initiation of treatment. In 2015, a retrospective analysis of charts of patients evaluated in a tertiary liver center of British Columbia reported delays of 66.3 to 119.5 months between diagnosis and referral to a HCV specialist and an additional delay of 6.8 months between the consultation and treatment initiation. As a result, only 25.6% of the referred patients started treatment. 12
In 2022, intravenous drug use remained the leading cause of HCV infection. The adverse social contexts of PWID, including homelessness, the need for income-generating activities to avoid drug withdrawal, and the lack of a supporting environment, impede their capacity to engage in the HCV care process.13,14 Altogether, delaying treatment, at the individual level, can result in disease progression, and among PWID populations, in the ongoing transmission of the infection. 15
Recently, owing to multiple advances in treatment and screening, the simplification of HCV management has been advocated for and included in several treatment guidelines.1,2,16 Point-of-care HCV antibody tests are currently on the market. Rapid HCV RNA testing has been developed and can be performed in central laboratories with tests, such as the Xpert HCV Viral Load assay (Cepheid, Sunnyvale, California, USA). 17 Liver fibrosis can be evaluated in 5 minutes through transient elastography, and biologic scores, such as FIB-4 or APRI, can be used to eliminate advanced fibrosis.18-20 With pangenotypic treatment, genotype determination is no longer required for the choice of an HCV regimen. Models of care integrating an accelerated HCV diagnosis with treatment processes were tested mostly in community-based, substance use treatment, and primary care settings.21-25 However, in many countries, treatment is not accessible in these settings and is rather mainly concentrated in hospital settings. 26 The feasibility of accelerated HCV care in hospital settings has not been extensively evaluated.
The primary aim of this study was to demonstrate the feasibility of a 1-day HCV pretreatment evaluation protocol and to compare the rates of evaluation completion, treatment rates, and delays in the initiation of treatment between this accelerated protocol and a historical standard evaluation process among treatment-eligible HCV-infected PWID. Throughout the course of this study, in 2018, treatment eligibility restrictions based on liver fibrosis were lifted, and all infected individuals, including those with mild or no fibrosis, became eligible for treatment. Hence, the protocol was amended to use this natural experiment to compare the rates of HCV treatment initiation between those who were or were not initially eligible within the accelerated arm.
Methods
Population
This was a prospective, non-randomized, open-label study that included retrospective controls. Participants in the retrospective component (historical cohort) were individuals who were examined at the Addiction Medicine Clinic of the Centre hospitalier de l’Université de Montréal (CHUM), a university hospital located in downtown Montreal close to many PWIDs’ dedicated resources, between October 2014 and June 2016. This clinic offers integrated medical and psychiatric services for patients with drug-related problems, including opioid agonist therapy (OAT). It implemented an HCV treatment program in 2010 with 2 dedicated HCV nurses within a multidisciplinary team of nurses, physicians, and social workers. Patients attending the clinic are either self-referred or referred by healthcare providers and various partner community-based organizations.
Participants in the prospective component (accelerated cohort) were recruited between March 2017 and May 2019, and the last follow-up visit occurred in May 2020. They were mainly recruited using the same referral pathways as those used in the Addiction Medicine Clinic. In addition, advertisements were posted in places frequented by PWIDs and in newspapers published by people experiencing homelessness. New patients seen directly at the Addiction Medicine Clinic during the accelerated cohort recruitment period were offered an opportunity to participate in the study before initiating an HCV evaluation.
To be eligible for the accelerated cohort, participants had to be 18 years old, had been injected at least once in the past year, HCV antibody positive, and unaware if they were eligible for treatment. Participants were excluded if they were already actively followed up in HCV care or were unable to provide informed consent. Participants who presented contraindications to medication (eg, pregnancy and breast-feeding) or transient elastography (eg, pacemakers and defibrillators) were excluded. The inclusion and exclusion criteria of the historical cohort were the same as those of the accelerated cohort, except for the possibility of obtaining informed consent.
Prospective intervention: Rapid evaluation
Participants were seen by a nurse and an infectious disease specialist at an appointment at the CHUM research center, which is located on the hospital premises.
The rapid evaluation protocol included standard blood tests (CBC, liver panel, etc.), rapid HCV RNA test (Xpert HCV Viral Load assay, Cepheid), transient elastography (FibroScan, Echosens, Paris, France), and HCV genotyping. Participants underwent a short biopsychosocial evaluation exploring their general medical history, HCV history, living conditions, history of substance use, and motivation to start treatment for HCV. Physical examination was performed to screen for the stigmata of cirrhosis. The results of the standard blood tests, HCV RNA, and transient elastography became available during the participant’s visit and allowed the investigator to determine whether the participant was treatable based on the reimbursement criteria at the time of the study (box). Information on potential treatment regimens and counseling was provided to the eligible participants (immediate treatment group). The participants were allowed to refuse treatment.
Evolution of criteria used in Quebec to determine eligibility to treatment.

As the Xpert HCV Viral Load assay was not approved in Canada at the time this study was conducted, the test was controlled in parallel with the COBAS AmpliPrep/COBAS TaqMan HCV quantitative test (version 2.0; Roche, Bâle, Switzerland), and the results were obtained generally 1 to 2 weeks after the initial visit and before obtaining the genotype result. On average, this was 3 to 4 weeks after the initial visit, at which time an appropriate DAA regimen was prescribed. There were no additional visits between the first visit and treatment initiation visit. The treatment initiation visit occurred once the medication was approved for reimbursement. The participant was seen again at weeks 2 and 4 of treatment, at the end of treatment (8 or 12 weeks depending on regimen), and then at weeks 12, 24, and 36 post-treatment.
Participants who did not fulfill treatment reimbursement criteria (delayed treatment group) were referred to the CHUM Addiction Medicine Clinic or Chronic Viral Infections clinic (if HIV-positive) for longitudinal follow-up of HCV infection and substance use. Participants were examined again at 6 months and 1 year to re-evaluate their treatment eligibility.
Protocol modification
In 2018, universal access to HCV treatment, regardless of fibrosis stage, was granted in Québec. The protocol was modified accordingly so that participants who were initially ineligible were actively re-contacted and offered treatment (delayed treatment group). Initiation and subsequent study visits were performed according to the same schedule as that for the immediate treatment group.
Outcomes
The primary outcome was the proportion of eligible patients who initiated treatment (immediate treatment group vs treatable historical controls). The secondary outcomes included the pretreatment HCV evaluation completion and time-to-treatment initiation.
The accelerated cohort participants were considered eligible for treatment if they fulfilled provincial reimbursement criteria at the time of their initial visit (box). The historical controls were considered eligible for treatment if they started medication, if treatment was prescribed but never initiated, if the physician documented in the record that the participant was treatable, and based on the investigator’s judgment, if the patient completed the evaluations but did not return to the clinic.
To examine how structural changes in treatment coverage affected outcomes, the proportion of patients in the delayed treatment group who could be successfully re-contacted after changes in policies was determined, and the proportion of patients who initiated HCV treatment was compared to those in the immediate treatment group.
Among the entire accelerated cohort, SVR results were determined in intention-to-treat (ITT) and modified ITT analysis, the latter excluding participants who died of unrelated causes, withdrew consent, were lost to follow-up, or had a reinfection before week 12 post-treatment, which was confirmed by a switch of genotype, subtype, or by sequencing.
Statistical analyses
The basic characteristics of the different subgroups are presented using descriptive statistics. We used chi-square and Fisher’s exact tests and the Mann–Whitney test for comparative analysis of categorical and continuous data, respectively.
Research ethics committee approval and registration
The protocol was approved by the research ethics committee of CHUM (approval number: 15.384), informed consent was obtained from all participants, and the study was conducted in accordance with the principles of the Declaration of Helsinki. It has also been registered at www.clinicaltrials.gov (NCT02755402).
Results
Accelerated cohort description versus historical cohort description
A total of 99 patients were enrolled in the accelerated cohort, and 76 fulfilled the inclusion criteria for the historical cohort (Figure 1). In both cohorts, most patients were men, and the median age was 42 years (Table 1). A higher proportion of patients in the historical cohort injected opioids in the previous year (76.3% vs 57.6%, P = .02) and were on OAT (63.1% vs 35.4%, P < .001), whereas more patients in the accelerated cohort than in the historical cohort injected drugs in the last month (53.9% vs 76.8%, P < .001). More patients in the historical cohort than in the accelerated cohort were homeless (42.1% vs 28.3%; P = .05). Approximately 30% of the patients in both groups had moderate fibrosis of grade F2 or higher.
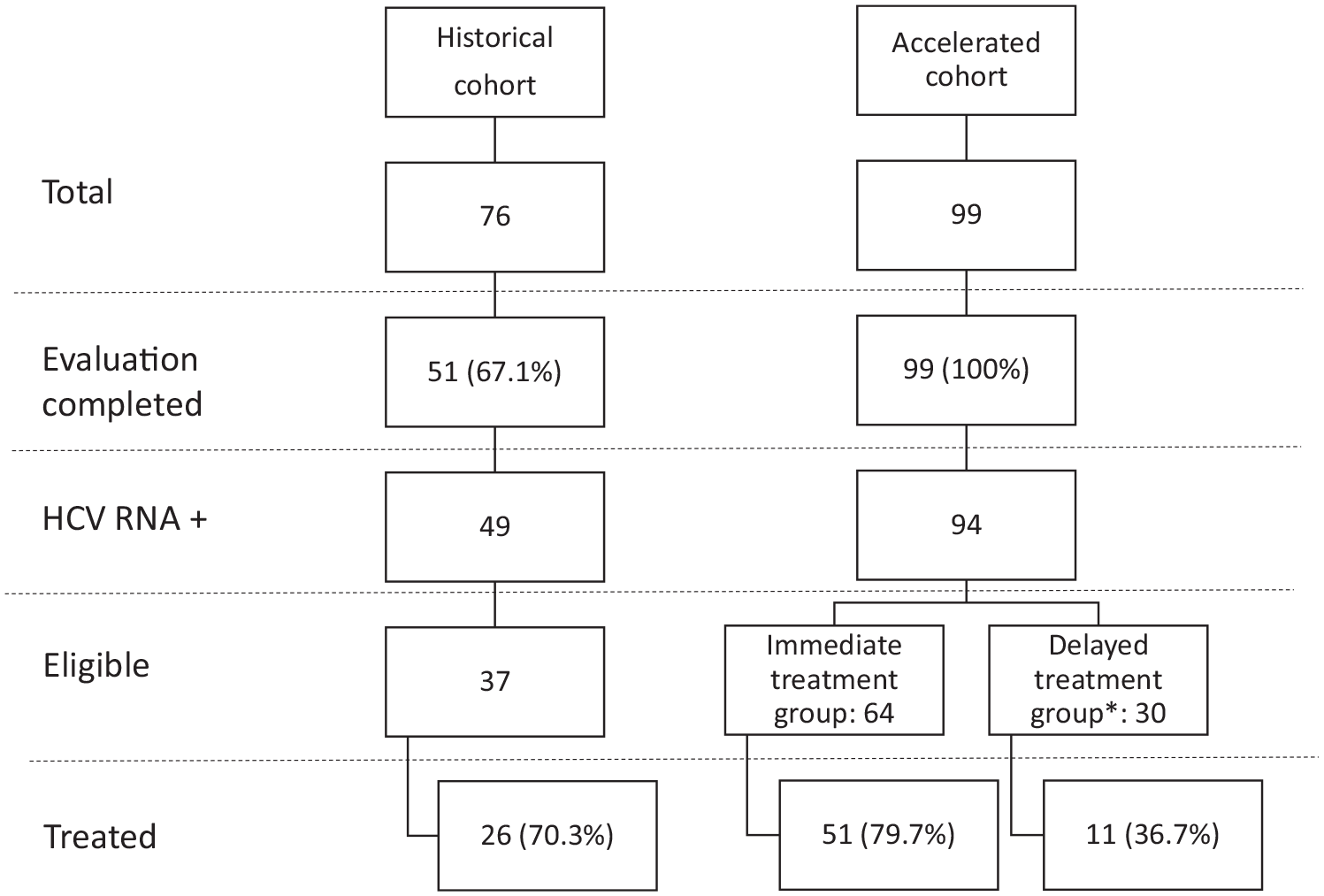
Study flow chart.
Baseline characteristics of patients from historical and accelerated cohorts.
An accelerated evaluation resulted in a higher proportion of evaluation completion (100% vs 67.1%; P < .001). Of the 99 accelerated cohort patients, 5 were HCV RNA-negative, 64 were initially eligible for treatment (immediate eligibility group), and 30 were initially ineligible (delayed eligibility group). Of the 76 patients in the historical cohort, 51 completed the pretreatment evaluation, and 37 were eligible for treatment.
HCV treatment initiation in the immediate eligibility group of the accelerated cohort versus historical cohort
There was no statistically significant difference in treatment initiation between the initially eligible participants in the accelerated cohort (immediate eligibility group) and the eligible participants in the historical cohort (51/64 [79.7%] vs 26/37 [70.3%]; P = .28) (Figure 2). Treatment initiation among the participants in these 2 groups was not associated with OAT (82.2% vs 71.4%, P = .21), IV drug use in the last month (80.6% vs 65.5%, P = .11), or homelessness (66.7% vs 79.7%, P = .17). The median time from the initial visit to treatment initiation was 69 days (IQR: 49-106 days) in the accelerated treatment group versus 219 days (141-416 days) in the historical cohort (P < .001).

Treatment initiation among the different groups.
Immediate and delayed eligibility groups of the accelerated cohort
Except for a significant fibrosis stage and a trend toward more patients injecting drugs in the last month among patients in the immediate treatment group, patients in the immediate and delayed treatment groups were similar (Table 2).
Baseline characteristics of patients from immediate and delayed treatment groups.
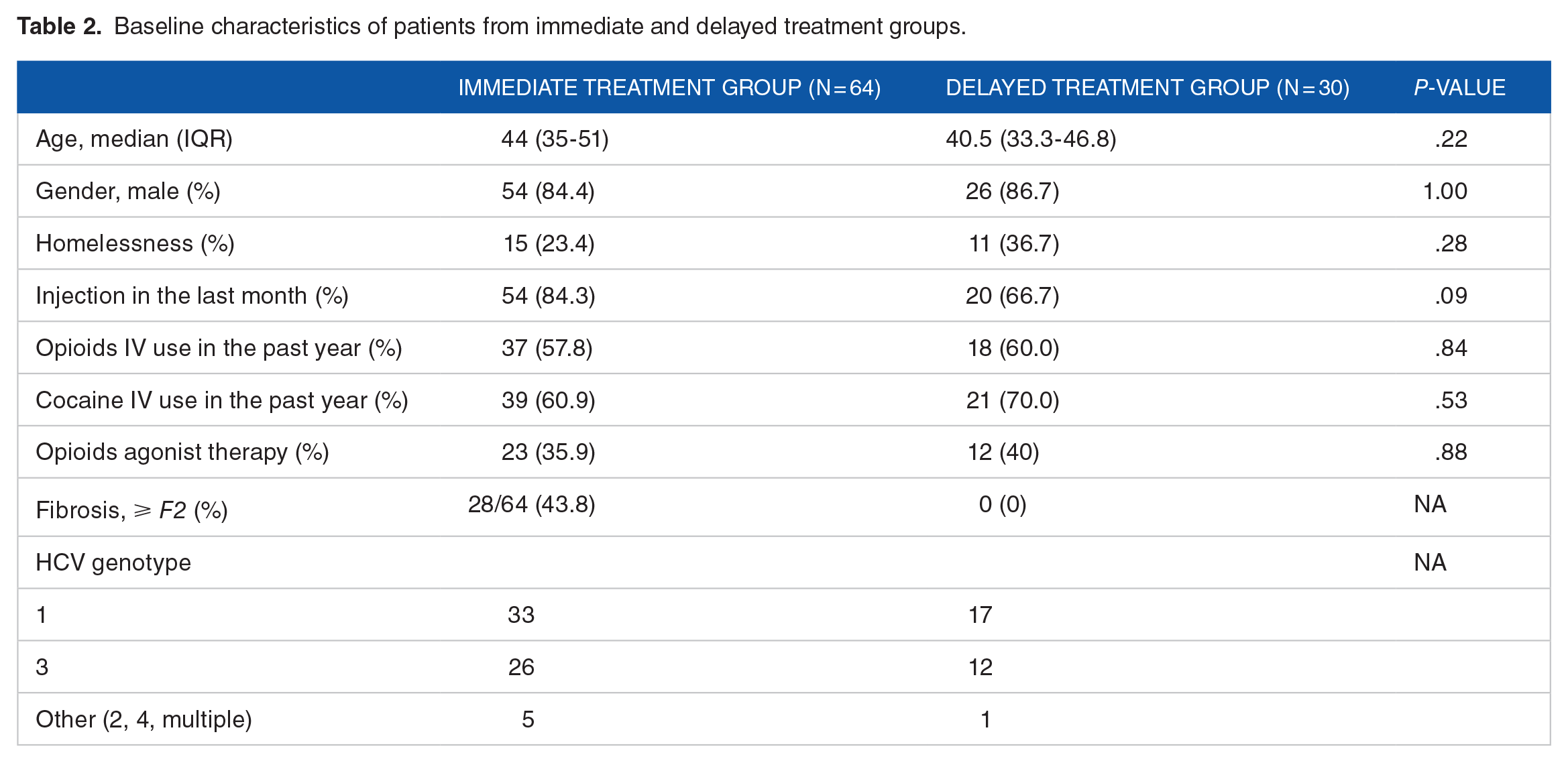
Patients in the immediate treatment group were significantly more likely to initiate treatment (79.7% vs 36.7%, P < .001) (Figure 2). Nine of the 30 patients in the delayed treatment group were lost to follow-up, while 10 were successfully re-contacted but were not treated for diverse reasons: 4 moved or started care at other sites, 2 did not complete the medication insurance process, 2 had treatments prescribed but never initiated, 1 wanted to delay treatment, and 1 withdrew consent.
The immediate-eligibility group included patients seen before (n = 29) and after (n = 35) universal access to treatment. Of the 35 patients seen after March 2018, 25 would not have been eligible if the criteria did not change. Among these 25 patients, 18 initiated treatment versus 11 of the 30 patients in the delayed treatment group (72.0% vs 36.7%, P = .01).
Sustained virological response
Of the 62 patients treated, 26 received sofosbuvir/ledipasvir, and 36 received sofosbuvir/velpatasvir. In the intention-to-treat (ITT) analysis, 82.3% achieved SVR. Three participants had undetectable disease at the end of treatment and were reinfected before week 12 post-treatment (confirmed by a switch in genotype), two were lost to follow-up, 2 died of reasons unrelated to treatment (overdose and pulmonary embolism), and one participant withdrew consent. Excluding these participants, only 3 had virological failure, resulting in an SVR of 94.4% in the modified ITT. Of those who had virological failure, 2 had incomplete treatment duration with sofosbuvir/ledipasvir (7 and 14 days). The third patient presented with genotype 3 infection and stage F3 fibrosis and underwent 12 weeks of sofosbuvir/velpatasvir therapy. Sequencing at the provincial laboratory confirmed relapse.
Discussion
This study demonstrates the feasibility of accelerating the HCV pretreatment assessment process in PWIDs disengaged from the healthcare system by using rapid tests (Xpert HCV Viral Load assay or transient elastography). It allows the pretreatment evaluation to be completed more often and can significantly speed up treatment initiation, with a median delay reduced threefold. Moreover, our results suggest a detrimental impact of administrative delays on treatment initiation, with only one-third of the participants initially not eligible ending up starting DAA after extension of the reimbursement criteria. Finally, it demonstrated a high SVR rate with only 1 virological failure after complete treatment.
Loss to follow-up is a major challenge at all steps of the HCV treatment cascade. For example, in the Netherlands, it is estimated that 14% to 64% of patients diagnosed with HCV but not treated are currently lost to follow-up. 27 It is also well-demonstrated that a significant proportion of patients diagnosed or linked to HCV care do not initiate treatment. Van Dijk et al. compiled data from clinical studies conducted during the DAA era and reported that only 12% to 77% (median = 29%) initiated treatment. 11 They also described that this proportion could be improved to 16% to 100% (median = 73%) with specific interventions, such as patient navigator or colocalization of services.11,28,29 In our study, we demonstrated that an accelerated approach can result in a significantly higher proportion of patients completing pretreatment evaluation. However, we could not demonstrate a significant increase in treatment initiation in the accelerated cohort compared with the historical cohort.
Although initially unplanned, the change in the reimbursement criteria gave us the opportunity to assess the potential effect of delaying treatment for administrative reasons. Despite efforts to trace back patients in the delayed eligibility group, only 36.7% could be treated. In a population that is difficult to link to the health care system, a delay in treatment is an additional risk of loss to follow-up, as was the case in one-third of our patients. This result is in the same range as those found in 2 studies conducted in the Netherlands. They showed that despite actively trying to trace back patients, only 29.2% and 17.4% of patients were lost to follow-up and were eligible for tracing.27,30 In a research setting, the staff may have time to actively seek out patients, but this is generally not possible to the same extent for clinical staff, who are often overloaded with patients already scheduled. Efforts are required to identify these patients, and many cannot be traced back. In addition, one-third of our patients were not lost to follow-up but did not initiate treatment, suggesting that when a person engages in care, it is important to complete the process, including initiating the treatment as soon as possible, as competing priorities can rapidly interfere.
Patients included in this study were generally considered challenging to treat: 78.8% injected drugs in the last month, only 35.4% on OAT, and patients aware of their HCV diagnosis but not linked to care. Nevertheless, 82.3% of patients achieved SVR in the ITT group and 94.4% of those in the mITT group achieved SVR. The latter result is similar to those reported in registration trials.31-34 Only 1 patient presented with virologic failure despite full treatment, but this patient had known risk factors for failure (genotype 3 with advanced fibrosis). 34
In recent years, many approaches to the management of HCV have been proposed, including decentralization of treatment to primary care clinicians or community resources, use of patient navigators or case managers, simplification of pretreatment algorithms, etc.1-3,16 Through reorganization of the testing procedure to avoid long waiting times and multiple appointments, our approach is intended to be patient-centered. We conducted our study in a hospital setting; however, our approach is adaptable to primary care or community settings, taking care of enough patients to justify the necessary equipment. In the ETHOS Engage study, investigators also relied on the Xpert HCV Viral Load assay and transient elastography in OAT clinics and needle and syringe programs. 35 In our study, access to transient elastography at the site accelerated the assessment of liver fibrosis at a time when at least moderate fibrosis was required for reimbursement of treatment. In many settings, this is no longer a prerequisite for reimbursements. Conversely, access to an HCV RNA test remains a potential barrier to treatment in 2022, as well as an opportunity for loss to follow-up.11,16 As the diagnosis of HCV is still performed in 2 steps, even when using reflex algorithms where samples from newly positive patients are sent immediately for RNA testing, there is usually a delay of several days between the screening test and confirmation of an active infection. In our study, the use of a delocalized HCV RNA test allowed for the rapid confirmation of a diagnosis of an active infection in patients who knew they were infected but did not know their RNA status. In fact, 5% were RNA-negative, even though they believed they were chronically infected.
The main limitation of this study is the use of the historical cohort as the control group, which may have tempered our ability to demonstrate a difference in treatment initiation between the accelerated and historical cohorts. One explanation relates to evaluation completion. Among the historical cohort, 32.9% of patients did not complete the pretreatment assessment, consequently precluding us to determine their eligibility for treatment, whereas eligibility could be determined for all patients in the accelerated cohort. In the historical cohort, the denominator of the proportion entering treatment, therefore, represents a group of patients sufficiently motivated to go through a longer evaluation process and probably more inclined to start treatment than those who dropped out along the way. Additionally, despite the fact that patients were recruited using the same methods and we tried to identify potential differences between the groups, unmeasured contextual and individual confounders probably remained. Motives to remain linked to care might have differed between patients in the 2 cohorts. Those of the historical cohort were referred to the clinic for substance use, and treatment for HCV was a secondary benefit of co-location of services at the same site, whereas HCV treatment was the primary reason for consultation for the accelerated cohort. Substance use management might have been a stronger motivation to stay linked to care than was HCV management. Furthermore, historical cohort patients were likely more stable, as a higher proportion was on OAT and fewer were injected in the last month. Finally, although patients were evaluated in a single visit with the accelerated approach of this study, the delay consecutive to awaiting the insurer’s response after the prescription of DAA may have negatively impacted the initiation of treatment in the accelerated cohort.
Despite accelerated evaluation, there was still a delay between the determination of eligibility and treatment initiation. In the present study, the median treatment duration was 69 days. This delay was multifactorial, but it was mainly due to the performance of the genotype test and the approval of the drugs for reimbursement. With the availability of pangenotypic drugs, genotype is no longer required to initiate treatment in Québec, and for patients with government insurance, approval times have significantly shortened over the last year. However, this last point is not the case in any jurisdiction.
Finally, our study recruited participants with known HCV antibodies. The ultimate fast-track algorithm should start with antibody testing in patients unaware of their status, but such an approach could prove to be inefficient in areas with a low prevalence, and given the limited staff resources, it is difficult to implement in the hospital environment. In addition, by 2022, a large number of patients with known HCV antibody positive remain untreated, confirming the relevance of our approach. Indeed, we targeted patients who had an antibody test but no HCV RNA test or those who had an HCV RNA test performed but did not show up for a first appointment. In the first case, the literature reports that around 30% of patients did not receive their RNA result. 11 Rates of missed first appointments are very variable. In screening studies conducted in emergency rooms, the proportions are often very low (14%-35%), while in other settings, they reach 27% to 91%.11,36 Approximately 50% of PWIDs show up for their first appointment. 11
Conclusions
We developed and demonstrated the feasibility of a hospital-based pretreatment evaluation model that can be used in jurisdictions where HCV care is still restricted to hospitals but could also be implemented in primary care settings. At a time when HCV pretreatment assessment and the treatment itself have significantly evolved, this protocol enhanced awareness of eligibility for treatment and reduced the time to initiation of treatment in HCV-positive PWID disengaged from the system. Finally, we demonstrated that, in addition to an accelerated evaluation, reducing administrative barriers to allow rapid initiation of the treatment itself is essential to avoid loss to follow-up.
Footnotes
Acknowledgements
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Gilead (protocol number: IN-CA-337-2100). Cepheid lent the GeneXpert machine. VML is funded by Chercheur-boursier clinicien Junior 2 – FRQ-S. JB holds the Canada Research Chair in Addiction Medicine.
Declaration of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VML is reporting having received advisor fees from AbbVie and research grants from Gilead Sciences and Merck, outside of the current work. JB is reporting having received advisor fees from Gilead Sciences and AbbVie and a research grant from Gilead Sciences, outside of the current work. SB, CWB, LCJ, MP and MEG have no conflicts of interest to report.
Author Contributions
Regarding authors’ contribution, VML and JB conceptualized and designed the study. The other authors commented on the study design. VML obtained funding, supervised the study and performed data analysis. All authors contributed to study acquisition and interpretation. VML, SB, CWB, and JB drafted the manuscript and the other authors critically revised the manuscript for important intellectual content. All authors approved the final version of the manuscript and agreed to be accountable for aspects of the work.
