Abstract
Objective:
Describe the experiences and perspectives among pregnant people with chronic HCV infection receiving ledipasvir/sofosbuvir (LDV/SOF) therapy during pregnancy.
Methods:
We conducted semi-structured, in-depth interviews within an open-label, phase 1 study of LDV/SOF therapy among pregnant people with chronic HCV infection. Participants took 12 weeks of LDV/SOF and were interviewed at enrollment and again at the end of treatment. We transcribed the interviews verbatim and coded them with NVivo software for subsequent inductive thematic analysis.
Results:
Nine pregnant people completed the study, leading to 18 interview transcripts. All participants identified as women. Eight women acquired HCV through injection drug use, and 1 through perinatal transmission. We identified 3 themes. (1) Treatment for HCV during pregnancy with LDV/SOF was tolerable and convenient. (2) Women described that taking investigational LDV/SOF increased their self-esteem and sense of well-being due to possible cure from HCV, and they felt that the experience of working hard to achieve cure may potentially prevent return to drug abuse in the future. (3) Women appreciated researchers and providers that gave non-judgmental care and communicated honestly, and preferred person-centered care that acknowledges women’s individual needs.
Conclusions:
Women stated that cure from HCV would be “life-changing,” and described antepartum treatment for HCV with LDV/SOF as tolerable and desired, when provided by non-judgmental providers. Antepartum treatment was found to be acceptable by study participants and should be further evaluated to combat the increasing HCV epidemic among young persons, including pregnant people.
Keywords
Introduction
Hepatitis C infection has increased significantly with the ongoing opioid epidemic, especially among women. From 2004 to 2014 there has been an estimated 4-fold increase of rates of acute hepatitis C virus (HCV) infection among women compared to a 2-fold increase among men.1,2 As well, from 2011 to 2014 there has been an estimated 68% increase in the proportion of infants delivered by HCV-infected people with approximately 1700 infected infants born annually in the United States. 3 Pooled analysis shows a 5.8% risk of vertical transmission of HCV, and interventions such as cesarean section do not prevent transmission.4-6
Treating chronic HCV among reproductive-aged and pregnant people is crucial to mitigating new infections and preventing perinatal acquisition. Direct acting antiviral agent ledipasvir/sofosbuvir (LDV/SOF) is an effective treatment for chronic HCV that has reasonable safety among pregnant animal models. 7 Concerningly, after discovering chronic HCV status due to HCV screening in pregnancy, people are often not able to obtain treatment postpartum.3,8-10 Pregnancy may represent a valuable opportunity of insurance access and retention in clinical care where people can obtain HCV treatment. While prior qualitative studies of non-pregnant people with chronic HCV have shown that people perceive their children and relationships with families as motivating influences to obtain testing and treatment, no prior work has investigated the perspectives of pregnant people with chronic HCV infection.8,11-14 Pregnant people were also excluded from the LDV/SOF efficacy trial. 15 Barriers to treatment in pregnancy include lack of efficacy data in pregnancy, stigma for chronic HCV and drug misuse by healthcare providers, and concerns that women are at higher risk for drug non-adherence.16-18
Qualitative interviews included in a recent phase 1 open-label trial of LDV/SOF in pregnancy investigate experiences and perspectives of participants, alongside efficacy and safety data. 19 Participants of the trial have critical insight on living with chronic HCV, acceptability of LDV/SOF, issues with adherence to treatment, and effective care and communication from providers and research staff. Through semi-structured qualitative interviews of participants of the phase 1 trial, we describe the experiences and perspectives among women with chronic HCV infection receiving ledipasvir/sofosbuvir (LDV/SOF) therapy during pregnancy
Methods
Qualitative interviews were included in the design of an open-label, phase 1 pharmacokinetic evaluation of LDV/SOF administration during pregnancy (ClinicalTrials.gov Identifier: NCT02683005). We chose qualitative methods alongside the pharmacokinetic investigation in order to investigate pregnant peoples’ complex experiences of living with and undergoing investigational treatment for chronic HCV. The University of Pittsburgh Institutional Review Board approved the study protocol. Food and Drug Administration Investigational New Drug Application (#129502) from the investigator (C.C.) at the University of Pittsburgh allowed the investigational use of LDV/SOF in pregnancy. All participants gave written informed consent before the screening procedures and were eligible if they had chronic HCV infection and were between 18 and 39 years of age and between 23 and 24 weeks of gestation at the time of enrollment. At enrollment, pregnant people completed a screening questionnaire including demographic data, self-reported risk factors, and route of HCV acquisition.
For the qualitative portion of the study, we interviewed all participants of the pharmacokinetic trial. Research team members experienced in the patient population and in qualitative methodology (E.K. and C.C.) designed semi-structured interview guides to capture perspectives regarding chronic HCV infection and investigational treatment during pregnancy. In order to ascertain experiences prior to study participation and experiences related to study participation itself, we interviewed participants at enrollment and after completion of the study medication. Enrollment and end-of-study interview guides are included in the Supplemental Appendix.
For the pharmacokinetic portion of this trial, participants initiated a 12-week course of ledipasvir 90 mg-sofosbuvir 400 mg fix-dose combination (LDV/SOF) starting between 23 and 24 weeks of gestation, and were monitored for pharmacokinetics and drug safety. Research staff called participants weekly to discuss drug adherence and any participant concerns. Three pharmacokinetic visits occurred at 25 to 26, 29 to 30, and 33 to 34 weeks gestation. Delivery data was collected if participants delivered at Magee Womens Hospital. All participants completed a postpartum visit 12 weeks after completing LDV/SOF including maternal and infant viral loads and we also conducted infant visits at 6 and 12 months. Compensation for each visit with the study team included $50 at screening, enrollment, and at the postpartum visit. They received $250 for each intensive pharmacokinetic visit and $150 during delivery hospitalization. Infant follow up visits were compensated at $50 for each visit. Transportation services were provided as needed, such as cab fare or bus fare. For full investigational drug study protocol including full inclusion and exclusion criteria, please see Chappell et al 19 in Lancet Microbe.
We enrolled pregnant people from October 1, 2016 to September 30, 2018 within Magee-Womens Hospital in Pittsburgh, Pennsylvania. Magee is a large, tertiary care maternity hospital with approximately 10 000 deliveries each year. We recruited pregnant people with print advertisements and through purposive sampling by referral from prenatal or substance use treatment providers. All study participants were interviewed one-on-one with semi-structured interviews at enrollment and after completing the study medication as described above. A 12-week interim is required after completing treatment to assess for cure from HCV, thus participants completed the qualitative portion of the study before learning if LDV/SOF use had cured them from chronic HCV.
Two experienced interviewers (F.C. and M.H.) conducted all interviews in-person within a private research office at Magee Womens Hospital. Interviewers had prior experience in allowing participants to lead the discussion and explore topics not included in the semi-structured guide, and the participants had no prior exposure to the interviewers. We recorded and transcribed them verbatim. Under the guidance of a senior qualitative researcher (J.C.), 2 trained researchers (Y.K. and E.F.) developed a qualitative codebook from all 18 transcripts using an editing approach to ensure that all relevant topics and themes are represented. 20 Codes were selected to describe content of interviews, for example, “Beliefs about Hep C’s Impact on Life,” and “Medication Adherence.” Y.K. and E.F. independently completed coding and met to compare the codes. To refine codes, discrepancies were discussed and resolved, codes were merged and altered in an interactive fashion to create a final codebook that both coders agreed upon. Any unresolved disagreements in interpretation were to be adjudicated by a third senior investigator (J.C.), but there were no unresolved disagreements.
During coding we monitored for thematic saturation, which occurs when researchers no longer identify new ideas in the data, and allows researchers to understand appropriateness of sample size and robustness of the data for the topic and population of interest. 20 We noted thematic saturation after coding the first 10 transcripts (5 enrollment transcripts and 5 end-of-study transcripts). NVivo software was used to organize and store qualitative data analysis. Y.K. performed thematic analysis by inductively assessing coded interviews for themes consistent with general qualitative inquiry.21-24 We sought to describe what participants experience with minimal inference from specific theory-based frameworks and thus chose a general qualitative inquiry approach. Thematic analysis of the transcripts focused on the components of the interview where participants discussed their perspectives on HCV care received before and during the study, and experiences with LDV/SOF therapy during the study. Identified themes were discussed with the larger study team (Y.K., C.C., F.C., J.C., M.H., E.K.) and compared to original interviews to ensure representative reporting. Final themes review participants’ experiences with HCV infection, experiences with LDV/SOF therapy during the study, and perspectives on the behaviors of researchers and providers.
Results
Nine pregnant people completed the study and all identified as women. All 9 women completed their enrollment and end-of-study interviews (Figure 1), resulting in 18 transcripts for analysis. Demographic data is reported in Table 1. All participants identified as white. Eight reported acquiring HCV through injection drug use and 1 reported perinatal acquisition. Most women had used recreational drugs (marijuana, cocaine, methamphetamines, or opioids) within the last year or at any time in their lives. Four women were using opioid maintenance therapy during the study, 2 were taking methadone and 2 were taking buprenorphine. We identified the following major themes. (1) Treatment for HCV during pregnancy with LDV/SOF was tolerable and convenient. (2) Women described that taking investigational LDV/SOF increased their self-esteem and sense of well-being due to possible cure from HCV, and they felt that the experience of working hard to achieve cure may potentially prevent return to drug abuse in the future. (3) Women appreciated researchers and providers that gave non-judgmental care and communicated honestly, and preferred person-centered care that acknowledges women’s individual needs. We discuss major themes and associated subthemes below.
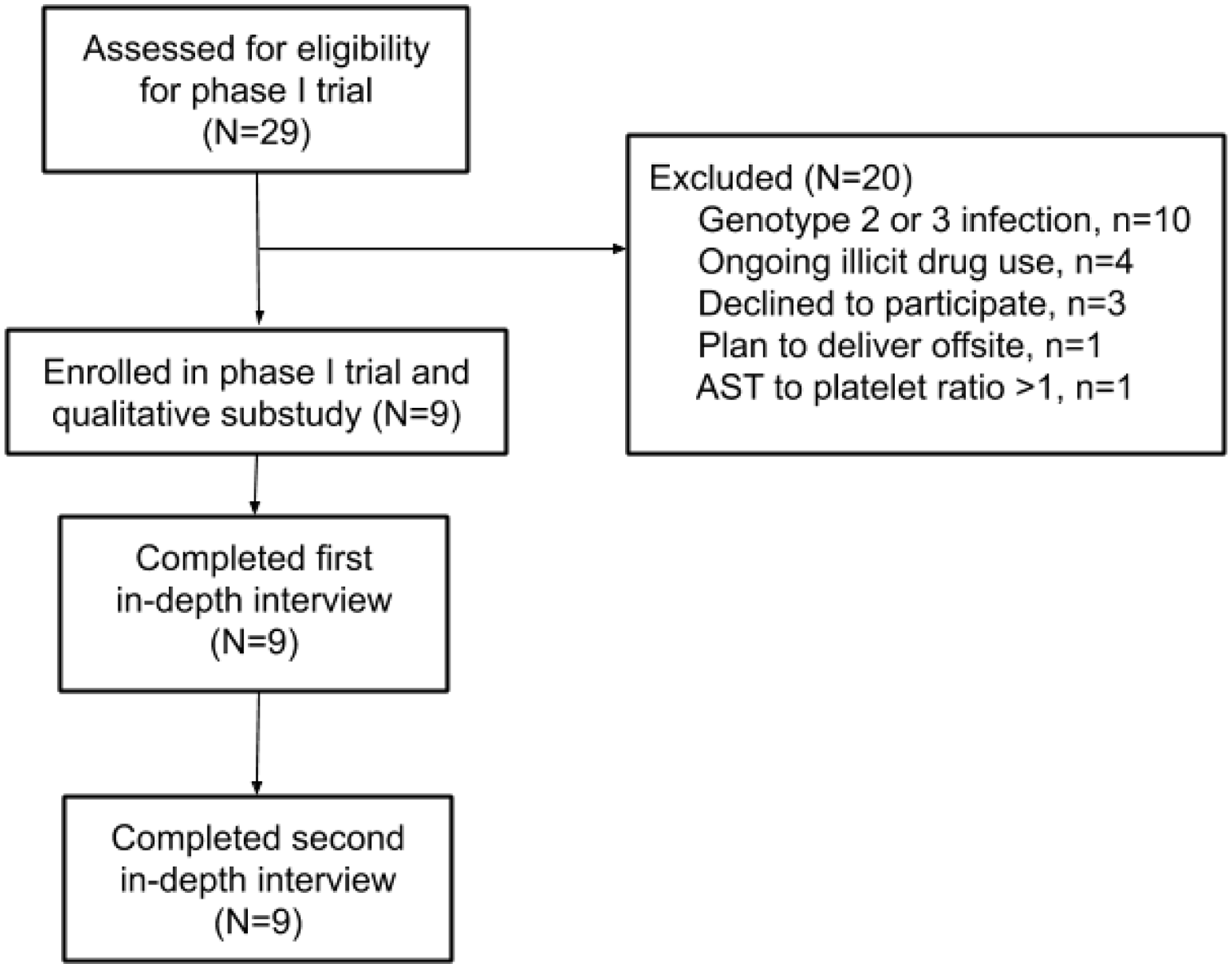
Flow diagram of participant inclusion and participation. For full inclusion/exclusion criteria and phase 1 open-label trial methodology, please see Chappell et al., Lancet Microbe. 19
Demographic characteristics.

Theme 1. Treatment for HCV during pregnancy with LDV/SOF was tolerable and convenient
Medication was tolerable, and women differentiated symptoms of pregnancy and side effects of the investigational drug
Women stated that it was easy to swallow the pill, and most participants reported no side effects from study medication. A few women noted that at the start of treatment they felt tired, but that this side effect was short lived and resolved before study completion. Contrastingly, other participants described feeling decreased fatigue immediately after starting the medication. A few participants reported headaches with the start of medication, which they attributed to the drug. However, participants further described how the headaches resolved after less than a month or by taking the medication with food. Other women noted at the start of pregnancy they experienced morning sickness, and they did not feel this was due to the study drug but due to pregnancy itself.
Treatment was convenient, and women discussed reminder techniques and reasons to forgo the daily pill
Most women discussed that it was very easy to adhere to medication during pregnancy. Many women had heard of others’ experiences with Interferon and were pleased to learn that LDV/SOF is an oral medication. Women discussed that taking it every day with their prenatal vitamin, or other daily medications (eg, subutex, psychiatric, or blood thinner medications), made it easy to recall taking the pill. Other women remarked that they were able to remember taking the medication despite caring for young infants and that being a mother was motivating to remember the pill. One woman stated that she took the medication in alignment with her baby’s schedule, always taking the medication shortly after changing her in the morning and stating that her daughter was “better than my alarm clock.” Many women discussed their motivation for cure and desire to avoid perinatal transmission as factors that facilitated adherence to LDV/SOF therapy. One woman said,
What made [remembering the pill] easy?
‘Cause it was saving my life and my baby’s. . . and I’m an adult. I can take medication. On time. Every day.”
Some women found that keeping a daily chart to record taking the medication was a helpful tool, while others reported that phone alarm reminders or taking it at the same time every day was helpful. Some women discussed that daily text reminders could be helpful, but few women utilized the daily text reminders that were made available through the study. Women also noted that the doctors explaining the effect of a missed dose as helpful, 1 participant described that “just knowing that it’s important that I take it every day . . .after talking to the doctors and them explaining. . .you can build up a resistance if you don’t take it regularly. . .”
Some women discussed the medication in the context of prior institutional involvement, for example 1 woman compared the medication to probation, “I have to take it, I have no choice.” Another participant discussed her history with rehabilitation centers and halfway houses, where the institutionalization of medication administration through a “nursing window” between “8 am and 8:30 am or you don’t get your meds and you get written up,” gave her the discipline to take daily medications on her own. Another participant noted that her partner set a reminder on his phone and that helped her to remember not only to take the medication, but also to take it at the same time every day.
Reasons to forget the medication included simply forgetting, travel, or return to opiate abuse. For example, some women did note that some days they “just forgot” to take the medication, and 1 participant stated that this is because it just wasn’t in her routine. When asked how her recall could be helped, she stated that she didn’t expect the study team to do more to help her remember, but that she felt she needed to be ultimately responsible for her own medications. Another woman described living in a shelter that held her medications for her, and when she would go to see her children on weekends it was harder to remember to take the medication because the weekend travel disrupted her routine. One participant experienced return to opiate abuse during the study, and she stated that she did not adhere during her relapse. This participant noted that she was admitted to a hospital for a painful hip infection, and she cited hip pain as the reason she returned to opiate abuse. In the following, she explains that her concern for the pregnancy and receiving medications during hospitalization were motivating factors to restart study treatment and to stop opioid abuse,
What triggered you deciding to not use?
Well my son, for one. I was scared of what I was doing to him with the drugs. . . and coming into the hospital and being admitted was a really big help because you know you can’t just up and leave and go use. . . and you know being treated for the infection and everything it was just easier cuz they give me my meds in the morning. . .”
Theme 2. Women described that taking investigational LDV/SOF increased their self-esteem and sense of well-being due to possible cure from HCV, and they felt that the experience of working hard to achieve cure may potentially prevent return to drug abuse in the future
Positive emotional effects from treatment with LDV/SOF
Women in general reported an improved sense of self-esteem related to taking study medication, stating that they “felt better” about themselves or found study participation to be a “self-esteem booster.” Women linked these positive feelings from study participation to hope for cure from HCV, 1 woman described that cure from HCV would be “life-changing.” When asked how cure from chronic HCV would make her feel, another woman responded with feelings of self-accomplishment and pride, stating that cure would make her feel, “ecstatic, grateful. . .kind of proud that I went through with something and accomplished it.”
Treatment for chronic HCV is potentially preventive against return to drug abuse
Among women whose acquisition was related to injection drug use, taking study drug and possible HCV cure was discussed in association with a departure from prior behaviors of drug abuse or sharing needles. When asked how her self-perception might change, 1 participant reported, “I think I’m. . . a bright thing and it’s a clean start.” Many participants discussed treatment and cure from HCV as a preventive factor for return to opioid abuse. For example, 1 participant explained, “I’m down to like barely detectable. . . I think it’s definitely gonna help me not wanna keep relapsing or using because this has been such a process trying to cure it. . . that’s not the life I wanna live anymore, I don’t wanna use, I don’t wanna be an active at addict anymore and now I really believe thinking and remembering this experience will definitely help me to stay on a straighter and narrow path.”
Other participants noted that seeing viral loads go down made them proud of themselves, and they felt they would be less likely to return to drug abuse since they did not want to contract HCV again. Many people did not think they could access treatment in the future if they did contract the disease again. All these factors were identified by participants as incentives to avoid return to opioid abuse.
Only 1 participant discussed that cure from HCV would not prevent her from using opiates in the future. In the following quote, that participant describes a prior experience of being in a study for Hepatitis C treatment and relapsing before taking therapy,
Whenever I first found out I had it, I went. . . to the study. . . It’s been a few years. . . .I got the blood work done, and did the whole consultation thing. But they were like, this is a one shot thing, and if you mess it up, you’re not going to be able to get treatment ever. So. . .I went for the consultation, did the blood work, and I started using. Like I didn’t really stop using, I just didn’t let my family know I was using. . . .it was really just to get my parents, my sister and stuff, off my back. To go talk to somebody and stuff. But I wasn’t ready to quit using drugs.
What prevented you from going through with the treatment?
It’s just I didn’t want to stop using drugs. I wasn’t ready to act like a mom to my son. I wasn’t ready to, I don’t know, I just wasn’t ready for any of that. Like, that sounds weird, but you would think, oh if a drug user is given the opportunity to not use, and they get help, why don’t they take it? ‘Cause they’re just not ready. . . .you have to be ready, and I wasn’t.”
She later stated that her biggest protective factor was “I’m not willing to lose my kids.” But she acknowledged that return to drug abuse is always possible with addiction, stating that “even though I don’t plan on using. . . I’m an addict, there’s always that chance that I could. . . be clean for twenty-something years and go back. I’ve seen it. I’ve seen people die after twenty years, going out once. . .so I work the program. I have support. I don’t associate with the people that use anymore.”
Theme 3. Women appreciated researchers and providers that gave non-judgmental care and communicated honestly, and preferred person-centered care that acknowledges women’s individual needs
Researchers and providers should practice non-judgmental and honest communication to encourage participant trust
Women commonly discussed a sense of stigma associated with chronic HCV diagnosis, but also felt that research staff were not participating in perpetuating this stigma. One woman described this lack of stigma as encouraging to her research participation. She stated, “people are just making me feel better. I don’t feel as dirty.” Another woman reported that the staff was good because they were, “. . .being understanding about addiction. None of them are actually addicts themselves but they related really good.” Another participant explained that, “I just appreciated that. . . it wasn’t like a condescending or patronizing conversation, it was a, “oh you wanna know more about this so I’ll give you as much information as I can.”” Women also discussed that prenatal providers that referred them for study participation were able to discuss their chronic HCV diagnosis without stigma and this promoted study involvement.
In addition to a lack of stigma, women noted that honesty from research staff was appreciated when discussing possible harm to the infant and that they could understand the lack of data. When asked to explain what the study had told her about possible fetal harm, 1 participant stated “. . .they kept it real and said you know we don’t know. So. . . that’s fine. I know that. I understand it.” Some women did note issues of perceived dishonesty that discouraged their participation such as the feeling that the referring obstetrician-gynecologist (OB/Gyn) was not well educated on the study itself, and another woman who felt that the study team misrepresented the number of people that had been treated and learning how few had been involved was troubling to her.
Women preferred person-centered care and acknowledgment of individual preferences for follow-up of chronic HCV and future pediatric care
Many participants identified that the research staff expressed empathy and concern for their whole person, not just their involvement in the research. One participant stated that she found the study team helpful when they called every week, “because. . . it wasn’t to remind me to take the medicine. I reminded myself to take the medicine, but just. . . them calling to just check to make sure that I am OK.” Another woman felt that she enjoyed the study staff because, “I make them laugh, so I’m happy here.” Still another participant described that the study was able to coordinate transportation for her, with “a taxi early in the morning and then take me home at night.”
Women had varied, individualized preferences for who they would have wanted to see to treat chronic HCV and they rarely had plans for pediatric follow-up. Two women wanted treatment from their OB/Gyn office, since it was felt to be convenient while obtaining prenatal care. One woman felt that she would prefer treatment through the study over all other options, due to the study’s altruistic focus and because she felt the study team had specialized expertise. Two women wanted to get care from a liver specialist, one of these women specifically stated that her primary care provider (PCP) and her OB/Gyn wouldn’t be “right for the job.” Another woman stated either a gastroenterologist or her PCP would be reasonable places to receive care for chronic HCV. Only 1 woman discussed preferences for written materials to learn more about chronic HCV. In terms of pediatric follow-up, it was rare that women had made pediatric plans. One woman explained, “I don’t know, ‘cause I didn’t even pick a pediatrician for real yet.” Women frequently noted that they wanted to ensure the infant had no evidence of disease transmission, but that they would address this after the baby was born with their pediatrician.
Discussion
Participants of this first study to evaluate treatment of Hepatitis C in pregnancy shared their experiences with chronic HCV therapy. Our results lend a critical platform to voice the experience of women living with chronic HCV. Participants desire HCV cure and feel it is a “life-saving” measure. They found that treatment during pregnancy was convenient, adherence to daily pill was feasible, and was facilitated by nonjudgmental interactions with providers and researchers. Given that rates of HCV infection are rising in the United States among reproductive aged women, our findings highlight an essential opportunity for testing and treatment during pregnancy that could meaningfully mitigate the HCV epidemic.1,2
This study reveals many consistencies with prior work and also highlights important differences related to chronic HCV in pregnancy. 6 There are no prior qualitative studies of pregnant people with chronic HCV, but prior qualitative interviews with women who have HCV and are undergoing treatment for opioid addiction revealed similar themes of stigma from healthcare providers and challenges to understanding the implications of diagnosis.8,11-14 In prior work, non-pregnant people who identify as women discussed a desire to obtain testing and treatment in order to live longer and spend more time with their children. They also discuss that return to drug misuse is a barrier to treatment adherence.8,11-14 Our findings also show that cure would contribute to a sense of self-esteem and accomplishment after completing treatment and we identify that treatment for HCV may be viewed by patients as a protective factor against return to drug misuse. 17
The participant population of pregnant people with chronic HCV taking HCV treatment is the strongest feature of our work and provides insight into the experiences, motivations, and perceptions of this population. We recognize, however, that pregnant people with chronic HCV infection who are not motivated to treat their infection during pregnancy or who may be more ambivalent or uncertain would likely describe different experiences, concerns, and needs; this would be a different study. While our population mimics epidemiologic data on new HCV infections in United States given that all participants are white and the majority had acquired HCV through injection drug use, we acknowledge that the views of non-white pregnant people with chronic HCV and those who have acquired HCV through other routes may also not be reflected in our findings. 25 We also acknowledge that gender diverse individuals bear children and our study may not reflect the views of people who do not identify as women or use the term mother. Although our interviewers and guide sought an open approach to exploring participants’ perceptions of HCV treatment including negative experiences, fears, and concerns, social desirability bias could have been an issue that may also limit our findings. We also note that during the recruitment for this study, there was significant national focus on the opioid epidemic and its implications and that may have enhanced motivation in the participants. Another limitation to the study is its structure within a pharmacokinetic study, where a priori recruitment and sample size strategies were determined by pharmacokinetic parameters. 19 However, in thematic analysis we noted thematic saturation after coding 10 interviews (5 pre-study and 5 end-of-study) and no new codes were generated in the last 8 transcripts. Thus, we feel that the sample is robust to represent thematic saturation.
This study is a novel view into the perspectives and self-reported desires of pregnant people with chronic HCV infection. Despite the limitations of the study, our findings illustrate several important steps for prenatal care providers, infectious disease specialists, public health personnel, and addiction treatment centers. Our work reveals that pregnant people desire effective and tolerable treatment for chronic HCV and that pregnancy may be a unique window of motivation and insurance access that reduces barriers to HCV testing and treatment. In this regard, clinicians should explore and support pregnant people’s interest in seeking HCV treatment during pregnancy. Pregnant people with HCV should be counseled clearly and offered reasonable avenues for follow-up of HCV infection, and treatment should be more widely accessible. To increase access, treatment through prenatal care providers rather than liver specialists, use of an effective pan-genotypic regimen in pregnancy, and decreased treatment monitoring protocols are proposed approaches that may facilitate treatment engagement and adherence and deserve further study.16,26 LDV/SOF treatment is just 12 weeks long, and safety data from the phase 1 trial supports ongoing investigation in oral therapy for chronic HCV infection during pregnancy. 19 Treatment during pregnancy for chronic HCV infection may represent an important opportunity to combat rising HCV rates among people with childbearing potential and children with perinatal acquisition of HCV.
Supplemental Material
sj-docx-1-sat-10.1177_11782218211046261 – Supplemental material for Experiences and Perspectives From Women Taking Investigational Therapy for Hepatitis C Virus During Pregnancy
Supplemental material, sj-docx-1-sat-10.1177_11782218211046261 for Experiences and Perspectives From Women Taking Investigational Therapy for Hepatitis C Virus During Pregnancy by Yasaswi Kislovskiy, Catherine Chappell, Emily Flaherty, Megan E Hamm, Flor de Abril Cameron, Elizabeth E Krans and Judy C Chang in Substance Abuse: Research and Treatment
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Institutes of Health/Eunice Kennedy Shriver National Institute of Child Health and Human Development (1R21HD089456-01), the National Institutes of Health/Office of Research on Women’s Health (5K12HD43441-14), and Gilead Sciences (CO-US-337-2117). Gilead provided the study drug, performed pharmacokinetic assays in their commercial laboratory, and provided consultation on the phase 1 trial study design. The National Institutes of Health had no role in the study design, collection, analysis, interpretation of data, writing of the report, or the decision to submit the report for publication.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yasaswi Kislovskiy and Catherine Chappell have served as consultants for Gilead Sciences. Elizabeth Krans and Catherine Chappell are receiving research funding from Merck.
Author Contributions
YK provided data analysis and manuscript preparation. CC composed study design, coordinated data collection, and assisted with manuscript review. EF assisted with data analysis and manuscript review. MEH and FAC assisted with study design, data collection, and manuscript review. EEK assisted with study design and manuscript review. JCC provided oversight of study design, data collection and analysis, and manuscript review.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
