Abstract
Background:
Given the immense burden of the widespread use of opioids around the world, exploring treatments that improve drug use outcomes, and craving and withdrawal measures in individuals with opioid use disorder is crucial. This pilot study examined the feasibility and preliminary efficacy of the L-type calcium-channel blocker isradipine (ISR) to improve drug use outcomes, and craving and withdrawal measures during buprenorphine (BUP)/ISR stabilization and subsequent taper in opioid-dependent individuals.
Methods:
Participants were stabilized on BUP sublingual tablets within the first 2 days of week 1, were then randomized and inducted on either ISR or placebo, gradually increasing the dose over the next 2 weeks, followed by a 10-day BUP taper during weeks 5-6, and ISR/placebo taper during weeks 7 to 8. Assessments included thrice-weekly measures of craving and withdrawal, as well as vital signs and urine drug screens. Medication compliance was assessed by monitoring number of missed clinic visit days.
Results:
Baseline characteristics of participants (n = 25; 60% male, 96% Caucasian, 48% employed, mean age 32.8 years) did not differ significantly between treatment groups (isradipine, n = 11; placebo, n = 14). During the stabilization phase (n = 19), ISR participants had significantly lower rates of illicit opioid-positive urines (treatment × visit: t = -2.16, P = 0.03), as well as reduction in craving intensity (t = –2.50, P = 0.01), frequency (t = –3.43, P < 0.01) and duration (t = –2.51, P = 0.01). ISR was well tolerated with mild adverse effects.
Conclusions:
This study was likely underpowered due to being a pilot trial. Although preliminary results suggest ISR may improve BUP-assisted treatment outcomes, concerns about high number of exclusions (n = 11 during taper phase) based on cardiovascular measures as well as ISR-induced changes in vital signs with the immediate release formulation may limit the feasibility of this approach.
Trial Registration:
Clinicaltrials.gov identifier NCT01895270. Registered 10 July 2013, https://clinicaltrials.gov/ct2/show/NCT01895270?id=NCT01895270&draw=2&rank=1
Background
Opioid analgesic use more than doubled worldwide between 2001-03 and 2011-13, with substantial increases in North America, western and central Europe and Oceania1,2 resulting in higher mortality rates from both prescription and illicit opioids.3,4 In the United States and Canada, rates of opioid misuse, overdose deaths, and the subsequent resurgence of heroin use among those with opioid addiction have reached epidemic proportions.5−7 Out of 20.5 million Americans aged 12 or older reporting a substance use disorder in 2015, about 2 million had prescription opioid use disorder and 591 000 had heroin use disorder. 8 Opioid addiction is a source of considerable health burden to society in terms of years of potential life lost, 9 along with increased disease burden in terms of overdose, transmission of infectious illness, mortality, lost workplace productivity, and criminal justice costs.10,11 Thus, exploring strategies to improve treatment outcomes in opioid-dependent individuals is of paramount importance.
There has been an expansion of medication-assisted treatment (MAT) for opioid use disorder in recent years, especially using opioid agonist medications methadone and buprenorphine. 12 Opioid agonist treatment provides the benefit of addressing cravings, suppressing illicit opioid use as well as retaining people in treatment. 13 However, rates of dropout from MAT programs are still quite high. In a randomized, prospective Bavarian study with 140 opioid-dependent, primarily heroin-addicted patients on opioid agonist treatment, overall retention rate at 6 months was only at 52.1%. 14 Treatment completion rates for methadone and buprenorphine in a 24-week trial was 74% and 46% respectively in the US. 15 Given that treatment retention is associated with a more positive health outcomes,16−19 these findings highlight the importance of identifying strategies to improve upon early retention of patients in MAT.
Meanwhile, dropout rates during and relapse rates following opioid detoxification are much higher 20 as compared to long-term agonist treatment. 21 One of the reasons for early dropout during buprenorphine detoxification include higher baseline levels of craving and smaller decreases in craving and withdrawal during stabilization, as well as higher craving and withdrawal rebound during buprenorphine taper. 22 Even with much higher rates of relapse, patients sometimes prefer and treatment providers recommend detoxification over longer-term MAT due to a variety of reasons, including cost of treatment, lack of access to services, lack of trained providers, lack of awareness of MAT options, and expressed negative beliefs in the family or community.23−25 Traditional methods of detoxification from opioids, including tapering off the opioid agonist methadone or buprenorphine and supportive treatment of symptomatology with the α2-adrenergic receptor agonists clonidine or lofexidine, as well as subsequent treatment with the injectable formulation of the opioid antagonist naltrexone (VivitrolTM) for relapse prevention, are limited not only by high relapse rates but also lack of efficacy in relieving subjective symptoms of cravings and withdrawals.26-37 Thus, exploring other detoxification strategies involving opioids is of great importance, not only for opioid-dependent pain patients and prescription opioid abusers, but also for providing a smooth transition from opioid agonists to naltrexone therapy or a drug-free state.
Calcium-channel blockers (CCBs) have been shown to alleviate opioid withdrawal in opioid-treated nonhumans. 38 Moreover, the L-type CCBs verapamil, nimodipine and nifedipine have been shown to alleviate withdrawal in clinical trials of opioid detoxification.39,40 Meanwhile, in our human laboratory model of opioid withdrawal, the L-type dihydropyridine CCB isradipine, dose-dependently reduced naloxone-induced discriminative stimulus, cardiovascular and self-reported effects among opioid-dependent participants. 41 When comparing these results with those of other CCBs 42 and alpha2-adrenergic agonists 43 tested in this model, isradipine showed greater dose-related efficacy to attenuate the behavioral effects of naloxone among opioid-dependent participants. Thus, this randomized, double blind, placebo-controlled pilot clinical trial examined the feasibility, tolerability and initial efficacy of isradipine to improve outcomes during initial stabilization on the partial opioid agonist buprenorphine and subsequent taper off buprenorphine among opioid-dependent individuals.
Methods
Participants
Twenty-seven opioid dependent individuals (aged 18-65 years) seeking treatment for opioid dependence were recruited from Central Arkansas from October 2013 through December 2015. Written informed consent was obtained from all individual participants prior to entry into the study proper. In order to participate in the study, subjects had to be available to attend clinic 6 days a week for approximately 30 to 60 minutes; fulfill DSM-IV (Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision) criteria for opioid dependence, as determined by a physician; submit a urine sample negative for benzodiazepines and barbiturates; meet stable orthostatic blood pressure requirements (ie, must have at least 1 orthostatic reading that contains all of the following: supine blood pressure of ⩾90 (systolic) and ⩾60 (diastolic) mm Hg, seated blood pressure of ⩾90 (systolic) and ⩾60 (diastolic) mm Hg, or an orthostatic change of <20 mm Hg systolic or <10 mm Hg diastolic on standing and a heart rate increase of <20 bpm; must have sitting heart rate of ⩾55 on at least 1 occasion); have no unstable medical condition or stable medical condition that would interact with study medications or participation; have no history of psychosis, schizophrenia or bipolar disorder; not be pregnant, no plans to become pregnant and have adequate birth control; have no present or recent use of medications including psychoactive drugs that would have major interactions with the study drugs; have liver function tests no greater than 3 times normal with blood urea nitrogen (BUN) and creatinine within normal range; have no significant electrocardiogram (ECG) abnormalities; have no physiological dependence on alcohol or drugs other than opioids, marijuana or tobacco; and have no pre-existing severe gastrointestinal narrowing. Eligibility was ascertained through a comprehensive evaluation, which included complete physical, neurological and clinical psychiatric examinations, routine lab studies, pregnancy test, and an ECG. Participants were compensated for their attendance under a low-cost contingency management procedure that utilized the “fishbowl.”44,45 Study completers could earn an average of approximately $235. This study was approved by the University of Arkansas for Medical Sciences Institutional Review Board.
Design and procedures
Potential participants initially underwent screening procedures to determine eligibility to participate in this 8-week randomized, double-blind clinical trial through a 1-week, centralized recruitment/screening procedure. Once participants were determined eligible, they completed an intake procedure with a research assistant and/or clinician and then randomized to receive isradipine or placebo using a computerized urn randomization program, balancing groups on primary opioid of abuse, score on the Opiate Withdrawal Symptom Checklist (OWSC), sex, and marijuana-use status. Only the research pharmacist and biostatistician were aware of medication assignment. All participants were inducted onto buprenorphine at the beginning of week 1 and then onto isradipine or placebo starting day 3 of week 1, reaching the maximum targeted dose of isradipine during week 3. Participants began a 10-day buprenorphine taper starting day 3 of week 5, and tapered off isradipine during weeks 7 and 8 (Table 1). Supervised urine drug screen samples, objective and subjective withdrawal symptom scores, self-reported adverse effects, body temperature, pupil diameter, and vital signs were obtained thrice weekly. An ECG and orthostatic vital signs were obtained prior to and 2 hours after the initial isradipine dose and after each scheduled isradipine dose increase during the study to determine any acute isradipine dose-related cardiovascular changes. Regardless of treatment group, all participants were scheduled to meet with a research therapist for 30 to 60 minutes to provide participants an opportunity to review critical issues and problem areas. Participants attended study visits Monday through Saturday to receive buprenorphine and study medication through the University of Arkansas for Medical Sciences’ Center for Addiction Services and Treatment (CAST), complete scheduled research assessments and attend any scheduled counseling session. At the end of the study, subjects were given treatment referrals in the community.
Study medication dosing schedule.

Assessments, urine drug screens, vitals and self-reported side effects collected thrice weekly.
Abbreviations: b.i.d., twice daily; BUP, buprenorphine; ISR, isradipine; mg, milligrams; PLA, placebo.
Drugs
All study medication was prepared by the UAMS Research Pharmacy. Buprenorphine tablets were placed into packets for dispensing by nursing staff at the UAMS Center for Addiction Services and Treatment. Isradipine and placebo (microcrystalline cellulose) were each placed in identical size 00 blue opaque capsules and placed into blister packs to allow for morning dispensing as well as a take home blister pack to take in the evening.
The dosing schedule for buprenorphine tablets and isradipine/placebo is shown in Table 1. Participants were typically inducted onto the targeted buprenorphine stabilization dose of 12 mg/day during the first 2 days of week 1 and continued to receive 12 mg/day (with a double dose of 24 mg administered on Saturday to cover Sunday) through weeks 2 to 4. The 10-day buprenorphine taper began on Monday of week 5, such that the buprenorphine dose was gradually reduced until Wednesday of week 6 when the final dose of 2 mg was administered. At the same time, starting on day 3 of week 1, participants began twice daily administration of immediate release isradipine (5 mg) or placebo. The isradipine dose was increased over a 2-week period until the maintenance dose of 10 mg twice daily was reached on day 3 of week 3. This target dose was selected based on its efficacy to block naloxone-induced behavioral effects among opioid-dependent participants. 41 Participants continued to receive this maintenance dose of isradipine (or placebo) until day 2 of week 7, after which the dose gradually tapered until the final dose of 5 mg/day was given on day 2 of week 8. All participants received a placebo on days 3 to 5 of week 8. In order to ensure medication compliance and control for medication diversion participants were observed at the CAST dosing window taking their isradipine/placebo morning dose, followed by their buprenorphine dose. Participants were required to demonstrate to the dosing nurse by oral inspection that the buprenorphine had dissolved, prior to leaving the clinic. Participants were given a daily packet of study medication to take in the evening. Participants were compensated for returning their weekend take-home medication packets on Monday mornings. Medication compliance was assessed via number of missed clinic visit medication days (Saturday dosing equaled 2 medication days) and quantitative urine riboflavin level of urine samples obtained on day 1 of week 5, as riboflavin (10 mg) was added to the study capsules for this purpose. Participants were discharged from the study if they missed 2 consecutive days of dosing.
Assessments
Substance use history was recorded at intake using a questionnaire that recorded the type of drug, amount of use, route of administration, number of years used regularly, date of last use, and age first tried. Thrice-weekly craving assessments were adapted from one developed previously for cocaine 46 and for opioids 47 using a validated Visual Analogue Scale (VAS) 48 and measured the following: (1) the intensity of desire for opioids in the past 24 hours on a scale of 0 (not at all) to 20 (a great deal), (2) the number of episodes of craving during the past 24 hours, (3) the length of craving episodes in the past 24 hours, and (4) whether craving increased, decreased or remained the same relative to the previous clinic visit assessment. Observer rated opiate withdrawal symptoms were recorded thrice weekly using the Objective Opiate Withdrawal Scale (OOWS) 49 which consists of 13 items describing withdrawal symptoms. For each symptom that was present during a 5-minute period and fit the given criteria, 1 point was scored. The OOWS has been shown to have good reliability with self-reported opiate withdrawal scales.49,50 Self-reported opioid withdrawal symptoms were measured thrice weekly using the Opiate Withdrawal Symptoms Checklist (OWSC), 51 which consisted of 22 items describing possible opioid withdrawal symptoms rated on a scale from 0 (not at all) to 4 (very much). Self-report assessments of opioids and other drug use were obtained at intake and on day 1 of each week using 7-day recall method instruments developed in previous studies,51,52 where participants were asked to report the amount used and method of use each day. Side effects of medications were assessed thrice weekly using a study medication side effects checklist to rate side effects specific to isradipine or buprenorphine from 0 (not at all) to 4 (very much). Physiological signs (vital signs, pupil diameter, and body temperature) were measured thrice weekly. Heart rate and blood pressure were taken using a Dinamap monitor, pupil diameter using a Colvar pupilometer, and body temperature measured using an AccuSystem FILAC*F-1500 or a WelchAllen Suretemp thermometer.
Because the contingency management procedure was, in part, based on current drug use of opioid, benzodiazepine, barbiturates, cannabis, cocaine, and amphetamine, Redwood multi-test drugs of abuse panel dipsticks were used to immediately test thrice-weekly supervised urine samples. Urine samples were rated positive if the quantity of drug or metabolite was ⩾ 500 ng/mL for methamphetamine and amphetamines; ⩾ 300 ng/mL for benzoylecgonine, benzodiazepines, barbiturates, morphine, and methadone; ⩾ 100 ng/mL for oxycodone; and ⩾ 50 ng/mL for THC. Ecstasy and PCP were also tested. If any dipstick reading was questionable, the participant was treated as though the urine was negative, for the sake of contingency management procedure. Samples with questionable results were analyzed using EMIT (Redwood Toxicology Laboratories, Santa Rosa, CA). Urine samples were rated positive by EMIT standards if the quantity of drug or metabolite is ⩾ 1000 ng/mL for amphetamines; ⩾ 300 ng/mL for benzoylecgonine, propoxyphene, and opiates; ⩾ 200 ng/mL for barbiturates and benzodiazepines; ⩾ 150 ng/mL for methadone; ⩾ 50 ng/mL for THC; ⩾ 25 ng/mL for phencyclidine; and ⩾ 0.04 g/dL for alcohol. Breath analysis for alcohol was also performed thrice weekly. Quantitative assessment of urine riboflavin levels was performed on urine samples obtained from participants on day 1 of week 5.
Data analyses
For computerized versions of assessments, a separate template containing appropriate assessments in timed sequence was constructed for each participant on a laptop. Computerized assessments had built-in out-of-range and other controls to ensure accurate initial entry. These templates were then converted to an Excel data file, checked for completeness and merged with other data files matched by subject ID. Data obtained on paper with pen as well as laboratory results were entered into a database using a double data entry procedure. Any inconsistencies were then corrected by checking source data for the correct entry. Data analyses were as follows:
Baseline characteristics of the 2 groups were compared using 2-sample t-tests for continuous variables (eg, age) and chi-square analyses for categorical variables (eg, sex, race) to determine whether any significant baseline differences have accrued despite randomization. Group differences in the number of weeks retained were assessed using a 2-sample t-test. The number of missed medication days were compared across groups using Wilcoxon rank sum test due to non-normal distribution.
Because there were essentially 2 phases to the study (ie, buprenorphine stabilization/ISR induction [weeks 1-4] and buprenorphine taper [weeks 5-6]), data for each phase were analyzed separately such that, for dependent variables obtained at several time points within a phase, data were entered into random regression models, also known as hierarchical linear models (HLMs), to determine whether scores changed differentially over time across treatment groups.53,54 For each phase, data for participants completing at least 2 assessments within that phase were included in the analyses. Thrice-weekly and weekly continuous data were analyzed longitudinally with PROC GLIMMIX in SAS version 9.4. Dichotomous urine results (ie, negative or positive) were analyzed longitudinally using the SAS procedure GENMOD, which allows an HLM modeling program for ordinal outcome measures. All available data were used in our analyses and no attempt was made to interpolate missing data. HLM methodologies fit a regression line for each subject, effectively interpolating missing data, before deriving final estimates. This approach of modeling repeated measures is specifically designed for use in repeated measures designs with missing data, allowing for intra-subject serial correlation and unequal variance and covariance structures over time. Solution of these problems, common to clinical trial data, is accomplished by incorporating available trend data for each individual with information on the behavior of the group from which the subject is drawn. If there were any significant baseline differences, the variable was added as a cofactor in the HLM analyses.
Orthostatic vital signs taken prior to and 2 hours following the initial and increased dose of isradipine were entered into a repeated measures mixed model with medication group (isradipine versus placebo), assessment time point (pre versus post) and week (time factor) as factors. A 2-step model was used to describe continuous variables during the buprenorphine taper phase (opioid withdrawal, cravings, side effects and adverse events) due to the presence of a significant amount of zero values. The first-stage model dichotomized the variables into zero versus above-zero and described the probability of having or not having a value. The second-stage model described the continuous variables, only when the values were above zero. All analyses employed a significance level of α ⩽ 0.05, and all tests are 2-tailed.
Results
Retention, subject characteristics, medication compliance
Of the 96 participants who consented for screening, 27 met eligibility criteria and were randomized to study medication condition (Figure 1; Consort flow diagram), 2 of which withdrew prior to receiving the first buprenorphine dose. Of these 25 participants who entered study proper, 6 participants did not undergo isradipine induction due to their blood pressure being outside dosing parameters. Further, 1 participant decided to use amitriptyline, and another missed 2 consecutive days of study medications and were both discharged from the study. A third participant voluntarily withdrew from the study during week 1 of induction (Figure 1).

Consort flow diagram.
Of the 16 participants that completed isradipine induction, 5 did not start buprenorphine taper due to missing 2 or more days of medications and were discharged from the study (Figure 1). Retention rates did not differ between treatment groups (isradipine = 3.8 ± 2.4 and placebo = 3.5 ± 2.8 weeks, P = .48) with 7 out of 25 participants (28%) completing the entire buprenorphine taper.
No baseline differences were observed between treatment conditions among the 25 participants that started the buprenorphine induction (Table 2). Overall participants had a mean age of 32.8 ± 10.07 years; 40% (10) of participants were female, 92% (n = 23) were Caucasian, 84% (n = 21) achieved at least a high school degree, and 48% (n = 12) were employed full time. Overall percentage of participants reporting using heroin was 12% (n = 3), morphine 8% (n = 2), oxycodone/oxymorphone 44% (n = 11), and other types of opioids 36% (n = 9). Most participants did not have a prior treatment history for opioid dependence, with 16% (n = 4) reporting prior drug detoxification, 4% (n = 1) reporting prior buprenorphine maintenance, and 4% (n = 1) prior methadone maintenance treatment.
Participant characteristics.

Abbreviations: ISR, isradipine; PLA, placebo; SD, standard deviation.
Denotes P value < .05.
Categorical variables analyzed by Fisher exact test due to low expected counts or Chi-square (X2) where counts were high enough.
Rank-sum test due to non-normality and unequal variance t-test due to heteroskedasticity.
Table 2 also shows baseline characteristics of those participants who participated long enough in the buprenorphine induction (n = 19) and buprenorphine taper (n = 11) phase respectively to be included in data analyses. In each phase there were no baseline differences between treatment groups. Duration of opioid use before treatment did not differ between isradipine and placebo groups. Only 2 participants reported prior opioid maintenance treatment, with 1 in the isradipine group reported both prior methadone and buprenorphine treatment. There was no difference between the 2 groups on the number of prior drug detoxifications (Table 2).
The 2 groups did not differ in the percentage of scheduled medication days missed (placebo = 13.73% ± 13.82 versus isradipine = 5.56% ± 5.12; P = .11).
Phase I: Buprenorphine and isradipine induction/stabilization
Drug use outcomes, opioid withdrawal, and craving measures
During the buprenorphine/isradipine induction phase, isradipine-treated participants were less likely to have illicit opioid-positive urines over time relative to placebo (treatment × visit interaction: t = –2.16, P = .031) (Figure 2). Participants in the isradipine group showed a more rapid decrease in craving intensity (t = –2.50, df = 188, P = .0133) as well as number (t = –3.43, df = 187, P = .0008) and duration of craving episodes (t = –2.51, df = 185, P = .0128) over time relative to placebo (Figure 3). Subjective ratings on the OWSC did not differ between groups over time (t = .24, df = 185, P = .8132; Figure 3, bottom right panel).

Opioid use.
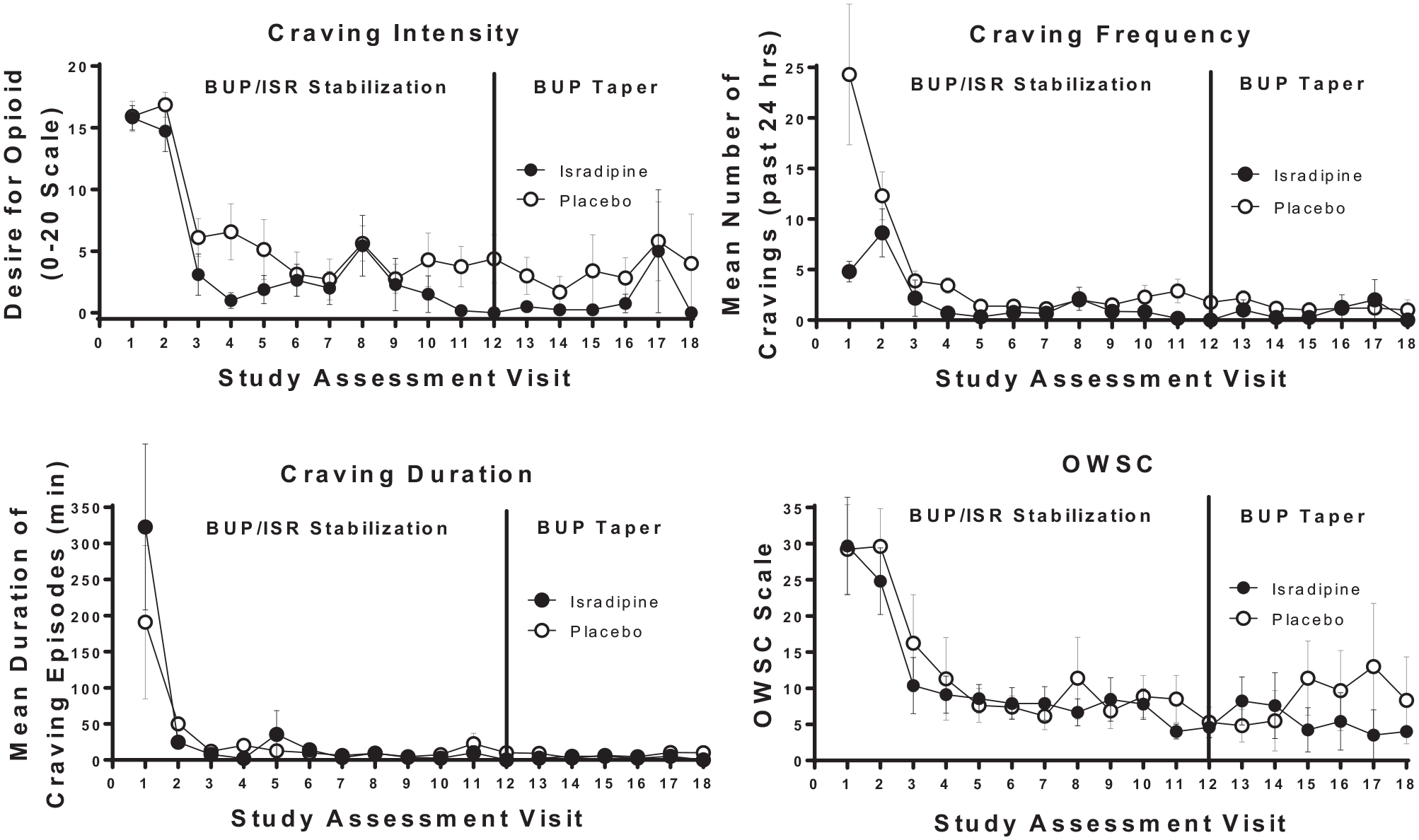
Secondary outcome measures.
Vital signs, side effects and adverse events
Similar trends for orthostatic vital signs were found for supine, sitting and standing measurements, so only those measured while seated are presented. Orthostatic diastolic blood pressure taken while seated pre- and 2 hours post the first scheduled isradipine dose and at each scheduled dose increase differed significantly by medication group, such that post-measures generally decreased and increased relative to pre-blood pressure measures in the isradipine- and placebo-treated subjects, respectively (t = –3.52, df = 31, P = .0013) (Figure 4). Orthostatic systolic blood pressure taken while seated 2 hours after the first scheduled dose of study medication similarly showed a trend toward decrease and increase in the isradipine and placebo groups, respectively (t = –1.76, df = 31, P = .0877), relative to pre-dose measures. Seated heart rate taken pre- and 2 hours post the initial dose or scheduled dose increase also differed significantly by medication group, such that post heart rate measures generally increased and decreased relative to pre-heart rate measures in the isradipine- and placebo-treated subjects, respectively (t = 2.17, df = 31, P = .0380) (Figure 4).
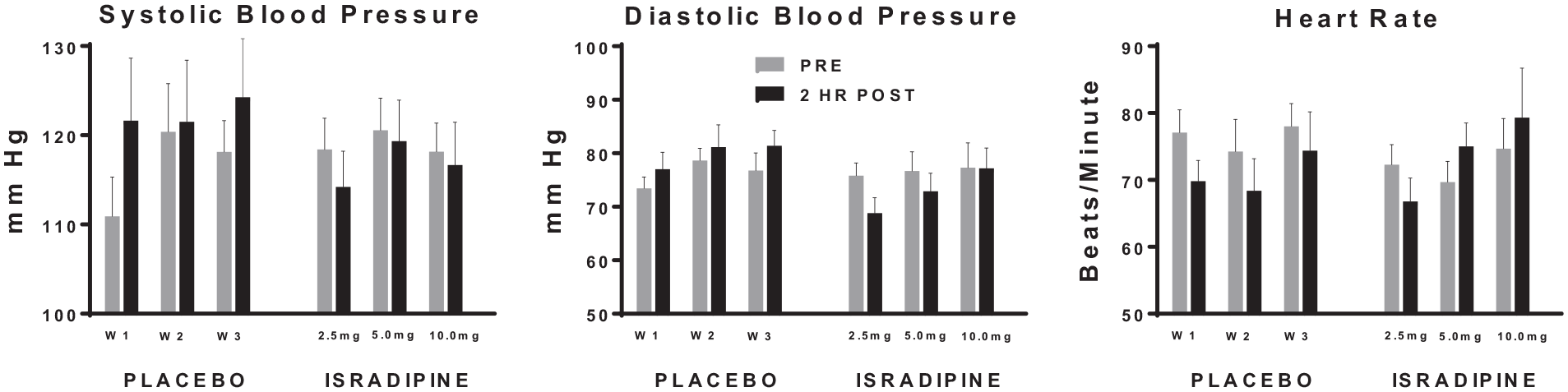
Vital sign changes.
Although ratings on the side effect checklist decreased significantly over time during the induction phase in a curvilinear fashion (t = –3.66, df = 23, P = .0013), there were no significant effects of treatment on side effects (t = 0.83, df = 158, P = .41) (data not shown).
No significant main effect of treatment (t = 0.88, df = 32, P = .38) or interactions between treatment and time (t = 0.54, df = 32, P = .59) occurred regarding pre- and 2 hours post ECG changes during this initial and scheduled dose increases (data not shown). QTc interval showed a trend for a pre/post main effect regardless of treatment with post measurements having a lower value (t = –1.87, df = 18, P = .0774).
Those events deemed at least possibly study related are shown in Table 3. Study related adverse events were generally in the mild range, with the exception of 1 participant in the isradipine group reporting moderate severity of swelling in both legs.
Summary of adverse events during induction phase.*

Number of times adverse events were attributed as possible, probable or definite. Events were collected by systematic assessment. Adverse events attributed as unrelated or unlikely were not reported here. No adverse events were reported by participants during the buprenorphine taper phase.
Phase II: Buprenorphine taper
Drug use outcomes, opioid withdrawal, and craving measures
During the buprenorphine taper phase, rates of illicit opioid-positive drug screens did not differ between isradipine and placebo groups (t = –0.04, P = .97) (Figure 2). There was significant main effect of study visit (t = 2.11, P = .03), such that rates of opioid-positive urine samples increased over time regardless of treatment condition.
During analysis of the second-stage model, there were significant main effects of visit on craving intensity (t = 2.62, df = 5, P = .05) and duration of cravings (t = 3.04, df = 4, P = .04) with craving measures in both treatment groups increasing over time. There was a trend towards differences by treatment (t = –1.95, df = 11, P = .07) as well as treatment by visit interaction (t = 2.02, df = 11, P = .06) with regards to the number of cravings (Figure 3). Subjective ratings on the OWSC did not differ between treatment groups over time (t = –0.81, df = 18, P = .43; Figure 3).
Side effects and adverse events
The second-stage model described the outcomes only when there was a reported side effect. When looking at buprenorphine side effects, there was only a trend toward an effect of visit (t = –2.01, df = 8, P = .0798; data not shown). There was no significant effect of isradipine treatment on side effects. When the side effects were combined, there was no significant effect of treatment (t = –0.81, df = 22, P = .43) or treatment by visit interaction (t = 0.71, df = 22, P = .48); however, side effects increased in a curved pattern over time from week 5-6.
No adverse events deemed at least possibly study related were reported during the buprenorphine taper phase.
Discussion
The results of this pilot study suggest that isradipine may improve treatment outcomes in participants being stabilized on buprenorphine. Due to the high rate of dropouts due to the pilot nature of this study, the results were underpowered. A recent study of factors influencing abstinence from opioids found that, in general, lower baseline levels of craving or greater decreases in craving and withdrawal during stabilization on buprenorphine predicted longer opioid-free intervals. 22 The finding in the present study that isradipine-treated participants had lower cravings and lower rates of positive illicit opioid-positive urines during the initial stabilization phase suggest potential utility of isradipine to improve initial treatment outcomes. This finding is also supported by a prior human drug discrimination study, in which Isradipine significantly attenuated naloxone-occasioned responding and naloxone-induced behavioral effects among methadone-maintained participants trained to distinguish between low dose naloxone and placebo. 41 Other L-type calcium channel blockers have also shown efficacy in reducing opioid cravings in individuals dependent on opiates and other substances. 40 As such, these findings demonstrate the promise of pharmacotherapeutic strategies to enhance initial MAT outcomes.
In contrast, during the buprenorphine taper no group differences occurred on craving intensity and duration, opioid-positive urine results, or retention, with only a trend towards less craving frequency in the isradipine-treated group. These findings suggest that isradipine may not be as efficacious in facilitating more positive outcomes during opioid detoxification; however, definitive conclusions likely cannot be made due to the high dropout rate during the taper. The reasons for this high drop-out rate likely include the fact that, instead of relying on participant symptoms to guide dosing, we employed very conservative orthostatic vital signs dosing parameters for isradipine dosing even when symptoms of hypotension were not present and, that the study protocol was very labor intensive, with participants not only attending clinic 6 days per week but also needing to stay for 3 hours at least once weekly during the first 3 weeks of the protocol. The fact that almost half of participants worked full time may have contributed to this increased drop-out rate. Nevertheless, the effect size estimate of differences between isradipine and placebo were quite large regarding urine results during the buprenorphine taper (1.0 for main effect of medication group), suggesting power of 80% to detect group differences with 20 participants per cell. Future research employing less stringent dosing criteria in a larger sample is necessary to evaluate the veracity of this effect size.
Opioid withdrawal consists of a particular set of symptoms (eg, nausea, cramps, sweats, and restlessness) that occurs when opioid-dependent individuals abruptly stop taking opiates, or after administration of an opioid antagonist like naltrexone. Withdrawal symptoms have been cited as the most common concern in opioid maintained individuals regarding coming off of opiate agonist treatment. 55 Alpha2-adrenergic agonists like clonidine, and the more recently FDA approved lofexidine 56 has been used to treat opiate withdrawal symptoms.57,58 Previous studies have also explored the role of calcium channel activation to be involved in the expression of opioid withdrawal and L-type calcium channel blockers (eg, nimodipine) to attenuate opiate withdrawal symptoms.39,59−61 Contrary to our expectations, however, withdrawal scores in our study did not differ across medication groups during either buprenorphine stabilization or taper, possibly for the reasons stated above.
Isradipine was quite well tolerated in our study with a modest decrease in both systolic and diastolic blood pressure and a compensatory increase in heart rate 2 hours after initial dose and after each dose escalation; effects that are consistent with the use of isradipine in normotensive individuals. 62 Reported adverse events were generally mild and consistent with isradipine’s side effects reported in other studies.63−65 Interestingly, some side effects were seen at a higher frequency in the placebo group, the implications of which are unclear.
Limitations and considerations
This study had several limitations given the nature of pilot trials. As discussed above, there were a significant number of dropouts during the taper phase which likely limited our ability to explore the effects of isradipine on treatment outcome. Our statistician was not blind to the drug conditions when conducting analysis which may have produced bias. 66 Also, our dosing parameters were quite stringent due to lack of knowledge regarding tolerability of opioid maintained participants for a hypotensive agent, which excluded potential participants from participating or continuing their participation, even in the absence of symptoms of hypotension. Because this was a phase I/II trial, participants had to attend clinic 6 times per week, which did not approximate “real-world” conditions. At the same time, immediate formulation of isradipine produced changes in vital signs and the extended formulation is no longer available. Moreover, calcium channel blockers may have low abuse potential, 41 which increases their potential utility as an opioid sparing agent as well as an adjunct medication during opioid detoxification. Thus, future research should consider focusing more on behavioral endpoints for determining tolerability as well as examining extended release or long acting formulations of calcium channel blockers to minimize potential side effects that would reduce tolerability.
Conclusion
This study adds to the growing literature that calcium channel blockers in general and isradipine, in particular, may help attenuate opioid craving and illicit opioid use in individuals stabilized on buprenorphine, thereby providing another potentially effective strategy for improving clinical outcomes in patients with opioid use disorder as well as those on a chronic opioid regimen. Based on these findings, future studies should employ an extended release version of isradipine if it becomes available again and incorporate procedures to ensure proper hydration in this population that is known to tend towards under-hydration. In addition, another dihydropyridine L-type calcium-channel blocker with a better cardiovascular side effect profile such as amlodipine may also be a good candidate to test in this model.
Footnotes
Acknowledgements
The authors thank Dr. Ben Guise for providing medical backup for the study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the R21DA035325 and DA039088 from the National Institute on Drug Abuse. This funding source had no other role other than financial support.
Declaration of conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s contributions
AHO, MJM, JT and JMG contributed to the study conception and design. Material preparation, data collection and interpretation of results were performed by NK, MJM, JDT JMG and AHO. Data analyses were performed by JDT. The first draft of the manuscript was written by NK with input from all authors; all authors commented on this and prior versions of the manuscript. All authors read and approved the final manuscript.
Author’s Note
A preliminary report of this work was presented at the Annual Meeting of the College on Problems of Drug Dependence in June 2016 in Palm Springs, California. Oliveto, A.H., McGaugh, J., Guise, J.B., Thostenson, J., and Mancino, M.J. (2016) Improving buprenorphine treatment outcomes with the L-type calcium channel blocker isradipine.
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institution (University of Arkansas for Medical Sciences Institutional Review Board, reference number 138823) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All subjects provided written informed consent prior to participating in the study.
Availability of data and materials
The datasets during and/or analysed during the current study available from the corresponding author on reasonable request.
