Abstract
Background and aims:
Studies assessing the cognitive performance effects of nicotine show inconsistent results and tobacco industry funding has been correlated with study outcomes. We conducted a systematic review of the primary literature assessing the cognitive performance effects of nicotine and assessed potential associations between tobacco and pharmaceutical industry affiliation and reported study conclusions.
Methods:
We searched PubMed, EMBASE, PsycINFO, BIOSIS, and Web of Science for peer-reviewed journal articles published between 2009 and 2016 that: (1) were randomized controlled trials; (2) investigated the effects of nicotine on cognitive performance in a laboratory setting; (3) administered nicotine to healthy adults (18-60 years); and (4) included participants were nonsmokers or minimally deprived smokers (⩽2 hours of abstaining from smoking). Study disclosures and tobacco industry documents were reviewed to determine industry funding.
Results:
Searches yielded 3,771 abstracts; 32 studies were included in the review. The majority of studies investigated the effects of nicotine on attention (n = 22). Nicotine had a non-uniform effect on attention: studies reported positive (41%; n = 9), mixed (41%; n = 9), and no effect (18%; n = 4). The majority of study authors had received prior tobacco industry funding (59%; n = 19), however over half of tobacco-industry funded authors did not report this (53%; n = 10).
Conclusions:
Nicotine does not appear to be associated with consistent cognitive performance effects. Although no association was found between reported outcomes and tobacco or pharmaceutical industry funding, findings likely underestimate the influence of industry funding due to strict inclusion criteria and incomplete data on pharmaceutical industry funding.
Clinical trial registration:
Not applicable
Introduction
Nicotine is an addictive substance that is typically consumed via cigarettes, 1 the primary commercial source of nicotine worldwide.2,3 The World Health Organization estimated that tobacco products cause 6 million deaths annually worldwide; frequency of tobacco-related deaths is expected to rise to 8 million per year by 2030. 4 Nicotine sustains and drives cigarette addiction and consumption and its related morbidity and mortality. 5
Nicotine from a cigarette is absorbed from the lungs into the blood stream. As a tertiary amine it binds to nicotinic cholinergic receptors in the central nervous system (CNS) and peripheral nervous system (PNS). Among other effects, binding of these receptors leads to the release of a neurotransmitter, dopamine, and activates the rewards pathway. Repeated consumption of nicotine can lead to neuroadaptation, dependence, mood/arousal modulation, and withdrawal symptoms if consumption is lowered or stopped. 5 Over the past decade, studies exploring the cognitive performance effects of nicotine have found inconsistent outcomes. 6 Some studies reported detrimental effects of nicotine while others reported nicotine induced cognitive enhancement.5–11
Tobacco companies argue that nicotine has positive effects on cognition. British American Tobacco Company (BAT), for example, recommends the use of nicotine to help with mood and concentration. 10 A consumer-oriented pamphlet from BAT aggregated statements from various groups supporting the use of nicotine, 10 which claimed the UK Royal College of Physicians stated that medicinal nicotine is “no more harmful than the existing use of caffeine” 12 and that the National Institute for Health and Care Excellence (NICE), UK, stated that, “Evidence is available from studies with up to 5 years follow-up which suggests that “pure” nicotine, in the form available in nicotine replacement therapy (NRT) products, does not pose a significant health risk.” 13 Advocates promoting e-cigarette use rely on claims that nicotine has cognitive benefits to advance the case that these products should be less unregulated and widely available.14,15
In 1994 a review of 101 studies on the effect of nicotine on human performance found nicotine could enhance finger tapping and motor sensory responses in the setting of a cognitive task associated attention. 16 This enhancement was not observed in terms of increased cognitive function. 16 Biased scientific conclusions may arise with industry funding, particularly for the tobacco and pharmaceutical industries, both of which produce and sell products containing nicotine.17,18 A 1997 follow-up found that funding by tobacco companies was significantly related to study outcome 19 after coding 91 studies for: 1) the reported conclusion on the cognitive performance effects of nicotine and 2) “acknowledged sponsorship” 19 divided into two categories, non-industry and tobacco industry. Studies (n = 35, 38%) with acknowledged tobacco industry support were significantly more likely to report “conclusions favorable to the tobacco industry.” 19 In 2010 a meta-analysis of 41 studies on the reported cognitive performance effects of nicotine in healthy volunteers found nicotine had significant effects on memory, motor abilities, and attention. 6 Although this study included funding source, only a small number of studies reported industry funding and the authors did not formally investigate a relationship between tobacco industry funding and reported study outcomes. 6 Overall, existing studies assessing the cognitive performance effects of nicotine have found inconsistent results that may be influenced by conflicts of interest, making it challenging to ascertain the effects of nicotine on cognition.
We expanded on past research by completing a systematic review of the primary literature on the cognitive performance effects of nicotine relative to human performance using studies published after the most recent 2010 meta-analysis. 6 We specifically focused on studies published after 2009 because tobacco industry priorities shifted when the FDA was granted regulatory authority over tobacco in 2009. 20 This regulatory change specifically allowed the FDA to regulate the amount of nicotine in tobacco products, 20 and created new urgency for generating evidence that nicotine had benefits as well as risks. At the time two-thirds of smokers supported nicotine reduction in cigarettes, 21 however the industry had found in internal research that nicotine was critical to smoker satisfaction and continuing use. 22 Evidence of possible benefits of nicotine use could provide counterarguments to regulation in the event that the FDA exercised this authority. This particular tobacco industry concern was validated in 2017 when the FDA announced its intention to reduce nicotine in combustible cigarettes to non-addictive levels. 23
We reviewed previously-secret internal tobacco industry documents, in addition to published funding disclosures, to assess whether study authors had received past tobacco industry funding. We hypothesized that studies with tobacco or pharmaceutical industry affiliation would be more likely to report nicotine having a beneficial cognitive effect relative to studies that did not.
This review addressed three primary research questions:
What are the reported cognitive performance effects of nicotine on the brain specific to human performance (eg, memory, attention, and motor sensory)?
Were there any reported adverse cognitive performance effects of nicotine on the brain? If so, what?
Did any of the included studies involve pharmaceutical and/or tobacco industry funding or affiliations? If so, how was the funding and/or affiliation reported (eg, disclosed funding, affiliation stated, no funding/affiliation was disclosed, funding/affiliation discovered by cross referencing author(s) to lists of tobacco company funded research)?
Methods
We conducted a systematic review of journal articles published between 2009 and 2016. Data collection was completed July 1-11, 2016 at the University of California San Francisco (San Francisco, CA USA).
Inclusion and exclusion criteria
We adapted inclusion and exclusion from a 2010 review on the effect of nicotine and tobacco smoking in human performance
6
as follows: 1) randomized double blinded placebo-control trial; 2) investigated the cognitive performance effects of nicotine after acute administration of nicotine; 3) nicotine was administered to healthy adults (18-60 years) who were either nonsmokers, non-deprived smokers, or minimally deprived smokers (
Outcomes
Participants completed cognitive tasks after nicotine administration. These tasks were classified into different performance domains, which made up the primary study outcomes. Following the classification strategy from the 2010 meta-analysis, 6 cognitive tasks were classified into one or more of the following performance domains: attention, long term memory, working memory, short term memory, reasoning, and motor sensory skills. Secondary outcomes included reported adverse events and effects such as a change in blood pressure, change in heart rate, and/or mood of the participants administered nicotine.
Search strategy
We conducted a literature search using PubMed, EMBASE, PsycINFO, BIOSIS, and Web of Science. Key words included: nicotine, tobacco products, smoking, cigarette smoking, mental processes, arousal, mental recall, reaction time/s, decision time/s, processing speed, performance, sensory, motor, psychomotor, attention, information processing, memory, cognition, electroencephalography, EEG, electroencephalogram/s, brain waves, alpha rhythm, beta rhythm, delta rhythm, gamma rhythm, theta rhythm, awake, asleep, slow-wave, sharp-wave, clinical trial/s, random allocation, humans, and not animal. Specific terms from these search threads were modified for certain databases in order to take advantage of unique search features and subheadings. The exact search threads for each database can be found in the Appendix.
Data collection
Data collection was completed in four phases.
Analytical strategy
Statistical tests were conducted using Stata. 29 Fisher’s exact tests calculated associations between funding source/affiliation and the type of reported cognitive effect (eg, positive versus negative/mixed/no effect). Reported outcomes from each study were categorized into four potential types: 1) positive, 2) negative, 3) mixed, or 4) no effect; for the statistical analysis, negative, mixed, and no effect were collapsed into one group. Industry affiliation was defined as the inclusion of at least one author who had ever worked for or received funds from either the tobacco or pharmaceutical industry. Funding sources or author affiliations for the tobacco industry could be disclosed, or undisclosed but identified through tobacco industry internal documents. Funding sources or author affiliations for the pharmaceutical industry were only sourced from author disclosures. An odds ratio and 95% confidence interval was calculated between the industry affiliation and the reported positive cognitive performance effect of nicotine.
Results
Study selection
We identified 3,771 abstracts from the literature search. From these, 54 full papers were extracted for full text review (see Figure 1). We excluded 22 studies for the following reasons:
Studies only explored the physiological effects of nicotine: Six studies were excluded due to only exploring the physiological effects of nicotine with no cognitive or behavioral outcome measure.
Study population was not appropriate: Five studies were excluded due to inclusion of nicotine deprived participants. One study was excluded because smoking participants never abstained from smoking. One study was excluded due to participants >60 years of age.
Study results were not usable: One study was excluded since for mixing outcomes of both healthy and patient participants. One study was excluded because it did not investigate a relevant performance domain. One study was excluded because all results were subjective.
Study design did not fit inclusion criteria: One study was excluded due to lack of randomization. Four studies were excluded because they were not double blinded.
Study intervention was not appropriate: One study was excluded because participants were only given placebos.
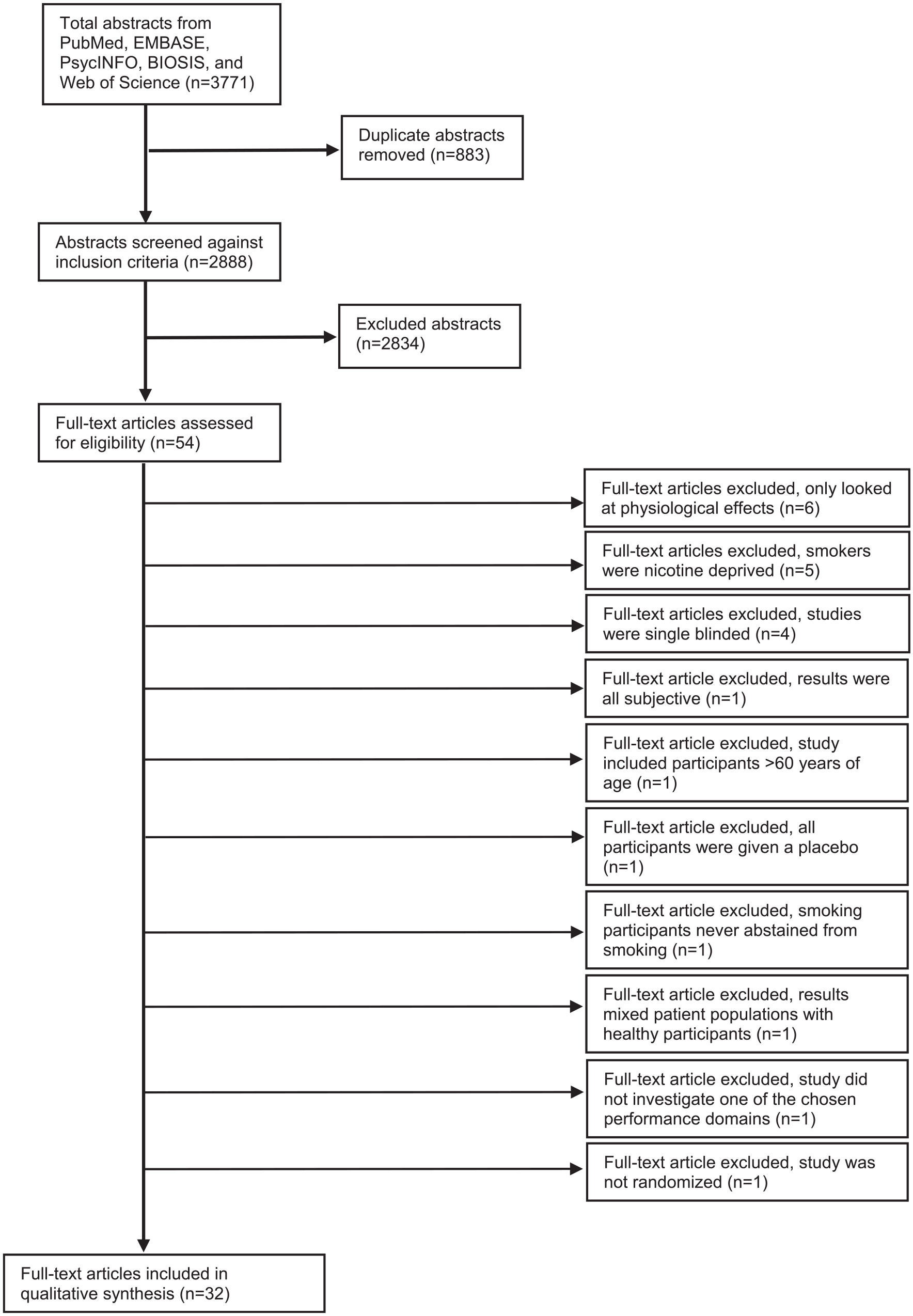
Prisma flow diagram.
Study characteristics
After exclusions, 32 papers were included in the final systematic review,30–61 the majority of which used a cross-over design (n = 27; 84%). Table 1 summarizes the study characteristics. The summary table of studies is provided in the Appendix.
Risk of bias assessments, all studies (n = 32).
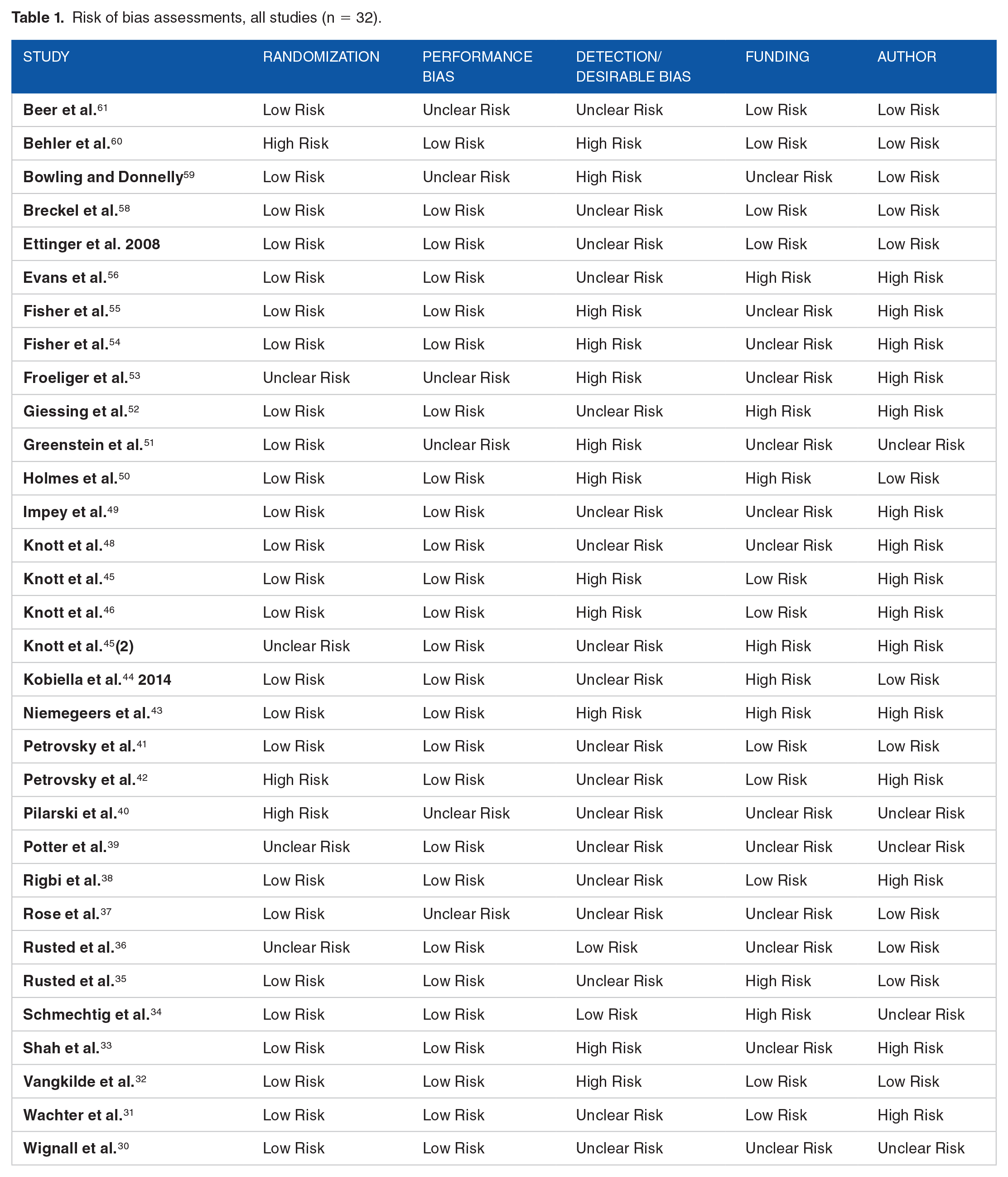
Risk of bias
We assessed risk of bias in six categories: (1) randomization; (2) performance; (3) detection/desirability; (4) funding; (5) author; and (6) publication. A summary of the analysis is provided in Table 1.
(1)
(2)
(3)
(4)
(5)
(6)
Outcomes
The majority of studies used either a patch or gum as the route of nicotine administration (n = 11 and 13, respectively). Of the remaining studies, 3 used nasal spray and 1 each used oral spray, lozenge, injection, cigarette, and chewing tobacco. No study used multiple routes of administration. Most included studies were located in Canada (n = 8; 25%), Germany (n = 7; 22%), or in the US (n = 7; 22%). Of the remaining studies, 5 (16%) were conducted in the UK, 2 in Australia, and 1 each in Israel, Denmark, and Belgium.
Performance domains and reported outcomes, all studies (n = 32).

Abbreviations: LTM, long term memory; MS, motor sensory; STM, short term memory; WM, working memory.
Our use of tobacco industry documents allowed us to identify additional undisclosed tobacco industry affiliations. If an author’s name was found on any of the unpublished tobacco funded research lists and they did not disclose the affiliation, then the study was coded as “undisclosed affiliated.” With these additional affiliations, a total of 19 studies were affiliated with either the tobacco or pharmaceutical industry. We found no statistically significant association between the funding source and the reported outcome on the cognitive performance effects of nicotine (OR = 0.804; 95% CI: 0.1 to 5.26).31,33–35,38,42–50,52–56
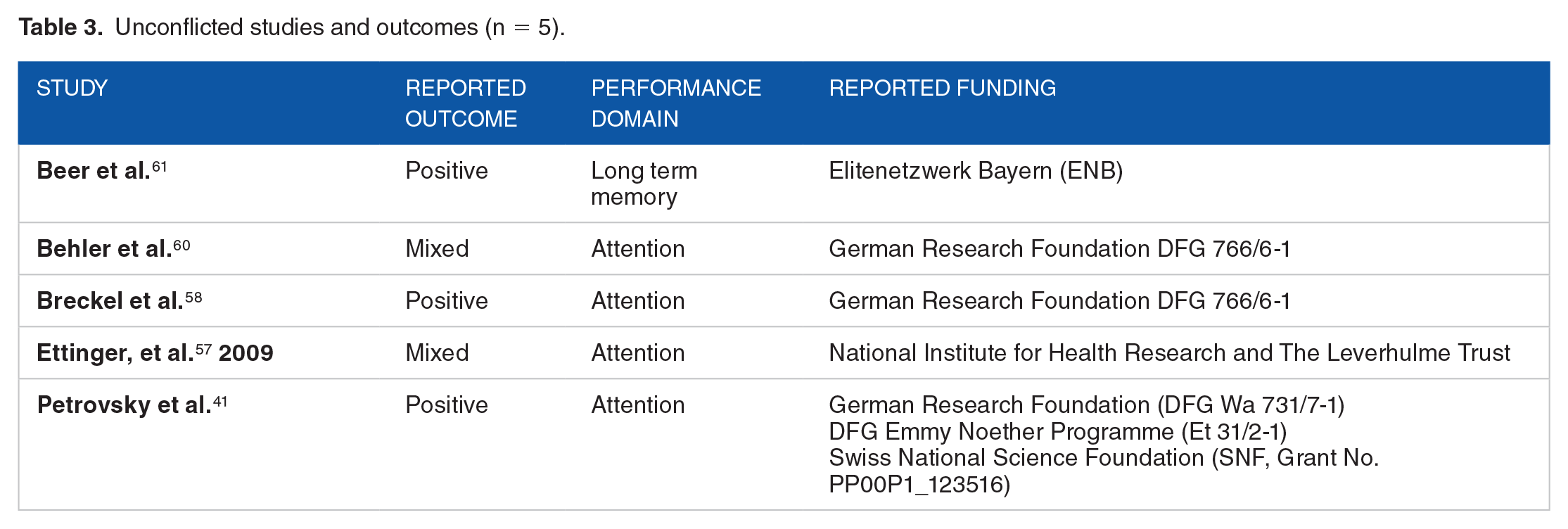
We also analyzed studies that were not affiliated with the tobacco industry (reported or unreported) and did not report a pharmaceutical industry affiliation. All studies coded as unconflicted reported funding from a government grant or entity, contained a conflict of interest statement which reported no conflicts, and did not include authors with unreported affiliations with the tobacco industry identified through internal industry documents. Undisclosed pharmaceutical funding could not be assessed. Five studies were coded as unconflicted; four of these studies investigated the effects of nicotine in the performance domain attention. Of these, two reported positive effects on attention and two reported mixed effects on attention. This outcome was similar to the distribution of reported outcomes in the performance domain of attention across all 32 studies included in this systematic review. Specifically, 22 of the 32 studies explored the performance domain of attention and the distribution of reported outcomes were: positive (n = 9 of 22; 41%); negative (n = 0 of 22; 0%); mixed (n = 9 of 22; 41%); and no effect (n = 4 of 22; 18%). A summary of the unconflicted studies is provided in Table 3.
Unconflicted studies and outcomes (n = 5).
Discussion
The objectives of this systematic review, which included 32 randomized controlled trials, were to assess the cognitive performance effects of nicotine among health adults in recent studies by updating a systematic review completed in 2010, 6 and to identify whether outcomes were associated with industry affiliations. Past studies suggest that nicotine administration might increase attention, based on a range of measures.6,16 Our review did not identify a similar effect, despite the high likelihood of publication bias. Although the majority of studies evaluated in this review focused on attention (n = 22, 69%), there was no consistent outcome for the cognitive performance effects of nicotine on attention. Our results could reflect publication bias, changes in study design, reliance on different cognitive tests, or use of updated technology.
We hypothesized that authors with current or past tobacco and/or pharmaceutical industry funding would be more likely to report positive effects on cognitive performance. No statistically significant association was observed. Less stringent inclusion and exclusion criteria than those adopted from the 2010 meta-analysis would have identified more papers of potentially lower quality, which has been associated with industry funding in the past 62 ; however including these papers would likely further increase publication bias and the inconsistency of research results.
Our findings also revealed that authors generally did not disclose their industry affiliations. Only one study in our systematic review reported tobacco industry sponsorship, although review of tobacco industry documents revealed that 10 additional studies had past tobacco industry affiliations. There is no consistent standard among journals regarding the time period for which authors must disclose support from organizations with a conflict of interest; required reporting may range from only support of the published manuscript to support over multiple years.18,63,64 When a longer time period was considered, authors had substantially more industry affiliations; considering that our sources only allowed us to identify past tobacco industry funding, and not past pharmaceutical industry funding, our results likely underestimate pharmaceutical industry affiliations.
This systematic review has limitations. The inclusion and exclusion criteria of the most recent meta-analysis on this topic were adopted to ensure consistency with prior work, however this choice meant that we included a small set of studies in the analysis. For example, novel nicotine delivery systems, such as electronic cigarettes, were not included in this systematic review because no studies using this mode of nicotine delivery met the inclusion criteria. The potential bias due to past industry affiliation may be understated given that we could not identify unreported pharmaceutical industry affiliations. The studies included in this systematic review are more rigorous randomized controlled trials, but less generalizable to the overall public, due to laboratory setting and conducted in a limited number of countries. Using less restrictive inclusion criteria could have identified additional studies and increased the likelihood of identifying potentially significant associations. In addition to the high risks of funding bias, our risk of bias assessment also found that all but two studies showed high risk of detection/desirability bias, and a risk of publication bias, raising the possibility that unpublished results may have shown insignificant effects, or significant effects that the authors or funders preferred not to publish.
This systematic review, like earlier research, found inconsistent results regarding the cognitive performance effects of nicotine, as well as indeterminate effects of past industry funding. Our findings identified underreported industry affiliations and the existence of publication bias, suggesting that research in this area may become more definitive with improved reporting of author affiliations. More than half of the papers with past tobacco industry affiliations did not disclose these relationships. Further research on the effects of nicotine with respect to cognitive performance, and its safety, is needed, particularly given that research on cognitive performance effects of nicotine, although inconclusive, are used selectively to advocate for consumption of tobacco products. Research on nicotine will continue to be difficult to assess without complete and transparent reporting of past and current industry affiliations.
Supplemental Material
neuro_supp_2019_xyz37821023beebd – Supplemental material for Cognitive performance effects of nicotine and industry affiliation: a systematic review
Supplemental material, neuro_supp_2019_xyz37821023beebd for Cognitive performance effects of nicotine and industry affiliation: a systematic review by Sarah V. Pasetes, Pamela M. Ling and Dorie E. Apollonio in Substance Abuse: Research and Treatment
Footnotes
Acknowledgements
The authors acknowledge Evans Whitaker, MD, for his assistance in searching the literature.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH CA140236 and TRDRP 26IR-0014 (Apollonio), NIH R01 CA-141661 (Ling), and a UCSF School of Pharmacy Pathway Project Grant (Pasetes). The funders played no role in the conduct of the research or preparation of the manuscript.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contribution
All authors conceived the protocol and manuscript, and completed data collection and analysis as described. SP completed the first draft; PL and DA supervised the project and revised the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
