Abstract
Deubiquitinating enzymes (DUBs) prevent ubiquitination by eliminating ubiquitin from their substrates. Deubiquitinating enzymes have important roles in a number of cell biology subfields that are highly relevant to diseases like neurodegeneration, cancer, autoimmune disorders, and long-term inflammation. Deubiquitinating enzymes feature a well-defined active site and, for the most part, catalytic cysteine, which makes them appealing targets for small-molecule drug development. Ubiquitin-specific protease 46 (USP46) is a member of the ubiquitin-specific protease family, the largest subfamily of DUBs. Over the past 10 years, some studies have steadily demonstrated the significance of USP46 in several biological processes, although it was identified later and early research progress was modest. Specifically, in the last few years, the carcinogenic properties of USP46 have become more apparent. In the current review, we provide a comprehensive overview of the current knowledge about USP46 including its characteristics, structure, inhibitors, function in diseases, especially in the nervous system, and the correlation of USP46 with cancers.
Introduction
Deubiquitinating enzymes (DUBs) are proteases that counteract the alteration caused by E3 ubiquitin ligases by removing the ubiquitin moiety from ubiquitylated substrates. Currently, it is known that DUBs are essential for controlling some physiological and pathological processes, including immunological homeostasis, carcinogenesis, and neurodegenerative illnesses. A growing body of research indicates that several DUBs may play unique roles in the development of tumors, including oncogenic, tumor-suppressive, or context-dependent activities. 1 These functions are mostly thought to be mediated by changes in the stability of proteins, enzymatic activity, or subcellular localization of their substrates. Significantly, a number of strong inhibitors that target the enzyme function of carcinogenic DUBs have been created, and preclinical models of their anticancer effectiveness indicate promise. 2 Therefore, investigating the distinct function of DUBs and their related effectors will offer fresh perspectives on the molecular foundation of cancer development. 3
There are about 100 DUBs in humans, and they are categorized into 7 groups based on their sequences and domain conservation including: ubiquitin-specific proteases (USPs), ubiquitin carboxy-terminal hydrolases (UCHs), ovarian tumor proteases (OTUs), Machado-Joseph disease proteases (MJDs), MIU-containing novel DUB family (MINDY) proteases, Jab1/MPN/MOV34 metalloenzymes (JAMMs), and Zn-finger and UFSP domain proteins (ZUFSPs), those are cysteine peptidases exception of JAMMs.3 -5 Among them, USPs is the largest group, with about 58 members. The size of the USP domains range from less than 300 to more than 800 amino acids. 6 It has been suggested that accessory domains, such as terminal extensions and the ubiquitin-associated domain (UBA), ubiquitin-interacting motif (UIM), and zinc finger ubiquitin-specific protease domain (ZnF-UBP), might provide USPs substrate specificity.6,7 The importance of several USPs in oncogenesis has been studied, and information about their pathological and physiological functions has recently started to accumulate. These USPs regulate cell cycle progression, injury to DNA repair, and tumor-related cellular signaling.6,8 -11
A recently found DUBs called ubiquitin-specific protease 46 (USP46) was shown to catalyze the deubiquitination of H2A and H2B for the initial time and may aid in the cultivation of Xenopus. 12 WDR48 controls the catalytic activity of USP46, which has a ubiquitin carboxyl-terminal hydrolase (UCH) domain. 13 Researches have demonstrated a connection between animal immobility and cell proliferation and USP46.14,15 The significance of USP46 in human malignancies has drawn more attention lately. It has been discovered that USP46 is crucial for the growth of tumor cells and is linked to metastasis. However, the existing body of knowledge regarding this protein is somewhat thin, and the information that is known regarding USP46’s function is scarce. Complex investigations are thus needed, especially those that deal with its traits and function. In this review, we provide the most comprehensive information related to USP46, including its characteristics, structure, inhibitors, function in diseases, especially in the nervous system, and the correlation of USP46 with cancers.
Characteristics and Structure of USP46
The human USP46 gene is located on chromosome 4p12 and spans approximately 1111 base pairs. The gene architecture is characterized by a complex exon-intron structure, comprising 11 exons separated by introns. The expected molecular size and theoretical isoelectric point (PI) of human USP46 are about 43 kDa and 6.39, respectively (calculated by EXPASy Compute PI/Mw). Based on an alignment of the amino acid structure of USP46 proteins from humans (Homo sapiens), black carp (Mylopharyngodon piceus), fathead minnow (Pimephales promelas), mice (Mus musculus), and zebrafish (Danio rerio) bcUSP46 is a molecule that has been genetically maintained and has a UCH region. According to phylogenetic study, the M. amblycephala USP46 and the bcUSP46 are the most closely related species. As a result, USP46 has evolved with great conservation. 16
Numerous USPs, as shown by previously published research, have modular architectures that include structural domains for localization and protein-protein interactions alongside the catalytic structural domains.
17
The finger, palm, and thumb domains are the 3 substructural domains that make up the main catalytic structure of USPs. The catalytic core is situated between the “palm” and “thumb” subdomains, whereas the “finger” holds the ubiquitin core, which supports the spherical domain of ubiquitin.
18
The prolonged finger domains, together with the palm and thumb domains, constitute the binding cavity for ubiquitin, which recognizes the extended ubiquitin tail and presents its C-terminus to the active-site cysteine in the catalytic central domain of USPs. This unique cysteine catalytic triad is found in the catalytic core domain, including Cys44, His313, and Asp329 (Figure 1). Jianping Yin et al determined the 1.9A ̊ resolution structure of a resolution structure of a covalently attached USP46 25–366 ubiquitin complex. The structure displays the broader fingers, thumb, and palm subdomains of the standard USP structure. The C-terminal sequence of ubiquitin substrate leads into the cysteine protease active site, where conjugated ubiquitin Gly76 is covalently bonded to the catalytic USP46 Cys44. The substrate is cradled between the fingers and the inner palm/thumb regions.
13
Moreover, a Z

The structure of USP46 catalytic domain was obtained from Homo sapiens (PDB ID: 5CVM). Purple is catalytic domain of USP46 (from His33 to Arg365). Yellow is ubiquitin binding to catalytic site of USP46. The blue is catalytic triad of USP46.
Furthermore, USP46 and USP12 are 2 small proteins that have the same active-site domain sequences and have a highly conserved catalytic domain with significant sequence similarity (93%). 20 A subfamily of USPs made up of USP1, USP12, and USP46 has a single USP domain and a common WDR partner called WDR48 (also known as USP1-associated factor 1; UAF-1) that, when bound, can increase the activity of these USPs. 21 The USP46-WDR48-WDR20 and the USP12-WDR48-WDR20 complex have a very comparable overall structure. 22 In addition, whereas USP1 cannot interact with WDR20, USP12, and USP46 may; further WDR48 and WDR20 can both independently and cooperatively stimulate the activities of USP12 and USP46.23,24 The structures of USP46 demonstrate that WDR48 interacts with the fingers subdomain of USP46. This binding may maintain the substrate binding, hence increasing USP46’s activity, without significantly altering the structure around the catalytic core. The USP46-WDR48 interface and the USP46-WDR48-Ub complex share nearly the same characteristics. 13 The interface between USP46 and WDR20 is facilitated through both hydrophobic and hydrophilic contacts. It involves numerous loops on WDR20’s top surface and 2 loops at the base of USP46’s palm subdomain. The residues of WDR20 implicated in the interactions with USP46 are rigorously conserved across species, according to sequence alignments. These findings imply that the complex formation and USP46 activity regulation are significantly influenced by the residues participating in the USP46-WDR20 interaction (Figure 2). 21

The structure of USP46-WDR48-WDR20 complex (PDB ID: 6JLQ). Blue: WD Repeat Domain 20 (WDR20), Purple: Ubiquitin-specific protease 46, Green: WD Repeat Domain 48 (WDR48).
Inhibitors of USP46
In accordance with its physiological and pathological involvement in several significant signaling pathways, USP46 has become a viable target for pharmaceutical intervention in a diverse array of cancers and neurological illnesses. Nonetheless, the creation of strong and focused USP46 inhibitors continues to be an appealing but difficult undertaking, in part because DUBs are so extensively conserved. 25 Discovering additional USP46 inhibitors is crucial because there is currently relatively little research on substances that might inhibit USP46.
Salidroside
For myocardial ischemia/reperfusion (I/R) damage, Salidroside exhibits an inhibitory action. 26 Myocardial infarction cell model simulation was achieved via I/R therapy of human cardiomyocytes. Cell viability, cell proliferation, and cell apoptosis were evaluated by CCK-8 assay, EdU assay, and flow cytometry analysis, respectively. Using qRT-PCR, the RNA expression levels of circ_0097682, miR-671-5p, F-box, and USP46 were found. The Western blotting test was used to quantify protein expression. Through the application of enzyme-linked immunosorbent assays, the amounts of interleukin (IL)-6, IL-1β, and TNF-α in the cell supernatant were determined. The administration of Salidroside alleviated the inhibitory impact that I/R had on the proliferation of AC16 cells, while also inducing apoptosis, inflammation, and oxidative damage in the cells. Salidroside reduced the expression of circ_0097682 in AC16 cells administered via I/R. Salidroside-mediated prevention of I/R-induced cellular damage included circ_0097682 expression reduced levels. Furthermore, in AC16 cells treated with I/R, circ_0097682 attached to miR-671-5p, and miR-671-5p inhibitors restored the effects of Salidroside pretreatment. Furthermore, miR-671-5p targeted USP46 in AC16 cells, and the addition of USP46 partly alleviated the effects of Salidroside pretreatment or circ_0097682 depletion in AC16 cells that had received I/R therapy. In conclusion, Salidroside prevented I/R-induced damage to AC16 cells by blocking the circ_0097682/miR-671-5p/USP46 pathway. 27
Galeterone
Ubiquitin-specific protease 46 is an enzyme for deubiquitinates androgen receptors (ARs), controlling AR signaling, and the P53 pathway. 28 Galeterone has been demonstrated to regulate the stability of the AR and MDM2 proteins in previous studies, while the exact mechanism is still unknown.29,30 After screening a wide panel of DUBs, Urszula L. McClurg et al found that Galeterone selectively inhibited USP12 (IC50 2.1–3.4 μM) and USP46 (IC50 3.4–4.2 μM), but had no impact on any other DUBs. Additional investigation revealed a strong functional relationship between USP12/USP46 silencing and Galeterone therapy. 28 Galeterone was able to prevent cell development even in AR-negative prostate cancer (PC) models by blocking the enzymatic activity of USP12 and USP46. This indicates the significance of USP12 and USP46 in controlling the P53 and AKT pathways.28,31 -34 Their findings suggest that galeterone or other drugs may be a very efficient antitumor therapy by inhibiting USP12 and USP46 activity; however, Galeterone benefits may be restricted in PC patients who express AR V (Table 1). 28
Small molecule inhibitors of USP46.

Function of USP46 in the Nervous System
Ubiquitin-specific protease 46 is crucial for synaptic function and plasticity in the central nervous system by deubiquitinating and stabilizing key synaptic proteins such as AMPA and GABA receptors. Dysregulation of USP46 has been linked to neurological disorders like major depressive disorder (MDD), intellectual disability, autism spectrum disorder (ASD), and epilepsy. Variants of the USP46 gene affect neurotransmitter systems, contributing to mood disorders, cognitive impairments, and increased seizure susceptibility. As a result, USP46 is a promising therapeutic target, with ongoing research focusing on developing specific inhibitors or modulators for treating these neurological conditions.
To elaborate the mechanism, most immediate excitatory transmission in the brain is accomplished by alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors (AMPARs), and regulating synaptic AMPAR levels is crucial for synapse growth and operation. GluA1–A4 pore-forming subunits may be aggregated into hetero-tetramers to create AMPARs. The biophysical and pharmacological features of the channel are determined by the makeup of its subunits and their associations with accessory subunits. 35 Alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors can be impacted by several posttranslational modifications, such as ubiquitination, which regulates receptor trafficking and degradation. 36 According to a research by Huo et al 37 in 2015, USP46 is the AMPARs’ DUB. They discovered that K63-type ubiquitination affects AMPARs and that both in vivo and in vitro, USP46 can deubiquitinate AMPARs. Expression of USP46 causes AMPAR ubiquitination in heterologous cells and neurons to significantly decrease, along with AMPAR decomposition rate diminution and surface AMPAR accumulation increase. In contrast, RNA interference (RNAi)-mediated knockdown of USP46 results in increased AMPAR ubiquitination and decreased surface AMPARs at neuronal synapses. Investigations of tiny stimulating postsynaptic currents repeatedly indicate that neurons expressing USP46-selective RNAi have weaker synapses. These findings show that AMPAR ubiquitination and degradation are regulated by USP46, which could be crucial for neurological activity and synaptic plasticity. 37 Ubiquitin-specific protease 46 integrates with GluA1 and PSD95 at synapses in developed astrocytes and is expressed in all regions of the brain, including the hippocampus, amygdala, cerebellum, and prefrontal cortex.15,37 Decreased mEPSC amplitudes, lower surface, and total levels of GluA1, and higher amounts of ubiquitinated GluA1 are the outcomes of USP46 knockdown, which is consistent with USP46’s function in deubiquitinating mammalian AMPARs. 37 All of these findings point to a conserved mechanism in which USP46 deubiquitinates AMPARs at synapses to prevent their breakdown and to encourage their recycling to the cell surface, which in turn impacts synapse function. 38
In addition, ubiquitin attaches directly to the cytoplasmic tail of glutamate receptors-1 (GLR-1), triggering clathrin-mediated endocytosis and subsequent destruction. Ubiquitin-specific protease 46 was the first DUB to control GLRs in Caenorhabditis elegans, as shown by a concentrated RNA interference screen. Ubiquitin-specific protease 46 dysfunctional mutants show reduced amounts of GLR-1 at synapses and elevated levels of ubiquitinated GLR-1. 39 Mutants losing USP46 show reduced levels of GLR-1 at synapses and elevated amounts of ubiquitinated GLR-1. 40 Ubiquitin-specific protease 46 loss-of-function mutants do not alter the abundance of GLR-1(4KR), a mutant receptor that is not ubiquitinated. This suggests that GLR-1’s abundance is regulated by USP46 deubiquitinating it. Because USP46 loss-of-function mutants exhibit decreased cell surface levels of GLR-1 and related abnormalities in GLR-1-dependent behaviors, USP46 control of GLR-1 is physiologically relevant. 38 Kowalski et al 40 suggested a hypothesis in which USP46 functions in endosomes to improve GLR-1 stability and recycling to the cell surface, based on evidence demonstrating USP46’s partial colocalization with endosomes. According to recent research, vertebrate GluA1(KR), which is not ubiquitinated, eludes lysosomal degradation and returns to the cell surface, supporting this concept. 41
In mice, USP46 is additionally involved in controlling the GABAergic system. Tomida et al 15 found that alterations in behaviors resembling depression, compatible with an antidepressive state, are also present in an inbred strain of mice (CS strain), known to have problems in circadian rhythms. Through quantitative trait locus mapping, a 3 bp deletion in a conserved lysine (ΔK92) in USP46 was found. The mice were CS. Significantly, depression-like behaviors are altered similarly in USP46 knock-out (KO) mice, 42 and these behaviors were restored in CS animals by wide expression of a wild-type USP46 transgene. 15 It is possible that USP46 activity is not entirely eliminated by the ΔK92 mutation since the behavioral effects in USP46 (ΔK92) mutant mice were not as pronounced as those in USP46 KO mice. 42 In fact, studies for in vitro deubiquitination showed that USP46(ΔK92) still possesses some enzymatic activity. 43
Ubiquitin-specific protease 46 is hypothesized to have a role in the regulation of brain activity, synaptic transmission, and synaptic plasticity by regulating glutamatergic AMPARs. 37 Ubiquitin-specific protease 46 is also related to the control of the GABAergic system in mice, which preserves rapid inhibitory transmission in the brain. 15 Mice that lack USP46 exhibit depressive-like b ehaviors 44 and express less glutamic acid decarboxylase (GAD67), an enzyme involved in the production of GABA. 15 All things considered, USP46’s role in controlling a variety of synaptic receptors emphasizes how crucial USP46 is for the development of synapses and the shape of neurons. 37 Although the exact mechanism is unknown, several other studies indicate that USP46 influences both presynaptic and postsynaptic components of the GABA system. One theory is that USP46 acts as a compensation mechanism to a basic deficiency in excitatory neurons’ AMPAR degradation, which might have an indirect effect on the GABA system. Future research should focus on determining if USP46 acts in GABA neurons and whether it directly deubiquitinates GABA system elements. 38
Dysregulation of USP46 in Cancer
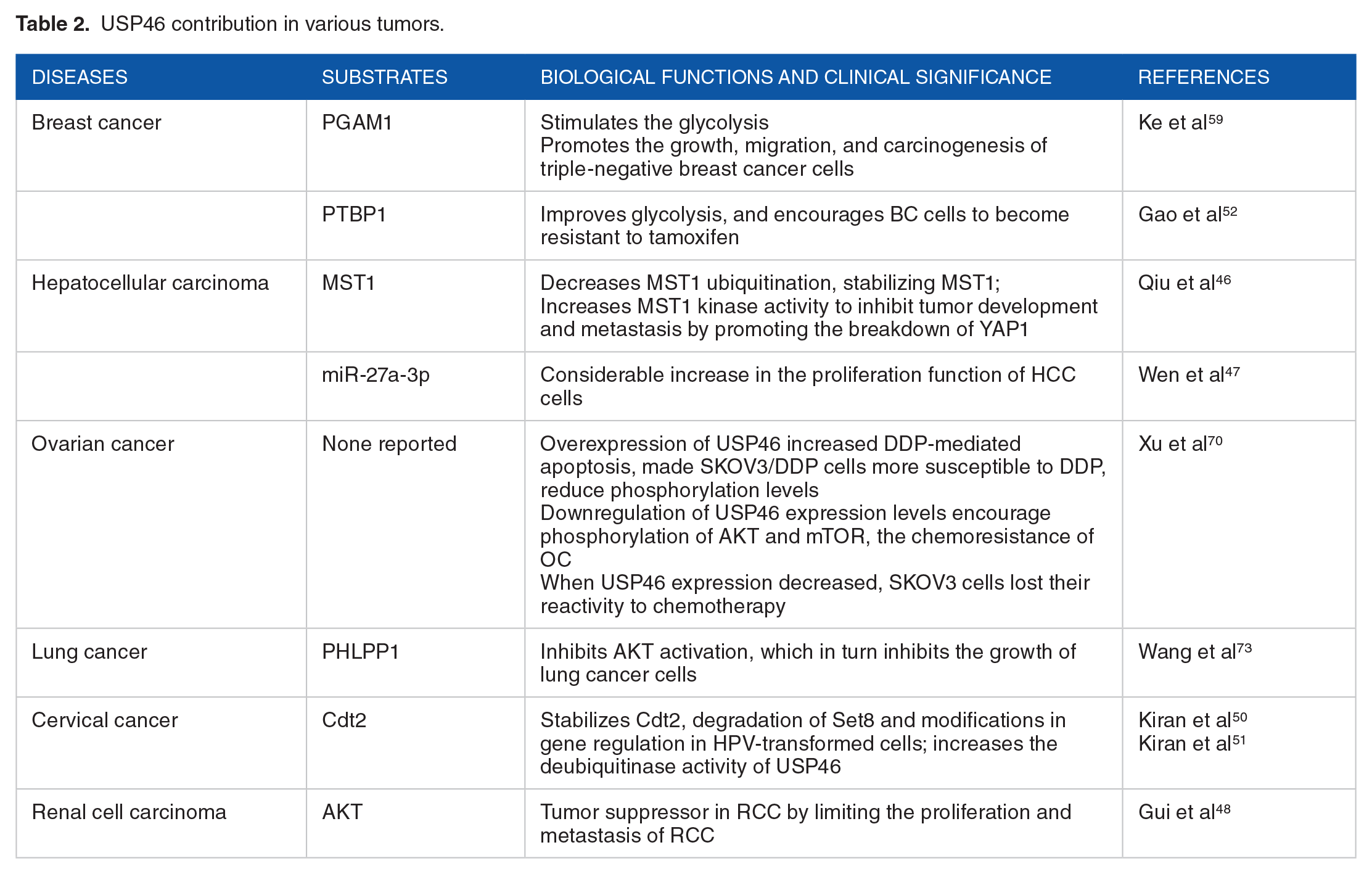
Ubiquitin-specific protease 46 is a multifunctional DUB that modulates certain substrates to regulate a variety of biological processes.12,40,43,45 Numerous studies have demonstrated the many functions of USP46 in the development and spread of tumors. It acts as a tumor suppressor in hepatocellular carcinoma (HCC),46,47 renal cell carcinoma (RCC), 48 and colon cancer, 32 inhibiting the progression of these cancers when highly expressed. However, in esophageal squamous cell carcinoma 49 and cervical cancer (CC),50,51 USP46 increases tumor growth. 52 The following section focuses on understanding the role of USP46 related to cancers.
Breast cancer
In the world, breast cancer (BC) is the most frequently recognized carcinoma in women and the leading cause of cancer-related deaths. Around the world, the prevalence of BC is steadily rising. Patients with BC make up to 36% of all cancer patients. In 2018, approximately 2.089 million women received a BC diagnosis.53,54 All across the world, the number of instances of this malignant tumor is rising, although it is more common in developed nations. Industrialized nations account for nearly half of all cases. 54 This trend is mainly due to the so-called Western lifestyle, associated with a poor diet, nicotinism, excessive stress, and little physical activity. 55 Estrogen receptor (ER+) BC represents up to around 70% of the BC subtypes. 56 Even with the significant therapeutic advantages that endocrine treatment has provided for ER+ BC patients, medication resistance and relapse continue to be significant issues.57,58 Thus, to lower the death rate for patients with BC, it is imperative to aggressively seek out novel therapeutic approaches as well as prompt diagnostic and prognostic techniques.
Ubiquitin-specific protease 46 has been demonstrated to be overexpressed in BC cells, and this was associated with a bad prognosis for the patients. Ubiquitin-specific protease 46 upregulation regulated phosphoglycerate mutase 1 (PGAM1)-mediated glycolysis, which in turn promoted the growth, migration, and carcinogenesis of triple-negative breast cancer (TNBC) cells. After USP46 was knocked down, cell death was triggered. 59
According to a recent study, USP46 is essential for controlling Tamoxifen resistance in BC cells. In particular, USP46 collaborates with PTBP1 to raise the protein levels of PTBP1, which in turn increase the PKM2/PKM1 ratio, improves glycolysis, and encourages BC cells to become resistant to tamoxifen. This pathway is particularly important in BC cells that express a lot of USP46. These results highlight the critical function of the USP46/PTBP1/PKM2 axis in inducing glycolysis and tamoxifen resistance in BC cells, providing a therapeutic approach that shows promise in addressing tamoxifen resistance in BC endocrine treatment. 52
Hepatocellular carcinoma
The third most frequent cause of cancer-related deaths globally is HCC, one of the most prevalent malignant tumors with a significant morbidity rate.60,61 To establish a theoretical foundation for the therapy of this illness, it is therefore very critical to investigate the molecular process underlying HCC growth and metastasis. In HCC tissues, USP46 is downregulated, and low USP46 levels are linked to a worse prognosis for HCC patients. In functional investigations, USP46 knockdown increased cell proliferation and invasiveness in vitro and in vivo whereas overexpression of USP46 decreased HCC cell proliferation and metastasis. Moreover, we discovered that USP46 inhibits YAP1 to reduce HCC cell growth and metastasis. The suppression of cell growth and metastasis brought on by USP46 overexpression was reversed by ectopic expression of YAP1. Scientifically, USP46 stimulates MST1 expression, which in turn antagonizes YAP1 to inhibit the development of HCC by promoting the breakdown of YAP1. Furthermore, USP46 directly binds to the MST1 protein and lowers its ubiquitination, stabilizing it. When combined, USP46 could represent a potential HCC tumor suppressor. In addition, USP46 functions as an MST1 DUB, enhancing MST1 kinase activity to inhibit tumor development and metastasis. These findings suggest that activating USP46 might be a viable therapeutic approach for HCC. 46
Through the use of RNA-seq, quantitative polymerase chain response, Western blotting, and luciferase report, USP46 was identified as the main target gene of miR-27a-3p in HCC. Ubiquitin-specific protease 46 mRNA is excessively overproduced, and this might be the primary cause of USP46’s poor expression in HCC. Over-expressing miR-27a-3p or downregulating USP46 in HCC cells resulted in a considerable increase in the proliferation function of HCC cells, whereas on the contrary, these actions dramatically reduced the proliferation of HCC cancer cells. Two potentially successful treatment approaches for HCC were activating USP46 expression or targeting suppression of miR-27a-3p. 47 Furthermore, it has been found that miR-27a functions as an inducer of tumors and is elevated in a number of human malignancies, including ovarian cancer (OC), lung cancer (LC), endometrial cancer, and colorectal cancer.43,62 -65 Noncoding RNAs, however, have been demonstrated to be important regulators in malignancies. But as of right now, no treatment approach based on noncoding RNAs is being used in clinical settings.63,66 -68 As a result, targeting the miR-27a/USP46 axis could represent a useful cancer treatment strategy.
Ovarian cancer
When gynecological malignancies are detected in women, OC is the primary cause of mortality. In general, it ranks as the sixth most common cause of mortality for women. The biggest challenge for treating advanced OC is resistance to clinical chemotherapy, even with the availability of several medications for treating and improving the prognosis of OC. Therefore, there is an urgent need for innovative treatment methods based on a deeper knowledge of the molecular characteristics of this malignancy as well as efficient preventive and detection measures.69,70
Ubiquitin-specific protease 46 expression amounts in DDP-resistant OC were also examined in vitro using the OC cell lines, SKOV3, and SKOV3/DDP. Xu et al found that USP46 expression levels were downregulated in OC chemoresistant tissues as compared to chemosensitive tissues, indicating that aberrant expression of USP46 might prove essential to the establishment of resistance in OC. The results showed that overexpression of USP46 increased DDP-mediated apoptosis and made SKOV3/DDP cells more susceptible to cisplatin (DDP). Contrarily, once USP46 expression was knocked down, SKOV3 cells lost their reactivity to chemotherapy. According to these results, USP46 could be a useful marker for identifying OC patients who are undergoing platinum-based chemotherapy and who are both chemoresistant and chemosensitive. In addition, Bcl-2 expression was elevated while Bax, caspase-3, and caspase-9 expression were downregulated upon USP46 suppression. On the contrary, these apoptotic-related factors’ expression levels had opposite effects due to the amplification of USP46. These results showed that USP46 could influence OC cells’ chemoresistance through the Bcl-2/caspase-3 signaling system. While USP46 overexpression may reduce phosphorylation levels, downregulation of USP46 expression levels may also encourage phosphorylation of AKT and mTOR. According to the previously reported results, controlling AKT/mTOR phosphorylation had an impact on the proliferative capacity of OC cells, which in turn controlled the chemoresistance of OC. Thus, USP46 might be a potential therapeutic target for DDP-resistant OC. 70
Lung cancer
With 11.6% of all instances identified, LC is the most often diagnosed kind of cancer worldwide, with growing incidence and mortality rates. There are 236 740 new instances of LC in the United States in 2022, and 130 180 people will die from the disease. 71 Lung cancer continues to be the primary cause of cancer deaths worldwide (18.4% of all cancer deaths), resulting in a large financial loss and social burden.71,72 Patients with LC now have a higher survival rate thanks to recent developments in cancer therapy, nevertheless there are still unmet therapeutic requirements. To effectively treat LC, it is essential to understand the molecular pathways that underlie carcinogenesis.
Ubiquitin-specific protease 46 expression has been reported to be downregulated in LC patients, indicating that USP46 might have a tumor suppressor function in LC. This finding is in line with data from research on RCC 48 and colorectal cancer. 32 In LC cells growing normally and in cells exposed to IR, USP46 effectively prevents cell division. On the contrary, radiation therapy combined with an AKT inhibitor inhibits the growth of LC cells, whereas downregulating USP46 promotes the growth of LC cells. These findings have great practical implications because they suggest that a combination of radiation and AKT inhibitors may be a useful treatment option for treating USP46-downregulated LC. The mechanism by which USP46 inhibits LC proliferation involves deubiquitinating Pleckstrin homologous domain leucine-rich repeating protein phosphatases 1 (PHLPP1), which in turn inhibits AKT activation. These results provide a strong basis for in vivo studies that will assess USP46’s precise function in the evolution of LC in more physiologically realistic mice models. It will be fascinating to look into whether USP46 plays a major role in the development of the main tumor, metastasis, or both. Future research will focus on these inquiries. 73
Cervical cancer
Cervical cancer is one of the main causes of cancer-related mortality among women. Over the past 30 years, the increasing proportion of young women affected by CC has ranged from 10% to 40%. 74 According to the World Health Organization (WHO) and International Agency for Research on Cancer (IARC) estimates, 529 000 new cases of CC were reported worldwide in 2008. 75 Cervical cancer is the fourth most common disease globally in terms of diagnoses and the fourth main cause of cancer-related mortality for women in 2018, with an estimated 570 000 cases and 311 000 deaths. 72 It is critical that we comprehend the pathogenic process and available therapeutic options for this malignancy.
In Human Papillomavirus-induced (HPV-induced) malignancies, E6 oncoproteins from high-risk variants are crucial for cell division and metamorphosis. According to Shashi Kiran’s 2018 investigation, the E6 of high-risk HPV specifically recruits USP46, a cellular deubiquitinase, to deubiquitinase and consolidate Cdt2/DTL, but not low-risk HPV. Cell proliferation is stimulated and the amount of Set8, an epigenetic writer, is limited by the stabilization of Cdt2, a component of the CRL4Cdt2 E3 ubiquitin ligase. The growth of HPV-transformed cells requires USP46, but not that of cells lacking HPV. Human cervical malignancies have higher levels of Cdt2, and xenografts with HPV-transformed tumors develop more slowly when USP46 is knocked down. One of the major functions of carcinogenic E6 is the engagement of a physiological deubiquitinase that regulates critical cellular proteins. Because of the significance of the E6-USP46-Cdt2-Set8 pathway in HPV-induced malignancies, USP46 is a potential for treatment of these cancers. 50
By 2022, Shashi Kiran et al demonstrated that, in CC biopsies, Cdt2 stabilization in HPV-induced cancers results in a reduction in H4K20me1 or H4K20me3, which can be identified through immunohistochemistry, as well as a CRL4-Cdt2 substrate, the histone H4K20 mono-methyltransferase Set8. E6 suppression reduces Cdt2 and raises Set8 in vitro in HPV-transformed cancer cell lines. Co-knockdown of Set8 demonstrates that amplification in Set8 is responsible for a portion of the alterations in gene expression brought about by E6 knockdown. This gene collection included both EGFR and EGFR regulated genes. Regarding the mechanism through which E6 stabilizes Cdt2, it could be seen that purified E6: USP46 complex exhibits significantly greater deubiquitinase activity in vitro than USP46 alone. This indicates that E6 may engage exclusively with USP46 regardless of other proteins and might serve as a stand-in for UAF1 and WDR20, 2 known activators of USP46. Three distinct but overlapping portions of the substrate are necessary for E6 to stabilize the substrate, according to deletion mapping of Cdt2: USP46. It is implied that interaction with DDB1 (and the remainder of the CRL4 complex) or with the substrate of the CRL4-Cdt2 E3 ligase is not required for E6: USP46 to interact with and stabilize Cdt2 because the helix-loop-helix region or the WD40 repeat motivated beta-propeller configuration of Cdt2 is irrelevant for the stabilization. The particular nature of the mechanism is shown by the discovery that 50 amino acid sequences within the 731 amino acid Cdt2 protein are crucial for the stabilization by E6. In conclusion, E6 causes degradation of Set8 and modifications in gene regulation in HPV-transformed cells, increases the deubiquitinase activity of USP46, and stabilizes Cdt2 by using numerous sites on Cdt2. 51
Renal cell carcinoma
One of the most common carcinogenic disorders of the urination process RCC is a heterogeneous, metastatic condition that is unresponsive to standard treatment approaches.76,77 Renal cell carcinoma causes about 140 000 fatalities yearly and accounts for around 95% of kidney cancers.72,76,78,79 Because of the inadequate evaluation, the frequency of RCC is gradually increasing. According to A. Sánchez-Gastaldo et al, 80 over 30% of patients with RCC are identified with metastasized tumors, and many patients subsequently experience recurrence following surgical therapies. Surgical resection therapy remains an effective and curative therapeutic method for RCC, even with the development of innovative immunotherapeutic techniques.81,82 Meanwhile, patients with RCC continue to have a very poor 5-year overall survival rate.83,84 It is consequently imperative to uncover the molecular processes underpinning the RCC regulatory system.
Ubiquitin-specific protease 46 has been shown by Dingwen Gui et al to operate as a tumor suppressor in RCC by limiting the proliferation and metastasis of RCC cells. It was evident that the USP46 high-expression group had a favorable prognosis and survival rate. In addition, the RCC patients’ USP46 mRNA expression was lower than that of the normal controls, and the tumor malignancy grade was inversely linked with USP46 expression. In vitro, ectopic USP46 expression significantly decreased tumor cell motility and cell proliferation, and vice versa. AKT pathway downregulation caused by USP46 contributed to abnormalities in cell migration and proliferation. They revealed a novel method by which USP46 suppresses the AKT signaling pathway, hence impairing RCC survival. Verification of AKT inactivation by USP46 is also employed in different tumor scenarios. These results not only offer a fresh perspective on the molecular control of RCC, but they also point to USP46 as a possible target in the development of RCC. To determine the functional effects of USP46 in RCC and to further understand the regulatory mechanism of USP46-mediated AKT inactivation, more research is required. 48
It will be fascinating to look into whether USP46 deletion alone or in conjunction with other known mutations is sufficient to cause cancers. In addition, it is yet unknown if cancer patients have recurring mutations in the USP46 gene and whether these changes are associated with other established driver mutations. PI3K/AKT gene alterations will probably be correlated in some way with a variety of cancer types. It is imperative that genetic data sets be thoroughly analyzed to offer more precise answers to these significant problems (Table 2).
USP46 contribution in various tumors.
Discussion
Deubiquitinating enzymes have grown in popularity as cancer therapeutic targets throughout the last 10 years. Ubiquitin-specific protease 46 is a significant DUB that regulates most, if not all, of the characteristics of cancer, including the signaling pathways that promote cell cycle progression, proliferation, apoptosis, and resistance to therapy. Over the last 10 years, a number of initiatives have improved our knowledge about USP46. Studies conducted on USP46 have demonstrated its important and diverse functions in both physiological and pathological conditions, especially in carcinogenesis, highlighting the fact that USP46 is becoming a desirable target for the treatment of some linked illnesses. The discovery that USP46 seems to have 2 roles in a variety of biological processes and clinical situations, however, emphasizes the necessity of assessing USP46’s function in many settings and illnesses.
We provided the key details about USP46 in this review, including its properties, structure, inhibitors, and function in various illnesses (Figure 3). More research on the regulation and function of USP46 in both healthy and pathological conditions will shed light on the protein’s specificity and activity, paving the way for the development of selective USP46 inhibitors as an effective treatment for cancer and other illnesses. Ubiquitin-specific protease 46’s potential as a therapeutic target is indeed promising, yet it remains largely unexplored in clinical trials or therapeutic development. The enzyme is pivotal in regulating synaptic function and has been linked to several neurological disorders, including MDD, intellectual disabilities, and epilepsy. In addition, USP46 has shown potential relevance in cancer research, where its role in modulating protein stability and cellular signaling pathways could influence tumor progression. Despite these implications, specific inhibitors or modulators of USP46 have not yet advanced to clinical trials. The development of selective USP46-targeted therapies requires further research to understand its mechanisms fully and to identify suitable compounds that can effectively modulate its activity while minimizing potential side effects.

Summary information of USP46 protein.
Thus, the goal of future research should be to find USP46-specific inhibitors that are more potent. The research and discovery of USP46 as an appropriate therapy is going to be facilitated by this improved understanding of USP46 functions and modulation for medicinal purposes.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Author Contributions
KHTP: Conceptualization, Methodology, Validation, Investigation, Writing—Original Draft, Visualization. MHT: Conceptualization, Visualization, Review. LBN: Conceptualization, Visualization, Review. PTVP: Writing—Review & Editing, Supervision. TKN: Writing—Review & Editing, Supervision.
Ethical Approval
Not applicable to this article.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
