Abstract
Background
The contact phase of coagulation is of physiologic/pathophysiologic importance, whenever unphysiologic polynegative substances such as cell fragments (microparticles) get in contact with blood. There are several clinically used inhibitors of intrinsic thrombin generation. Here the inhibitory concentrations 50% (IC50) of these anticoagulants are measured by the highly specific thrombin generation assay INCA.
Methods
Unfrozen pooled normal citrated plasma in polystyrole tubes was supplemented at 23°C in duplicate with 0–2 IU/ml low molecular weight heparin (dalteparin), 0–2 IU/ml unfractionated heparin, 0–500 KIU/ml aprotinin, or 0–40 mM arginine. 50 μl plasma or 1 IU/ml thrombin standard were pipetted into a polystyrole microtiter plate with flat bottom. 5 μl SiO2/CaCl2 - reagent (INCA activator) were added and after 0–30 min incubation at 37°C 100 μl 2.5 M arginine, pH 8.6, were added; arginine inhibits hemostasis activation and depolymerizes generated fibrin within 20 min at 23°C. The in the physiologic 37°C incubation phase generated thrombin was then chromogenically detected. The intra-assay CV values were < 5%.
Results and Discussion
The approximate IC50 were 0.01 IU/ml dalteparin, 0.02 IU/ml heparin, 25 KIU/ml aprotinin, and 12 mM arginine. The efficiency of any anticoagulant on intrinsic thrombin generation should be measured for each individual patient.
Abbreviations
IIa, thrombin; δA, increase in absorbance; APTT, activated partial thromboplastin time; CRT, coagulation reaction time (at 37°C in water-bath); F-wells, polystyrole microtiter plates with flat bottom; IC50, inhibitory concentration 50%; INCA, intrinsic coagulation activity assay; IU, international units; KIU, kallikrein inhibiting unis; LMWH, low molecular weight heparin; mA, milli-absorbance units; PSL, pathromtin SL®; RT, room temperature (23°C); U-wells, polystyrole microtiter plates with round bottom.
Introduction
Low molecular weight heparins (LMWH) are essential drugs for patients inside and outside the hospital; unfortunately, the global hemostasis assay APTT is not sensitive to LMWH [Fareed et al. 2004]. There is clinical need for a simple physiologic global hemostasis test that monitors the anticoagulant power of LMWH. The only LMWH - routine assay currently available is the anti-Xa assay, that is available in a clotting [Denson and Bonnar, 1973] and in a chromogenic version [Teien et al. 1976]. However, the target therapeutic dose range in the anti-Xa assay is 0.4–0.7 IU/ml heparin in the anti-Xa assay but only 0.2–0.4 IU/ml heparin in the APTT [Kitchen, 2000]. This discrepancy indicates that the Xa activity added in the anti-Xa assays might be supra-physiological and that the other pharmacologic actions of the heparins than just the inhibition of activated factor X are not reflected by the anti-Xa assay. Recently, a new test for thrombin activity in plasma was developed [Stief,2006; Stief et al. 2006]. This assay uses (I) a chromogenic thrombin substrate at final concentrations < 0.6 mM and (II) arginine at final concentrations > 1 M, resulting in highly specific thrombin determination. Of diagnostic importance are the following chromogenic thrombin tests that all base on this new technique:
basal thrombin activity (IIa) recalcified coagulation activity assay (RECA) intrinsic coagulation activity assay (INCA) extrinsic coagulation activity assay (EXCA).
As equipment only a normal microtiterplate reader is required; these only 1 or 2 measuring point - based tests are easy to handle with high reproducibility (all have intra-assay CV values less than 5%), they are fast and economical, and can thus be used in routine diagnostic.
Material and Methods
INCA coagulation reaction time
The INCA is a new simple global chromogenic hemostasis test that requires only a two-point determination of thrombin generation in the important ascending part of the thrombin activity curve, i.e. the ratio between thrombin at a second time point divided by thrombin at a first time point should be > 1.
50 μl unfrozen plasma (1 part 106 mM citrate + 9 parts of venous blood; centrifuged at 2800 g (4000 rotations per minute at 23°C) are pipetted into flat bottom polystyrole microtiter plate wells (F-wells, Polysorp®, NUNC, Wiesbaden, Germany; article nr. 446140). The INCA is started by addition of 5 μl SiO2/CaCl2 reagent (freshly with 278 mM CaCl2 1:10 diluted Pathromtin SL®, DadeBehring, Marburg, Germany). Always H2O-rinsed completely emptied new disposable polypropylene tips for the Eppendorf-multipette® are used: if this disposable tip of the multipette for addition of the INCA-activator to the reaction well is not rinsed, the intrinsic thrombin generation is strongly inhibited; some releasable plastic material in the new tip seems to act like a heparinoid; if the untreated polystyrole F-well plate (Polysorp®) is replaced by an irradiated one (Maxisorp®) or a U-well plate (for sample volumes < 25 μl U-wells result in higher precision than F-wells), then this plastic material inhibits the intrinsic coagulation to some extent.
INCA inhibition by heparins
Unfrozen pooled normal citrated plasma in 5 ml polystyrole tubes was supplemented with 0–2 IU/ml dalteparin (a LMWH; Fragmin P®, Pharmacia, Erlangen, Germany) or with 0–2 IU/ml unfractionated heparin (Roche, Basel, Switzerland) and kept at 23°C. 50 μl samples were pipetted into F-wells, and the INCA was started by addition of 5 μl SiO2/CaCl2 reagent. After 3 min coagulation reaction time (a CRT of 3 min is the initial time point of thrombin generation) at 37°C in the water-bath, 100 μl 2.5 M arginine, pH 8.6 (Sigma, Deisenhofen, Germany) were added. The plate was withdrawn from the water-bath. After 20 min (23°C = RT) 10 μl 3.85 mM chromogenic thrombin substrate HD-CHG-Ala-Arg-pNA (Pentapharm, Basel, Switzerland) in H2O were added and δA/t was determined by a microtiterplate reader with a 1 mA resolution (Milenia-DPC, Los Angeles, USA). The result was standardized against 1 IU/ml IIa in 6.7 % human albumin (Kabi, Stockholm, Sweden) replacing the plasma sample.
INCA kinetic in plasma supplemented with heparins
20 μl unfrozen pooled normal plasma, supplemented with 0 IU/ml, 0.063 IU/ml, 0.13 IU/ml, 0.25 IU/ml dalteparin or 0 IU/ml, 0.063 IU/ml, 0.13 IU/ml, 0.25 IU/ml, or 0.5 IU/ml unfractionated heparin were incubated in U-wells (NUNC) with 2 μl SiO2/CaCl2 reagent. After 0–30 min (37°C), 50 μl 2.5 M arginine, pH 8.6, were added. The plate was withdrawn from the water-bath. After 20 min (23°C) 20 μl 0.77 mM chromogenic thrombin substrate in 2 M arginine was added and the linear δA/t (RT) was determined.
INCA inhibition by aprotinin
Unfrozen pooled normal plasma in polystyrole tubes was supplemented with 0–400 KIU/ml aprotinin (Bayer, Leverkusen, Germany). The INCA was performed with 20 μl plasma samples in U-wells and 2 μl INCA-activator. After 0–30 min (37°C), 50 μl 2.5 M arginine, pH 8.6, were added. After 20 min (RT) 20 μl 0.77 mM chromogenic substrate in 2 M arginine was added and the linear δA/t (RT) was determined. Additionly, the APTT of the aprotinin-supplemented plasma samples was measured by a Behring Coagulation Timer®.
INCA inhibition by arginine
Unfrozen pooled normal plasma in polystyrole tubes was supplemented with 0–40 mM arginine (Braun, Melsungen, Germany) or alkaline arginine (pH = 8.7; Sigma). 50 μl samples were tested in the INCA (F-wells).
Influence of fibrin on anticoagulants in INCA
Unfrozen pooled normal plasma with 2.8 g/l fibrinogen was unsupplemented and supplemented with purified human fibrinogen (Haemocomplettan, Aventis, Frankfurt, Germany; the preparation contained about 1% soluble fibrin [Stief, 2000] that might act as antithrombin I [Mosesson, 2005]) to a final active fibrinogen concentration of 4.0 g/l. The plasmas were then unsupplemented or supplemented with 0.01 IU/ml dalteparin, 0.02 IU/ml heparin, 25 KIU/ml aprotinin, or 12 mM arginine. The INCA was performed as described above. Addition of 0.1% Triton X 100® (Sigma) final test concentration accelerates the decrease of the fibrin-related turbidity about 7fold, i.e. instead of 20 min arginine reaction time 3 min is sufficient before starting the IIa detection phase of the assay. A turbidity increase in plasma occurs within minutes, if the final thrombin activity exceeds 0.02 IU/ml [Stief,2007 in press].
Results
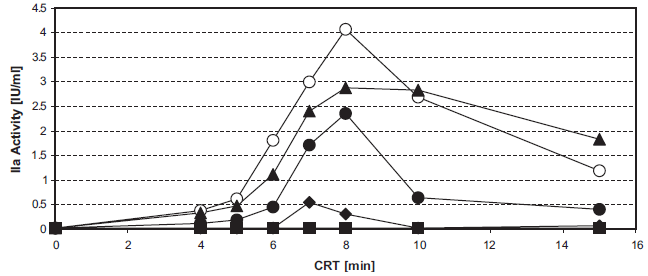
The action of heparins on INCA is shown in Figure 1. The 50% inhibitory concentrations (IC50) were about 0.01 IU/ml for LMWH and 0.02 IU/ml for unfractionated heparin (Fig. 1a). In therapeutical heparin concentrations (about 0.2 IU/ml), the thrombin generation in the INCA at CRT 20 min does not exceed 0.5 IU thrombin/ml sample (Fig. 1b, c). The INCA curves are evaluated in their important pre-maximum phase.
Inhibition of intrinsic thrombin generation by heparins. Figure 1a. IC50 determination. Unfrozen pooled normal plasma, supplemented with dalteparin (●) or unfractionated heparin (▪) was analyzed in the INCA (50 μl sample into F-wells). The INCA was performed with a thrombin generation time = coagulation reaction time of 3 min (initial phase of thrombin generation). 10 μl 3.85 mM CHG-Ala-Arg-pNA in H2O were added and AA/t was measured. 1 IU/ml IIa had 12.2 mA/min RT. Reaction kinetic of dalteparin. 20 μl unfrozen pooled normal plasma, supplemented with 0 IU/ml (O), 0.063 IU/ml (♦), 0.13 IU/ml (▴), or 0.25 IU/ml (A) dalteparin was incubated with 2 μl SiO2/CaCl2 reagent. After 0–30 min CRT at 37°C in the water-bath 50 μl 2.5 M arginine, pH 8.6, was added. After 20 min (RT) 20 μl 0.77 mM chromogenic thrombin substrate in 2 M arginine was added and the linear AA/t (RT) was determined. Reaction kinetic of unfractionated heparin. 20 μl unfrozen pooled normal plasma, supplemented with 0 IU/ml (O), 0.063 IU/ml (▪), 0.13 IU/ml (♦), 0.25 IU/ml (▴), or 0.5 IU/ml (•) heparin was incubated with 2 μl SiO2/CaCl2 reagent. After 0–30 min CRT at 37°C in the water-bath 50 μl 2.5 M arginine, pH 8.6, was added. After 20 min (RT) 20 μl 0.77 mM chromogenic thrombin substrate in 2 M arginine was added and the linear δA/t (RT) was determined.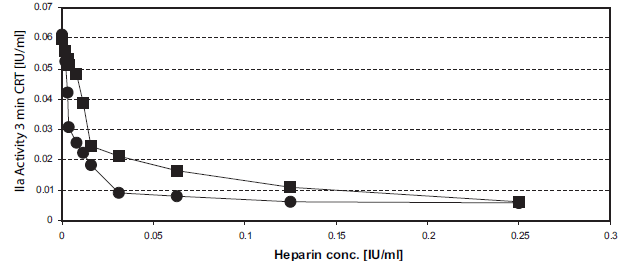
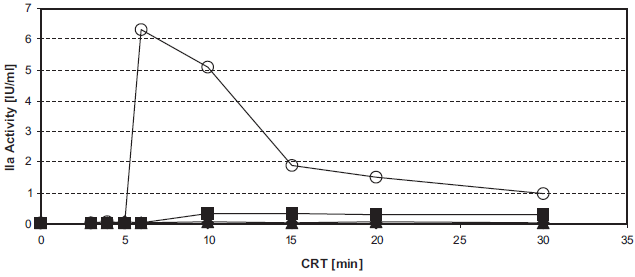
The IC50 of aprotinin on the INCA is about 25 KIU/ml (Fig. 2). This reflects the extreme sensitivity of the INCA: in the usual APTT the aprotinin concentration that prolongs the normal APTT 1.5 fold is 300 KIU/ml.
Inhibition of intrinsic thrombin generation by aprotinin. Figure 2a. 20 μl unfrozen pooled normal plasma, supplemented with 0 KIU/ml (O), 25 KIU/ml (ô), 50 KIU/ml (▵), 100 KIU/ml (□), 200 KIU/ml (▴), or 400 KIU/ml (•) aprotinin, were tested in the INCA (U-wells).
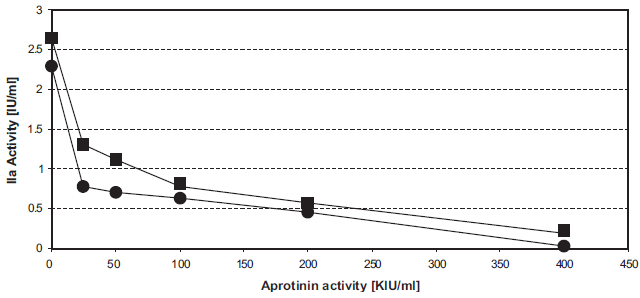
CRT = 10 min (•), CRT = 15 min (▪).
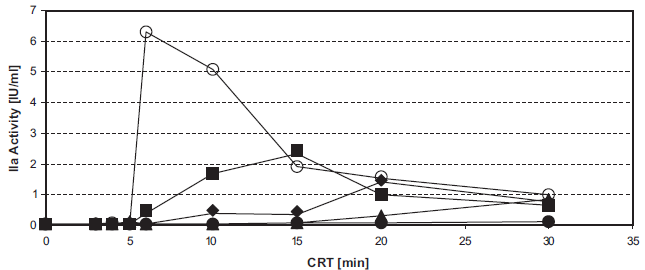
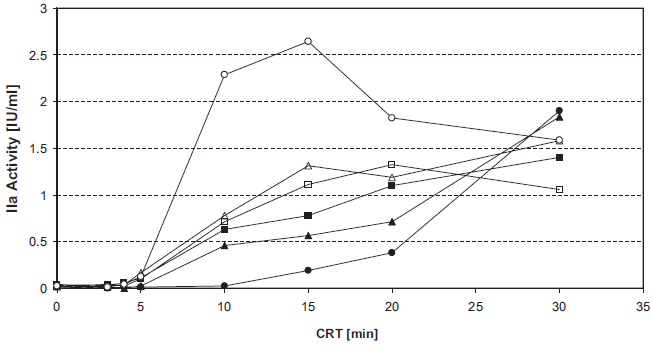
Arginine dose-dependently inhibits INCA (Fig. 3). The IC50 values were about 12 mM for commercially available arginine for i.v. infusion and about 8 mM for alkaline arginine (at pH 8.7).
Inhibition of intrinsic thrombin generation by arginine. Figure 3a. Unfrozen pooled normal plasma was supplemented with 0–40 mM arginine. 50 μl samples in F-wells were tested in the INCA: 0 mM (ô), 5 mM (▴), 10 mM (•), 20 mM (♦), or 40 mM (▪) arginine.
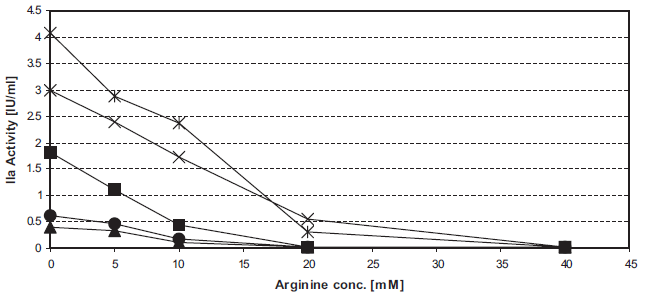
4 min CRT (▴), 5 min CRT (•), 6 min CRT (▪), 7 min CRT (X), 8 min CRT (*).
Normal plasma supplemented with 1.2 g/l purified fibrinogen containing 1% soluble fibrin resulted in a 50% decrease of IIa–generation: plasmatic fibrin entraps generated thrombin, that is why fibrin can be considered as antithrombin I (8). This decrease in IIa-generation was independent of added anticoagulant.
Discussion
The APTT does not reflect the efficiency of some important clinically used anticoagulants, such as e.g. the LMWH [Fareed et al. 2004]. The SiO2 amount used to trigger the INCA is about 100fold below the contact activator amount used for the APTT, and in contrast to the usual global coagulation test APTT the plasma matrix in the INCA is not significantly changed (only 1 part of reagent to 10 parts of citrate plasma).
The INCA monitors the anticoagulant potency of anti-factor Xa or anti-thrombin drugs [Tobu et al. 2004]: LMW-heparins are powerful inhibitors of IIa generation, especially via inhibition of factor Xa. In an INCA test version with an assay incubation at 37°C prolonged to 12 min (INCA-12) patient APTT values of about 40 s correspond to about 1 IU/ml IIa generation, APTT values of 50–60 s to about 0.2 IU/ml IIa (36 s = 100% of norm APTT; 4.3 ± 1.4 IU/ml IIa = 100% of norm IIa generation) [Stief et al. 2006].
The usual plasmatic unfractionated heparin concentrations in therapeutic anticoagulation are about 10fold higher than the IC50 values observed here in the INCA. The superior anticoagulant capacity of LMWH compared to unfractionated heparin might be due to some contact activating (pre-kallikrein to kallikrein folding) potential of unfractionated heparin, a polynegatively charged molecule of a molecular mass > 15000 Dalton [Kongsgaard et al. 1992]. In many clinical situations cells can fragment into phospholipid-microparticles that activate the intrinsic pathway of hemostasis and are thus of great pathophysiologic importance [Soriano et al. 2005; Boulanger et al. 2006].
10–20 mM arginine are common plasmatic concentrations in the so-called arginine-test in internal medicine [Appleton et al. 2002; Cylwik et al. 2005]. Therefore, arginine might be a new therapeutic option in patients with severe disturbances of the coagulation system [Lee and Downey; 2000].
It is suggested to measure the efficiency of any anticoagulant on intrinsic thrombin generation for each individual patient.
