Abstract
Background:
Patients with inborn errors of metabolism related to Methylenetetrahydrofolate reductase (MTHFR) gene variants are at an increased risk for microvascular complications resulting from diabetic retinopathy. Early intervention with targeted nutritional support, particularly folate supplementation, may help stabilize metabolic function and slow the progression of diseases such as diabetes and hypertension, especially before irreversible structural damage occurs.
Objectives:
To assess the effects of the folate supplement Ocufolin® on serum markers in patients with Type 2 diabetes (T2D) and mild diabetic retinopathy (MDR).
Design:
Prospective Cohort Study.
Methods:
Ten patients with both MDR and MTHFR polymorphisms (C677T or A1298C) were enrolled in the present study and received Ocufolin® to address errors in folate methylation metabolism. Patients were excluded if they had a history of other ocular or systemic diseases. Serum biomarkers associated with clinical chemistry (homocysteine, high-sensitivity C-reactive protein [hs-CRP], myeloperoxidase [MPO], fasting insulin, triglycerides, total cholesterol, high density lipoprotein [HDL], low density lipoprotein [LDL], Oxidized-LDL [Ox-LDL], vascular endothelial growth factor [VEGF], D-Dimer, hemoglobin A1c [HbA1c] and glutathione) were measured pre-and post-intervention.
Result:
Treatment with Ocufolin® resulted in a 23% decrease in serum homocysteine (P = .005), an 18% decrease in hsCRP, a 13% decrease in fasting insulin, a 15% increase in D-Dimer (P = .03) and a 47% decrease in VEGF (P = .04). Additionally, there was a 9% increase in glutathione, a 2% increase in MPO, a 6% reduction in triglycerides, a 3% increase in total cholesterol, a 4% increase in HDL, a 4% increase in LDL, an 8% increase in Ox-LDL. HbA1c did not change.
Conclusion:
Normalizing folate metabolism through Ocufolin® significantly improved key blood-based biomarkers in patients with diabetic retinopathy. These metabolic improvements may underlie enhanced retinal perfusion and reduced oxidative stress, suggesting potential adjunctive therapeutic benefits for managing vascular retinopathies in this population.
Introduction
Cellular metabolism depends on adequate intake and processing of vitamins, minerals and other nutrients. Disruptions to these processes, and to folate metabolism in particular, can lead to conditions characterized by hypertension, inflammation, and impaired neuroplasticity.1,2 One of the mechanisms driving these pathological changes is an increase in the vasotoxic compound homocysteine.3,4 Folate is a crucial mediator in the conversion of homocysteine to methionine in the purine synthesis pathway, and without folate in its methylated form L-methylfolate, homocysteine concentrations will rise. 5 Elevated homocysteine levels are associated with increased inflammation and oxidative stress which contribute significantly to retinal vascular damage.6,7 Inadequate folate intake often exacerbates these conditions, necessitating supplementation that is often unachievable through diet alone.8,9 Folate supplementation, typically by L-methylfolate, can restore disrupted vascular endothelial function, especially in the presence of hyper-homocysteinemia.6,10 Given the retina’s high metabolic demands, nutritional interventions targeting folate metabolism may protect against ischemia and oxidative injury caused by endothelial insult, potentially slowing or reversing early metabolic dysfunction in retinal vascular conditions such as diabetic retinopathy.10 -13
C-reactive protein (CRP) and vascular endothelial growth factor (VEGF) are additional biomarkers which increase early in vasculopathic conditions associated with impaired folate metabolism, and are thought to represent oxidative stress and inflammation within retinal and cerebral tissues.14 -16 Targeted restoration of folate metabolism may mitigate retinal vascular disease progression through reduction of the associated inflammatory processes linked to these biomarkers.
The present study investigated the impact of normalizing folate metabolism in patients with retinal vascular disease and an inborn error to folate metabolism caused by polymorphisms in the methylenetetrahydrofolate reductase (MTHFR) gene. MTHFR gene mutations significantly impact folate metabolism, leading to elevated homocysteine levels, impaired endothelial function, and increased susceptibility to diabetic complications, including diabetic retinopathy.11,17 Patients with MTHFR polymorphisms often exhibit increased oxidative stress and inflammation, factors that exacerbate diabetes-related vascular damage, and so, represent a population that may benefit the most from supplementation. 3 Ocufolin® has been developed to alleviate ischemia in the visual system for patients with MTHFR polymorphisms by optimizing folate-dependent metabolic pathways with essential vitamins and cofactors which stabilize methylation.11,13,18,19 Thus, the present study aimed to assess the effects of the folate supplement Ocufolin® on serum inflammatory markers in patients with Type 2 diabetes (T2D), (MTHFR) gene variants, and diabetic retinopathy.
Methods
The study was approved by the Institutional Review Board (IRB) of the University of Miami and adhered to all ethical standards set forth in the Declaration of Helsinki. The present study conforms to the “Improving the reporting quality of nonrandomized evaluations of behavioral and public health interventions: the TREND statement.” 20 All participants provided written informed consent after being made aware of the potential risks, uncertainties, and anticipated benefits of participation. Patient recruitment took place at the Bascom Palmer Eye Institute at the University of Miami. Inclusion to the present study required the presence of at least one MTHFR polymorphism (C677C, C677T; A1298A, A1298C) and mild diabetic retinopathy (MDR). The presence of the MTHFR polymorphism was determined through serum genetic screening performed by MyGenetx Laboratory (Franklin, Tennessee, USA). The diagnosis of MDR was confirmed by clinical retinal specialists (JT and ZY) using the criteria established by the American Academy of Ophthalmology and the American Diabetes Association (ADA). 21 Following confirmation of both MTHFR polymorphisms and MDR, participants underwent a comprehensive review of their medical history, including a structured interview and medication reconciliation, to assess for exclusion criteria. Exclusion criteria included: cloudy corneas, glaucoma, severe cataracts, other retinal vascular diseases, or systemic diseases such as systemic sclerosis or lupus erythematosus. A total of ten patients were enrolled. Demographic information for this cohort is available in Table 1.
Demographic Information of the Study Cohort.

Abbreviations: HbA1c, hemoglobin A1c; HCY, homocysteine; MTHFR, methylenetetrahydrofolate reductase.
Patients received Ocufolin® in a regimen of 3 capsules taken orally once daily for a duration of 6 months and took the regimen at home. Adherence to the intervention was monitored by interview at scheduled follow-up visits to ensure consistent supplement intake. The ingredients are listed in Table 2. 18 Baseline fasting samples were collected initially and after 6 months of Ocufolin® use, measuring the markers: serum homocysteine (HCY), high-sensitivity hs-CRP, myeloperoxidase (MPO), fasting insulin, triglycerides, total cholesterol, High-Density Lipoprotein (HDL), Low-Density Lipoprotein (LDL), Oxidized Low-Density Lipoprotein (OX-LDL); plasma D-Dimer and VEGF; whole blood hemoglobin A1c (HbA1c), and glutathione. Quest Diagnostics performed all clinical laboratory assessments. Descriptions of each test may be found in the Appendix Table A1.
Ingredients of the Medical Food Ocufolin®.

Statistical analyses were conducted using Excel (version 2020; Microsoft, Redmond, WA, USA) and SPSS Statistics software (version 27; IBM Corp., Armonk, NY, USA). The Wilcoxon signed-rank test was used to compare the measurements before and after the intake of medical food from the same participants. The Wilcoxon signed-rank test does not assume normality in the data. A P-value of <.05 was considered statistically significant.
Results
Effects on Folate Metabolism
It has been previously shown that increased serum homocysteine (HCY) is the best marker of impaired folate metabolism. 22 To determine the effects of Ocufolin® on folate metabolism, baseline and 6-month follow-up serum levels HCY were measured in patients taking Ocufolin® for 6 months. The current study found that supplementation yielded a significant 23% decrease (P = .005, Wilcoxon signed-rank test) in serum HCY levels in patients with mild diabetic retinopathy and MTHFR polymorphisms (Figures 1 and 2).
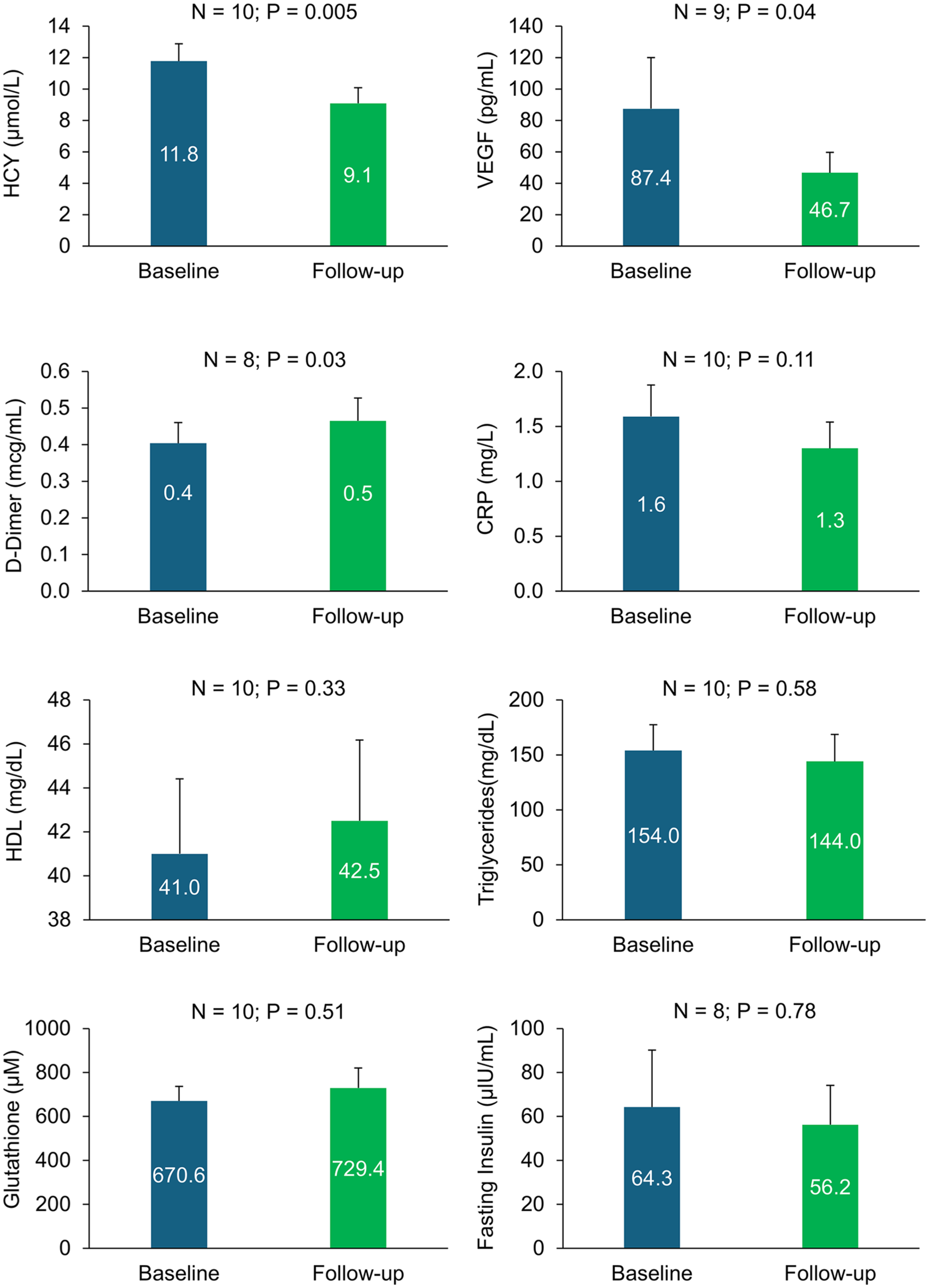
Change in biomarkers after 6 months of Ocufolin use in patients with early diabetic retinopathy carrying MTHFR polymorphism. HCY, VEGF, and D-Dimer were significantly decreased after 6 months of intake of Ocufolin (P < .05), while other biomarkers showed positive trends.

Change in biomarkers after 6 months of Ocufolin® use in patients with early diabetic retinopathy carrying MTHFR polymorphism. HCY, VEGF, and D-Dimer were significantly decreased after 6 months of intake of Ocufolin (P < .05), while other biomarkers showed positive trends.
Effects on Systemic Inflammation
hs-CRP is a general marker of inflammation associated with a variety of diseases. 23 To determine the effects of Ocufolin® on systemic inflammation, of serum hsCRP in this population of diabetic patients was measured at baseline and 6-month follow-up. We found an 18% reduction in hsCRP (Figures 1 and 2, P = .11). D-Dimer is another inflammatory marker used to assess acute inflammation via the coagulation cascade. 24 D-Dimer levels were significantly increased by 15% (P = .03) after 6 months relative to baseline measurement (Table 3; Figures 1 and 2).
All Biomarker Results.

Abbreviations: Hs-CRP, high sensitivity c-reactive protein; VEGF, vascular endothelial growth factor; MPO, myeloperoxidase antibody; HDL, high density lipoprotein; LDL, low density lipoprotein; Ox-LDL, oxidized LDL; HbA1c, hemoglobin A1c.
Effects on Insulin Metabolism
To determine the effects of 6 months of supplementation on insulin metabolism, we measured the levels of fasting serum insulin in all subjects (Table 3; Figures 1 and 2). Participants experienced a 13% decrease in fasting insulin levels. Hemoglobin A1c (HbA1c) was also measured in all subjects to examine Ocufolin®’s influence on overall blood sugar levels. HbA1c levels were unchanged after 6 months of supplementation (Table 3).
Effects on Angiogenesis
VEGF is a well-accepted marker of angiogenesis. 25 To identify the effects of Ocufolin® on angiogenesis, we measured plasma levels of VEGF (Table 3; Figures 1 and 2). We found a 47% decrease in plasma VEGF, suggesting decreased angiogenesis and a potential decrease in neovascularization in these subjects. The largest decreases were in patients with the highest baseline plasma VEGF.
Effects on Oxidative Stress
Glutathione is an important antioxidant for protecting cells from oxidative damage to the mitochondria. Mitochondrial neuroprotection is particularly relevant in diabetic retinopathy, where oxidative stress plays a significant role in disease progression. 26 Our findings indicated a 9% increase in whole blood glutathione levels, highlighting a systemic improvement of oxidative stress (Table 3). Myeloperoxidase (MPO) activity is an indirect measurement of oxidative stress in the body. 27 In the current study, MPO levels increased by 2% (Table 3).
Effects on Lipid Metabolism
Lipid dysregulation is a major indicator of metabolic imbalance, particularly in metabolic syndrome and T2D. 28 To determine whether Ocufolin® is beneficial to lipid metabolism, we measured the levels of triglycerides in this patient cohort after 6 months of therapy. Triglycerides were reduced by 6% (Table 3; Figures 1 and 2). Total cholesterol increased by approximately 3% (Table 3). Low density lipoprotein (LDL) levels were increased by 4% after 6 months. Oxidized LDL (Ox-LDL) levels increased by 8%. High density lipoprotein (HDL) increased by 4%.
Discussion
To the best of our knowledge, this is the first comprehensive biomarker study of a medical food supplement aimed at addressing inborn errors of folate metabolism. The present study found that Ocufolin®, which contains high doses of vitamins B2, B6, L-methylfolate, B12, vitamin C, zinc, lutein, and n-acetylcysteine, can normalize metabolism and improve serum biomarkers related to inflammation and oxidative stress.
Overall, a 6-month trial of Ocufolin® in mild diabetic retinopathy patients with MTHFR variants resulted in significant reductions in the inflammatory markers HCY, CRP, VEGF, and glutathione, along with improvements in fasting serum insulin. Reductions in HCY, CRP, and VEGF could potentially lower retinal vasculopathy risk,14,29 -32 and improved fasting insulin indicates improved insulin resistance and overall metabolic health. 33 These changes likely reflect the impact of increased flow-mediated dilation, improved perfusion, reduced oxidative stress, and reduced ischemia.6,32,34,35 Further research is necessary to elucidate the optimal impacts of nutrient supplementation on metabolism and physiology in patients at risk for vasculopathy. Moreover, the current study’s sample size was 10 patients, and so future studies will expand upon this limited patient pool.
Homocysteine (HCY) 23% Reduction
HCY is a crucial intermediate in the methylation cycle, affecting the synthesis of cysteine, cystathionine, and methionine. 36 Elevated HCY is a biomarker for poor folate metabolism, endothelial dysfunction, systemic vascular health, and small vessel ischemia in the CNS. Elevation of HCY leads to inflammation and brain atrophy.37,38 High HCY is linked to early hypertension, stroke, and the MTHFR C677T polymorphism.39,40 In addition, iatrogenic vitamin deficiencies can raise HCY, necessitating supplementation beyond diet. 40 Meta-analysis shows increased mortality risk above even 1 µmol/L HCY, with significant ischemic stroke risk starting around 50% of the normal upper limit. 41
Folate supplementation improves flow-mediated dilatation in hyper-homocysteinemia. 6 Systemically, the China Stroke Primary Prevention Trial found that low-dose folate reduced ischemic stroke incidence in hypertensives, particularly with the MTHFR variant. 42 In the eye, elevated HCY is also a risk factor for retinal microvascular conditions, disrupting the blood-retinal barrier and causing vision loss. 14 B-vitamin supplementation lowers HCY, reducing macular degeneration risk more effectively than AREDS/AREDS2 formulations. 43 Vitamins B2, B6, L-methylfolate, and B12 are essential for regulating HCY. 44 Vitamin D deficiency can also impair folate transport and absorption. 45 Recent studies suggest optimal serum HCY-targets below 8 µmol/L to mitigate vascular risks. 41
hs-CRP 18% Reduction
Serum CRP is an acute-phase protein biomarker of vascular breakdown, released during tissue injury, and contributes to endothelial dysfunction, vascular stiffness, and hypertension. 46 Elevated CRP levels damage the blood-brain barrier (BBB) and the blood-retinal barrier.30,31 CRP is elevated in diabetic retinopathy, particularly in proliferative forms.47,48 In this study, the average hs-CRP fell 18% after 6 months of Ocufolin®, likely due to vitamins B1, L-methylfolate, C, D, lutein, and n-acetylcysteine. Supplemental L-methylfolate and thiamin (B1) have shown promise in reducing CRP.49,50 Antioxidants, including lutein and vitamin C, also lower CRP.51,52 Moreover, vitamin D deficiency is linked to increased CRP and vitamin D supplementation reduces CRP and other inflammatory markers in Type 2 diabetes.53,54 Multivitamin formulations may effectively lower CRP,55,56 potentially restoring normal metabolic states and protecting the visual system.
D-dimer 15% Increase
The D-dimer antigen is the product of cleaved fibrin polymers during terminal fibrinolysis of the coagulation cascade. 24 Clinical assays were developed to detect this antigen as an indicator for chronic clot formation such as deep vein thrombosis or pulmonary embolism in emergency departments.24,57 Elevated D-dimer levels have since been linked to adverse outcomes in aortic dissection patients. 58 D-dimer has also been linearly correlated to other inflammatory markers such as CRP and immune responses such as elevated neutrophil counts and elevated levels of inflammatory cytokines.59 -61 This means it might be an effective surrogate for acute inflammation. However, D-dimer elevation has been caveated in many clinical assessment tools. The most widely used assays are considered highly sensitive for the presence of fibrinolysis, but not specific for the presence of any single pathology, including deep vein thrombosis.57,62 D-dimer levels were elevated in the current study. This may reflect acute inflammation in some patients after 6 months, but could also be the result of unrelated changes in patients’ overall health.
VEGF 47% Reduction
VEGF is a family of vasoactive proteins secreted by the vascular endothelium during anoxia and oxidative stress to promote new vascular growth, enhance perfusion, and reduce ischemia. 63 Elevated VEGF increases retinal capillary permeability, exacerbating ischemia. 32 While beneficial in most tissues, ocular neovascularization can lead to glaucoma, retinal detachment, retinal edema, and blindness. 63 VEGF elevation is linked to the onset and progression of diabetic retinopathy (DR) and neovascular AMD. 63 Diabetic patients typically have higher serum VEGF levels, especially those with microvascular complications. 64 There is ongoing debate regarding the most predictive VEGF test, with some studies showing plasma VEGF correlating well with vitreous levels for predicting proliferative DR progression. 65 B6 and folate reduce VEGF expression by altering DNA methylation. Additional lowering of retinal VEGF through B6 and folate supplementation may be beneficial.66,67 B12 supplementation also lowers VEGF and reduces retinal neuron death. 68 Other nutrients like n-acetylcysteine, vitamin C, and vitamin D also show effectiveness in lowering VEGF.69,70 Collectively, these may offer important neuroprotective benefits for diabetic retinopathy. In this study, average plasma VEGF may have decreased potentially due to these vitamins. Patients with elevated VEGF showed a more significant reduction, warranting further exploration.
Glutathione 9% Increase
Glutathione is the primary antioxidant that protects cells from oxidative stress caused by reactive oxygen species (ROS). It quenches ROS and is crucial for mitochondrial protection. Most plasma glutathione is synthesized in the liver. 71 Glutathione plays key roles in antioxidant defense, cell cycle regulation, and apoptosis. Severe depletion can lead to oxidative damage and necrotic cell death.72,73 Impaired synthesis is linked to various conditions, including diabetes.71,74 Retinal tissues exhibit high metabolic rates, and glutathione depletion can lead to endothelial dysfunction and inflammation.72,73 Nutrients that effectively increase glutathione levels include L-methylfolate, B2, B6, lutein, n-acetyl cysteine, vitamin C, selenium, zeaxanthin, and zinc,34,35 and Ocufolin® contains all of these.
Fasting Serum Insulin, 13% Reduction
Insulin regulates carbohydrate metabolism, and insulin resistance impairs its many functions, affecting all body cells. 75 Elevated fasting insulin is a key biomarker for insulin resistance and can be more predictive of diabetes than elevated glucose.76,77 High fasting insulin is associated with increased blood viscosity and reduced perfusion. 78 Errors in folate metabolism affect glucose metabolism and inflammation. Furthermore, MTHFR C677T polymorphisms correlate with elevated fasting insulin and inflammatory markers.79,80 Fasting insulin levels in metabolic syndrome are strongly linked to CRP. 81 In this study, the fasting serum insulin reduction potentially reflects improved metabolic function through vitamins L-methylfolate, B12, C, N-acetylcysteine, and zinc.
Triglycerides 6% Reduction
Triglycerides are key lipids linked to cardiovascular and neurovascular diseases. Their elevation is associated with obesity, insulin resistance, metabolic syndrome, and type 2 diabetes. 82 There is a debate over whether triglycerides are causes or biomarkers of disease. High triglyceride levels coupled with low HDL-C increase cardiovascular risk. 82 Meleth et al 83 found higher serum triglycerides in patients with severe non-proliferative diabetic retinopathy (NPDR) and diabetic macular edema (DME), and Chew et al 84 noted that elevated triglycerides were linked to earlier onset of retinal hard exudates while lowering serum lipids reduced disease risks. There is consensus that dietary changes are initially preferred over medications. 82 Folate and vitamin D supplementation have been found to reduce serum triglyceride levels. 85 N-acetylcysteine has been shown to lower serum and liver triglycerides. 86
Footnotes
Appendix
Biomarker Information.

| Test | Test source | Test method | Reference range | Sample type |
|---|---|---|---|---|
| MTHFR | Lifetime sciences | PCR—single taqman genotype assay | Wild type; C677C, C677T; A1298A, A1298C | Buccal swab |
| Homocysteine | Quest diagnostics | Electrochemiluminescence | <10.4 umol/L | Serum |
| Hs-CRP | Quest diagnostics | Immunoturbidimetric | <1.0 mg/L, lower relative CVR | Serum |
| 1.0-3.0 mg/L, average relative CVR | ||||
| 3.1-10.0 mg/L, higher relative CVR | ||||
| >10.0 mg/L, infection or inflammation | ||||
| Serum VEGF | Quest diagnostics | Enzyme ImmunoAssay | 31-86 pg/mL | Plasma |
| Total glutathione | Quest diagnostics | Kinetic, BioAgilytix diagnostics | 373-838 μM | Whole blood |
| Myeloperoxidase antibody (cardio IQ) | Quest diagnostics | Turbidimetreic immunoassay | <470 pmol/L | Plasma |
| D-dimer | Quest diagnostics | Immunoturbidimetric assay | <.50 μg/mL | Plasma |
| Total cholesterol | Quest diagnostics | Spectrophotometry | <200 mg/dL | Serum |
| LDL cholesterol | Quest diagnostics | Martin-Hopkins calculation | <100 mg/dL | Serum |
| Ox-LDL, cardio IQ | Quest diagnostics | Enzyme linked immunosorbent assay | <60 U/L, optimal | Plasma |
| 60-69 U/L, moderate | ||||
| ⩾70 U/L, high | ||||
| HDL cholesterol | Quest diagnostics | Spectrophotometry | ⩾50 mg/dL | Serum |
| Triglycerides | Quest diagnostics | Spectrophotometry | <150 mg/dL | Serum |
| Fasting insulin (cardio IQ insulin) | Quest diagnostics | Immunoassay | <18.4 uIU/mL | Serum |
| HbA1c | Quest diagnostics | Turbidimetric inhibition immunoassay | <5.7% | Whole blood |
Abbreviations: MTHFR, methylenetetrahydrofolate reductase; PCR, polymerase chain reaction; hs-CRP, high sensitivity C-reactive protein; CVR, cardiovascular risk; VEGF, vascular endothelial growth factor; LDL, low density lipoprotein; Ox-LDL, oxidized LDL; HDL, high density lipoprotein; HbA1c, hemoglobin A1c.
All tests in the present table were taken at a fasting level apart from the MTHFR swab.
Acknowledgements
Special thanks are extended to the patients who participated in this study.
Ethical Considerations
The study protocol was reviewed and approved by the Institutional Review Board (IRB) at the University of Miami (IRB ID 20070492) and conducted in accordance with the tenets of the Declaration of Helsinki.
Consent to Participate
Informed consent was obtained from all participants prior to their inclusion in the study.
Author Contributions
Jianhua Wang: Conceptualization, methodology, supervision, funding acquisition. Andrew Hoover: Statistical analysis, manuscript revision. Justin H. Townsend: Data collection, statistical analysis, manuscript drafting. Zohar Yehoshua: Review and editing of the manuscript. Kirill Stremousov: Technical support, data curation. Juan Pablo de Rivero Vaccari: Scientific advising, interpretation of inflammasome-related data. Hong Jiang: Clinical evaluation, manuscript revision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by research grants from Global Healthcare Focus LLC (Wang), NIH Center Grant P30 EY014801, NINDS Grant 1R01NS111115-01 (Wang), and the Research to Prevent Blindness (RPB).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Juan Pablo de Rivero Vaccari is a co-founder and managing member of InflamaCORE, LLC, and has licensed patents on inflammasome proteins as biomarkers of injury and disease as well as on targeting inflammasome proteins for therapeutic purposes. Juan Pablo de Rivero Vaccari is a Scientific Advisory Board Member of ZyVersa Therapeutics Inc.
Data Availability Statement
Data supporting the findings of this study are available upon reasonable request from the corresponding author.
