Abstract
Background:
Chronic non-specific low back pain (CNSLBP) is a debilitating condition with unclear underlying mechanisms. The presence of systemic biomarkers associated with inflammation in nonspecific low back pain (NSLBP) has been inconsistently reported primarily through invasive blood sampling.
Objective:
This study evaluates the use of saliva as an alternative medium for assessing inflammatory biomarker levels in patients with CNSLBP.
Design:
Prospective cross-sectional pilot study.
Methods:
Twenty-five patients with CNSLBP and 25 age and sex matched asymptomatic participants were selected according to specific inclusion and exclusion criteria. The primary outcome was determination of the levels of inflammatory biomarkers in unstimulated saliva samples of CNSLBP patients relative to controls using Luminex™ 200 technology. Secondary outcomes were pain, disability and anxiety/stress levels of participants.
Results:
In CNSLBP patients, 9 biomarkers interferon γ (IFNγ), interleukin-2 (IL-2), IL-4, IL-5, IL-10, IL-13, IL-12p40, IL-12p70, and tumor necrosis factor α (TNFα) were comparable to controls (P = .25-.94). However, 4 pro-inflammatory mediators were significantly elevated, exhibiting medium to large effect sizes: IL-1β (P = .028, Cohen’s d = 1.62), IL-6 (P = .001, d = 1.0), IL-8 (P = .002, d = 0.86), and MCP-1 (P = .001, d = 0.77). Additionally, IL-1Ra levels were significantly higher, though with a small effect size (P = .03, d = 0.43). A significant correlation (P = .02) was observed between VAS pain scores and MCP-1 levels.
Conclusion:
Saliva represents a viable medium for assessing key inflammatory biomarkers in patients with chronic non-specific low back pain (CNSLBP). Elevated levels of proinflammatory cytokines, IL-1, IL-6, IL-8, and the nociceptive chemokine MCP-1 were observed in comparison to asymptomatic controls, with MCP-1 showing a positive correlation with self-reported pain intensity. Future studies utilizing unstimulated saliva samples may further investigate changes in inflammatory biomarker levels to monitor treatment outcomes.
Introduction
Non-specific low back pain (NSLBP) is the most common type of low back pain (LBP) and a leading cause of disability worldwide.1,2 Various structures in the lumbosacral spine, including intervertebral discs, facet and sacroiliac joints, and surrounding soft tissues, may serve as pain generators due to degenerative changes or trauma.1,3 However, the pathophysiological mechanisms underlying the onset and persistence of CNSLBP, which may lead to chronicity, remain poorly understood.4 -6
Research into the pathophysiology of NSLBP has examined the role of inflammatory mediators in both pain initiation and progression. Elevated levels of inflammatory cytokines, such as interleukins (eg, IL-1, IL-6), interferon-gamma (IFN-γ), and tumor necrosis factor alpha (TNFα), have been detected in blood samples of NSLBP patients, suggesting an inflammatory component to the condition.7 -13 However, findings on the qualitative and quantitative presence of these biomarkers remain inconsistent.
For instance, blood levels of the inflammatory mediator TNFα in LBP patients have been reported as significantly elevated in several studies7,14,15 while others found no difference compared to healthy controls.15,16 Similarly, serum/plasma levels of IL-1β and IL-6 have been described as only marginally elevated, 17 unchanged relative to controls15,18,19 stable over time, 20 or even reduced. 21 Likewise, plasma concentrations of nociceptive chemokines, typically elevated in nociceptive and spinal disorders,22 -24 have been reported to be reduced in patients with chronic low back pain. 21
Importantly, these studies have included heterogeneous patient cohorts with respect to LBP etiology, pre-existing comorbidities, and/or prior treatment with various conventional medical therapies. Indeed, systematic reviews generally report weak, and at best, moderate evidence supporting the role of inflammation in NSCLBP.6,24 -26 Furthermore, inconsistencies persist regarding changes in inflammatory biomarker levels following therapeutic interventions such as manual therapy.27 -30
A common limitation of existing studies assessing inflammatory biomarker levels in NSLBP is their reliance on single time point blood sampling via phlebotomy. Although blood samples obtained by phlebotomy are standard for routine diagnosis, this relatively invasive procedure is often unsuitable for research purposes that may require multiple sampling over the course of short periods (eg, 12- or 24-hour cycles). Such an approach is important due to diurnal (circadian) variations in the production of inflammatory cytokines.31 -33 Outside a hospital setting, where prolonged catheter-facilitated sampling may be employed, repeated phlebotomy can pose challenges in participant recruitment, and local inflammatory responses at venipuncture sites may confound biomarker determinations34,35
Considering these challenges, saliva may serve as a potential alternative source of inflammatory biomarkers, offering several advantages over blood. Saliva collection is non-invasive, easily accessible for multiple collections, does not require specialized personnel, and allows for self-collection. Furthermore, the collection methods are well-established and have been extensively researched. 36
Although most current studies on salivary biomarkers of inflammation have focused on oral pathologies 37 changes in the concentrations of both pro- and anti-inflammatory cytokines, such as IL-1β, TNFα, IL-6, IL-8, IL-4, and IL-10, have also been reported in the saliva of patients with systemic disorders, including rheumatoid arthritis, diabetes, cancer, and kidney or lung diseases 38 . Recent genomic research suggests that salivary RNA analysis using real-time polymerase chain reaction (RT-PCR) may enable early diagnosis and detection of inflammatory conditions. 39 Salivary biomarker levels may feasibly reflect a patient’s overall systemic health, making them useful for monitoring therapeutic outcomes. While there is still some debate about whether saliva can fully replace blood testing, it provides a convenient, non-invasive biofluid with the convenience of self-collection of samples over predefined intervals and periods, offering strong diagnostic and prognostic potential.40,41 To our knowledge, no comprehensive studies have yet explored whether the salivary profile of inflammatory biomarkers in patients with NSLBP differs from that in asymptomatic subjects.
This study aims to investigate whether specific inflammatory biomarkers can be detected and quantified in saliva samples from patients with chronic nonspecific low back pain (CNSLBP) and, if so, whether they correlate with self-reported outcomes of pain and disability. The insights gained from this study could reduce reliance on blood samples and potentially facilitate larger randomized controlled clinical trials. Such trials are essential for further elucidating the role of inflammation in the pathophysiology of CNSLBP and for evaluating the effects of treatment interventions over time.
Methods
This study is designed as a prospective observational pilot investigation conforming to the STROBE statement for cross-sectional studies 42 (Supplemental file 1)
Participants
Patients were recruited from the outpatient teaching clinics of the Canadian Memorial Chiropractic College (CMCC) in Toronto, Canada. Patients with CNSLBP who met the inclusion criteria were identified consecutively by the Clinical Research Coordinator (JL) using the clinic’s electronic records. All participants had a diagnosis of CNSLBP based on history and physical examination, with no requirement for routine X-rays.1,2,43 They were under the care of primary supervising clinicians and, as per their management plans, had not been scheduled for a follow-up treatment for at least 1 week prior to their participation in the study.
Upon study enrollment, potential participants underwent a rigorous screening process in consultation with clinician-intern teams to rule out red flags and confirm the diagnosis of CNSLBP. They then completed intake questionnaires to verify eligibility based on the study’s inclusion and exclusion criteria (Table 1).
Inclusion and exclusion criteria for patient cohort.

A cohort of age- and sex-matched healthy control participants was recruited through local advertisements and word of mouth. Controls met the same exclusion criteria as patients and were additionally excluded if they had MSK complaints within the past 6 months with a VAS score above 1.
The study protocol was approved by the CMCC Research Ethics Board (certificate #2203B01).
Sample Size
Convenience samples of 32 consecutive patients with CNSLBP and 30 asymptomatic controls were identified through the initial screening process and were enrolled in the study. Ultimately, several participants were excluded from both groups due to various issues that precluded their inclusion in the study. In the end, 25 patients with CNSLBP and 25 asymptomatic participants completed the study (Figure 1).

Flow diagram illustrating the enrollment of 32 potential patients with non-specific chronic low back pain (CNSLBP) and 30 asymptomatic controls. Exclusion of participants from both cohorts due to different reasons is indicated.
Saliva Sample Collection
All sample collections occurred between 10:00 AM and 12:00 PM. Participants received a study information sheet and had the opportunity to discuss concerns with the principal investigator before providing written informed consent. At presentation for sample collection, they were assigned a deidentifying code number. They then completed study intake forms designed to collect demographic information and confirm that they met the inclusion and exclusion criteria. Additionally, participants filled out 3 validated questionnaires: the visual analog scale 44 (VAS) to assess their self-reported pain level, the Modified Oswestry Disability Index45,46 (ODI) to obtain a measure of functional capability, and the Bournemouth Back Questionnaire 47 (BQ) to obtain insight to stress/anxiety levels.
Non-stimulated saliva samples were collected using the passive drool method. 36 A script detailing the collection procedure was used to provide standardized instructions to all participants. All collections occurred in a room equipped with a sink, and participants were asked to rinse their mouths with water before sitting back and relaxing for 5 minutes, a practice intended to remove or minimize possible mediator contribution due to acute stress.48,49 Each participant was given a chilled sterile saliva collection tube (NORGEN Biotech, Thorold, ON, Canada) labeled with their unique study code number. They were instructed to passively collect saliva in their mouth for 2 to 3 minutes, then bring the tube close to their mouth and drool into it without spitting. If necessary, the tubes were re-placed on ice and the process was repeated for the duration of 10 minutes. The aim was to collect 3 ml of saliva. At the conclusion of the collection, the tubes were capped, placed on ice and transferred to the laboratory within 1 hour for immediate processing.
Cytokine Determinations
All personnel involved in sample processing, data generation and analyses were blinded to the identity of participants. The volume of raw saliva collected from each participant was measured and recorded. A sample of 1.0 ml was centrifuged at 4°C (3000 rpm) for 10 minutes to remove any cellular debris. The resulting supernatant was then divided into 200 µL aliquots and stored at −80°C. The remaining volume of each sample was left on ice for immediate use in a second arm of the study. At the conclusion of all participant recruitment, 200 µL frozen aliquots of all 50 samples were shipped on dry ice to Eve Technologies Laboratory in Calgary, AB, Canada for assessment of inflammatory biomarker levels.
Determinations were performed using The Luminex™ 200 system (Luminex, Austin, TX, USA) for multiplexed quantification of human cytokines, chemokines and growth factors. Fifteen markers: granulocyte-monocyte colony-stimulating factor (GM-CSF), interferon γ (IFNγ), interleukin 1beta (IL-1β), interleukin-1 receptor antagonist (IL-1Ra), IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p40, IL-12p70, IL-13, monocyte chemoattractant protein-1 (MCP-1) and tumor necrosis factor α (TNF-α) were simultaneously measured in the samples using Eve Technologies Human Focused 15-Plex Discovery Assay® (MilliporeSigma, Burlington, Massachusetts, USA). Detection limits for these markers ranged from 0.1 to 5.39 pg/ml. All determinations were done in duplicate, and the mean values were used in analysis.
Data Analysis
Statistical analysis of the data was conducted using PAST 3.18 beta software. 50 The primary outcomes for the present study were the 15 inflammatory biomarker levels listed above. Distributions of the biomarker levels for both study groups were visually inspected to identify serious departures from normality. Secondary outcomes included self-reported pain level (VAS), 44 functional scores using the Modified Oswestry Disability scale (ODI), 45 and stress/anxiety scores using the Bournemouth Questionnaire (BQ). 47 Mean levels of the mediators were compared between subjects with CNSLBP versus those who were asymptomatic using t-tests for independent samples, unless the distributions showed marked departures from normality, in which case non-parametric Mann-Whitney U tests were used. Values of P < .05 were considered significant. Differences in mean levels of the biomarkers were expressed using a standard deviation metric Cohen’s d (difference in means/SDpooled) with values of 0.2 considered small, 0.5 medium and ⩾0.8 large. 51 Correlations between inflammatory mediators in patients’ saliva samples and secondary outcome measures were quantified using Pearson’s correlation coefficients with 95% confidence intervals (95% CI).
Results
Following initial screening, 32 patients and 30 control participants were identified. Of these, 4 patients and 2 controls were disqualified for not meeting at least one of the 24-hour exclusion criteria (Table 1). Among the remaining participants, 2 patients were unable to provide sufficient saliva, and 1 reported unexpected menstruation. In the control group, 1 participant also reported unexpected menstruation, 1 developed neck pain, and another disclosed a recent thumb surgery that had not been revealed during initial screening (Figure 1). Consequently, 25 participants from each group, CNSLBP and controls, were included in the study and provided saliva samples successfully.
Participant characteristics are shown in Table 2. The 2 groups were comparable in terms of age, sex, and body mass index (BMI). The CNSLBP participants exhibited relatively high levels of pain and functional disability. Additionally, their self-reported stress levels were significantly (P = .001) elevated.
Demographic data, and baseline scores of pain, disability, and the Back Bournemouth Questionnaire.

Abbreviations: Back BQ, the back Bournemouth questionnaire; BMI, body mass index; ODI, Modified Oswestry disability index, SD, standard deviation; VAS, 11-point visual analog scale (average VAS/week).
Significant difference between groups (P = .001)
Inflammatory Mediator Concentrations in Salivary Samples
The Luminex assays measured simultaneously 15 inflammatory biomarkers (GM-CSF, IFNγ, TNFα, IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-13, IL-12p40, IL-12p70, and MCP1) in saliva samples from both CNSLBP patients and asymptomatic controls. GM-CSF was undetectable in all samples from all participants. Among the remaining 14 biomarkers, 9 (IFNγ, IL-2, IL-12p40, IL-12p70, TNFα, IL-4, IL-5, IL-10, and IL-13) showed comparable levels between CNSLBP patients and asymptomatic controls (Table 3). The mean concentrations of these biomarkers, along with their t statistics and P-values demonstrated consistency between the study groups (t 24) = 0.03-0.19; P = 0.28 to 0.92) and no significant between-group differences (Cohen’s d = 0.01-0.17).
Evaluation of salivary inflammatory biomarkers in asymptomatic controls compared to patients with CNSLBP.
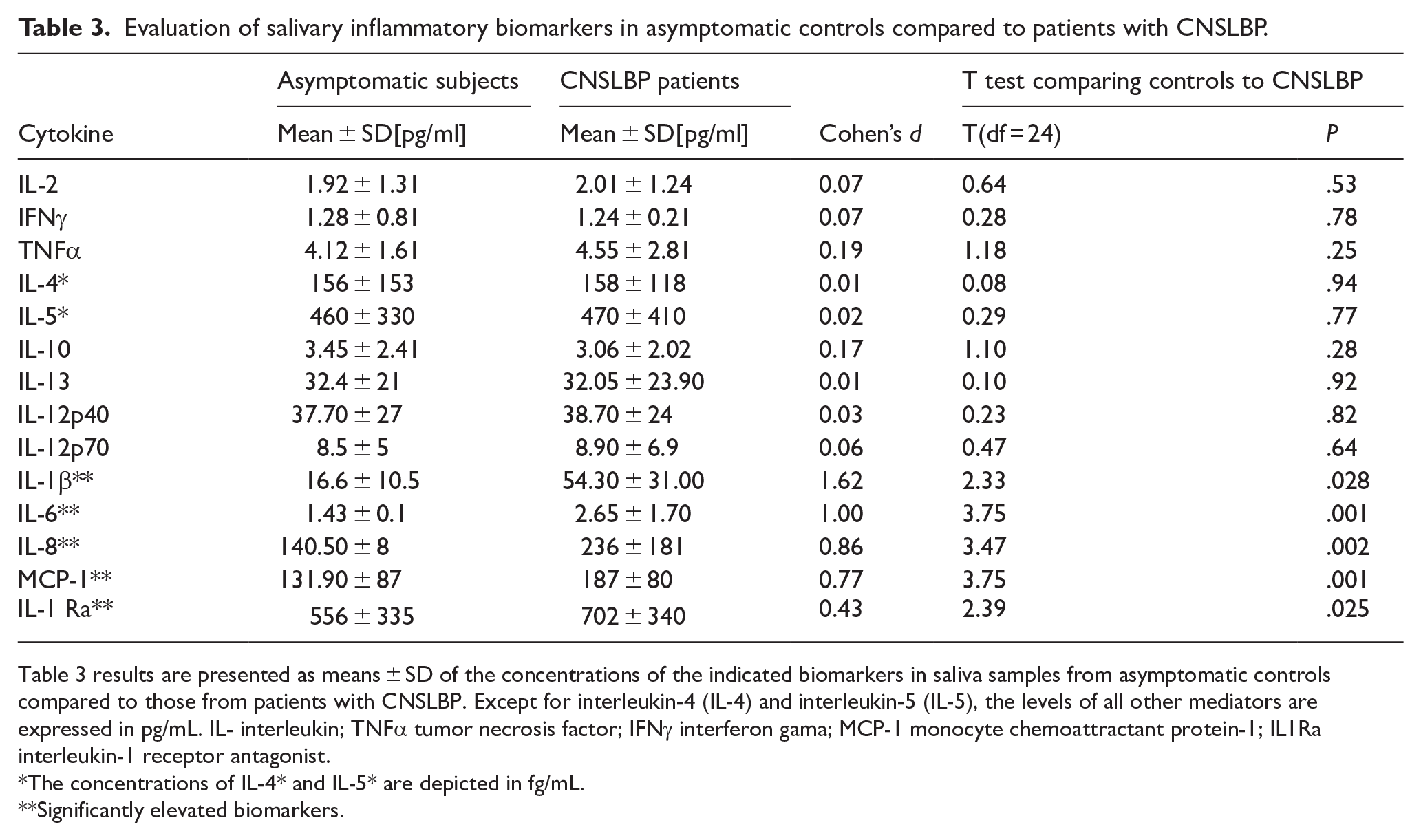
Table 3 results are presented as means ± SD of the concentrations of the indicated biomarkers in saliva samples from asymptomatic controls compared to those from patients with CNSLBP. Except for interleukin-4 (IL-4) and interleukin-5 (IL-5), the levels of all other mediators are expressed in pg/mL. IL- interleukin; TNFα tumor necrosis factor; IFNγ interferon gama; MCP-1 monocyte chemoattractant protein-1; IL1Ra interleukin-1 receptor antagonist.
The concentrations of IL-4* and IL-5* are depicted in fg/mL.
Significantly elevated biomarkers.
Five inflammatory biomarkers commonly characterized as pro- (IL-1, IL-6, IL-8, MCP-1) or anti-inflammatory (IL-1Ra) showed significant differences between samples collected from the CNSLBP patients and asymptomatic controls (Table 3). A graphical comparison of their mean concentrations to the controls is shown in Figure 2.
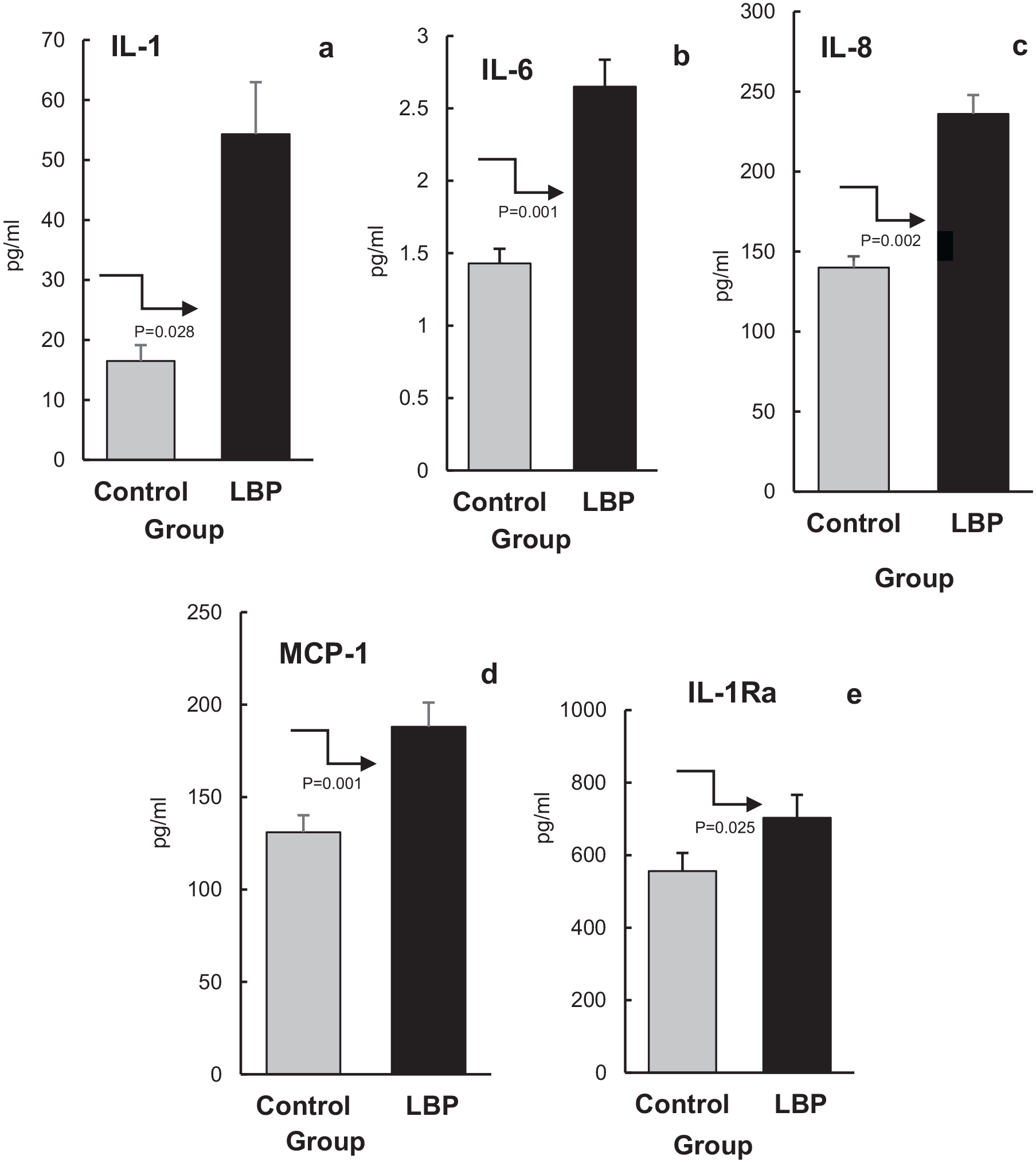
Mean levels (±SEM) of salivary inflammatory markers IL-1, IL-6, IL-8, MCP-1, and IL-Ra (panels a, b, c, d, and e respectively) in patients with non-specific low back pain (CNSLBP) and asymptomatic controls. The arrows point to the difference between the groups, and the significance of the difference between the groups is indicated by the P values underneath the arrows.
The mean level of IL-1β in samples from CNSLBP was 54.30 ± 31 (95%CI 28.1- 80.50) pg/ml compared to 16.60 ± 10.50 (95%CI 13.60-19.60) pg/ml in saliva from asymptomatic participants (Table 3, Figure 2a). The differences between these outcomes were statistically significant (P = .028) and the magnitude of change between their averages was large (d = 1.62). The mean levels of IL-6 and IL-8 in saliva from patients with CNSLBP were significantly higher compared to controls (P = .001 and .002, respectively). The mean IL-6 concentrations in saliva from patients with CNSLBP was 2.65 ± 1.7 (95% CI: 2.19-3.12) pg/ml versus 1.43 ± 0.10 (95% CI: 1.28-1.57) pg/ml in asymptomatic control subjects (Table 3, Figure 2b). Mean salivary IL-8 in CNSLBP patients was 236 ± 181 (95%CI: 184.50-287.50) pg/ml compared with 141 ± 8 (95%CI: 114.50- 165.50) pg/ml in asymptomatic participants (Table 3, Figure 2c). Cohen’s d values for the differences in the means of IL-6 and IL-8 levels in the study groups were 1.00 and 0.86 indicating large effect sizes, respectively (Table 3).
The levels of MCP-1 in salivary samples were also significantly higher (P = .001) in patients with CNSLBP compared to controls (Table 3, Figure 2d). Specifically, the mean salivary concentration of MCP-1 in patients was 187.70 ± 80 pg/ml (95% CI: 165.40-209.60), whereas in controls it was 131.90 ± 87 pg/ml (95% CI: 106.80-157.10), bordering on a large effect size (Cohen’s d = 0.77; Table 3).
In contrast to the previously mentioned pro-inflammatory mediators, the levels of anti-inflammatory cytokines (IL-4 and IL-10) did not differ from those in the control group (Table 3). Conversely, the mean concentration of salivary IL-1Ra in patients was significantly elevated (P = .025, Table 3 and Figure 2e) compared to that in asymptomatic participants. Specifically, the mean IL-1Ra levels were 702 ± 340 pg/ml (CI: 607.60 - 796.40 pg/ml) in patients compared to 556 ± 335 pg/ml (CI: 457.40-650.60 pg/ml) in controls. The effect size of this difference was small (d = 0.43, Table 3).
Relationship of Inflammatory Cytokines to Secondary Outcomes
An evaluation of the relationship between self-reported pain scores (VAS) and the levels of the 5 inflammatory biomarkers determined to be significantly elevated in patients, (IL-1β, IL-1Ra, IL-6, IL-8, and MCP-1) showed a significant correlation (P = .02) only with MCP-1 concentrations (Table 4). Correlations between VAS scores and the other 4 inflammatory mediators were much smaller in magnitude and close to 0 with a wide 95% CI covering values above and below 0. Similarly, none of the above mediators were significantly correlated with ODI (P range 0.14-0.69) or BQ (P range .33-.99) scores (data not shown).
Relationship between VAS pain scores and inflammatory mediator levels in saliva from patients with CNSLBP.

Pearson correlation coefficient (r) and the significance of correlation analysis (P) were calculated to establish the relationship between the degree of self-reported pain scores (VAS) and levels of 5 mediators determined to be significantly elevated in patients with CNSLBP.
Discussion
In patients with CNSLBP, levels of IFNγ and IL-2 (both products of Th1 cells) remained comparable to those of the control group. This aligns with our earlier studies utilizing blood cultures from CNSLBP patients, which showed unaltered or even reduced levels of IFNγ, and IL-2 compared to asymptomatic subjects. 13 Furthermore, in the current study, the concentrations of both subunits of IL-12 (IL-12p40 and IL-12p70) were also not significantly different from those in the controls (Table 3). This suggests that the IL-12-regulated production of Th1-derived pro-inflammatory cytokines like IFNγ 54 may remain unchanged in the inflammatory processes associated with CNSLBP.
Measurable levels of the Th2 products, IL-4 and IL-5 were also comparable to those in controls, hovering just above the detection limit of the assay. Moreover, levels of salivary IL-10 and IL-13 did not differ between the patient and control groups (Table 3). Thus, evaluating salivary levels of Th2 cytokines (ie, IL-4, IL-5, IL-10, and IL-13) as inflammatory biomarkers in patients with CNSLBP may not provide significant insights.
Interestingly, TNFα, a proinflammatory cytokine produced primarily by activated monocytes, was not elevated in saliva samples from patients with CNSLBP (Table 3). This contrasts with studies of blood samples, which consistently report significantly elevated TNFα levels7,10,13 -15 but aligns with research on acute LBP patients, where no baseline differences in TNFα levels were found compared to pain-free controls.11,55 This variability may stem from differences in study protocols and patient characteristics, not the least the heterogeneous nature of CNSLBP. Additionally, the accurate measurement of immunoreactive TNFα can be influenced by the presence of its soluble receptors, sTNFR1 and sTNFR2. 56 Previous studies have shown that sTNFR2 production is significantly elevated in CNSLBP patients 13 Under stress, the concentrations of both sTNFR1 and sTNFR2 rise significantly, 57 and chronic low back pain is known to be associated with increased stress levels, 58 as also reported in our current study (Table 1). These findings suggest that while TNFα production might be altered in CNSLBP patients, its salivary levels could not reflect these changes, due to elevated concentrations of sTNFR1 and sTNFR2. Unfortunately, the current Luminex panel did not include the assessment of these media.
As noted, salivary levels of IL-1 family cytokines (IL-1β, IL-1Ra) and IL-6 were significantly increased in the patient cohort (Figure 2a and e) and Figure 2b. These findings support the notion that increased production of these inflammatory biomarkers is associated with CNSLBP. Of particular interest is the highly significant increase in salivary concentrations of the potent nociceptive mediators IL-8 and MCP-1 (Figure 2c and d). IL-8 activates neutrophils and regulates their migration to sites of tissue damage or inflammation under the regulatory influence of MCP-1.59,60 The role of neutrophil activation was recently examined in patients with LBP. Parisien et al 61 found that transient upregulation of the neutrophil-mediated inflammatory response protects against the transition from acute to chronic LBP. However, prolonged and excessive IL-8 production might result in neutrophil-mediated tissue damage and exacerbation of the inflammatory response62,63 necessitating feedback mechanisms involving other cytokines. Increased production of IL-6, as observed in this study (Figure 2b), might facilitate the transition of activated neutrophils to monocytes. 64 The primary regulatory mechanism for this process involves the dominant anti-inflammatory cytokine IL-10 which inhibits IL-8 production in human neutrophils. 65 Studies from our and other laboratories have reported significantly reduced IL-10 production in patients with LBP.9,13,24 Thus, it may be suggested that elevated levels of salivary IL-8 result from insufficient negative feedback mechanisms involving anti-inflammatory mediators like IL-10. While these observations shed some light on the complex interactions of immune biomarkers in the pathophysiology of CNSLBP, more work is required using a wider array of inflammatory mediators for a more comprehensive understanding of mechanisms involved and contributing to clinical applications.
An association between increased levels of biomarkers such as IL-1, IL-6, TNFα, IL-8, IL-17, C - reactive protein (CRP) and pain intensity has been reported in blood samples from acute,10,11,55 chronic 13 and radicular pain. 66 In the current study, however, only MCP-1 levels in saliva from CNSLBP patients were found to be correlated with VAS scores (Table 4). It has been shown previously that inducible production of MCP-1, a member of the CC chemokine ligand family (CCL2), is significantly and protractedly elevated in patients with CNSLBP. 22 MCP-1 is the primary chemokine regulating the migration and infiltration of inflammatory macrophages to the site of injury 60 The significant correlation between VAS scores and salivary MCP-1 levels, along with the absence of similar relationships with other inflammatory biomarkers (IL-1β, IL-6, and IL-8), suggests that MCP-1 may play a more direct role in the development and maintenance of pain in patients with CNSLBP. These findings highlight MCP-1′s potential as a specific biomarker for nociception and related inflammation in CNSLBP. The clinical significance of this finding remains to be investigated further.
None of these findings, however, address how inflammatory biomarker levels might change with disease progression or treatment interventions, as the study was limited to baseline measurements. Nor do the results clarify whether elevated inflammatory biomarkers in NSLBP are associated with specific tissues as sources of pain generation. To our knowledge, aside from research on the role of inflammatory mediators in degenerative disc pathology and pain,67 -69 little work has examined their associations with other tissues implicated in NSLBP pathology, such as the facet joints, sacroiliac joints, and associated soft tissues. 1 In the absence of such studies, elevated levels of inflammatory cytokines in the saliva of CNSLBP patients, like findings in blood samples, may offer only a general indication of the potential pathogenic mechanisms involved in CNSLBP.
The cellular origin of inflammatory mediators in the saliva of patients with CNSLBP may be diverse. Parenchymal constituents of salivary glands are largely of epithelial origin but also include distinct subsets of immunocytes, such as lymphocytes and monocytes, which can produce inflammatory molecules upon appropriate signaling.70,71 Additionally, inflammatory mediators may be transported to salivary glands via the bloodstream, reflecting systemic events. Salivary glands, being highly vascularized, facilitate the exchange of blood constituents. Absorption of small, blood-transported proteins, including cytokines, by the acinar cells of the glands allows for their exocrine secretion into the oral cavity through active or passive diffusion.72,73 Notably, CD14+16+ monocytes, which represent a significant percentage of mononuclear cells in human saliva 74 are major producers of inflammatory mediators in patients with chronic LBP. 75 These observations may direct future parallel studies to evaluate and compare the levels and relationships of inflammatory mediators in saliva compared to blood of low back pain patients. A possible association between salivary inflammatory biomarkers and those found in circulation has been suggested, for example, in a study involving a population of elderly adults. 76
Strengths and Limitations
The strength of this study lies in its rigorous inclusion and exclusion criteria, the use of well-established and standardized unstimulated saliva collection procedures, and the application of Luminex™ technology for the simultaneous analysis of multiple inflammatory biomarkers. These methodological approaches enhance the reliability of the results and conclusions.
However, the study has limitations which must be considered when interpreting the findings. First, the small sample size (n = 50) limits statistical power, preventing generalization beyond this pilot study. Second, the patient cohort was not stratified based on psychosocial characteristics, radiological findings, or past treatment approaches. Diagnoses were made by primary clinicians based on patient history and physical examination, with treatment plans varying according to clinical presentation and suspected underlying pathology. While the exclusion criteria helped standardize the cohort to some extent, they could not fully address the inherent heterogeneity of the patient population. Moreover, uncontrolled variables such as diet, lifestyle factors (including sleep time and period), and prior treatments may have influenced biomarker levels.
Another limitation was the restrictive cytokine panel, which included only 14 inflammatory biomarkers. A broader, customized panel would have provided a more comprehensive assessment of the complex interplay between pro- and anti-inflammatory cytokines and could contribute to a more comprehensive understanding of the pathophysiology involved.
Finally, all saliva collections were conducted between 10:00 AM and 12:00 PM for practical reasons. Given the circadian fluctuations in cytokine production,31 -33 some biomarker levels might have been underestimated, potentially affecting interpretation of inflammatory status. Saliva offers the possibility of self-collection of samples, which facilitates mediator assessments at different times over predetermined periods, potentially overcoming the issue of circadian fluctuations of mediators of interest.
Future studies should address these limitations by employing larger, stratified cohorts, collecting saliva samples across a 24-hour cycle, and using expanded biomarker panels. These improvements would enhance the study’s significance and generalizability.
Conclusion
Despite these limitations, the findings suggest that inflammatory biomarker levels can be effectively determined in unstimulated saliva samples of patients with non-specific chronic low back pain (CNSLBP). Proinflammatory cytokines IL-1, IL-6 and IL-8 were found elevated, along with MCP-1, a nociceptive chemokine which was found to correlate with self-reported pain intensity. However, no significant correlations were found between any of the elevated mediator levels and ODI or BQ scores.
Thus, given the simplicity of saliva collection, future studies could utilize repeated sampling throughout predetermined study periods to enhance data reliability and explore treatment outcomes. Limitations identified in this study provide valuable guidance for future research aiming to refine the assessment of salivary inflammatory mediators in CNLBP.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719251355038 – Supplemental material for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain
Supplemental material, sj-docx-1-bmi-10.1177_11772719251355038 for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain by H. Stephen Injeyan, Julita A. Teodorczyk-Injeyan, Sheilah Hogg-Johnson, Shadi Rashed, Joyce Lee and Glen Harris in Biomarker Insights
Supplemental Material
sj-docx-2-bmi-10.1177_11772719251355038 – Supplemental material for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain
Supplemental material, sj-docx-2-bmi-10.1177_11772719251355038 for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain by H. Stephen Injeyan, Julita A. Teodorczyk-Injeyan, Sheilah Hogg-Johnson, Shadi Rashed, Joyce Lee and Glen Harris in Biomarker Insights
Supplemental Material
sj-docx-3-bmi-10.1177_11772719251355038 – Supplemental material for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain
Supplemental material, sj-docx-3-bmi-10.1177_11772719251355038 for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain by H. Stephen Injeyan, Julita A. Teodorczyk-Injeyan, Sheilah Hogg-Johnson, Shadi Rashed, Joyce Lee and Glen Harris in Biomarker Insights
Supplemental Material
sj-docx-4-bmi-10.1177_11772719251355038 – Supplemental material for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain
Supplemental material, sj-docx-4-bmi-10.1177_11772719251355038 for Salivary Biomarkers of Inflammation in Patients With Chronic Non-Specific Low Back Pain by H. Stephen Injeyan, Julita A. Teodorczyk-Injeyan, Sheilah Hogg-Johnson, Shadi Rashed, Joyce Lee and Glen Harris in Biomarker Insights
Footnotes
Acknowledgements
We are grateful for the support and cooperation provided by supervising clinicians at the outpatient clinics of CMCC. Specifically, we wish to acknowledge the insightful help of Drs C. DeGraauw, R. Gringmuth, L. Wiltshire, A. Romanelli and J. Goldin in our efforts to recruit patients for the study.
Ethical Considerations
The study was approved by the Research Ethics Board of the Canadian Memorial Chiropractic College (certificate# 2203B01) in accordance with The Tri-Council Policy Statement, TCPS 2 (2022), of Canada.
Consent to Participate
All participants signed the study Informed Consent form.
Consent for Publication
Informed Consent signed by participants explicitly contained the statement
Author Contributions
Conceptualization, H.S.I. and J.A.T-I; Recruiting, J.G.L. and G.H.; Methodology, H.S.I. and S.R. Data analysis, J.A.T-I. and S.H.J; Investigation, H.S.I. and J.A.T-I; Writing- original draft, J.A.T-I. and H.S.I; Writing -review and editing H.S.I, J.A.T-I, S.H.J. and G.H. All authors have read and agreed to the present version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by funds from the Canadian Memorial Chiropractic College.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets obtained and analyzed in the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
