Abstract
Background:
Prostate cancer (PCa) patients with biochemical recurrence (BCR) following radical prostatectomy or radiation therapy often present with very low prostate-specific antigen (PSA) levels (⩽0.5 ng/mL). Accurate detection of recurrence at such low levels is crucial for guiding treatment decisions.
Objectives:
To assess the diagnostic efficacy of [18F]PSMA-1007 PET/MR (PSMA-PETMR) in detecting BCR of PCa in patients with very low PSA levels.
Design:
A prospective study conducted between May 2021 and January 2023, with data subsequently analyzed retrospectively after a 2-year follow-up.
Methods:
The cohort comprised 157 PCa patients with BCR, of whom 52 had PSA levels ⩽ 0.5 ng/mL and underwent PSMA-PETMR imaging. The imaging protocol incorporated multiparametric MRI (mpMRI) and PET acquisitions, with lesion classification following PSMA-RADS version 1.0. Detection rates of recurrent lesions, including local recurrence, lymph node metastasis, and skeletal metastasis, were evaluated.
Results:
PSMA-PETMR exhibited a 63.5% detection rate for recurrent PCa at low PSA levels, surpassing traditional diagnostic methods. Thirty-four local recurrences, 12 metastatic lymph nodes, and 4 skeletal metastases were identified. Follow-up imaging enhanced the detection rate to 73.1% by reclassifying initially equivocal findings. PSMA-PETMR influenced clinical decision-making in 17% of patients by facilitating personalized treatment strategies.
Conclusion:
PSMA-PETMR significantly improves the detection of recurrent PCa in patients with very low PSA levels, offering precise lesion localization and supporting personalized treatment approaches. Further studies are needed to optimize its clinical use and validate its long-term benefits.
Introduction
Prostate cancer (PCa) is a significant health concern worldwide, ranking as the second most frequently diagnosed cancer in males and the fifth leading cause of cancer-related mortality. In 2020, approximately 1 414 000 new cases and 375 304 deaths were attributed to PCa, highlighting the ongoing need for better diagnostic and treatment approach.1,2 Despite advancements in detection and treatment, managing PCa recurrence or metastasis remains a challenge, especially as recurrent disease often requires a highly individualized and multifaceted therapeutic approach. Following primary treatment, 20% to 50% of PCa patients experience biochemical recurrence (BCR).3,4 Patients with BCR vary widely with respect to medical parameters such as tumor stage, Gleason score, primary treatment modality, preoperative PSA levels, and surgical margins. Early and accurate detection is pivotal for improving patient outcomes.
The American Urological Association and European Association of Urology provided guidelines emphasizing prostate-specific antigen (PSA) levels as critical markers for recurrence, 5 with BCR defined as a PSA level ⩾ 0.2 ng/mL post-prostatectomy or increasing by 2 ng/mL after radiation therapy.4,6 This definition is critical for standardizing follow-up care and determining the appropriate clinical interventions for patients with recurrent disease. Given the complexities in managing BCR, an integrated, patient-specific, multidisciplinary approach that considers the nuances of disease progression is essential for effective management. 7
In this context, prostate-specific membrane antigen (PSMA) PET/CT and PET/MRI scans using radiotracers such as [68Ga]Ga-PSMA-11 and [18F]PSMA-1007 have become important tools for early detecting PCa recurrence.8,9 Studies have demonstrated that in patients with low PSA levels, [18F]PSMA-1007 provides higher detection rates than do other radiotracers such as [68Ga]Ga-PSMA-11, as the favorable pharmacokinetics and longer half-life of [18F]PSMA-1007 allows for more precise lesion localization. These imaging modalities are particularly advantageous due to their high sensitivity and specificity in detecting recurrence. PSMA agents increase the diagnostic accuracy of imaging, especially in patients with very low PSA levels (<0.5 ng/mL), enabling earlier localization of recurrent disease and more precise therapeutic planning. 10
The diagnostic accuracy of PSMA-PET imaging is especially critical in the patients with low PSA levels, where traditional imaging techniques such as CT or MRI alone are often insufficient for precise localization of recurrent lesions. 11 This advantage is particularly relevant for early-stage recurrences or small metastases that may be undetectable through conventional imaging approaches. 12 By offering superior spatial resolution and functional imaging capabilities, PSMA-PET/MR allows clinicians to detect subtle changes in tumor biology, which can guide more accurate and individualized treatment plans. 13 Furthermore, the use of multiparametric MRI (mpMRI) in combination with PET offers functional and anatomical data, increasing the precision of lesion characterization and improving overall patient management. 14
This study aims to evaluate the diagnostic efficacy of [18F]PSMA-1007 PET/MR (PSMA-PETMR) in detecting BCR in individuals with very low PSA levels (⩽ 0.5 ng/mL), with the goal of improving early detection and management of BCR of PCa. This approach could lead to more personalized, effective treatments, ultimately improving patient outcomes by influencing disease progression and survival.
Materials and Methods
Patient recruitment
This study was conducted prospectively in a cohort of BCR PCa patients, with the data analyzed prospectively and then retrospectively after a 2-year follow-up. BCR was defined as a PSA level > 0.2 ng/mL measured more than 6 weeks after prostatectomy, or a PSA increase of more than 2 ng/mL above the nadir following external beam radiation therapy (EBRT).15,16 All participants had undergone radical prostatectomy as a curative treatment. The study protocol was reviewed and approved by the Institutional Review Board of the study hospital (TPEVGH IRB No.: 2021-02-008BC). All enrolled patients were thoroughly informed about the study and provided written informed consent. This study was performed in line with the principles of the Declaration of Helsinki.
[18F]PSMA-1007 PET/MRI protocol
Participants were instructed to fast for at least 6 hours before undergoing the imaging procedures. The protocol began with intravenous administration of the [18F]PSMA-1007, with the dosage based on the participant’s body weight (4 MBq/kg). PET/MRI (SIGNA PET/MR, GE Healthcare, Waukesha, Wisconsin, USA) imaging of the body from the thighs to the skull commenced 90 minutes after the injection. This system uniquely combines 3T MR imaging capabilities with PET imaging, employing state-of-the-art silicon photomultipliers (SiPM) for increased photodetection.
The acquired PET data were reconstructed into dynamic, multi-frame images using a sophisticated 3-dimensional time-of-flight (TOF)-enabled ordered subsets expectation maximization (OSEM) iterative algorithm. This process included the incorporation of a point-spread function kernel, which significantly improves image resolution.
mpMRI began with axial T1-weighted images for a basic anatomy overview, followed by axial, sagittal, and coronal T2-weighted images for precise mapping of the prostate and surrounding areas. Axial diffusion-weighted imaging with an apparent diffusion coefficient map was used to identify potential cancer areas by detecting restricted diffusion. The study also included dynamic contrast-increased MRI scans with a gadolinium-based contrast agent to examine the prostate’s vascular patterns and improve lesion visibility, capturing 30 dynamic phases. Whole-body MRI assessments were expanded using diffusion-weighted imaging and 3D T1-weighted axial scans before and after contrast to detect metastatic disease and provide a comprehensive view, highlighting the protocol’s thorough approach in evaluating prostate cancer. The integrated PET and T1-weighted axial scan images were obtained for a comprehensive evaluation of prostate cancer, focusing on both local tumor characteristics and potential disease spread. By combining PET with various MRI techniques, the aim was to offer a detailed, comprehensive view of the tumor, which is essential for formulating an effective treatment plan.
Image analysis, interpretation, and strategies for patient management
Analysis of the PET and mpMRI images in this study was conducted through a meticulous and collaborative approach. Initially, the mpMRI scans were independently reviewed by a radiologist, who identified and staged any tumors within the prostate. This crucial step provided the foundation for further analysis. Following this initial review, a collaborative assessment was performed by both a radiologist and a nuclear medicine physician.
To evaluate lesion uptake on PSMA-PETMR, we used a semi-quantitative approach using the maximum standardized uptake value (SUVmax), corrected for body weight. This value was derived from the area of highest uptake within the lesion, with the region of interest (ROI) labeled on a GE AW workstation. PSMA-PETMR interpretation adhered to the PSMA-RADS version 1.0 criteria 17 ; lesions categorized as 4 (high focal PSMA uptake in areas typical of prostate cancer without corresponding MRI lesions) and 5 (high focal PSMA uptake coinciding with definitive anatomic lesions on MRI) were considered positive findings.
Subsequent treatment protocols were developed by a multidisciplinary team specializing in prostate cancer. Patients with positive lesions received treatment with either local EBRT combined with short-term androgen deprivation therapy (ADT; 26 individuals), local EBRT alone (5 individuals), or ADT alone (2 individuals). For those with equivocal (PSMA-RADS 3A or 3B) or negative findings, a minimum follow-up period of 12 months (range, 14-30 months) was implemented, along with imaging assessments (via PSMA-PETMR or mpMRI) to monitor for any progression or changes. However, not all participants received histopathological confirmation of their findings.
Statistical analysis
Statistical analysis was performed using MedCalc software version 20.100 (MedCalc Software Ltd., Ostend, Belgium). Continuous variables (PSA levels, PSA doubling time [PSAdt], and SUVmax) are presented as the mean and standard deviation (SD). Comparisons between groups (PSMA-PETMR-positive vs PSMA-PETMR-negative patients) were conducted using the independent samples t-test. Categorical variables (Gleason scores and recurrence site classification) were compared using the chi-square test. Correlation analysis between PSA levels, PSAdt, and SUVmax were conducted using Pearson correlation coefficients, depending on the data distribution. P ⩽ .05 was considered statistically significant.
Results
Patient demographics and PSA characteristics
From May 2021 to January 2023, 157 patients with histologically confirmed PCa and BCR following radical prostatectomy or radiation therapy were enrolled in this study. Among these patients, 54 patients with PSA levels ⩽ 0.5 ng/mL (mean: 0.34 ± 0.1 ng/mL; range: 0.11-0.50 ng/mL) had their results retrospectively reviewed and analyzed. Two patients with initial positive findings were excluded from the analysis due to inconclusive results after a follow-up period of 1.5 years. Ultimately, 52 patients were included in the final analysis. The duration between radical prostatectomy and PSMA-PETMR imaging ranged from 3 to 172 months, with a mean of 48.9 ± 44 months. The pathological staging of the tumors was as follows: pT2N0M0 (n = 16), T3aN0M0 (n = 24), pT3N0M0 (n = 11), and pT3aN1M0 (n = 1; Table 1).
Clinical characteristics of patients (n = 52).

Abbreviation: PSA, prostate-specific antigen.
PSMA-PETMR detection rate and comparison with mpMRI
The PSMA-PETMR imaging exhibited a high detection rate in patients with low PSA levels. Among the 52 patients, 33 were confirmed positive by PSMA-PETMR, resulting in a detection rate of 63.5%. This substantial detection rate is especially remarkable given the low PSA levels (⩽0.5 ng/mL) in the cohort, highlighting the sensitivity of PSMA-PETMR for early detection of recurrent PCa.
Simultaneous mpMRI yielded negative findings in 9 of these 33 PSMA-PETMR-positive patients. This discrepancy suggests that PSMA-PETMR may detect lesions that mpMRI fails to identify, emphasizing the greater sensitivity of PSMA-PETMR imaging (Figure 1). Comparative analysis revealed that patients with positive PSMA-PETMR results had significantly higher PSA levels (0.36 ± 0.10 ng/mL) compared to those with negative findings (0.30 ± 0.11 ng/mL; P = .014; Table 1). However, no statistically significant correlation was observed between PSAdt and PSMA-PETMR results (P = .32), indicating that PSAdt does not significantly affect the detection capability of PSMA-PETMR in this patient population.

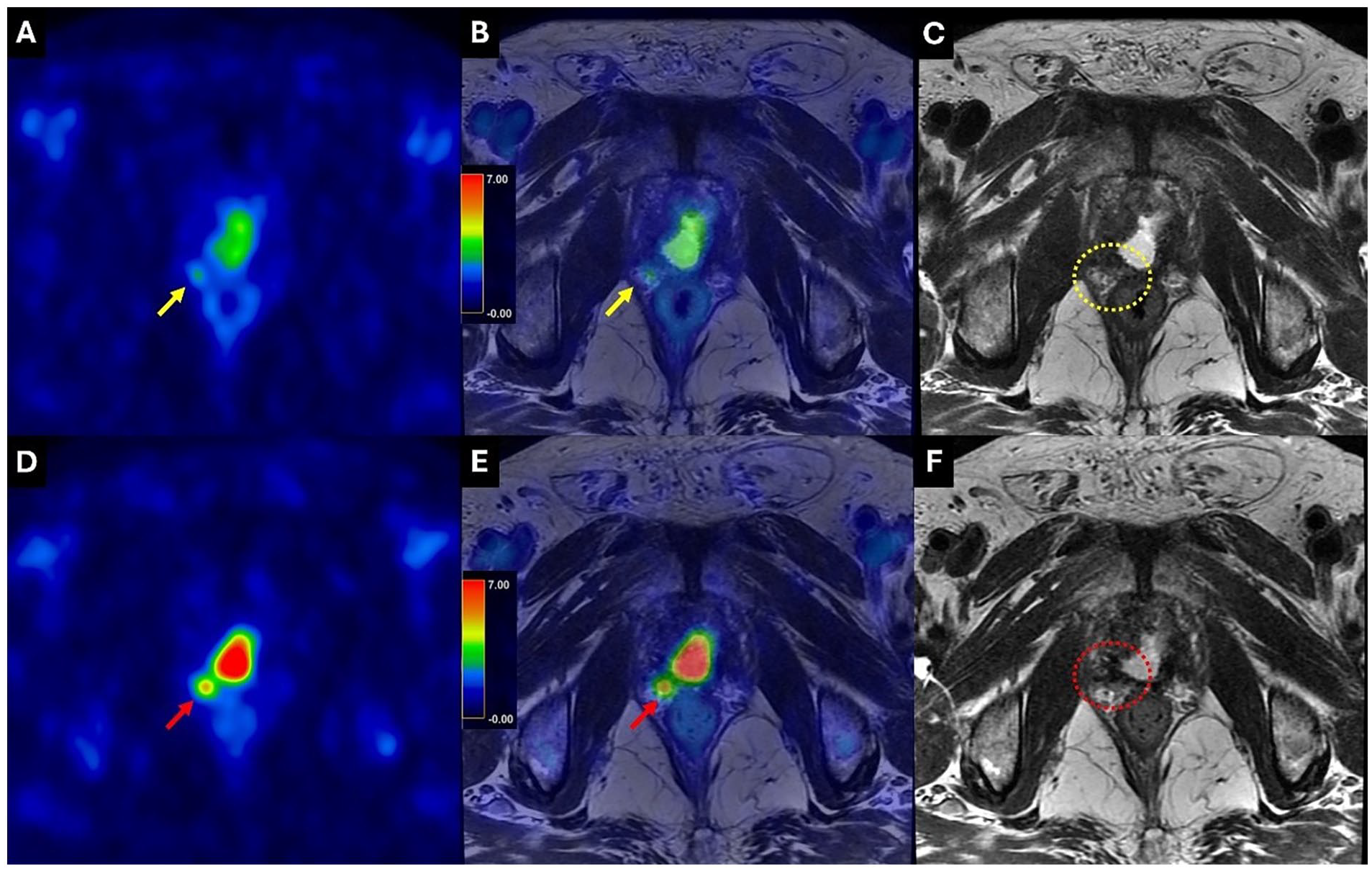
Demonstration of local recurrent lesions adjacent to the urinary wall detected using PSMA-PETMR study. (A-C) A 67-year-old patient with PCa underwent a PSMA-PETMR study due to a gradual increase in PSA (0.39 ng/mL; PSAdt: 4.2 months). (A) Axial fused PSMA PET/MRI showed a lesion with intense and confluent PSMA uptake (SUVmax 12.3) in the posterior urinary bladder wall (yellow arrow). However, (B) T2WI-MRI and (C) the fusion image of DWI with T2WI-MRI identified 2 adjacent lesions (yellow arrows). (D-F) An 81-year-old patient with PCa underwent a PSMA-PETMR study due to a gradual increase in PSA (0.23 ng/mL; PSAdt: 13.8 months). (D) Axial fused PSMA PET/MRI revealed a lesion with moderately increased PSMA uptake (SUVmax 3.27) in the posterior urinary bladder wall (red arrow). However, simultaneous (E) T2WI-MRI and (F) the fusion image of DWI with T2WI-MRI only showed vague signals (red dashed circles).
Further comparison of the patients with positive and negative PSMA-PETMR results showed a distinct distribution of Gleason scores (GS) and PSAdt (Table 2). In the PSMA-PETMR-positive group, 26 patients had a GS of 7, while 7 had a GS > 7. The mean PSAdt was slightly higher in the PSMA-PETMR-positive group (9.59 ± 6.44 months) than in the negative group (9.05 ± 6.38 months).
Gleason scores and PSA doubling time in patients with positive and negative PSMA-PETMR results.

Abbreviations: GS, Gleason Scores; PSA, prostate-specific antigen; PSAdt, PSA Doubling Time; PSMA-PETMR, [18F]PSMA-1007 PET/MR.
P = .009.
Local recurrence and metastasis findings
PSMA-PETMR imaging identified 34 local recurrent lesions in 31 patients, accounting for 59.6% of the cohort. The majority of these lesions (56%) were located at anastomotic sites. The low urinary activity of [18F]PSMA-1007 enabled the clear identification of 7 lesions near the urinary bladder wall, which constituted 20% of the total findings. Additionally, 1 lesion adjacent to the posterior urinary bladder wall was accurately identified as a recurrence in the vas deferens.
PSMA-PETMR imaging detected 12 metastatic lymph nodes in 10 patients, representing 24% of all detected lesions. All metastatic lymph nodes were confined to the pelvic cavity, with no extra-pelvic detections. The short-axis diameter of these lymph nodes ranged from 2 to 8 mm. Notably, 1 lymph node classified as PSMA-RADS category 5 had a short-axis diameter of 8 mm and an SUVmax of 16.69. Furthermore, 6 patients with nodal metastases were concurrently identified with local recurrence.
Four skeletal metastases were detected in 4 patients, representing 8% of all detected lesions. These metastases were located within the pelvis and included 2 lesions classified as PSMA-RADS category 5, 1 as category 4, and 1 as category 3 (Table 3).
Detection of lesions and SUVmax in various recurrence sites.
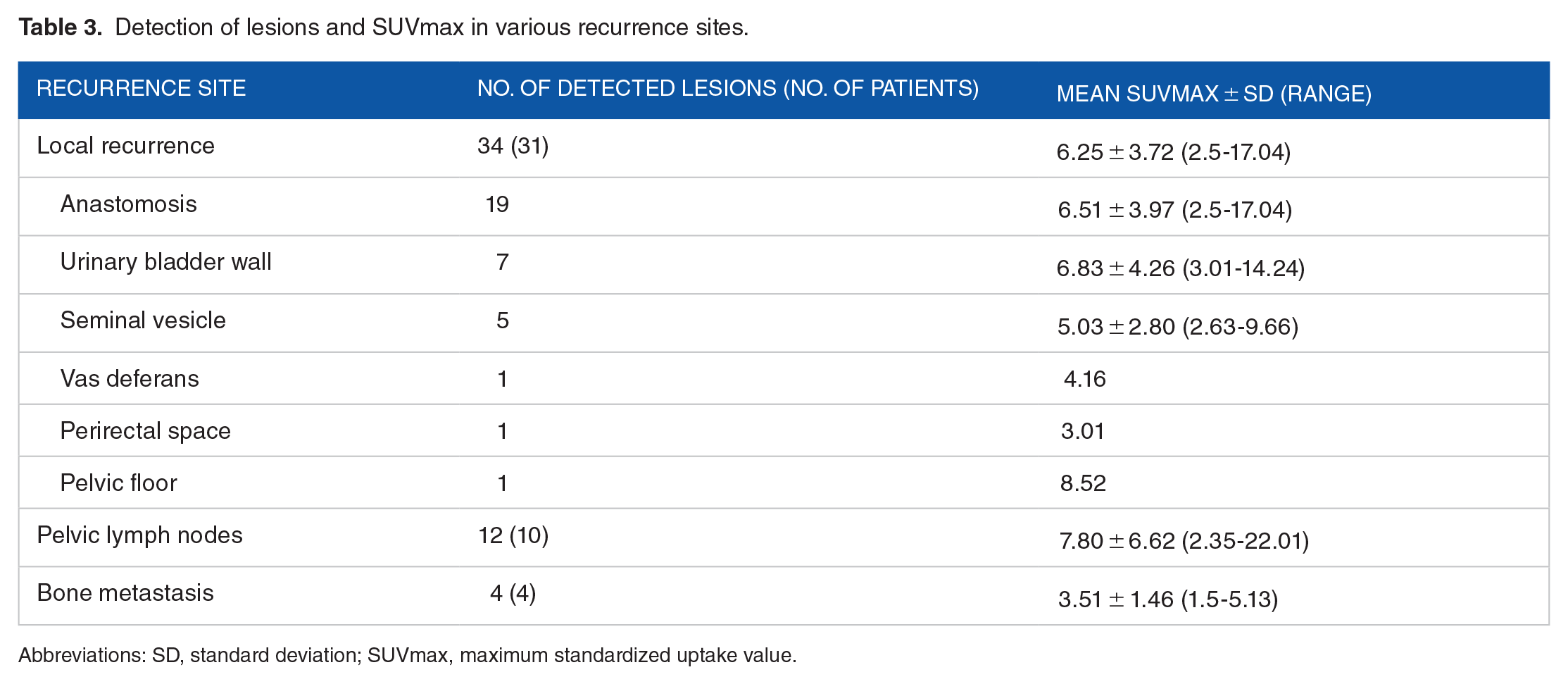
Abbreviations: SD, standard deviation; SUVmax, maximum standardized uptake value.
Longitudinal follow-up and reclassification
The longitudinal follow-up and reclassification of initially equivocal lesions (PSMA-RADS score 3) showed the dynamic nature of recurrent prostate cancer and the importance of continuous monitoring. Initially equivocal lesions were found to be positive in 5 patients during follow-up scans performed 14 to 26 months later. This reclassification increased the detection rate from 63.5% to 73.1%, demonstrating the clinical importance of longitudinal monitoring in patients with indeterminate initial findings.
The characteristics of these reclassified lesions, including their locations, PSA levels, PSAdt, and SUVmax values, are detailed in Table 4. For instance, 1 patient (#1) initially had an equivocal lesion at the anastomosis and a PSA of 0.26 ng/mL and a PSAdt of 1.9 months. Upon re-evaluation 26 months later, this lesion was confirmed as positive, with an SUVmax of 2.5. Similarly, patient #2 had an equivocal lesion near the urinary bladder wall (PSA, 0.23 ng/mL; PSAdt, 2.4 months) that was later confirmed as positive through mpMRI with an SUVmax of 3.82 after 12 months.
Reclassification of initially equivocal (Category 3) lesions as true-positive on follow-up.

Abbreviations: m, month; mpMRI, multiparametric MRI; PSA, prostate-specific antigen; PSAdt, PSA Doubling Time; SUVmax, maximum standardized uptake value; PSMA-PETMR, [18F]PSMA-1007 PET/MR.
The reclassification of initially equivocal lesions as true positives upon follow-up indicates the need for continued study and development of more sensitive imaging technologies. These findings suggest that PSMA-PETMR imaging can significantly improve patient outcomes by enabling earlier detection and intervention, thus reducing the burden of recurrent prostate cancer. In this study, 7 patients were classified as PSMA-RADS categories 3A or 3B. Category 3A included 2 patients with local recurrences and 2 with nodal metastases, all confirmed as true positives during follow-up. Due to the high false-positive rate of rib lesions on [18F]PSMA-1007 PET, our institution implemented stricter interpretation criteria, classifying rib lesions as category 3B only if uptake scores were ⩾2 or comparable to other lesions. As a result, rib lesions were largely excluded from this study. Among the 3 patients with category 3B lesions outside the ribs, only 1 was confirmed to have bone metastasis during follow-up. Although no significant differences in clinical indicators were observed between the 5 true-positive and 2 false-positive category 3 lesions, PSAdt showed a nearly significant trend (6.24 vs 16.0 months, P = .052; data not shown), suggesting that a more rapid PSA increase may indicate a greater probability of true-positive findings in equivocal lesions.
Statistical analysis and correlations
In-depth statistical analysis was performed to elucidated the correlations between various clinical parameters and the efficacy of PSMA-PETMR imaging. No significant correlation was found between PSAdt and the detection rate of PSMA-PETMR (P = .32), suggesting that the rate of PSA increase does not affect the sensitivity of PSMA-PETMR in detecting recurrent lesions at low PSA levels. Furthermore, the analysis showed no significant correlations between PSA level-SUVmax and PSAdt-SUVmax of the detected lesions (P = .85 and 0.20, respectively). This finding indicates that the SUVmax, which measures the intensity of radiotracer uptake, is not influenced by PSA kinetics, reaffirming the reliability of PSMA-PETMR imaging, regardless of PSA dynamics (Table 5).
Comparison of clinical characteristics between patients with positive and negative PSMA-PETMR results.

Abbreviations: GS, Gleason Scores; PSA, prostate-specific antigen; PSAdt, PSA Doubling Time; PSMA-PETMR, [18F]PSMA-1007 PET/MR.
P = .014.
Discussion
Our research demonstrates a high detection rate (up to 63.5%) for PSMA-PETMR in patients with BCR of PCa and low PSA levels (⩽0.5 ng/mL), highlighting the diagnostic value of PSMA-PETMR in early PCa recurrence, particularly when PSA levels are low and traditional imaging methods lack sensitivity. The observed detection of local recurrences and metastases, especially near the bladder, demonstrates the advantage of reduced urinary excretion with [18F]PSMA-1007, allowing clearer pelvic imaging and improving diagnostic precision for guiding treatment strategies.
In this retrospective analysis of these cases 2 years after the initial PSMA-PETMR scans, we found that initially equivocal lesions (PSMA-RADS score 3) were confirmed positive in 5 patients, raising the detection rate to 73.1%. This finding shows the sustained efficacy of this imaging modality in detecting disease recurrence over time and emphasizes the importance of follow-up imaging in patients with initially indeterminate results. Moreover, PSMA-PETMR imaging has shown potential in both detecting recurrent prostate cancer and guiding more precise therapeutic strategies, hence improving patient outcomes. Before the advent of PSMA-PET, standard treatment for BCR patients included radiotherapy to the prostate fossa (with or without lymphatic drainage field), ADT alone, or continued PSA monitoring. The introduction of PSMA-PETMR has profoundly influenced therapeutic approaches. In this study, 17% of patients (9/52) had their treatment plans adjusted based on serial PSMA-PETMR findings. Among them, 3 patients (5.7%) with local recurrences were successfully treated with targeted radiotherapy, and 4 patients (7.7%) with initially equivocal category 3 lesions were confirmed as recurrent with follow-up scans. Additionally, 2 patients (3.8%) initially classified as negative were later identified with local recurrences via follow-up imaging and were successfully treated. These findings emphasize the capability of PSMA-PETMR not only in precise disease detection but also in guiding individualized treatment approaches, ultimately contributing to improved patient outcomes.
Our findings have several important clinical implications. Firstly, the detection of very small lesions with mild tracer uptake, initially categorized as equivocal, shows the difficulties associated with PSMA-PETMR imaging in patients with low PSA levels. For instance, 1 patient (Table 4, patient #1) had very small lesions with a slight elevation in uptake, making definitive categorization difficult. Follow-up PSMA-PETMR scans after 2 years showed a significant increase in tracer uptake, confirming their pathological nature (Figure 2). Notably, the higher rate of local recurrence observed in this study (59.6%) compared to previous studies is partly attributed to the higher proportion of patients with advanced pathological stages (pT3a or above, 69%). More importantly, the lower urinary background activity of [18F]PSMA-1007 compared to other PSMA radioligands provides a substantial advantage in detecting local recurrence. These findings emphasize the clinical value of [18F]PSMA-1007 in precisely identifying local recurrence and encourage further research focusing on high-risk patients using PSMA-1007 PET.
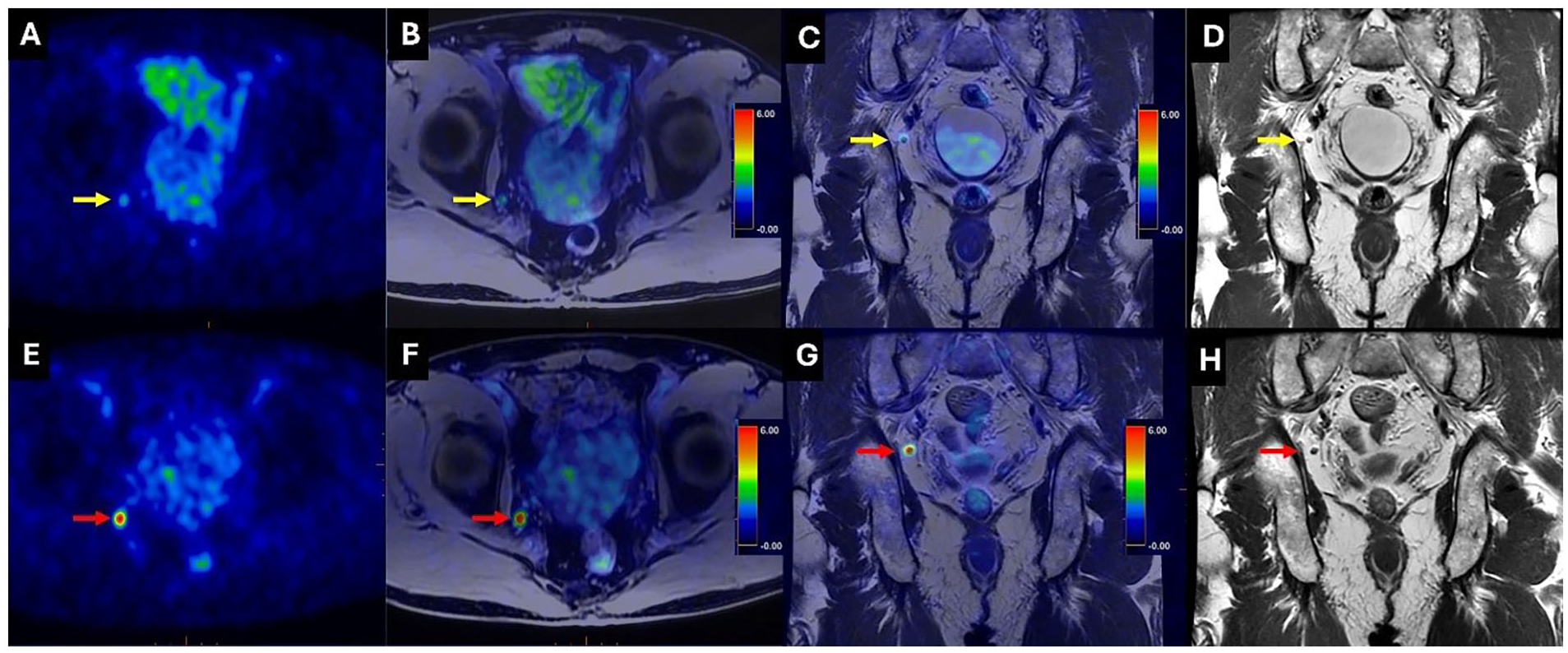
PSMA-PETMR study for a 67-year-old patient with PCa who exhibited a gradual increase in PSA levels (0.26 ng/mL; PSAdt: 1.9 months). (A) Axial PSMA PET; (B) fused PET with T2WI-MRI; and (C) axial T2WI-MRI. A focus of vague uptake (SUVmax 2.16) was noted adjacent to the right posterior bladder neck abutting the surgical anastomosis (yellow arrows). A PSMA-RADS score of 3 was assigned. A follow-up PSMA-PETMR study was performed 26 months later due to a progressive rise in PSA levels to 1.81 ng/mL. (D) Axial PSMA PET; (E) fused PET with T2WI-MRI; and (F) axial T2WI-MRI. The vague lesion exhibited progressive change in PSMA uptake (SUVmax 5.05; red arrows). Local recurrence was confirmed, and the patient underwent targeted radiation therapy followed by ADT. However, simultaneous T2WI-MRI failed to detect this lesion in both the initial and follow-up scans (C and F; yellow and red dashed circles).
In another case (Table 4, patient #2), high urinary radioactivity hindered the accurate interpretation of the initial scan. A distinct lesion identified in a subsequent MRI imaging became visible upon reevaluation of the initial PSMA-PETMR scan, which had been masked by urinary activity. This finding emphasizes the critical role of complementary imaging modalities and the need for radiologists to be vigilant about potential artifacts that may obscure true-positive findings in PSMA-PETMR scans. Moreover, the confirmation of very small lymph nodes and skeletal metastases upon follow-up imaging demonstrates the importance of thorough image analysis. Two patients (Table 4, patient #3, #4) had small lymph nodes that initially raised clinical suspicion but could not definitively identified as pathological. Follow-up PSMA-PETMR scans conducted 2 years later confirmed these as lymph node metastases (Figure 3). Similarly, small sacral lesions initially detected were later confirmed as metastatic in follow-up scans (Table 4, patient #5; Figure 4). These cases highlight the need for follow-up imaging to confirm and validate initial suspicions, especially for small and difficult-to-interpret lesions. We included these cases to highlight common diagnostic challenges in clinical practice and provide insights for interpreting equivocal findings and guiding follow-up strategies. These examples demonstrate the essential factors for improving diagnostic accuracy of PSMA-PETMR scans in patients with very low PSA levels and represent patterns that clinicians may frequently encounter, aiding in the prevention of overlooked diagnoses.
PSAM-PET study for a 68-year-old patient with PCa who presented with a gradual increase in PSA levels (0.23 ng/mL; PSAdt: 4.6 months). (A) Axial PSMA PET; (B) axial fused PET with TIWI-MRI; (C) coronal fused PET with T2WI-MRI; and (D) coronal T2WI-MRI. An indistinct lesion (SUVmax 2.8) was observed in right internal iliac region (yellow arrows). A PSMA-RADS score of 3 was assigned. A follow-up PSMA-PETMR was performed 15 months later due to a progressive rise in PSA levels to 0.48 ng/mL. Progressive changes in the right internal iliac lesion (SUVmax 7.3) were identified in the corresponding images ((E-H) red arrows).

PSMA-PETMR study of a 78-year-old patient with PCa who presented with a gradual increase in PSA levels (0.4 ng/mL; PSAdt: 19 months). (A) Sagittal fused PET/MRI; and (B) T2WI-MRI. An indistinct lesion (SUVmax 2.61) was observed in the sacrum (yellow arrows). A PSMA-RADS score of 3 was assigned. A follow-up PSMA-PETMR was conducted 26 months later due to a progressive rise in PSA levels to 1.09 ng/mL. Progressive changes in the sacral lesion (SUVmax 4.64) were identified in the corresponding images ((C and D) red arrows).
Compared to other imaging modalities, [18F]Fluciclovine PET/CT, an amino acid analog, has been used for detecting recurrent PCa in patients18-20 with low PSA levels (⩽1 ng/mL). 21 Clinical studies, such as the FALCON trial, have demonstrated its potential in improving the PSA response compared to conventional imaging techniques. 22 However, its sensitivity declines at very low PSA levels. Our results suggest that [18F]PSMA-1007 performs better in this patient population by reducing urinary interference, a common limitation of [18F]Fluciclovine and other tracers with renal excretion.
Previous studies report detection rates of 38% to 45% for [68Ga]Ga-PSMA-11 in patients with PSA levels below 0.5 ng/mL.23,24 One of the key advantages of [18F]PSMA-1007 is its hepatobiliary excretion pathway, in contrast to the renal excretion observed with [68Ga]Ga-PSMA-11, which leads to clearer imaging of the pelvic region.25,26 This reduced urinary interference is crucial for identifying local recurrences near the bladder and urethra, areas where conventional tracers often fall short due to tracer accumulation in the urine. When comparing [18F]PSMA-1007 to other [18F]-labeled tracers such as [18F]DCFPyL and [18F]rhPSMA-7, the minimal renal with [18F]PSMA-1007 offers distinct advantages for pelvic imaging by minimizing urinary bladder activity and increasing the clarity and accuracy of detecting local recurrences. Nevertheless, [18F]DCFPyL has shown effectiveness in detecting liver metastases, making it particularly useful in scenarios where liver involvement is suspected. 27 Additionally, [18F]DCFPyL has been noted for its high tumor uptake and favorable biodistribution, making it a strong candidate for whole-body imaging.28-31 Similarly, [18F]rhPSMA-7 combines the benefits of both renal and hepatobiliary excretion, facilitating the detection of lesions across various regions. Each tracer possesses distinct advantages, with [18F]PSMA-1007 excelling in detailed pelvic imaging, [18F]DCFPyL noted for its overall sensitivity and whole-body imaging capabilities, and [18F]rhPSMA-7 recognized for its balanced excretion properties. 28
The lack of significant correlations between variables highlights the complexity of PCa recurrence and the multifactorial nature of its detection. PSMA-PETMR imaging may be influenced by a variety of biological and technical factors, necessitating an integrated diagnostic approach. Such strategies that combine clinical, biochemical, and imaging data are essential for thorough assessment of recurrent disease and tailoring individualized treatment plans. For example, recent studies have demonstrated that a neural network model combining the Prostate Health Index (PHI) and mpMRI significantly improves the sensitivity for detecting clinically significant PCa. 32 Another study has shown that both Proclarix and PHI accurately detect the presence of PCa. 33 Our analysis confirms the reliability of PSMA-PETMR as an independent diagnostic tool capable of detecting recurrent PCa across a spectrum of PSA dynamics. Its independence from PSA kinetics underscores the robustness and utility of PSMA-PETMR in diverse clinical scenarios. Moreover, the lack of correlation with PSA kinetics suggests that PSMA-PETMR may be especially useful when PSA levels alone do not fully reflect disease status, improving the overall diagnostic process.
The potential significant impact of PSMA-PETMR imaging on personalized treatment strategies is evident from our findings. 34 The detection of recurrent disease at low PSA levels allows for timely and targeted interventions, such as local EBRT and ADT, which can significantly improve patient outcomes.34,35 The ability to plan treatment according to the precise location and character of recurrent lesions underscores the value of PSMA-PETMR imaging in the clinical management of PCa. By enabling early detection and accurate staging, PSMA-PETMR imaging facilitates the development of personalized treatment plans that are tailored to the specific needs and conditions of each patient, thereby improving the effectiveness of therapeutic interventions.36,37 Additionally, this approach minimizes unnecessary treatments and reduces potential side effects by focusing on targeted therapy, thereby improving the quality of life for patients. The precision of PSMA-PETMR imaging also supports ongoing monitoring and timely adjustments to treatment plans, which is crucial for managing recurrent PCa effectively.
PSMA PET/MR and PSMA PET/CT each possess unique advantages and limitations. PET/CT is easily accessible and generally faster, providing both metabolic and anatomic information that is crucial for initial staging and restaging of PCa. The CT component, however, exposes patients to ionizing radiation and offers limited soft tissue contrast, which can be a limitation in certain clinical situations. 10 Conversely, PET/MR offers better soft tissue contrast attributable to the MRI component, which is particularly advantageous for assessing local recurrences and pelvic lesions.38,39 Additionally, MRI does not involve ionizing radiation, making it a safer option for repeated imaging, especially in younger patients or those requiring frequent follow-up scans. However, PET/MR is less accessible, more expensive, and generally requires more time than PET/CT, thus restricting its use in some clinical settings. The choice between PET/CT and PET/MR should be based on the specific clinical scenario, patient characteristics, and the available resources, with PET/MR being preferable for detailed soft tissue evaluation and PET/CT for its accessibility and speed.
Despite the promising results, our study has several limitations that should be noted. First, the sample size was relatively small, with only 52 patients included in the final analysis. This limited number may affect the generalizability of our findings to a broader population of PCa patients with BCR. Second, our study was conducted at a single medical center in northern Taiwan, which may introduce selection bias and constrain the applicability of the results to other settings or populations with different demographic or clinical characteristics. Third, not all participants underwent histopathological confirmation of their PSMA-PETMR or mpMRI results. The absence of confirmatory pathology could lead to potential misclassification of lesions, hence compromising the accuracy of our findings. Fourth, while PSMA-PETMR demonstrated a high detection rate, the effect of false-positive results cannot be entirely ruled out, particularly in cases of very low PSA levels where non-specific uptake might occur. Finally, the retrospective nature of the follow-up for initially equivocal lesions could introduce bias and affect the interpretation of the dynamic changes observed in these lesions. Further prospective studies with larger cohorts and multi-center involvement are warranted to validate our findings and improve the clinical utility of PSMA-PETMR imaging in the management of recurrent PCa.
Conclusion
Our study revealed that the use of PSMA-PETMR significantly improves the detection of recurrent PCa in patients with low PSA levels (⩽0.5 ng/mL), providing precise localized anatomical and functional data that enhance clinical decision-making. The integration of this advanced imaging modality into clinical practice has the potential to significantly improve patient outcomes through early detection and personalized treatment approaches. Further research and longitudinal studies are essential to optimize its application and validate its long-term efficacy in managing BCR of PCa. The continued evolution of PSMA-PETMR imaging techniques and their integration with other diagnostic modalities will likely play a pivotal role in advancing the management of recurrent PCa, ultimately leading to better patient care and increased survival rates.
Footnotes
Acknowledgements
The authors would like to acknowledge the National Science and Technology Council (NSTC 113-2314-B-075-038-MY2) and the Melissa Lee Cancer Foundation for their generous support.
Abbreviations
BCR biochemical recurrence
EBRT external beam radiation therapy
GS Gleason scores
mpMRI multiparametric MRI
PCa prostate cancer
PSA prostate-specific antigen
PSAdt PSA doubling-time
PSMA prostate-specific membrane antigen
PSMA-PETMR [18F]PSMA-1007 PET/MR
SUVmax maximum standardized uptake value
