Abstract
Background:
Urinary tract infection (UTI) affects half of women at least once in their lifetime. Current diagnosis involves urinary dipstick and urine culture, yet both methods have modest diagnostic accuracy, and cannot support decision-making in patient populations with high prevalence of asymptomatic bacteriuria, such as older adults. Detecting biomarkers of host response in the urine of hosts has the potential to improve diagnosis.
Objectives:
To synthesise the evidence of the diagnostic accuracy of novel biomarkers for UTI, and of their ability to differentiate UTI from asymptomatic bacteriuria.
Design:
A systematic review.
Data Sources and Methods:
We searched MEDLINE, EMBASE, CINAHL and Web of Science for studies of novel biomarkers for the diagnosis of UTI. We excluded studies assessing biomarkers included in urine dipsticks as these have been well described previously. We included studies of adult patients (≥16 years) with a suspected or confirmed urinary tract infection using microscopy and culture as the reference standard. We excluded studies using clinical signs and symptoms, or urine dipstick only as a reference standard. Quality appraisal was performed using QUADAS-2. We summarised our data using point estimates and data accuracy statistics.
Results:
We included 37 studies on 4009 adults measuring 66 biomarkers. Study quality was limited by case-control design and study size; only 4 included studies had a prospective cohort design. IL-6 and IL-8 were the most studied biomarkers. We found plausible evidence to suggest that IL-8, IL-6, GRO-a, sTNF-1, sTNF-2 and MCR may benefit from more rigorous evaluation of their potential diagnostic value for UTI.
Conclusions:
There is insufficient evidence to recommend the use of any novel biomarker for UTI diagnosis at present. Further evaluation of the more promising candidates, is needed before they can be recommended for clinical use.
Background
Urinary tract infections (UTIs) are common across all age ranges and healthcare settings, with a lifetime risk of 50% to 60%. 1 They are amongst the most common indications for antibiotics in the USA, 2 and in England 3 where they cost the NHS in the UK £316 million annually in emergency admissions for older adults alone. 4 Over-treatment of suspected UTI and unnecessary treatment of asymptomatic bacteriuria (ASB) drive antibiotic overuse, and selection for antimicrobial resistance.5,6 Receipt of antibiotics for UTI results in carriage of resistant bacteria, which may persist for up to 12 months, 7 and treatment of ASB increases the risk of recurrent infection. 8 Thus current antimicrobial guidelines support prompt but targetted antibiotic prescribing, especially for older, multimorbid or frail patients, reliant on timely and accurate diagnosis of infection. 9
Diagnosis of UTI is traditionally based on presence of typical symptoms, positive urine dipstick and growth of uropathogenic bacteria on urine culture. 10 Each of these components presents problems. Many patients at risk of UTI may not experience typical symptoms, especially older adults, or those living in residential homes, where 40% of UTIs are incorrectly diagnosed. 11 Even in younger women with uncomplicated UTI, the specificity of ‘typical’ symptoms such as dysuria, frequency and urgency is low, ranging from 20% to 45%. 12
Urine dipstick tests to detect nitrates (presence of bacteria) and leucocytes are quick, simple and readily available across community and hospital settings. Nitrates have moderate specificity for infection (85%-98%) 13 but are insufficient to rule-out UTI with a sensitivity of 30% to 40% . Presence of leucocyte-esterase is only modestly improves post-test probability of UTI, and is not diagnostic of infection in isolation.1,4 Due to the prevalence of ASB, urine dipsticks are unreliable in both older adults and catheterised patients. 14 Accordingly, though widely used, results are commonly discounted in routine clinical practice. 15
There are, too, limitations, in the diagnostic performance of urinary culture. Up to a third of urine cultures are contaminated by skin and/or faecal flora introduced during sampling 16 and this has the potential to both obscure a true infection and give a positive culture result in the absence of infection. Further, cultures typically take 24 to 72 hours to report so it is usually necessary to make an antibiotic prescribing decision before culture results are available and they cannot distinguish infection from asymptomatic bacteriuria, especially in the elderly. 17
Urine biomarkers could aid accuracy of UTI diagnosis and be developed into cheap, rapid point of care tests (POCTs), useful in settings without ready access to laboratory facilities. The last systematic review of urine biomarkers (in 2009) identified interleukins, notably IL-6 and IL-8, as potential candidates, but these had only been evaluated in a small number of studies. 18 Over the last decade there has been a rapid expansion in biomarker technologies but there is insufficient evidence as to how these perform. In this review we therefore aimed to synthesise evidence of urine biomarkers for the diagnosis of UTI. We primarily used urine culture as our reference standard as this is widely used and easily comparable between studies. As a secondary aim we explored how urine biomarkers can distinguish UTI from ASB, given urine culture cannot differentiate these 2 conditions.
Methods
Our review protocol was registered with PROSPERO in November 2019: CRD42019156071.
Search
We searched Medline, Embase, CINAHL and Web of Science from inception until 11th April 2022 for studies with combined Medical Subject Headings and free text search terms in 3 main themes: urinary tract infection (eg, cystitis, UTI, bacteriuria); biomarkers (eg, biomarkers, immunoglobulins); and urine testing (eg, urinalysis, urine*, test*). The full search strategy is available in Supplemental Table 1.
Eligibility
Participants: We included studies of adult patients (⩾16 years) with a suspected or confirmed urinary tract infection (including cystitis and pyelonephritis) or bacteriuria. We excluded studies not specifying the ages of included patients, of children under the age of 16, or where data for any patients under the age of 16 could not be disaggregated.
Index tests: We included studies of urine biomarkers. We considered a biomarker to be any substance which can be measured in the urine which may be indicative of medical state. 19 This may arise through a biological or pathogenic process and includes markers of immune response and bacterial activity. This does not include detection of bacteria. We excluded studies of leucocyte esterase and nitrites, as they have been thoroughly studied, have modest test accuracy,1,13,14 and are insufficient for making a final diagnosis in clinical practice.14,15,20
Reference standard: We included studies with microscopy and/or culture as a reference standard and we did not specify a threshold for infection after culture as our aim was to offer a wide perspective on the available evidence for novel biomarkers. There is not one agreed threshold level of bacteria for diagnosing all UTIs21,22 and microscopy only can be useful for ruling out bacteriuria. 23 We excluded studies using clinical signs and symptoms or dipstick only.
Types of studies: We included prospective cohort studies assessing diagnostic accuracy, cross-sectional studies, and case-control studies with a healthy control group. Although case-control studies risk exaggerating the differences between groups, by excluding cases for which diagnosis is difficult or unclear, and overestimate prevalence (spectrum bias), we included studies with this design as we did not expect to find a large number of cohort studies.
Settings: We did not exclude studies based on their clinical setting.
Selection of studies
The Cochrane Collaboration Covidence platform was used for study screening. 24 Two authors (GE, GH, ELAC, KH, AVDB, JV, MPS, AES, AC) screened each study according to prespecified inclusion and exclusion criteria and we resolved disagreements by discussion with a third reviewer. We screened titles and abstracts initially and obtained full texts for potentially relevant studies. We hand searched reference lists in relevant systematic reviews for relevant studies.
Data extraction
We extracted study information, participant characteristics, index test description and process, statistical analysis, and results using a data extraction form designed by GE and piloted by GE, KH and MPS. One author (GE, AC, MPS, KH, AES) chosen at random performed data extraction using a standardised and piloted data extraction form. This was checked by a second author, chosen by availability, for accuracy.
Risk of bias assessment
One author (GE, AC, MPS, KH, AES) assessed the risk of bias in the procedures of each included study using an the QUADAS-2 framework. 25 This was checked by a second reviewer and any disagreements were resolved through discussion. One author reviewed the appraisal of each included study for consistency.
Analysis
Due to study heterogeneity and a paucity of diagnostic accuracy data we were unable to perform meta-analyses as initially intended. We have summarised our results narratively.
Results
Our database search identified 4446 unique references; we excluded 4206 based on the title and abstract leaving 240 for a full text review. We included 37 studies in our descriptive analysis (see Figure 1 for PRISMA flowchart). The most common reasons for exclusion were a lack of information about the ages of participants or confirmed inclusion of children. The 37 studies included 4009 adults and measured 66 different biomarkers (see Table 1 and Supplemental Table 2).

Prisma flow diagram.
Biomarker abbreviations.

Risk of bias assessment
Our risk of bias assessment using the QUADAS-2 tool is presented in Table 2. Overall, we found little high quality prospectively collected evidence. We found 4 studies with a prospective cohort design.26-29 In one of these 28 we had low concerns in all domains apart from bias in the conduct of the index test as it wasn’t clear whether the threshold was pre-specified. This was the only study which recruited a random sample of patients presenting with a suspected UTI. 28 We rated one of these studies with a high or medium risk of bias in all domains due to a lack of reporting of the study process. 26
Quality assessment summary using QUADAS-2 tool. Red = high risk of bias, or high concern of applicability to the research question, Orange = medium risk or concern. Green = low risk or concern.

Most of our included studies (33/37) had a case-control design. We rated most (26/33) of these studies were rated as having a high risk of bias for patient selection (26/33) and conduct of the index test (31/33) because they either did not use consecutive or random sampling or did not report their sampling method, and the index test was interpreted with knowledge of the results of the reference standard (culture).
Across all studies, we rated the risk of bias due to the conduct or applicability of the reference standard to be low or medium as we excluded studies not using culture and microscopy as a reference standard. We had minimal concern about the applicability of the index tests to our question, which was intentionally broad.
Objective 1: Potential urine biomarkers for the diagnosis of UTI
The diagnostic accuracy of 64 biomarkers compared with urine culture was investigated in 36 studies including 3979 participants. Three biomarkers (MCR, IL-8, IL-6) were evaluated as part of a cohort study, and a further 7 biomarkers were each studied in 3 or more case-control studies (see Tables 3 and 4); the results for these biomarkers are summarised below. Supplemental Tables 2 and 3 detail our findings for the remaining 54 biomarkers which were studied either once or twice in 27 case-control studies. Supplemental Table 4 summarises the biological function of each biomarker.
Results for IL-6 and IL-8.
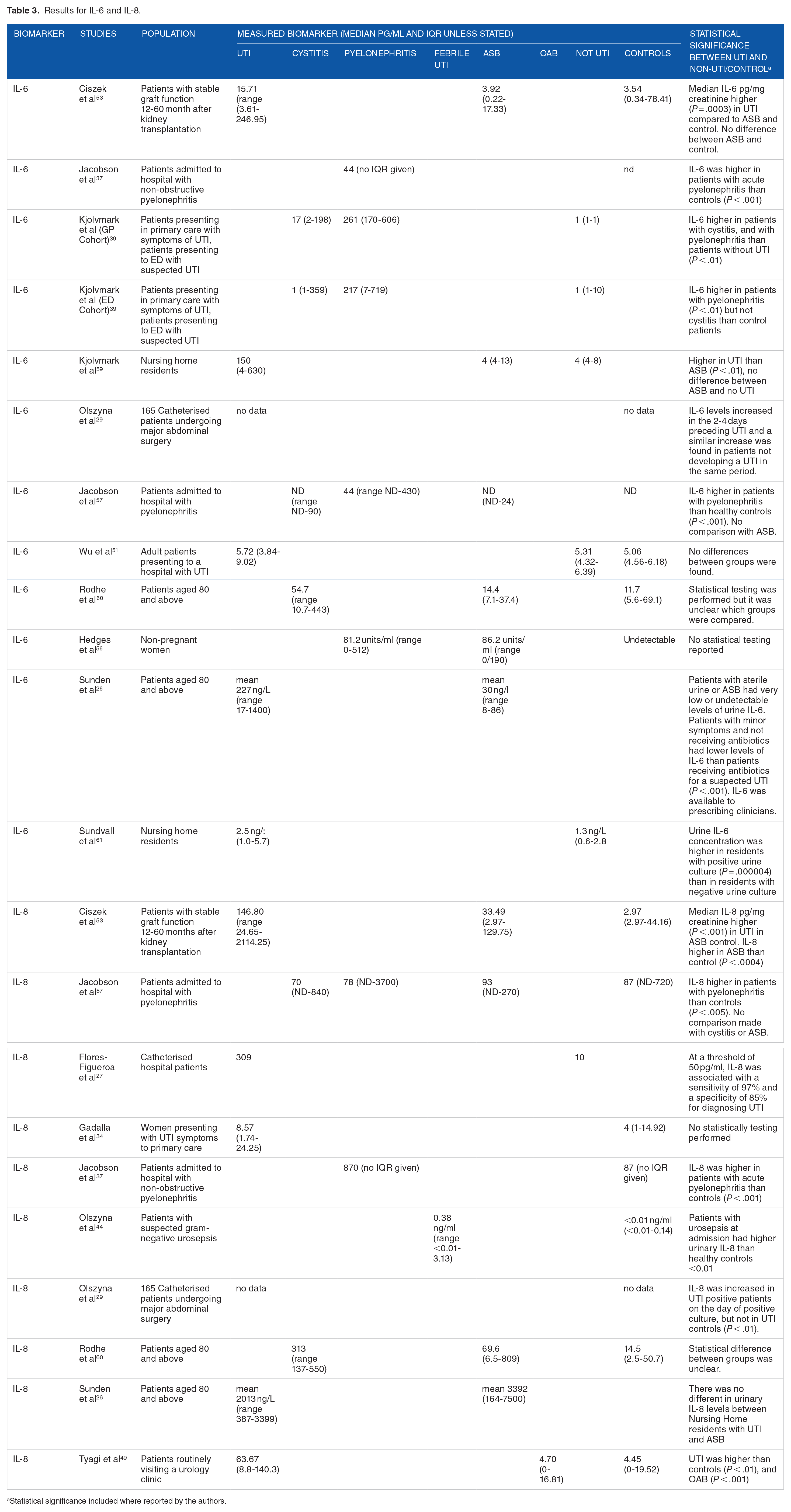
Statistical significance included where reported by the authors.
Biomarker table for biomarkers to diagnose UTI investigated 3 times or more.

Statistical significance included where reported by the authors.
Myeloperoxidase (MPO) to creatinine ratio (MCR)
One cohort study measured myeloperoxidase (MPO) to creatinine ratio (MCR) (measured in ng MPO to g creatinine) in 253 adult outpatients with suspected UTI. 28 In samples which were culture positive for 1 or 2 pathogens, log2MCR values were higher than those in patients with sterile urine (mean 8.6 ng/g (SD 2.5) vs 5.4 (SD 1.5), P = .001). Accordingly an MCR of 194.0 ng/g or above had a sensitivity of 66%, specificity of 95% and PPV of 95% for positive culture.
IL-8
We found 2 cohort studies of catheterised in-patients and 7 case-control studies, conducted in hospital in-patients, out-patients and general practice. Figure 2 displays median IL-8 levels measured for each condition across these studies.
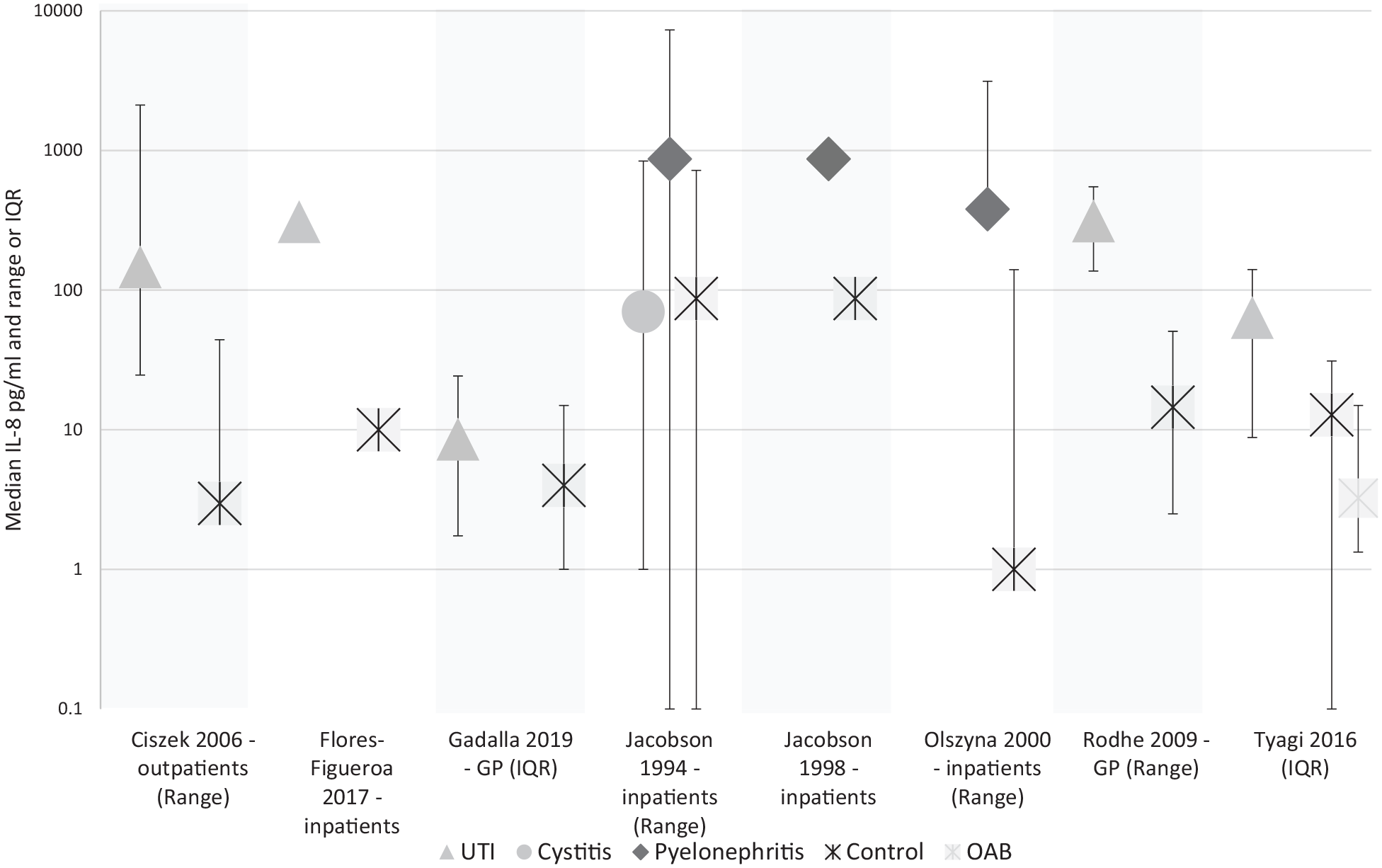
Median IL-8 titre in pg/ml in studies reporting medians. Olszyna et al. 29 not shown. Variance shown in the study title.
In a cohort study of 16 catheterised in-patients, 27 a threshold of 50pg IL-8 per ml of urine was associated with a sensitivity of 97.1% (95% CI 77.1-99.9) and a specificity of 85.3% (95% CI 62.3-95.3) for diagnosing UTI.
In a second cohort study of 165 patients catheterised following major abdominal surgery, 29 mean IL-8 concentrations were higher at the point of positive urine culture than baseline, and also higher than in patients without a positive culture. No precise figures were given.
The results from the case-control studies are summarised in Table 3. Urinary IL-8 was between 10 and 36 times higher in samples from patients with pyelonephritis37,57 and febrile UTI, 44 and between 2 and 49.4 times higher in samples from patients with cystitis or a non-specific UTI (ie, a UTI not specified as upper or lower tract) than in those from healthy controls.34,49,53,60 One study, including 8 participants with cystitis found similar median urinary IL-8 in cystitis and control groups. 57
IL-6
We found 1 cohort study, 1 cross-sectional study and 7 case-control studies investigating IL-6, conducted across in-patient, nursing home and primary care populations. See Figure 3 for median IL-6 levels reported in these studies.

In the cohort study, no increase in mean IL-6 the urine of catheterised patients on the day of UTI diagnosis was detected. This study found that urinary IL-6 increased in all patients in the 8 days following catheterisation. 29
In the cross-sectional study, urinary IL-6 was found to be higher in nursing home residents with positive urine culture (median concentration of 2.5 ng/l (range 1.0-5.7)) compared with those with negative urine culture 1.3 ng/l (range 0.6-2.8) P < .001.
The results from the case-control studies are summarised in Table 3. Three studies found higher level of IL-6 in samples from patients with pyelonephritis than samples from healthy control subjects.37,39,57 Six studies compared samples from patients with cystitis or a non-specific UTI with those from controls.39,51,53,57,59,60 In 3 studies, samples from patients were between 3.8 and 37.5 times higher in samples from patients than those from controls.53,59,60 The remaining 3 found equivocal results, 39 or no difference between groups.51,57
Other biomarkers
We found consistent evidence that biomarkers CXCL-1 and sTNFR types 1 and 2 were elevated in samples from patients with UTI
Three studies found median chemokine (C-X-C Motif) ligand 1 (CXCL-1) to be between 10 and 38 times higher in samples from a mixed population of patients with UTI, cystitis, febrile UTI compared with controls.44,49,60 Median levels of sTNFR-1 were between 2 and 10 times higher in samples from patients with pyelonephritis or febrile UTI than in those from healthy controls, whilst median sTNRF-2 was between 3 and 7 times higher.37,43,58
We found consistent evidence that IL-1B and IL-10 biomarkers were not elevated in samples from patients with UTI
We found 3 studies each which demonstrated no elevation of IL-1B43,49,60 or IL-10 marker levels in samples from patients with UTI.37,43,60
We found contradictory evidence for 2 biomarkers: secretory IgA (sIgA) and IL1-RA
Two studies found samples from patients with recurrent UTI or UTI not specified as upper or lower tract to have mean levels of sIgA between 3.3 and 40 times higher than healthy controls.32,48 Two studies found no difference in mean concentration of urinary sIgA between cystitis and non-specific UTI groups, and controls respectively.35,55
Two studies from the same trial found median IL1-RA concentrations to be 200 times lower (P < .001) in patients with UTI than controls, albeit with large ranges. One study found median IL1-RA in samples from patients with UTI to be half (P < .05) that of samples from healthy controls. 49 One study found no differences. 43
Objective 2: Biomarkers to distinguish between asymptomatic bacteriuria and UTI
We found 9 studies, including 1058 patients, and evaluating 14 biomarkers for differentiating urine from patients with positive culture and symptoms (UTI) and positive culture and no symptoms (asymptomatic bacteriuria (ASB)) (Tables 3 and 4). One study had a cohort design, whilst the rest had a case-control design. Seven studies compared ASB to an non-specific UTI group26,53-55,57,60,61, 1 study compared ASB to a cystitis and a pyelonephritis group, 59 and the final study compared ASB with a pyelonephritis group only. 56 Fourteen biomarkers were investigated; IL-6 7 times, IL-8 4 times, and the remainder only once.
All of these studies compared biomarker levels in patients with a positive urine culture and symptoms to patients who had a positive urine culture without symptoms. No study attempted to differentiate UTI and ASB in patients who were unable to report symptoms. In 1 study, 26 biomarkers levels were compared between patients with UTI symptoms prescribed antibiotics and those not receiving antibiotics, although the biomarker was made available to clinicians making treatment decisions, meaning there may be an incorporation bias.
IL-8
Four studies analysed IL-8, one of which had a cohort design and sampled a nursing home population. 26 The cohort study took monthly urine samples from 35 patients with ASB (defined as 2 consecutive urine samples 4 weeks apart with ⩾105 CFU/ml of the same pathogen with controlled somatic diseases) and compared levels of both biomarkers before and during episodes of UTI. During ASB, the mean IL-8 concentration in urine was 2013 ng/l (range 387-3999) in comparison to 3392 (164-7500) during UTI. No statistical difference was found.
In one case-control study urine samples from patients with cystitis 60 had a median concentration of IL-8 4.8 times higher than those from patients with ASB. One study including renal transplant patients with non-specific UTI during follow up demonstrated that those with UTI had a median concentration of IL-8 4.4 times higher than samples from patients with ASB (P < .001). 53 A study including patients with pyelonephritis and cystitis found that they had comparable Il-8 concentrations to patients with ASB. 57
IL-6
Six studies analysed IL-6, including one cohort study. 26 In the cohort study, the mean IL-6 concentration in urine from patients with ASB was 30 ng/l (range 8-86) in comparison to a mean of 227 (17-1400) during UTI (P = .017).
In a second phase of the cohort study, 254 suspected UTIs in 84 patients were analysed using IL-6 testing in combination with a urine culture. Patients with minor symptoms and not receiving antibiotics had lower levels of IL-6 than patients receiving antibiotics for a suspected UTI (point estimates not given) (P < .0001). 26 At a threshold of 25 ng/ml, urinary IL-6 was associated with a sensitivity of 57%, a specificity of 80%, a PPV of 52% and an NPV of 83% for differentiating treated UTI from ASB.
Two case-control studies found 4 53 and 37.5 59 times higher median urinary IL-6 in samples from patients with non-specific UTI than those with ASB (see Table 3). One study found median IL-6 at 3.8 times the concentration in samples from patients with cystitis compared to those from patients with ASB 60 , whilst another found no difference. 57 Two studies found no differences in urinary IL-6 between samples from patients with pyelonephritis and those with ASB.56,57
Others
Results from the remaining 12 biomarkers are summarised in Supplemental Table 2. Three biomarkers: uHPB, CXCL-1 and sTREM-1 were elevated in samples from patients with UTI compared to those with ASB. In the study analysing sTREM-1, 39/70 participants with a UTI (56%) had a urine culture, whilst the remainder we diagnosed with a positive dipstick and bacteria in the urine sediment.
Discussion
We found 37 studies investigating 64 individual urine biomarkers for the diagnosis of UTI. The quality of the available evidence was limited by study design and heterogeneity, but a handful of biomarkers emerged as viable candidates for new diagnostic tests. In 8 studies IL-8 was consistently raised in UTI patients compared to controls, and in 1 study rose 24 hours earlier than a UTI diagnosis was made. CXCL-1 was also markedly higher in samples from patients with UTI compared to those from controls across 3 studies, albeit all were case-control. A singular cohort study suggested MCR may have a good ability to rule-in UTI, but the sensitivity was low. In the majority of studies, IL-6 was also associated with infection compared to controls, and was the only biomarker to consistently demonstrate higher levels in UTI compared to ASB, including in older adults.
The role of specific cytokines in the bladder’s response to infection is a major unknown. Interestingly IL-8, IL-6 and CXCL-1 are released by macrophages, the largest resident immune cell population in the bladder, and the urothelium. 62 Early in pathogenic E. coli infection, shedding of superficial urothelial cells helps clear bacteria, but also exposes deeper layers of the urothelium, triggering cytokine release. A delicate balance exists between bacterial clearance and preserving tissue integrity. Taken together, high levels of these cytokines may be early indicators of invasive infection and help differentiate this from asymptomatic bacteruria.
Our results incorporate a major expanse in evidence since a previous systematic review in 2009, 18 which only identified 11 urine biomarkers. The previous review also focused on serum biomarkers, including IL-6, IL-8 and procalcitonin, which cannot differentiate UTI from other sources of infection. The evidence for urinary IL-6 and IL-8 is mirrored in a recent systematic review of febrile children with UTI; urinary IL-6 had a pooled sensitivity of 77% (95% CI 69%-83%) and specificity of 87% (95% CI 86%-92%), and urinary IL-8 had a pooled sensitivity of 87% (95% CI 82%-91%) and specificity of 90% (95% CI 87%-93%). 63
Due to the nature and quality of evidence available there are some limitations to our findings. There was virtually no data on diagnostic performance, hence we were unable to perform quantitative analysis, either between biomarkers, or compared to dipstick or culture. The majority of biomarkers were only studied once and the large heterogeneity between study design, population ages and included conditions make comparisons less meaningful. We did not use a pre-specified threshold for number of colony forming units per ml of urine in our reference standard. This reflects the lack of consensus on this threshold internationally.21,22 We also included microscopy as a possible, albeit far from perfect, reference standard, and found one study which used microscopy only as the reference standard for some patients. The diagnostic accuracy of microscopy is unclear due to a lack of specific evidence and its sensitivity may be as low as 50%, 64 however it may be used to rule out the presence of bacteria. Both choices allowed us to capture the widest range of evidence possible regarding novel biomarkers, but may have reduced comparability of evaluations. Furthermore, most of our included studies had a case-control design increasing the risk of selection and ascertainment bias. All of the studies comparing biomarkers in symptomatic UTI versus ASB used patient-reported conventional UTI symptoms to distinguish between the 2 conditions. This limits the applicability to patients with atypical symptoms, or those who cannot communicate, such as nursing home residents.
Our results are a stepping-stone for future research, in particular prospective, rather than case-control studies. Larger cohort studies could help determine diagnostic performance and thresholds for testing. Our focus would be on settings where near-patient testing has the potential for largest impact, for example, within primary care, where urine dipsticks are usually the only other test available, and delays in receiving urine culture results are longest. Research addressed at how novel biomarkers may differentiate between ASB and UTI need careful design. Currently there is no universal reference standard, and there is also an overlap in patient populations, especially in older adults, as patients with ASB develop active infection. Combined panels of urine biomarkers may also increase diagnostic performance. In one study an algorithm using IL-8 and 3 other biomarkers (MMP9, NGAL and IL-1β) had a modest ability to rule in infections (positive Likelihood ratio 6.29, 95% CI 2.04-19.36), 34 although the diagnostic thresholds used in these algorithms were not specified. Future research may consider the diagnostic value of measuring multiple biomarkers from a single sample, and the added value of this for clinicians. Ultimately, well powered and carefully design randomised trials of urinary biomarkers in practice are needed to establish how they can help identify those patients who would most benefit from antibiotics.
Conclusion
This systematic review provides justification for the further investigation of a number of novel urinary biomarkers, notably CXCL-1, IL-6, IL-8, MCR and the sTNFRs. Primary care based prospective studies are needed to establish diagnostic performance and utility in clinical practice.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-docx-1-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Supplemental Material
sj-docx-4-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-docx-4-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Supplemental Material
sj-docx-6-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-docx-6-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Supplemental Material
sj-xlsx-1-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-xlsx-1-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Supplemental Material
sj-xlsx-3-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-xlsx-3-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Supplemental Material
sj-xlsx-5-bmi-10.1177_11772719221144459 – Supplemental material for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection?
Supplemental material, sj-xlsx-5-bmi-10.1177_11772719221144459 for What is the Diagnostic Accuracy of Novel Urine Biomarkers for Urinary Tract Infection? by George Edwards, Anna Seeley, Adam Carter, Maia Patrick Smith, Elizabeth LA Cross, Kathryn Hughes, Ann Van den Bruel, Martin J Llewelyn, Jan Y Verbakel and Gail Hayward in Biomarker Insights
Footnotes
Acknowledgements
The authors would like to acknowledge Nia Roberts for performing the literature searches, and Marta Wanat, Marta Santillo, Tomasz Dobryzki, Joe Kang, Barbora Benkova, Nahoko Kasai, and Olga Romanova for helping translate papers not published in English.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
