Abstract
Background:
Preterm neonates, particularly extremely preterm, are susceptible to respiratory distress syndrome (RDS) due to surfactant deficiency. Single nucleotide polymorphisms (SNPs) in the antioxidant enzymes influence the balance between antioxidant and oxidative stress molecules.
Objectives:
To ascertain the role of SNPs of antioxidant enzymes and oxidative stress biomarkers in preterm neonates with RDS.
Design:
Observational, cross-sectional study.
Methods:
Preterm neonates diagnosed with RDS receiving external surfactant within 24 hours were considered as the cases and those without RDS were the control group. Umbilical cord blood and peripheral blood samples before administering surfactant (day 1), and on days 2 and 3 were collected. Plasma malondialdehyde, 8-hydroxy-2-deoxy guanosine (8-OH-dG), advanced oxidation protein products (AOPP), total antioxidant capacity (TAC), visfatin, reduced glutathione, and chaperonin 60 were evaluated using enzyme-linked immunosorbent assay. SNPs in manganese superoxide dismutase (MnSOD), copper/zinc superoxide dismutase (Cu/Zn SOD), glutathione peroxidase (GPX1 and GPX3), catalase (CAT), glutathione S-transferase (GSTP1) were evaluated using real-time polymerase-chain-reaction. The receiver-operating characteristics curve was used for predicting the accuracy of biomarkers using the area under the curve (AUC) and 95% confidence intervals (95% CI).
Results:
GSTP1, MnSOD, and eNOS (rs1799983) SNPs were observed to significantly influence the oxidative biomarker concentrations in the entire study population. SNPs in GSTP1, MnSOD, and eNOS (rs1799983) were significantly associated with differences in oxidative stress biomarkers. MnSOD (rs4880) significantly increased the risk of pulmonary complications in neonates with RDS. DNA damage product (8-OH-dG) concentrations before surfactant administration has the best predictive accuracy (AUC: 0.8; 95% CI: 0.7-1; P = .001) for pulmonary complications with a cut-off value of 5008.8 pg/mL. TAC concentrations are significantly greater on day 2 and day 3 amongst neonates receiving surfactant compared to the control group. AOPP in the umbilical cord blood was observed to significantly predict the severity of RDS (AUC: 0.8; 95% CI: 0.6-1; P = .01) with an optimal cut-off value of 88.78 µmol/L.
Conclusion:
We observed that SNPs in eNOS and MnSOD significantly influence the production of oxidative stress biomarkers in preterm neonates. Baseline 8-OH-dG concentrations best predict the risk of pulmonary complications and AOPP concentrations in the umbilical cord blood predict the risk of RDS severity.
Introduction
Respiratory distress syndrome (RDS) is a common neonatal disorder associated with an increased risk of morbidity and mortality with an estimated 8% admission rate in intensive care units in the Gulf Co-operation Council countries and 860 deaths/annum in the United States.1,2 Even in neonates with birth weight above 1 kg, mortality has been observed to be around 30% to 40%. 3 Neonatal RDS also called hyaline membrane disease has been identified due to surfactant deficiency and lung immaturity, with the following risk factors: male gender, cesarean section delivery, maternal diabetes mellitus, preterm birth, and having an APGAR score of less than 7.4,5 External surfactant administration forms the mainstay of pharmacological management with mortality reduction by 10%.6-8 Additionally, antenatal corticosteroids between 24 and 34 weeks of gestation have also been shown to effectively reduce the incidence of RDS. 9
Preterm neonates with RDS are at a high risk of oxidative stress and consequently damage by free radicals. 10 An imbalance between molecules that promote oxidation (predominantly reactive oxygen and nitrogen species), and the antioxidants results in oxidative stress. 11 Pro-oxidant molecules damage DNA, lipids, and proteins resulting in respiratory cell death and thus RDS. 12 Oxidative damage of the lipids are indicated by plasma malondialdehyde (MDA) concentrations, while 8-hydroxy-2-deoxy guanosine (8-OH-dG) is an indicator of oxidative DNA damage, and protein damage is indicated by the concentration of advanced oxidation protein products (AOPPs); while total antioxidant capacity (TAC) reflects the overall antioxidant status. A few studies have evaluated the role of oxidative stress biomarkers in neonates with RDS. The limitations identified with the existing literature include an absence of a study evaluating all the critical oxidants and antioxidants, particularly concerning the external surfactant administration, and there are no studies from the Arabian Gulf region.
Key antioxidant enzymes involved in neutralizing the reactive oxygen and nitrogen species include manganese superoxide dismutase (MnOD), copper/zinc superoxide dismutase (Cu/Zn SOD), glutathione peroxidase (GPX), catalase (CAT), and glutathione S-transferases (GSTM1, GSTP1). 13 Single nucleotide polymorphisms (SNPs) in MnSOD (rs4880 and rs5746136), Cu/Zn SOD (rs8192287), GSTP1 (rs1695), GPX1 (rs1050450), GPX3 (rs1946234), and CAT (rs1001179) genes have been identified with reduced antioxidant activity in several healthy and diseased populations.14-17 However, a few studies have been published evaluating the possible associations of single nucleotide polymorphisms (SNPs) of the antioxidant enzymes in neonates with RDS. Similarly, there is hardly any data regarding the influence of genetic polymorphisms of the antioxidant enzymes in neonates with RDS. The hypothesis on the identified polymorphisms in antioxidant enzymes in the preliminary reports in neonatal RDS will be clinically meaningful only if we can establish their association with increased oxidative stress markers. Until now, there are no studies that had evaluated the association between SNPs of antioxidant enzymes with oxidative stress biomarkers in neonates with RDS, particularly those receiving external surfactant. Hence, we carried out the present study to address these gaps.
Methods
Ethics
This observational, cross-sectional study was carried out after obtaining approval from the Institutional Ethics Committee of Arabian Gulf University, Royal College of Surgeons in Ireland-Medical University of Bahrain (RCSI-MUB), and Salmaniya Medical Complex, Kingdom of Bahrain. The study was carried out in the neonatal intensive care unit of Salmaniya Medical Complex from October 2021 till February 2022, after obtaining consent from either of the parents. We adhered to the Declaration of Helsinki guidelines.
Study population
Preterm neonates (<37 weeks of gestational age) of either gender diagnosed with RDS within 24 hours of birth receiving external surfactant as a part of the standard of care formed the case group. RDS was diagnosed with the presence of clinical and radiological features as follows: tachypnea, nasal flaring, grunting, and subcostal, intercostal, and/or suprasternal retractions at birth along with chest X-ray findings. RDS grading was carried out based on the X-ray findings as follows: grade I—reticulogranular pattern present diffusely over the lung fields, grade II—the presence of air bronchogram overlapping the heart, grade III—an extension of the air bronchograms beyond the cardiac silhouette, and grade IV—a white lung. 18 Those with major congenital abnormalities were excluded. Preterm neonates without any diagnosis of RDS/sepsis/chronic lung disease/congenital heart disease formed the control group.
Study procedure
Following written consent, eligible neonates were recruited and the following details were collected: demographics (gestational age, APGAR scores at first, fifth, and tenth minutes, per rectal temperature, birth weight, length, head circumference, and chest circumference), maternal history [mode of delivery (natural/induced/Cesarean section), singleton/twin/triplets, administration of antenatal corticosteroids], drug-related details (name, dose, frequency, and duration) and outcome (discharged alive/died). For those with RDS, details regarding whether they have developed pneumothorax, pulmonary hemorrhage, or chronic lung disorder were obtained. Half a milliliter of umbilical cord blood was collected at the time of their birth, and half a milliliter of peripheral blood was collected each before surfactant administration (day 1), day 2, and day 3 following the surfactant for the analysis of oxidative stress biomarkers. For the control group of neonates, umbilical cord blood at the time of birth, and peripheral blood samples on days 1, 2, and 3 of post-natal life were collected. Half a milliliter of blood was collected for genotyping of the antioxidant enzymes for all study participants.
Analysis of oxidative markers and antioxidants, and genotyping of SNPs
Enzyme-linked immunosorbent assay kits were used for evaluating the following oxidative biomarkers: 8-OH-dG (ThermoFisher: catalog number—EIADNAD), TAC (ABCAM: catalog number—ab65329), MDA (assayed by measuring thiobarbituric acid reactive substances) (R&D systems: catalog number—KGE013), AOPPs (MyBioSource.com: catalog number—MBS028634), chaperonin 60 (MyBioSource.com: catalog number—MBS702807), reduced glutathione (MyBioSource.com: catalog number—MBS727656), and visfatin (ABCAM: catalog number—ab264623). SNPs in MnSOD (rs4880 and rs5746136), Cu/Zn SOD (rs8192287), GSTP1 (rs1695), GPX1 (rs1050450), GPX3 (rs1946234), CAT (rs1001179), and eNOS (rs2070744 and rs1799983) were evaluated using TaqMan® SNP genotyping assays through the real-time polymerase chain reaction. Extraction of the total genomic DNA from the peripheral white blood corpuscles was carried out using QIAamp® DNA Blood Mini Kit (Qiagen), following which the DNA concentration was measured using a Nanodrop spectrophotometer. The allelic discrimination method was used for evaluating the SNPs using the StepOne Plus® real-time PCR system (Applied Biosystems; Foster City, CA) according to the instructions of the manufacturer) using TaqMan® assays.
Statistical analysis
Descriptive statistics were used for representing the demographic variables. Kruskal-Wallis H and Mann-Whitney U tests were used for evaluating the numerical variables between the groups, and Wilcoxon signed-rank sum and Friedman test were for within the group differences. The Chi-square test of association with Yates correction and Fisher’s exact probability test were used for analyzing the statistically significant differences between the categorical variables. The receiver-operating characteristics (ROC) curve was used for evaluating the significance of oxidative biomarkers in predicting pulmonary complications in neonates with RDS. The area under the curve (AUC) with a 95% confidence interval (CI) was used as the effect estimate for predictive accuracy. The optimal cut-off point was established using the Youden index. The sample size of the present study was calculated with the estimates of total antioxidant capacity reported in a previous study (cases: 2.58 + 0.81 and controls: 3.3 + 1.1 mmol/L), 19 power of 80%, and 5% level of two-sided significance, 40 was the estimated sample size for each group. A P-value of ⩽.05 was considered statistically significant and 95% confidence intervals (CI) were used for effect estimates. SPSS (IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp.) was used for statistical analysis.
Results
Demographics
Eighty neonates were recruited in this study and a summary of their demographic characteristics is mentioned in Table 1. Neonates with RDS were significantly younger in terms of their gestational age compared to the control group. Seven (17.5%) neonates received 2 doses while the remaining (43, 82.5%) received a single dose of surfactant.
Demographic characteristics of the study participants.
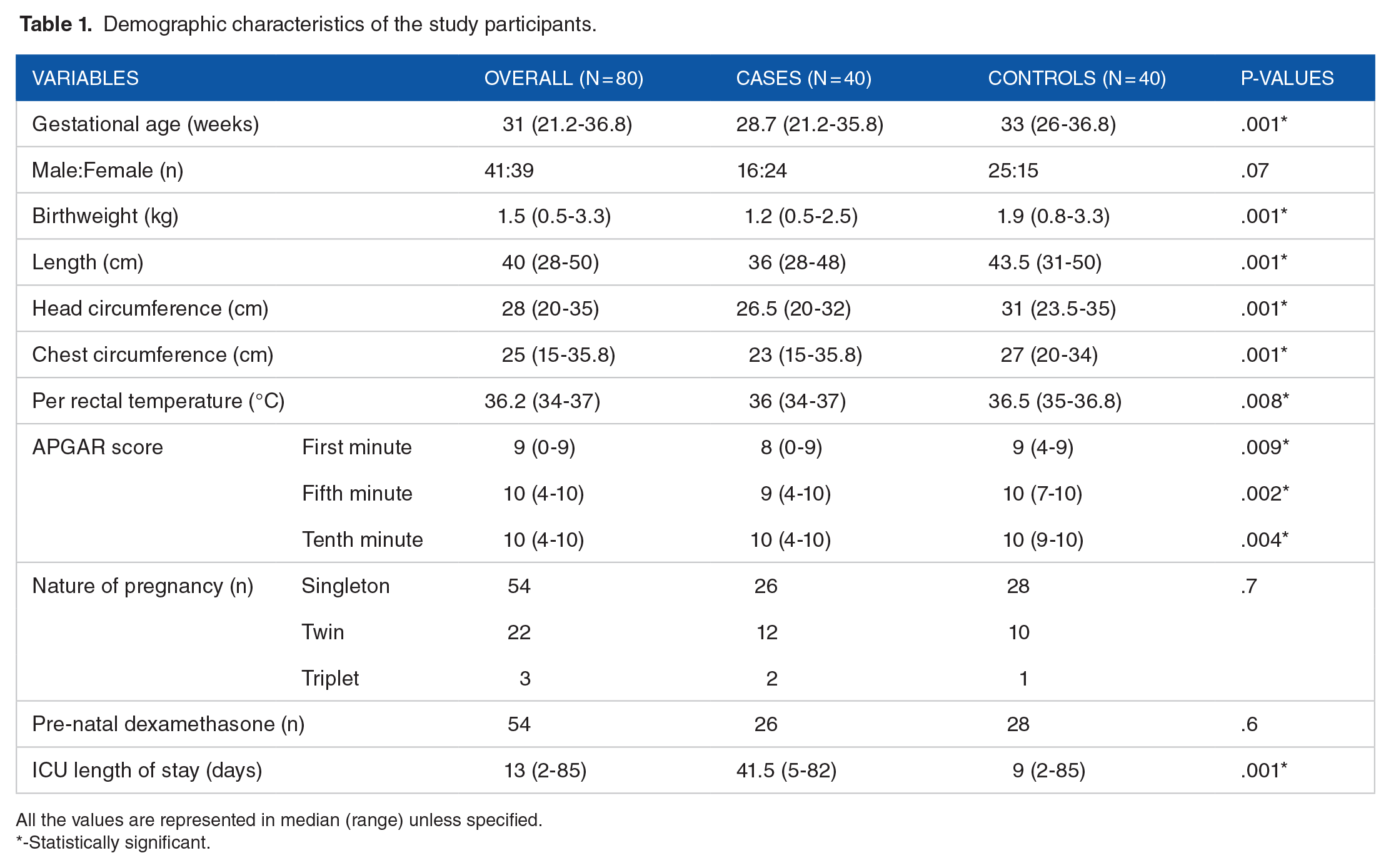
All the values are represented in median (range) unless specified.
-Statistically significant.
SNPs and oxidative biomarkers
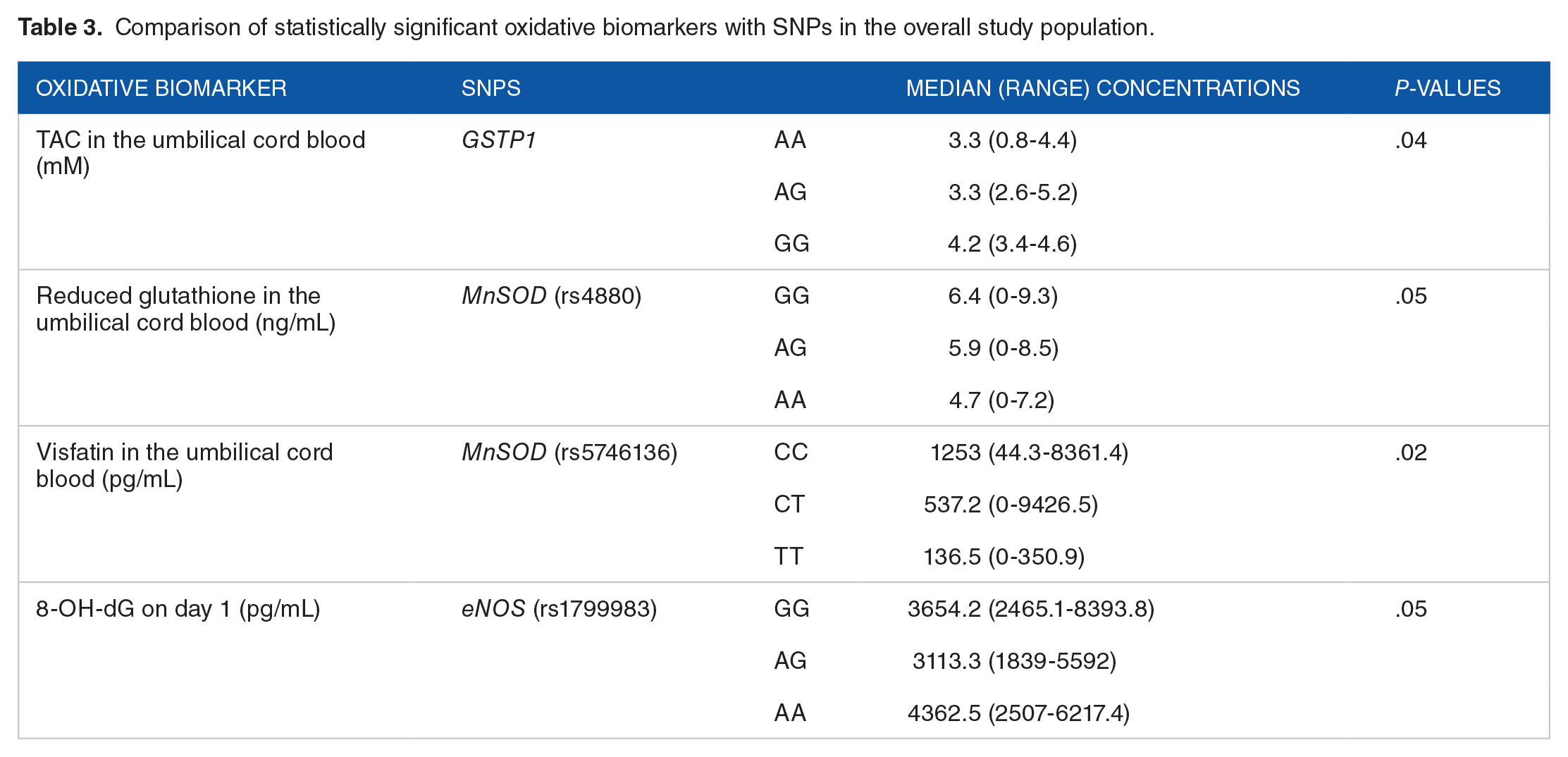
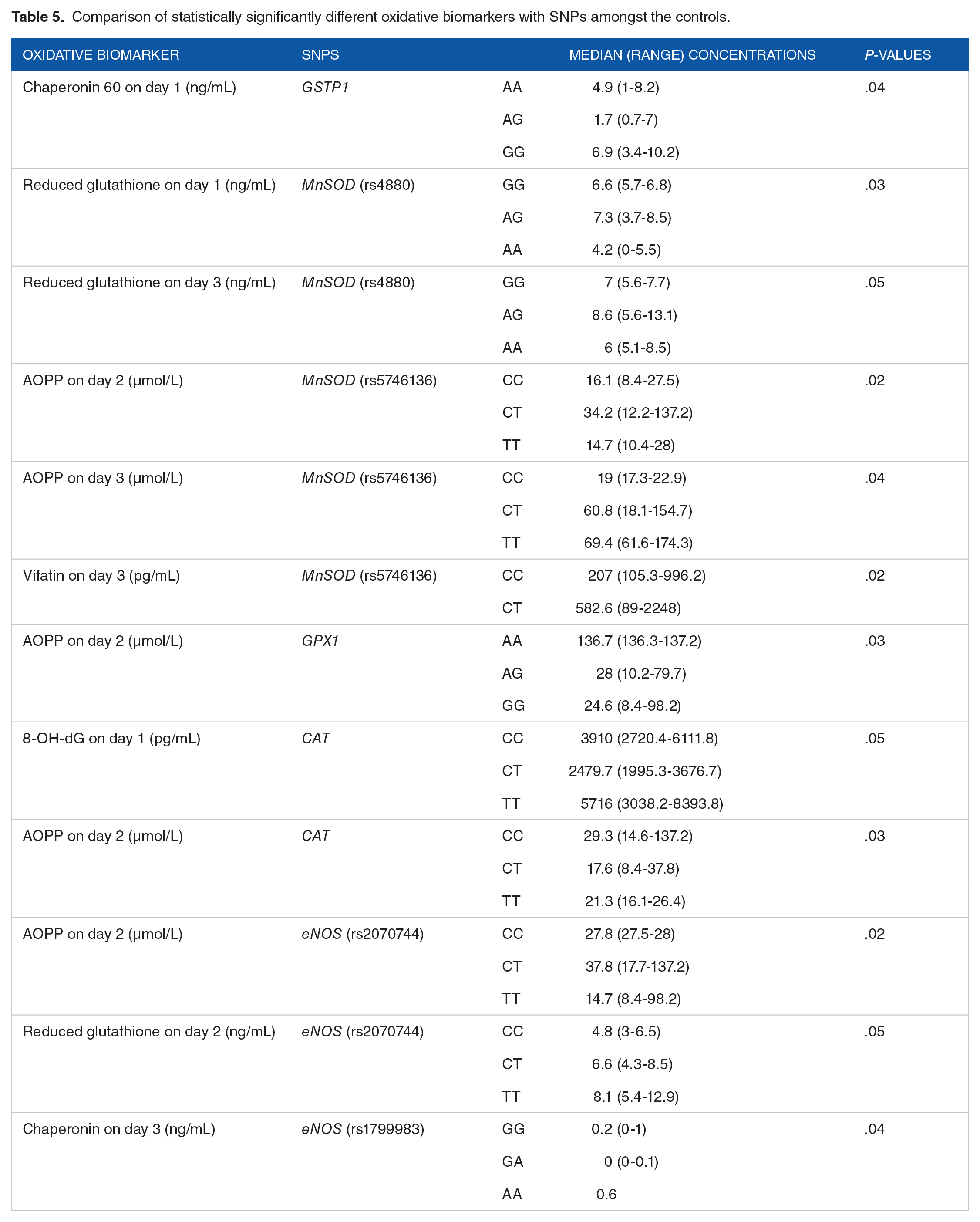
The prevalence of SNPs amongst the study participants is depicted in Figure 1. No significant differences were observed in the distribution of evaluated SNPs between cases and the control group (Table 2). GSTP1, MnSOD, and eNOS (rs1799983) SNPs were observed to significantly influence the oxidative biomarker concentrations in the entire study population (Table 3). Several SNPs have been observed with statistically significant differences in the concentrations of the biomarkers amongst the neonates with RDS (Table 4) and the control group (Table 5).

Prevalence of SNPs in the study participants. The horizontal bar chart depicts the number of study participants in each category of genetic polymorphism; Homozygous 1 for GPX3 was AA, eNOS 2 (rs1799983) was GG, eNOS 1 (rs2070744) was CC, CAT was CC, GPX1 was AA, Cu/Zn SOD was GG, MnSOD 2 (rs5746136) was CC, MnSOD 1 (rs4880) was GG, and GSTP1 was AA; Homozygous 2 for GPX3 was GG, eNOS 2 (rs1799983) was AA, eNOS 1 (rs2070744) was TT, CAT was TT, GPX1 was GG, Cu/Zn SOD was AA, MnSOD 2 (rs5746136) was TT, MnSOD 1 (rs4880) was AA, and GSTP1 was GG; Heterozygous for GPX3 was GA, eNOS 2 (rs1799983) was AG, eNOS 1 (rs2070744) was CT, CAT was CT, GPX1 was CG, Cu/Zn SOD was AG, MnSOD 2 (rs5746136) was CT, MnSOD 1 (rs4880) was AG, and GSTP1 was AG.
Comparison of SNPs between neonates with RDS and controls.

All the values are expressed in total numbers.
Comparison of statistically significant oxidative biomarkers with SNPs in the overall study population.
Comparison of statistically significant oxidative biomarkers with SNPs amongst neonates with RDS receiving surfactant.

Comparison of statistically significantly different oxidative biomarkers with SNPs amongst the controls.
Association of SNPs and oxidative stress biomarkers with pulmonary complications
Four developed pneumothorax, 2 developed pulmonary hemorrhages, and 5 developed chronic lung disorder among the neonates with RDS. AA genotype in MnSOD (rs4880) was observed with a significantly greater risk of any of the composite pulmonary complications (Table 6). Similarly, MDA in the umbilical cord blood, 8-OH-dG on day 1, and AOPP and MDA on day 3 were significantly different between the neonates that developed any of the composite pulmonary complications and those that did not (Table 7). The ROC analysis revealed DNA damage product (8-OH-dG) concentrations before surfactant administration have the best predictive accuracy (AUC: 0.8; 95% CI: 0.7-1; P = .001) for the composite pulmonary complications (Figure 2). An optimal cut-off value of 5008.8 pg/mL for the baseline 8-OH-dG was observed to significantly predict the risk of pulmonary complications in neonates with RDS.
Association of SNPs with pulmonary complications amongst RDS neonates.

Statistically significant.
Comparison of statistically significant oxidative biomarkers amongst those with pulmonary complications in the RDS neonates.

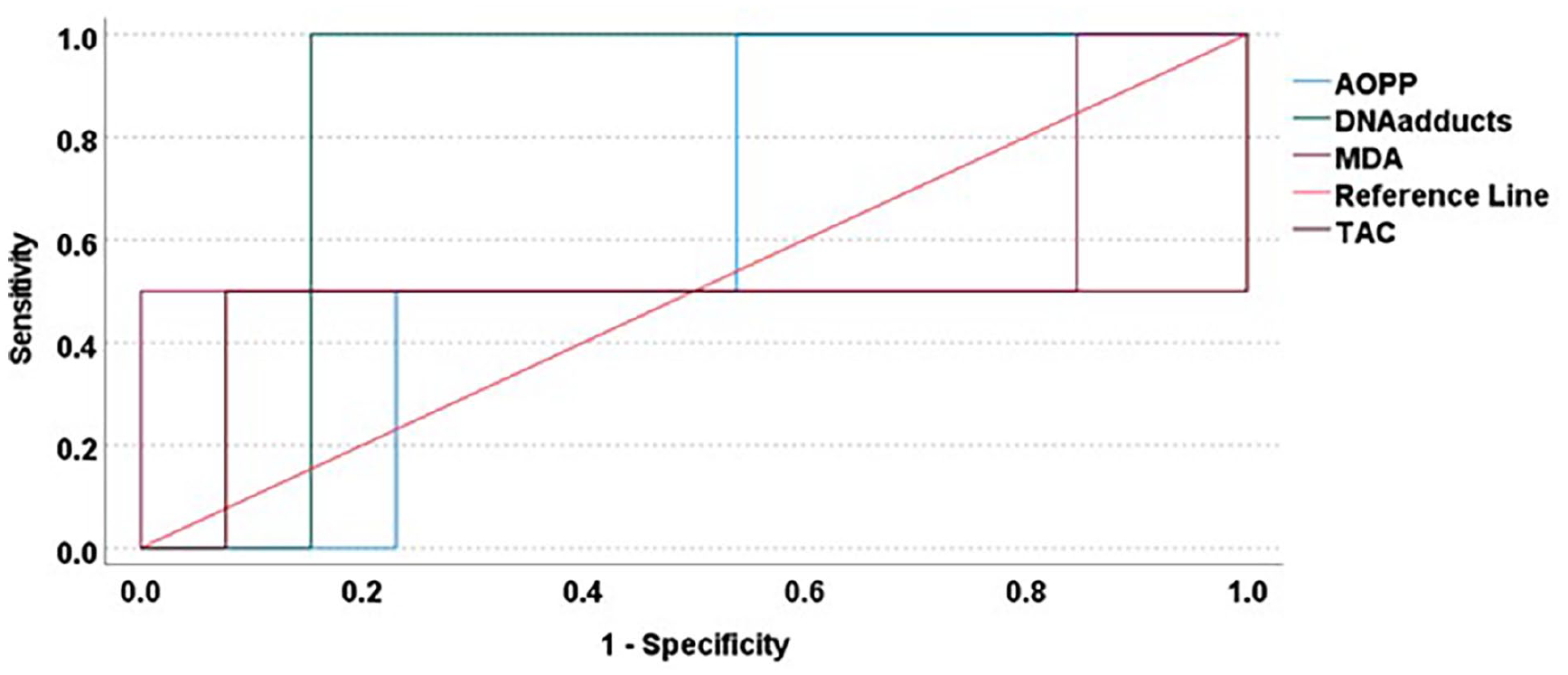
ROC analysis of oxidative biomarkers predictive of composite pulmonary complications. The diagonal line in this ROC plot indicates the reference line while the other color-coded lines represent each of the oxidative biomarkers. AOPP – Advanced oxidation protein products; DNA adducts (8-OH-dG) – 8-hydroxy 2’-deoxyguanosine; MDA – Malondialdehyde; TAC – Total antioxidant capacity.
Association of SNPs and oxidative stress biomarkers with other variables
Seven neonates with RDS each had grades 1 and 3, while 13 neonates each had grades 2 and 4. No significant association was observed between the SNPs and RDS gradings. AOPP in the umbilical cord blood was observed to significantly predict the severity of RDS (AUC: 0.8; 95% CI: 0.6-1; P = .01) with an optimal cut-off value of 88.78 µmol/L (Figure 3). No significant differences were observed in any of the oxidative stress biomarkers between those that received antenatal dexamethasone and those that did not. Two neonates died precluding the statistical analysis of differences between any of the oxidative biomarkers/SNPs for mortality.

ROC analysis for evaluating oxidative stress biomarkers with RDS severity. The diagonal line in this ROC plot indicates the reference line while the other color-coded lines represent each of the oxidative biomarkers. AOPPU – Advanced oxidation protein products in the umbilical cord blood; DNA adducts (8-OH-dG) – 8-hydroxy 2’-deoxyguanosine in the umbilical cord blood; RGLU – Reduced glutathione in the umbilical cord blood; TACU – Total antioxidant capacity in the umbilical cord blood;TBARSU – Thiobarbituric acid reactive substances in the umbilical cord blood; VisU – Visfatin in the umbilical cord blood.
Biomarkers of oxidative stress amongst neonates
Comparison of oxidative stress biomarkers in the umbilical cord blood, and at various time points from the peripheral blood of the neonates with RDS receiving surfactant is represented in Table 8. DNA damage products were observed to be significantly greater on days 1, 2, and 3 compared to umbilical cord blood. Similarly, TAC was significantly more on days 2, and 3 following surfactant therapy. On the contrary, chaperonin 60 was significantly greater on day 3 while visfatin was significantly greater in the umbilical cord blood.
Oxidative biomarkers in neonates with RDS receiving surfactant.
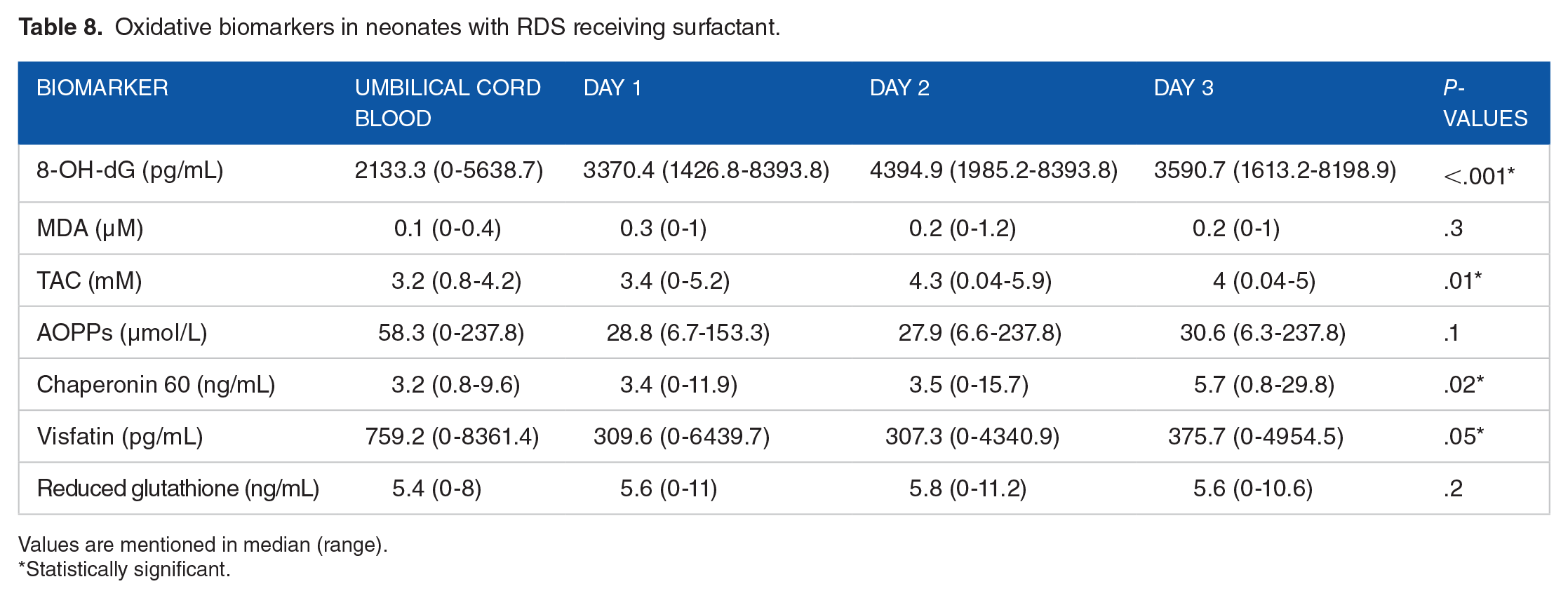
Values are mentioned in median (range).
Statistically significant.
Comparison of the concentrations of the biomarkers of oxidative stress amongst the control group of neonates was not significantly different at any of the time points (Table 9).
Oxidative biomarkers in control neonates.

Values are mentioned in median (range).
A comparison of biomarker concentrations of oxidative stress is depicted in Supplemental Figure 1. TAC concentrations are significantly greater on day 2 and day 3 amongst neonates receiving surfactant compared to the control group.
Discussion
Key findings of the present study
We evaluated the concentrations of the biomarkers of oxidative stress and prevalence of SNPs of the antioxidant enzymes in 40 neonates with RDS compared to 40 controls. SNPs in GSTP1, MnSOD, and eNOS (rs1799983) were significantly associated with differences in the oxidative stress biomarkers in the entire study population while eNOS, MnSOD, Cu/Zn SOD, CAT, and GPX3 in neonates with RDS, and GSTP1, eNOS, MnSOD, GPX1, and CAT in the controls. MnSOD (rs4880) significantly increased the risk of pulmonary complications in neonates with RDS. The administration of external surfactant in neonates with RDS showed a significant improvement in terms of improved TAC but not regarding other oxidative biomarkers. Baseline 8-OH-dG concentration before external surfactant administration has the best predictive accuracy for identifying the risk of pulmonary complications while AOPP in the umbilical cord blood for predicting the severity of RDS.
Comparison with other studies
Negi et al observed a positive association of risk of RDS with protein carbonyl, MDA, and 8-OHdG, and an inverse relation with TAC, in comparison with healthy newborns. 20 However, the control group of neonates had a median (range) gestational age of 34.6 (34.1-35.2) weeks compared to the present study where the median gestational age amongst the controls was 33 weeks and the lower limit of the range was 26 weeks. It is well known that gestational age influences the quantum of antioxidant enzymes and molecules. 21 Secondly, we did not have a group of healthy preterm neonates as a comparator which might have led to this difference in the finding. Another study that included healthy control as a comparator was Elkabany et al 19 where the authors observed that the serum levels of MDA, AOPPs, and 8-OHdG were significantly higher in neonates with RDS like the lower levels of TAC, copper, and zinc in the umbilical cord blood. The authors of that study also evaluated the concentrations of the oxidative stress biomarkers on day 3 and found that they were still significantly greater than the healthy control group of neonates. The management protocol for RDS (INSURE) is like that of the present study and we also have observed that despite external surfactant administration except for TAC, no significant improvement was observed in other biomarkers. However, the authors have also observed elevated levels of 8-OHdG and AOPPs with severe RDS and mortality, unlike the present study. This could be explained by the differences in the population included in their study as only one had the pulmonary complication (while 7 in the present study) and 6 neonates out of 40 died in their study while only 2 in the present study. However, the authors observed that 8-OH-dG could predict the severity of RDS grade with a cut-off value of 2418 pg/mL and mortality with a cutoff value of 7250 pg/mL whereas we observed a cut-off value of 5008.8 pg/mL for predicting the pulmonary complications. Similarly, the authors have also observed an AOPP cut-off value of 47 ng/mL for predicting the RDS severity and 95 ng/mL for predicting mortality while we observed a cut-off value of 88.78 µmol/L for AOPP in the umbilical cord blood for predicting the RDS severity. AOPP and 8-OH-dG are advanced oxidation products that are indicative of the intensity of oxidative stress and correspondingly inflammation. So, the present study stresses the importance of early diagnosis of RDS and the need for timely intervention using an external surfactant to reduce the incidence of chronic respiratory complications. Marseglia et al recently reported that visfatin (an adipocytokine) can be a novel biomarker indicative of oxidative stress in neonates with RDS. 22 The authors in that study included preterm neonates that underwent cardiopulmonary resuscitation and observed that serum visfatin concentrations were proportional to the oxygen concentrations administered. We did not observe any significant differences with this biomarker in the study population.
There are limited studies evaluating the role of single nucleotide polymorphisms in the antioxidant enzymes in neonates. Giusti et al 23 through a retrospective study observed that rs8192287 Cu/Zn SOD polymorphism independently protects the neonates against bronchopulmonary dysplasia, RDS, and intraventricular hemorrhage, while MnSOD polymorphisms increased the risk of bronchopulmonary dysplasia. We also observed that SNP in MnSOD was significantly associated with the risk of pulmonary complications following RDS. Hence, it may be prudent to consider more aggressive treatment in this sub-population with the SNP in the MnSOD enzyme. Poggi et al 24 observed that polymorphisms in endothelial nitric oxide synthase (eNOS) (rs2070744 and rs1799983) significantly influence the risk of RDS in neonates. In the present study, we did not observe any statistically significant difference (P = .09) when analyzed as individual genotypes, but with the combination (CC with CT+TT), a trend toward statistical significance was observed (P = .06). Probably, the small sample size in the present study might have limited the significance of this association.
To the best of our knowledge, this is the first study evaluating the concentrations of oxidative stress biomarkers and their association with SNPs of the antioxidant enzymes in preterm neonates. Despite some differences between neonates with RDS and the control group, eNOS and MnSOD polymorphisms were observed to exert significant differences in the entire study population, as well as amongst the cases and controls. MnSOD is localized in mitochondria, the most important oxygen metabolizing cellular component resulting in the production of reactive oxygen species. 25 Hence SNPs in MnSOD are likely to produce a functional impact on the oxygen scavenging system thereby increasing the risk of cellular damage induced by the oxidative stress molecules. This reiterates the significance of evaluating the presence of SNPs in MnSOD to determine candidates for more aggressive treatment.
Strengths and limitations
This is the first study evaluating the association between oxidative biomarkers and SNPs of the antioxidant enzymes in neonates with RDS, in comparison with the control group. The present study has also evaluated the role of SNPs and oxidative biomarkers in pulmonary complications in neonates with RDS. However, the inclusion of a group of healthy preterm neonates not admitted to the intensive care unit might provide additional information on the strengths of the association of SNPs and oxidative biomarkers with RDS. We could also not evaluate the influence of oxygen concentrations and the type of mechanical ventilation. Also, a larger sample size might have provided more conclusive evidence on the association of SNPs with various biomarkers and clinical outcomes. Also, it is not feasible to obtain neonates in the extremely preterm category without RDS and so despite the entire study population being preterm, the control groups were slightly older.
Conclusion
We observed that SNPs in eNOS and MnSOD significantly influence oxidative stress biomarkers in preterm neonates. Baseline 8-OH-dG concentrations best predict the risk of pulmonary complications and AOPP concentrations in the umbilical cord blood predict the risk of RDS severity.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719221137608 – Supplemental material for Evaluation of Genetic Polymorphisms of the Antioxidant Enzymes and Biomarkers of Oxidative Stress in Preterm Neonates With Respiratory Distress Syndrome Receiving External Surfactant
Supplemental material, sj-docx-1-bmi-10.1177_11772719221137608 for Evaluation of Genetic Polymorphisms of the Antioxidant Enzymes and Biomarkers of Oxidative Stress in Preterm Neonates With Respiratory Distress Syndrome Receiving External Surfactant by Kannan Sridharan, Mona Al Jufairi, Aamal AbdulGhani Mahdi Hejab, Abdulraoof Al Madhoob, Reem Al Marzooq, Safa Taha, Muna Jaber Mulla Aljishi, Ameera Abdulhadi, Eman Al Ansari, Masooma Abdulla Ali, Maryam Ali Ahmed Naser, Ola Al Segai and Kevin Dunne in Biomarker Insights
