Abstract
Background:
MicroRNAs are short nucleotide sequences that contribute to the regulation of various biological functions and therefore their roles have been investigated in many pathologic conditions such as epithelial to mesenchymal transition in cancer and fibrosis; among them, miR-138 has been mostly studied in cancer biology and is well-known for its suppressing effect on cancer progression. Being able to suppress major pathways involved in EMT, miR-138 could be a good candidate to be investigated in fibrotic responses too. Based on our previous studies, and the capability of miR-138 to target and regulate several components of the EMT pathway; we hypothesized a role for miR-138 in systemic sclerosis. Accordingly, the gene expression of miR-138 was assessed to find any alterations in the whole blood of the SSc patients.
Methods:
Blood was collected from 70 patients with systemic sclerosis (equally divided between 2 groups of limited and diffuse categories) and 30 healthy individuals as controls. RNA was immediately isolated from the fresh whole blood; afterward, the resulting RNA was reverse transcribed into cDNA and then the relative expression of miR-138 was compared between the patients and the controls by the means of qPCR, and specific TaqMan primer and probes.
Results:
The relative expression of miR-138 was significantly lower in patients with systemic sclerosis compared to the controls. No significant difference was observed between the limited and diffuse patient groups. ROC curve analysis showed an appropriate diagnostic value of miR-138 in effectively differentiating SSc patients from the healthy controls.
Conclusion:
miR-138 is likely involved in the pathogenesis of SSc and with further evaluations may be utilized as a diagnostic biomarker in SSc. Also, targeting miR-138 in future studies could be promising for finding a novel treatment option for patients with SSc.
Introduction
Systemic sclerosis (SSc) is an inflammatory disease diagnosed by various symptoms manifesting in different body organs, resulting from either vascular damage or autoimmunity; on top of that, are fibrotic lesions that could be identified in almost all parts of the body.1,2 In fact, the most prominent characteristic of SSc is skin thickening due to fibrosis which results from excessive production of the extracellular matrix in connective tissue.3,4 Internal body organs are also affected by fibrosis which leads to organ failure and mortality. 5 The exact etiology of this disease is not identified yet, although like other inflammatory diseases a combination of genetic predisposition and environmental factors, especially industrial pollutants are thought to be involved. 6 SSc affects women 5 times more than men, and also is more prevalent among some ethnicities or populations. 7 Subsequently, the prevalence of SSc shows a remarkably different distribution among various regions; it seems that SSc is most prevalent in the United States and is less prevalent among eastern countries.8,9 Besides, it affects African-American people with more severe symptoms and comorbidities. 7 Nevertheless, still there are many undiscovered aspects of the underlying pathogenic mechanisms and etiology of this disease. 10 Hence, the diagnosis and treatment of SSc are challenging, due to the lack of proper and specific biomarkers and medical interventions capable of effectively treating this illness.6,11,12 The majority of the currently employed medical interventions in the treatment of SSc only focus on addressing the clinical symptoms that the patient presents with, rather than targeting a fundamental aspect of the disease pathogenesis, this may lead to inappropriate management of patients with complex comorbidities.13,14 As a result, researches are ongoing to elucidate all aspects of the SSc pathogenesis. Among the new fields of study, are the microRNAs; these small sequences of about 20 nucleotides have been found to contribute to many cellular functions such as metabolism, proliferation, senescence, and death.15,16 Therefore, they are implicated in various pathologic conditions and they have been investigated in many illnesses such as cancer, autoimmunity, impaired development, etc.17-19 Being able to target and regulate many genes involved in prominent signaling pathways progressing in hard-to-cure illnesses like cancer, miRNAs have been widely studied in those pathways contributing to major events in the pathogenesis of cancer.20,21 As we believe that fibrotic disorders like SSc utilize the same cellular key pathways employed by cancer,22-25 we sought to investigate the well-known components of cancer in our patients suffering from SSc. Of the most prominent shared pathways between cancer and fibrosis is the epithelial to mesenchymal transition (EMT)26-29; this phenomenon not only underly the proliferation, invasion, and metastasis in cancer, but also is the main cause of fibroblast differentiation into myofibroblasts as the major event in fibrotic disorders, ultimately leading to organ dysfunction. 30 One of the well-studied miRNAs in cancer is miR-138 which is capable of targeting and regulating the EMT pathway at many points.26,31-37 It has been found that this miRNA could target TGF-β-associated signaling molecules in cancer; which is also the key regulator in fibrosis and SSc.38-40 Accordingly, we proposed a possible role for miR-138 in SSc and decided to assess its gene expression in the blood of SSc patients to find out whether this miRNA is dysregulated in SSc or not. And to further evaluate any possible alterations to be employed as a diagnostic biomarker.
Methods
Patients: This was a cross-sectional randomized case-control study that was conducted between June 2017 and November 2017. The minimum sample size was calculated based on the prevalence of SSc, which is estimated to be 15 per 100 000 individuals, 41 with a precision of 0.05 and type one error of 0.05. 42 Equal numbers of patients either with limited or diffuse SSc (30 from each subtype, 60 in total) were randomly selected from the cohort of the Firuzgar Hospital, Tehran, Iran. All the patients filled out an informed consent. The diagnosis of SSc and categorization of the patients between its 2 subtypes was performed by an expert rheumatologist with years of experience in SSc, according to ACR/EULAR criteria. The history of the patients was thoroughly reviewed and their laboratory findings, as well as X-rays and high-resolution computed tomography (HRCT), were obtained (Figure 1). Individuals with a history of other complications and comorbidities such as malignancies and infections or those receiving certain drugs that would possibly affect the results were excluded. Due to the very few numbers of male SSc patients, they were omitted and the female patients only were enrolled. The mean age of the patients was 51 years and their average BMI was 24.4. The controls (30 individuals) were randomly chosen among the healthy female individuals who were referred to the laboratory for checkups, those matching the age of the patients with normal laboratory findings and no known history of any illnesses were enrolled. All the methods and procedures conducted in this study were confirmed by the ethics committee of the Iran University of Medical Sciences (ethics code: IR.IUMS.FMD.REC.1397.316).

Comorbidities (A) and autoantibodies (B) found in SSc patients enrolled in the current study, the bars represent the percent of the affected individuals with each comorbidity.
RNA isolation and cDNA synthesis
Whole blood was drawn from the patients into EDTA vials by an expert laboratory technician considering the skin and vascular problems of the patients. Immediately, the samples were transferred to the laboratory and underwent RNA isolation steps from the whole blood using RNAzol BD (MRC, USA); 500 µl of blood was added to 1 ml of the RNAzol reagent and the whole process was carried out according to the manufacturer protocols. The quality and concentration of the isolated RNA samples were evaluated using gel electrophoresis and the NANODROP instrument (Thermo Fischer, USA). Subsequently, fresh isolated RNA was used as the template for synthesizing cDNA using TaqMan advanced microRNA synthesis kit (Thermo Fischer, USA) following the manufacturer’s instructions, then the resulting cDNA was kept at −20°C to be later employed in the qPCR tests.
qPCR
The relative gene expression of miR-138 was assessed in the whole blood of the patients and the controls using TaqMan advanced microRNA assays specific for miR-138 and miR-199-5p as the internal control according to the manufacturer and previous studies. Also, cel-miR-39 (Qiagen, Germany) was used as the external control. All the qPCR tests were performed in duplicates along with no template controls and no primer controls using RotorGene Q real-time PCR system (QIAGEN, Germany).
Statistical analysis
The relative gene expression was calculated and compared between the study groups using the 2−ΔΔCT with the miR-199-5p as a normalizer. qPCR data were compared using one-way ANOVA and Tukey’s post hoc, correlations were assessed using Pearson’s correlation tests. All data were analyzed and plotted using GraphPad PRISM software; the data are represented as mean ± SD and the P values equal to or below .05 were considered statistically significant.
Results
It was observed that the relative expression of miR-138 was considerably decreased in both limited and diffuse patients in comparison to the controls (P values ⩽ .0001) (Figure 1). Also, there was a further slight reduction in the miR-138 gene expression in diffuse patients relative to the limited patients (P-value = .9184) (Figure 2).

Relative gene expression of miR-138. The relative expression of miR-138 is significantly downregulated in the 2 SSc subsets in comparison to controls. (****P-value ⩽ .0001).
Since we observed a remarkable downregulation of miR-138 in SSc patients, we sought to evaluate whether this miRNA could be utilized as a biomarker capable of differentiating the SSc patients from the healthy controls. Hence, ROC curve analysis was performed on the gene expression data; it was found that miR-138 gene expression is effectively useful in the diagnosis of the patients. Figure 2 illustrates the diagnostic value of miR-138 gene expression data using the ROC curve plots in different states; a good Area Under the Curve (AUC), as well as acceptable sensitivity and specificity, was obtained as shown in Figure 3 and Table 1.
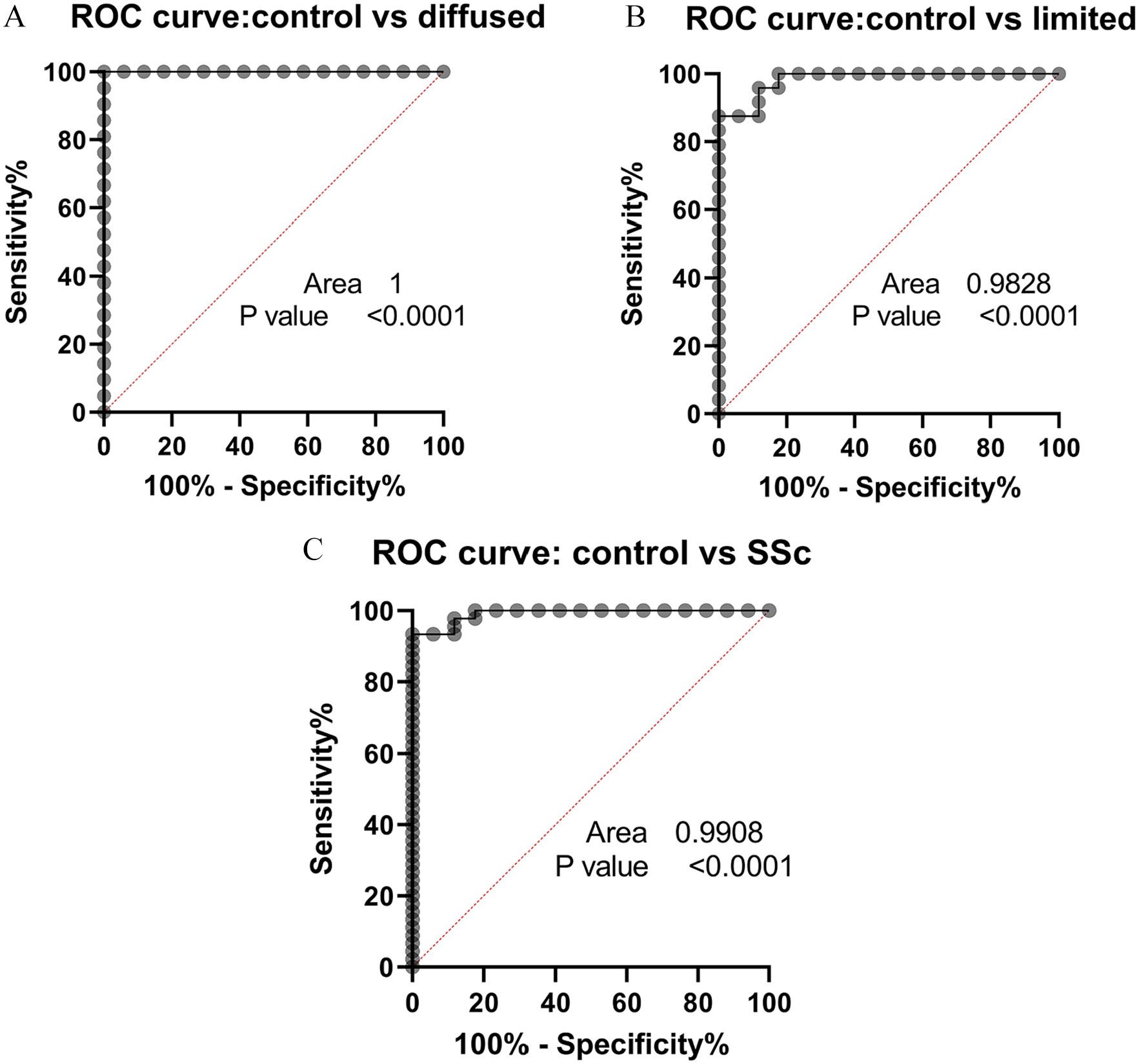
ROC curve analysis of miR-138 relative expression. (A) This plot demonstrates the good diagnostic value of miR-138 for identifying the SSc patients with the diffuse subtype, from healthy controls. (B) This plot illustrates a relatively good diagnostic value for miR-138 relative expression in identifying the SSc patients with limited subtype from healthy individuals. (C) This graph was plotted to evaluate the capability of miR-138 in discriminating SSc patients with both limited and diffuse subtypes. P values ⩽ .05 were considered significant.
ROC curve characteristics for 3 individual comparisons.

In order to gain a better insight into the role of miR-138 in systemic sclerosis and how it could affect the disease symptoms and complications we conducted a global comparison of the miR-138 expression between individual subsets based on showing a specific symptom or having a specific auto-antibody; The results of this analysis are presented in Figure 4 in details. Altogether, it seems that having high levels of miR-138 could be associated with a lower incidence of PAH, Digital ulcers, and Myositis, as well as decreased autoantibodies against DNA topoisomerase, ribonucleoproteins, and RNA polymerase III; whereas upregulation of miR-138 correlates with increased incidence of ILD, Telangiectasia, and ACA autoantibody (Figure 4). Also, it seems that the expression of miR-138 in female cases is lower than that of the male cases, although the number of male cases was 7 compared to 60 females.

The differences in miR-138 expression between pairs of different subsets. Each pair of bars represents the expression of miR-138 in a specific subset based on the comorbidities or autoantibodies found in SSc patients.

A proposed mechanism for miR-138 in the pathogenesis of SSc. Any chronic tissue damage such as those associated with inflammatory and rheumatologic illnesses could induce prolonged activation of the healing responses leading to fibrotic lesions; the cardinal mediator in wound healing is TGF-β which triggers several cell signaling pathways to enhance cell proliferation, survival, and production of extracellular matrix components in the fibroblasts undergone differentiation into myofibroblast as a result of EMT. Over time this aberrant activation of the healing pathways could result in undesired consequences in those patients who are genetically susceptible to developing SSc. In this context, miR-138 seems to be downregulated by the TGFβ and some other epigenetic factors such as lncRNAs; as a result, miR138 possibly plays a substantial role in the regulation of EMT by targeting several major contributors such as the PI3K/AKT pathway, ZEB2 as a master regulator of mesenchymal genes and MMP2/MMP9, which are responsible for tissue remodeling.
Discussion
In this study, the relative expression of miR-138 was assessed in the whole blood of SSc patients with either limited or diffuse subtypes of the disease for the first time. It was observed that this miRNA is downregulated in the whole blood of the SSc patients in comparison to the healthy controls; it was even further decreased in patients with the diffuse subtype. subsequently, it was demonstrated in ROC curve analyses that the relative expression of miR-138 could be utilized as a reliable diagnostic biomarker for effectively diagnosing both subtypes of SSc. It could not be served for discriminating the limited subtype from its diffuse counterpart. The selection of this miRNA in the current study was based on previous research regarding the various possible functions of miR-138 in several cancers. We aimed to evaluate miR-138 expression since we had focused on the TGF-β signaling pathways, especially the non-canonical (non-SMAD) pathways, such as the PI3K-AKT-mTOR pathway which contributes to the EMT process. 43 EMT is the main underlying mechanism responsible for the activation of fibroblasts in fibrotic disorders as well as cancer cell survival, proliferation, and invasion. 44 In this regard here we summarize some of the main findings achieved so far, especially in the field of cancer research, in order to propose a role for miR-138 in fibrotic disorders such as SSc. 27 Although the reports are not consistent; altogether, they provide an insight into different roles that miR-138 may play in the context of fibrotic disorders. Some studies demonstrated that miR-138 is a negative regulator of the EMT pathway which serves an anti-tumor role,33,45 while some other studies consider it as a pro-tumor factor by inducing the EMT.46-49 In support of our theory, Wu et al 50 have demonstrated that miR-138 inhibits EMT in mice models of idiopathic pulmonary fibrosis (IPF) through direct targeting of the ZEB2 transcription factor, which is responsible for activating the transcription of mesenchymal markers and repressing the epithelial markers; therefore, it is possible that the downregulation of miR-138 in our study somehow has resulted in the overactivation of the ZEB2. Also, it was revealed that the lncRNA PFAR promotes fibroblast activation through the downregulation of miR-138. 51 Interestingly, It has been shown that the expression of miR-138 is downregulated by the TGF-β1 in the process of EMT in the primary lung cancer cells 38 ; TGF-β on the other hand is recognized as the master inducer of fibrosis which could explain the downregulation of miR-138 in SSc. 52 Besides, miR-138 was demonstrated to be able to reduce the proliferation and invasion of the prostate cancer cell lines through inhibition of the Wnt/β-catenin pathway which is also involved in fibrogenesis and SSc.53,54 Also, in renal and lung carcinoma cell lines and patient samples, it was observed that the expression of miR-138 is reduced which results in the overexpression of SOX4 in the EMT pathway,.32,55 Interestingly, miR-138 was shown to be upregulated in chondrocytes from mice models of rheumatoid arthritis, which propose a role for this miRNA in rheumatologic diseases like SSc; this finding is consistent with our data as well. 56 Additionally, it was demonstrated that miR-138 could directly bind to and regulate HIF1-α which subsequently results in the inhibition of the MAPK and AKT signaling pathways in the endothelial cells and malignant melanoma cells as well.34,57 Furthermore, in cell cultures mimicking the human coronary artery endothelial cell (HCAEC) injury which were treated with miR-138 mimics, it was shown that miR-138 is capable of suppressing the PI3K-AKT pathway as well as upregulating IL-4 and IL-10 which are known to dampen inflammatory responses involved in fibrosis. 58 In line with our theory, Liu et al 39 have shown that in human airway muscle cells, miR-138 directly binds to and downregulates PDK1, which serves a key role in the activation of the PI3K-AKT pathway thus inhibiting the whole pathway and cell proliferation. Furthermore, in human fibroblasts obtained from hypertrophic scars, it was shown that miR-138 targets and regulates PPARβ which contributes to the activation of the PI3K-AKT pathway. 59 Moreover, in a study conducted to elucidate the effect of miR-138 on pulmonary arterial hypertension (PAH) which is among the comorbidities associated with SSc, it was observed that miR-138 was downregulated which results in increased apoptosis (leading to tissue remodeling) in human artery smooth muscle cells, through upregulation of Mst1 as a miR-138 target.35,60 Even MMP2 and MMP9 which are among the major contributing factors in the extracellular matrix remodeling were shown to be targeted and inhibited by miR-138. 36 Besides, miR-138 was shown to be upregulated in chondrocytes from mice models of rheumatoid arthritis, which propose a role for this miRNA in rheumatologic diseases like SSc; although this finding is opposed to our results regarding the downregulation of miR-138 in SSc. 56 Overall, these data and our results suggest that miR-138 has a role in the regulation of signaling pathways involved in EMT, which is a key contributing factor in the pathogenesis of both cancer and systemic sclerosis (Figure 5). Our study was conducted in a single cohort and the number of patients enrolled was quite small; indeed, increasing the sample size and gathering data from other cohorts in the country would enhance the robustness of our results and help with drawing a comprehensive conclusion. In the current study, only gene expression of miR-138 was evaluated in whole blood and no functional analysis was performed so as to elucidate the exact role of miR-138 in the pathogenesis of SSc, nor the putative targets were identified which is a major limitation in our study. It is suggested to assess the expression of miR-138 in clinical samples such as skin or lung fibrotic lesions to further gain insight into the local expression of this miRNA in SSc. Also, investigating the epigenetic factors affecting miR-138 expression, such as long non-coding RNAs (eg, HOTAIR and PFAR) would be beneficial in understanding the etiology of SSc.51,56 Further evaluation of miR-138 role in the pathogenesis of SSc could also pave the way for introducing this miRNA as a novel therapeutic target too. 27
Conclusion
MiR-138 is significantly downregulated in patients with SSc, especially in those with diffuse subtypes, suggesting a role for miR-138 in SSc which definitely need further evaluations and confirmation in larger populations. On the other hand, Given the appropriate AUC of the ROC curves, we suggest that miR-138 expression could be included in the diagnosis of the SSc patients along with other criteria or biomarkers.
