Abstract
Objective:
Autism Spectrum Disorder (ASD) is a common neurodevelopmental disorder whose cause remains unknown. Oxidative stress is one of the possible causes of many disorders, including neurological ones. This study aims to measure some oxidative stress biomarkers (Malondialdehyde “MDA,” Advanced Oxidation Protein Product “AOPP,” Glutathione “GSH”) within Syrian children with ASD.
Methods:
MDA, AOPP & GSH were measured in the plasma of a total of 60 children. The ages of the children ranged from 1 to 13 years old. Thirty children had ASD and were compared with 30 controls that don’t have ASD. Fifteen of the controls were siblings of an ASD child, while the remaining 15 had no relations with ASD.
Results:
MDA and AOPP plasma levels were higher in ASD children compared with non-related controls (P = .0001). However, there were no significant differences between MDA and AOPP plasma levels in ASD children in comparison with related controls (P > .05). Alternatively, GSH plasma levels were lower in ASD children compared with both related and non-related controls (P = .0001).
Conclusion:
Further studies are needed to investigate more regarding the diagnostic use of oxidative stress biomarkers, and the therapeutic use of antioxidants in children affected with the autism spectrum disorder.
Introduction
Autism Spectrum Disorder (ASD) is a complex neurodevelopmental disorder characterized by neurological, psychological, and behavioral deficits. The main symptoms of this disorder include a lack of social skills and repetitive stereotyped behavior, which appear after a certain age. 1
Developmental disorders are defined as; severe and chronic disabilities due to mental or physical impairment or both. Such disorders begin during the early developmental period and often last forever; leading to significant limitations in social, occupational, and daily areas. Examples of developmental disorders include ASD, attention deficit hyperactivity disorder (ADHD), intellectual disability, movement disorders, cerebral palsy, Asperger’s disorder, and vision and hearing impairments as well as others. 2
One in 44 (2.3%) 8-year-old children have been identified with ASD according to an analysis of 2018 data, published in CDC’s Morbidity and Mortality Weekly Report (MMWR) Surveillance Summaries in December 2021. This means that there is a 241% increase in autism prevalence since 2000. 3
One potential reason for this significant increase, is that the cause of ASD is still unknown. However, there are several proposals and studies regarding the possible causes of this disorder. One of those theories is focused on the levels of oxidative stress.
Oxidative stress, or oxidoreductive stress, is a state of imbalance between the production of oxidative molecules (prooxidants) and cellular antioxidants in the body, in favor of the oxidants, resulting in several damages. It is not only limited to direct cellular injury due to increased oxidants, but may also interfere with signal transduction and regulation of gene expression by indirect mechanisms. 4
Normal aerobic metabolism generates dangerous reactive intermediates from the reduction of oxygen, the so-called reactive oxygen species (ROS), 5 which include both free radicals and oxygenated non-radical molecules, such as: Superoxide anion radical (O2–•), hydrogen peroxide (H2O2), hydroxyl radical (•OH), singlet oxygen ( 1 O2). 6
ROS, at low to medium levels, are useful in regulating processes and are involved in maintaining homeostasis as well as a wide range of cellular functions. However, high levels of ROS can cause multiple damages, to lipids, proteins, and DNA. . .. 7
The oxidation of these macromolecules results in multiple end products, the concentrations of which can be measured to assess oxidative stress in vivo. 8
Lipid oxidation is often assessed by measuring malondialdehyde (MDA), protein oxidation by measuring advanced oxidation protein products (AOPP), and DNA/RNA oxidation by measuring 8-hydroxy deoxyguanosine in DNA, and 8-hydroxyguanosine in RNA. As for the antioxidant defense in the body, the biggest and most important change is the decrease in the concentration of glutathione (GSH).9-11
The Essa et al study that was conducted in the Sultanate of Oman, found that levels of MDA and protein oxidation (protein carbonyl formation) were significantly higher within autistic patients than in their age-matched controls (P < .0001). 12 While the Yenkoyan et al study 13 did not find a significant difference in AOPP levels between the ASD group and controls with no ASD (P > .05).
In the González-Fraguela et al study 14 GSH levels were lower in the ASD group in comparison with controls with no ASD (P ⩽ .001). Moreover, in their study, Frustaci et al 15 found in their study that ASD patients showed decreased blood levels of reduced glutathione (27%), glutathione peroxidase (18%) and increased concentrations of oxidized glutathione (45%) relative to controls, whereas superoxide dismutase showed no association with ASDs.
This research aims to investigate the relationship between oxidative stress and ASD in Syrian children, after a previous study investigated the role of lead, manganese, and zinc in ASD on Syrian children affected by the Syrian crisis. 16 It will also compare the levels of the oxidative stress biomarkers in the ASD children, with their levels in the siblings with no ASD.
Methods
Participants
This case-control study consisted of 30 children with ASD and 30 children with no ASD identical in age and sex. The control group was sub-divided into 2 groups;
15 children with no ASD, who are related (siblings) to one of ASD children.
15 children with no ASD, without any relationship between them and the ASD children.
Children were diagnosed based on DSM-5 criteria. They were assessed on the Childhood Autism Rating Scale (CARS), 17 a tool that aids in the diagnosis of ASD, completed by specialist doctors, who depended on parent reports and direct observations.
The ages of the participating children ranged between 1 and 13 years. The gender of the participants was predominantly males. Patients were recruited from associations concerned with the rehabilitation of children with neurodevelopmental disorders. All subjects completed a questionnaire included: age of diagnosis, autism rating scale, maternal and paternal age at contraception, consanguineous marriage, premature birth, smoking and alcohol intake during pregnancy, history of mental illness in the family of the participants, and the presence of chronic diseases in mother or child. The protocol of the study was approved by the Ethics Committee at Damascus University (No. 471 on May 12/2021). Moreover, written informed consent was obtained from the parents of the examined children before joining the study. Blood sampling was also performed in the presence of parents and with their consent. The duration of the study, from finding the population to be studied to finalizing the assays, was from September 20/2021 to November 22/2021.
Inclusion criteria
Children with ASD: Based on a specialist doctor’s diagnosis.
Children under 13 years old.
Exclusion criteria
Children over 13 years old.
Children with cardiovascular, kidney or liver diseases.
Children with diabetes.
Children who have suffered head injuries.
All of these are determined based on a specialist doctor’s diagnosis.
Collection of blood samples
Five milliliters of venous blood samples were collected from the cubital vein, using lithium heparin tubes. The blood samples were then centrifuged and plasma was collected and divided into 3 parts, one for each biomarker. For MDA assay, butylated hydroxytoluene (BHT) was added to the plasma “3µmol/1 mL plasma” immediately after centrifuging and before the beginning of the assay process, the samples were stored after centrifuging at −20°C.
Estimation of malondialdehyde (MDA) in plasma
Malondialdehyde levels were analyzed using MDA Kit from BioVision Inc. USA, using Colorimetric Detection according to the manufacturer’s instructions. The principle is based on the reaction between a specific chromogenic reagent and MDA at 45°C. The assay yields a strong, yet stable colorimetric signal (OD 586 nm) that is proportional to the amount of MDA in samples.
Estimation of advanced oxidation protein products (AOPP) in plasma
AOPP levels were analyzed using AOPP Kit from Cell Biolabs Inc. USA, using Colorimetric Detection according to the manufacturer’s instructions. The unknown AOPP-containing samples or Chloramine standards are first mixed with an assay reaction initiator that begins a color development process. After a brief incubation, a stop solution is added and the samples and standards can be read with a standard colorimetric plate reader. The AOPP content in unknown samples is determined by comparison with the predetermined Chloramine standard curve. The absorbance of each well was read immediately on a spectrophotometer microplate reader using 340 nm as the primary wavelength, and the (0 μM) Chloramine standard as an absorbance blank.
Estimation of glutathione (GSH) in plasma
Glutathione levels were analyzed using GSH Kit from BioVision Inc. USA, using Colorimetric Detection according to the manufacturer’s instructions. The process is based on an enzymatic cycling method in the presence of GSH and a chromophore. The reduction of the chromophore produces a stable product, which can be followed kinetically at 450 nm. Therefore, its absorbance is directly proportional to the amount of GSH in the sample. The assay was performed using a spectrophotometer microplate reader, with 450 nm as the wavelength to measure the absorbance.
Statistical analysis
The Statistical Package for Social Sciences (SPSS) “26th version” was used to process the data.
Shapiro-Wilk test was used to see the normality of the distribution. Kruskal-Wallis test was used to compare MDA/AOPP/GSH levels between the 3 groups (ASD children, related and non-related controls), then all the pairwise between the study groups were compared. The Spearman correlation coefficient test was conducted to study the relationship between the levels of the 3 biomarkers (MDA, AOPP, and GSH) and the rating scale of ASD (mild, moderate, severe) in the ASD group, also it was used to study the relationship between (consanguineous marriage, Premature birth) and the occurrence of ASD. A par value of .05 or lower was assumed significant.
Results
The sociodemographic data
Table 1 shows the characteristics of the study population, and Table 2 shows the age ranges in the study population.
The characteristics of the study population.

Age ranges (in years) in the study population.

When statistical comparison test was done, the P-value was more than .05, which means there is no significant differences between the children ages and mothers ages in the study groups.
MDA levels in the study groups
The levels of MDA in the study groups are presented in Figure 1. MDA was elevated in ASD and related control with no significant difference between these 2 groups (P > .05), MDA levels were the lowest in non-related control, with a significant difference between this group and each of the other 2 groups (P = .0001).
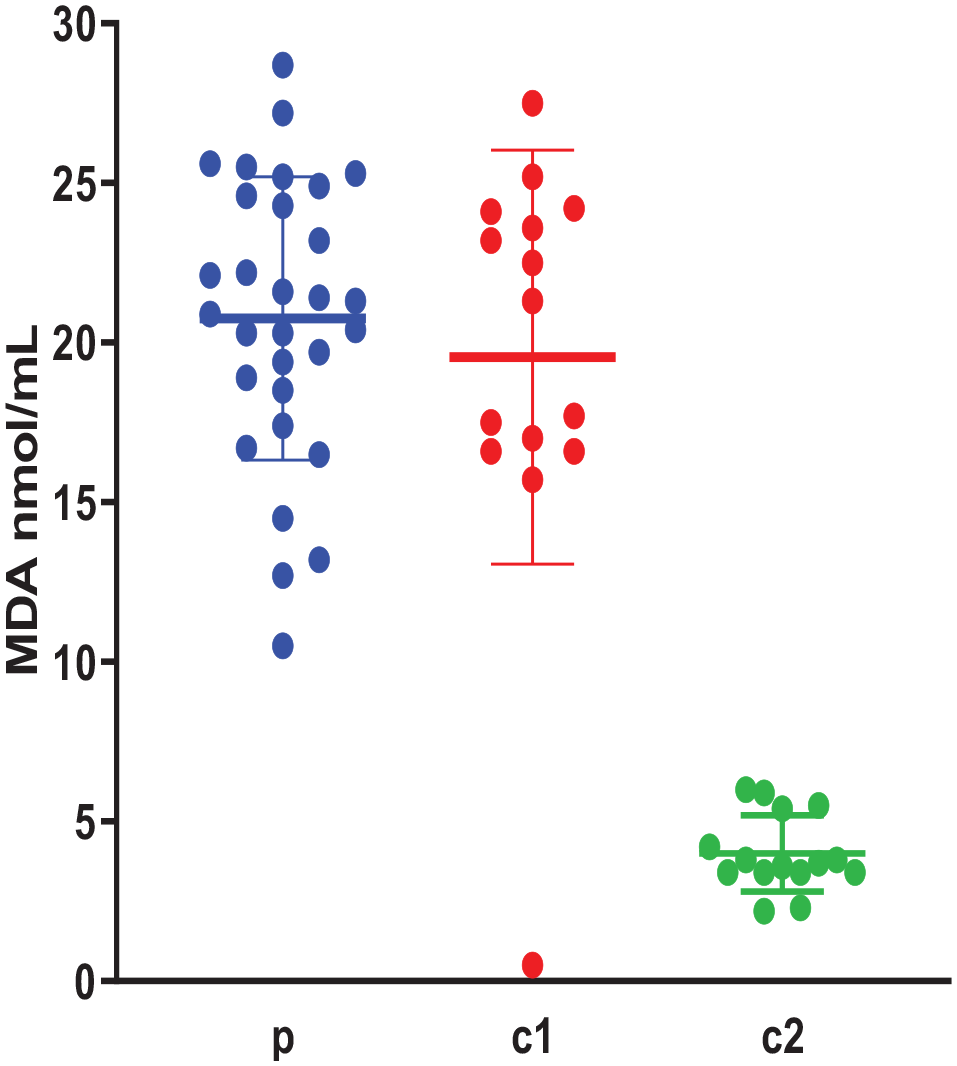
Plasma MDA levels (nmol/mL) in the study groups (n = 60); p: ASD children, c1: Siblings controls, c2: Non-related controls. Data are expressed as mean of 3 different experiments ±SEM. Kruskal-Wallis with post hoc test indicated no significant difference between the groups: p and c1 (P > .05) and significant differences between the groups: p and c2, c1 and c2 (P = .0001).
AOPP levels in the Study Groups
The levels of AOPP in the study groups are presented in Figure 2. AOPP was elevated in ASD and related control with no significant difference between these 2 groups (P > .05), AOPP levels were the lowest in non-related control, with a significant difference between this group and each of the other 2 groups (P < .05).
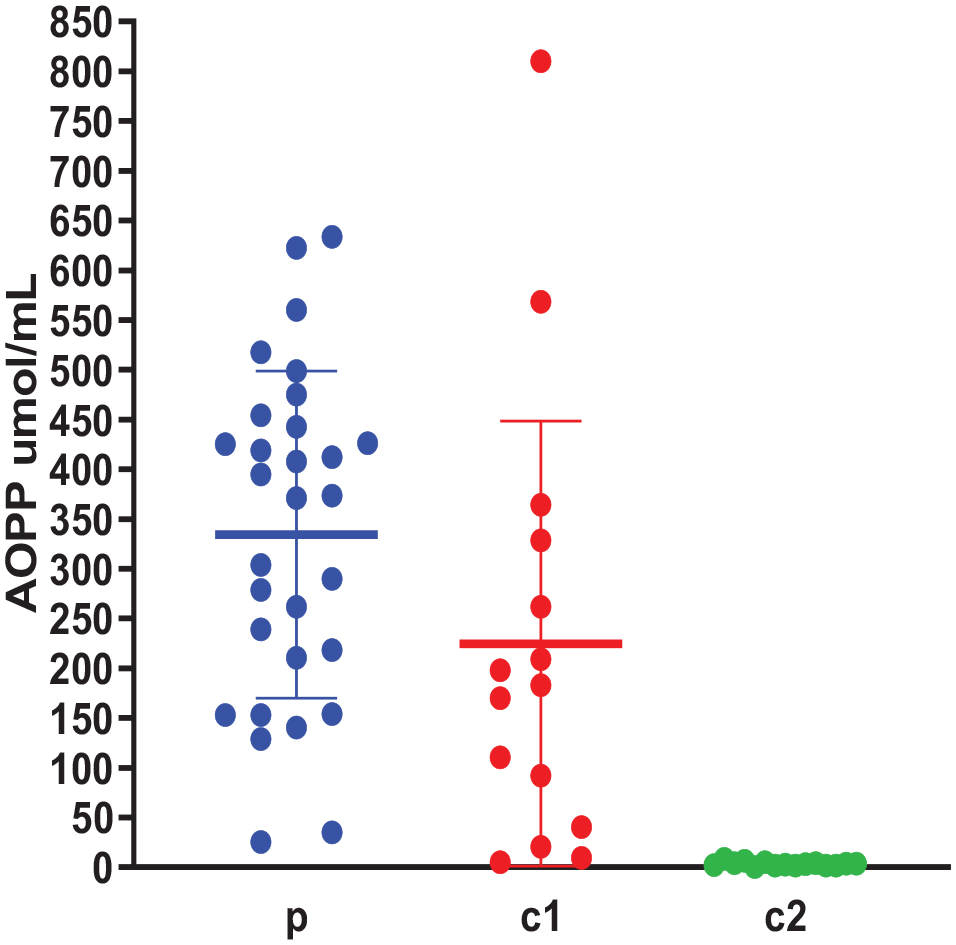
Plasma AOPP levels (µmol/mL) in the study groups (n = 60); p: ASD children, c1: Siblings controls, c2: Non-related controls. Data are expressed as mean of 3 different experiments ±SEM. Kruskal-Wallis with post hoc test indicated no significant difference between the groups: p and c1 (P > .05) and significant differences between the groups: p and c2, c1, and c2 (P = .0001).
GSH levels in the Study Groups
The levels of GSH in the study groups are presented in Figure 3. GSH was lower in ASD when compared with controls, with a significant difference between the ASD group and each of the other 2 controls (P < .05). GSH levels were higher in the controls (related and non-related) with no significant difference between these 2 groups (P > .05).
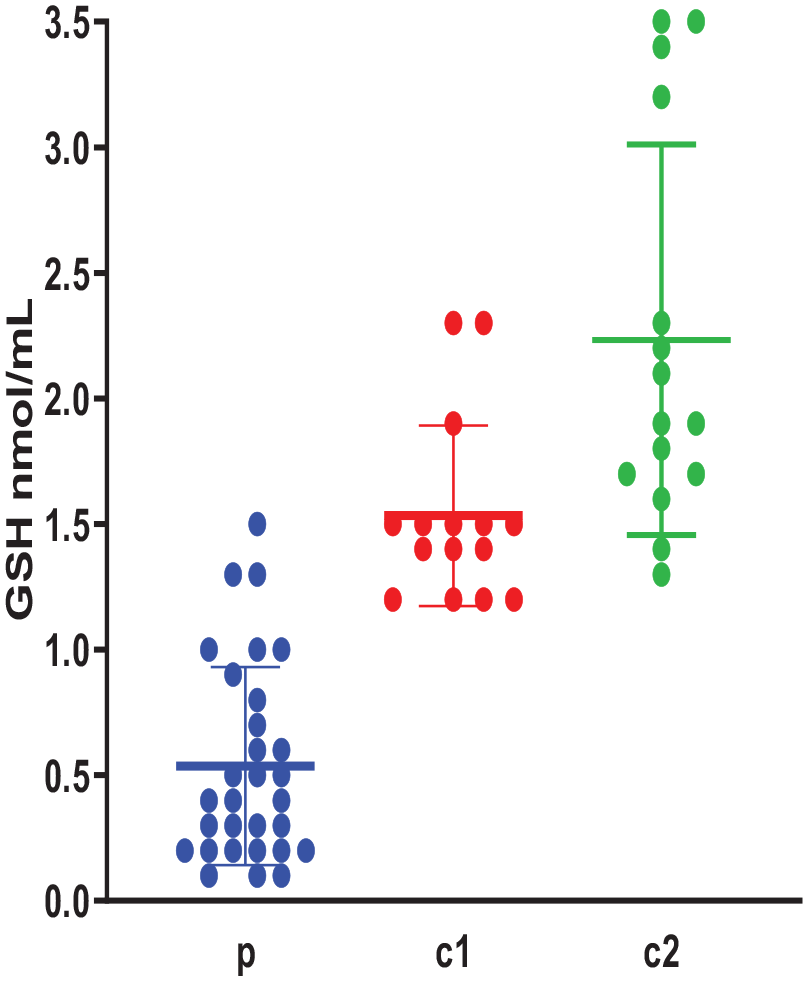
Plasma GSH levels (nmol/mL) in the study groups (n = 60); p: ASD children, c1: Siblings controls, c2: Non-related controls. Data are expressed as mean of 3 different experiments ±SEM. Kruskal-Wallis with post hoc test indicated significant differences between the groups: p and c1, p and c2 (P = .0001) and no significant difference between the groups: c1 and c2 (P > .05).
Correlation between MDA, AOPP & GSH levels and the rating scale of ASD
Based on the Spearman correlation coefficient test, shown in Table 3, it was found that AOPP and GSH levels are correlated with the severity of ASD (P < .05). While MDA levels did not correlate with the severity of ASD (P > .05).
Correlation between MDA, AOPP & GSH levels and the rating scale of ASD.

Relation between (consanguineous marriage, premature birth) and occurrence of ASD
Based on the statistical analysis, it was found that each of these 2 population characteristics (consanguineous marriage, premature birth) are not correlated with the occurrence of ASD (P > .05).
Discussion
Our study determined the levels of 3 biomarkers in ASD children and compared it with their siblings that do not have ASD, in addition to the non-related controls that also don’t have ASD themselves. Our study demonstrated that there were no significant differences in MDA and AOPP levels between ASD children and their siblings with no ASD (P > .05), hypothesizing that the vulnerability to oxidative stress is related to kinship, in addition to the exposure to environmental factors.
However, higher MDA and AOPP levels were observed in children with ASD in comparison with non-related control (P = .0001), these results were similar to the results of Meguid et al, 18 Omotosho et al, 19 and Altun et al 20 studies, which found significantly higher levels of MDA in children with ASD when compared with control with no ASD (P < .0001). Furthermore, our findings corresponded with the results of Ahmad et al study, 21 which found higher levels of AOPP (+120.6%) in children with autism spectrum disorder when compared with the control (P ⩽ .05), denoting the relation between oxidative stress and ASD. However, from our results, it is clear that this relation is not a causal one, considering the elevated levels of these oxidative stress biomarkers in the sibling controls with no ASD.
MDA is not only a common indicator of oxidative stress, it has toxic effects on different types of cells, including red blood cells, endothelial cells, fibroblasts, and hippocampal neurons. 22 Therefore, MDA may interfere with the pathophysiology of autism spectrum disorder, as it is a highly neurotoxic compound that has a much longer life in the body than reactive oxygen species. It impairs brain function and causes cell death by 2 mechanisms: apoptosis and necrosis. 23 It also causes damage to cortical neurons; By cross-linking proteins, and depolarizing the mitochondrial membrane, carbonyl stress leads to neuronal damage by stimulating Calcium influx, calcium overload, generating reactive oxygen species (ROS), as well as, activating the programed cell death signaling pathway. Therefore, its increased accumulation within cells may contribute to the occurrence of autism spectrum disorder, as well as various neurodegenerative diseases. 22
Despite the high MDA levels in the siblings with no ASD in our study, it is possible that it did not reach the toxic concentration responsible for causing neuronal damage to cells. As some studies have found that the toxic effects of MDA depend on concentration and time in experimental animals, 22 which may also be true in humans.
Another biomarker of lipid oxidation is 8-isoprostane, which forms as a result of non-enzymatic oxidation of arachidonic acid in phospholipids by free radicals, and it was measured in biological fluids of autistic children by El-Ansary and Al-Ayadhi, 24 Ming et al, 25 and Qasem et al 26 who found higher levels of 8-isoprostane than the normal range in ASD groups, which confirm the hypothesis of higher exposure to oxidative stress in autistic children.
During oxidative stress, proteins are major targets of ROS, due to their overall abundance in biological systems. Oxidation of proteins results in modification of their structure and function, and these modified proteins are often irreparable. This results in a lot of damage, because proteins are primarily responsible for most of the functional processes inside cells, and this is one of the reasons why oxidative stress plays a role in many acute and chronic diseases. 27
AOPP is a group of carbonylated protein products formed by the reaction of proteins with chlorinated oxidants, such as HOCl and chloramine, during oxidative stress. 28
AOPP may contribute to autism spectrum disorder by activating monocytes and providing proinflammatory cytokine production. Also, acute inflammation that occurs during severe autism spectrum disorder may also accelerate AOPP formation. This may explain why higher levels of AOPP were observed in children with a severe degree of autism spectrum disorder compared to the group of children with mild and moderate autism and the controls (P < .05) in Ahmad et al study 21 and in our study, which found that there was a significant correlation between the levels of AOPP and the severity of autism spectrum disorder if it was mild, moderate or severe (P < .05).
However, Yenkoyan et al study did not correspond with the results of our study, as it did not observe an increase in AOPP levels in children with autism spectrum disorder compared to the control (P > .05). 13 One of the reasons for this may be the small sample size in the Yenkoyan et al study (N = 10), which could affect the interpretation and efficacy of the results.
Aside from AOPP, there is another protein oxidation biomarker called Dityrosine (DT). DT is a tyrosine dimer and a marker of tyrosine residue oxidation. It has been proposed to be a good indicator of protein oxidation in the Osredkar et al 10 and Anwar et al 29 studies which found dityrosine to be an important predictor of ASD. Similarly, our study also found ASD diagnosis to be associated with higher levels of dityrosine expression. Another study by Anwar et al 30 also found higher levels of DT (88%) in ASD children but no changes regarding 3-nitrotyrosine—a marker of tyrosine nitration when compared with controls with no ASD, suggesting only selected oxidative damage and no increase in protein nitration is present in ASD cases.
In this study, low GSH levels were observed in children with autism spectrum disorder compared to both control groups (related and non-related controls) (P = .0001).
When compared with other organs, the brain is particularly vulnerable to oxidative stress for several reasons: (1) Human brain cells use 20% of the oxygen consumed by the body, and they make up only 2% of the total body weight, which indicates that the brain generates many more free radicals than other tissues. (2) Different areas of the brain contain high concentrations of iron, which can stimulate the generation of free radicals. (3) The brain is rich in fats with unsaturated fatty acids, which are targets for oxidation. (4) The brain has low to moderate protective systems (antioxidants) compared to the kidneys or liver. 31
Recent studies conducted on children with ASD showed that an imbalance between oxidants and antioxidants may contribute to this disorder. Especially since the central nervous system is more sensitive to oxidative stress as mentioned, and because of the selectivity of the blood-brain barrier, which reduces the spread of some antioxidants such as vitamin E (alpha-tocopherol) in the brain. 32
One study about the predictive value of some oxidative stress biomarkers in children with ASD by El-Ansary et al found that lipid peroxide is a good marker, and correlated well with ASD occurrence. Whereas the predictive value of some antioxidants in ASD cases was categorized into worthless markers (catalase, reduced and total vitamin C), fare markers (SOD and oxidized glutathione), good markers (Glutathione peroxidase, GSH) and an excellent marker (vitamin E). 33
Glutathione is one of the most important intracellular defense systems against oxidation and redox processes, Moreover, there is evidence that glutathione itself is a neurotransmitter, and that any change in glutathione levels or its oxidation state will negatively affect the activity of the central nervous system; contributing to many neurodegenerative disorders, including ASD.21,34 This was supported by the finding which showed that the increase in oxidative stress in autism spectrum disorder, was accompanied by a decrease in the function of antioxidants in addition to a decrease in the levels of glutathione 34 which corresponded with the results of our study.
Similar to our study, Chen et al study indicated that the ASD patients showed decreased blood levels of GSH and GSH/GSSG ratio, and increased concentrations of oxidized glutathione (GSSG) relative to control with no ASD. 35 The results of Al-Gadani et al 36 and Adams et al 37 studies, along with our study, demonstrate clear evidence that low levels of antioxidants such as glutathione may be a risk factor and may promote the development of the autism spectrum disorder.
Despite the decrease in glutathione levels in the related control in our study, this decrease was relatively small. When analyzed and compared with the non-related control there was no significant difference (P > .05), however, when compared with the results of the ASD group, a significant difference was observed (P = .0001). This may be one of the reasons that led to the protection of the siblings’ group from oxidative damage to neurons compared to their siblings who have autism spectrum disorder. Which can indicate an important role of glutathione in preventing ASD occurrence.
Double-blind, placebo-controlled trials of potent antioxidants—vitamin C or carnosine—significantly improved autistic behavior. Benefits from these and other nutritional interventions may be due to the reduction of oxidative stress. Understanding the role of oxidative stress may help illuminate the pathophysiology of autism, its environmental and genetic influences, and offer new treatments, and potential preventive measures. 38
Conclusion
This study further indicates the existence of the relationship between oxidative stress and children with autism spectrum disorder. It indicates the possibility that this disorder may be occurring as a result of the imbalance between the oxidants and antioxidants in the body. In addition, this study hypothesizes about the protective role of reduced glutathione (GSH). Furthermore, this study encourages the use of antioxidants as one of the therapeutic strategies for ASD treatment. However, it was found that MDA and AOPP biomarkers cannot be used to diagnose autism spectrum disorder, as this requires more accurate and specific biomarkers.
Strength of this study
This is the first study of its kind in Syria, attempting to study the association between oxidative stress and autism spectrum disorder, by assessing 3 important biomarkers (MDA, AOPP and GSH). Also, it is the first to compare results from ASD children with their siblings with no ASD, demonstrating that the increase in the oxidation is not a direct cause of ASD, but rather it is the decrease in the antioxidants. Finally, this study points to the role of glutathione in the protection of these siblings from the occurrence of ASD.
Limitation of the study
A small sample size was a drawback in this study, but logistical and financial factors limited the ability to reach more autistic children.
Footnotes
Acknowledgements
We are grateful to the ASD associations in Damascus for their cooperation.
