Abstract
The hepatocyte growth factor (HGF)/c-met axis plays a crucial role in cancer development by promoting cellular proliferation, motility, and morphogenesis, as well as angiogenesis. Different cellular distributions of both the ligand and the receptor in benign vs malignant lesions indicate this biological system as a candidate for a diagnostic biomarker of malignancy occurring in endocrine glands, such as the thyroid and pituitary. Furthermore, the HGF/c-met expression may help to identify a subset of patients eligible for potential targeted therapies with HGF/c-met inhibitors or antagonists in thyroid tumour, as well as in other malignancies. This may be relevant for iodine-refractory cancers, the treatment of which is still a major challenge. With this in mind, HGF/c-met expression in thyroid cancer tissue may be useful for prognostic and therapeutic stratification of patients.
Introduction
Differentiated thyroid carcinoma (DTC) represents the most frequent endocrine cancer, and its incidence has been increasing in the past few decades. According to the epidemiological data of Surveillance, Epidemiology, and End Results (SEER), the annual incidence of thyroid cancer has increased from 5.9 to 17.3 cases per 100 000 individuals from 2005 through 2009. 1 Incidence in 2012 in the United States rose to 15 cases per 100 000 individuals per year, and it has been estimated that by 2019 it will increase to 23.8 cases per 100 000 individuals per year. 2 However, the mortality rate of thyroid cancer has remained stable over time, confirming the overall favourable long-term prognosis of this cancer. 1 Also, the epidemiologic distribution of thyroid cancer histotypes has changed over the past 20 years.3,4 The percentage of aggressive follicular and undifferentiated carcinomas has drastically reduced in favour of the papillary counterpart, often diagnosed at less than 1 cm in maximum diameter (the so-called microcarcinoma), whereas noninvasive follicular neoplasms have recently been declassified from thyroid cancers.3,5 Also, the clinical presentation of DTC has been changing, evolving towards an increasing number of cancers being diagnosed at early stage or serendipitously, which is associated with less aggressive behaviour and better prognosis.6,7 Less than 20% of patients present with advanced or aggressive cancers, with recurrent and/or metastatic disease that significantly impairs survival.8,9 Finally, about 5% of patients with clinical DTC develop iodine-refractory cancer, which is associated with a survival rate lower than 10% at 10 years. 10 Given these observations, progress in the management of DTC may be achieved by translation of molecular findings in thyroid cancer to the clinical setting. First, recognizing the molecular characteristics that are associated with a more aggressive behaviour of the tumour and a poorer outcome may help to identify patients at higher risk of recurrence and/or distant metastases. Second, identification of proliferative, antiapoptotic, or angiogenic pathways, which are activated in metastatic and/or refractory cancers, may provide the basis for molecular-targeted therapies. In this light, we reviewed data on hepatocyte growth factor (HGF)/c-met axis expression/activation in thyroid disease and on its possible usefulness as a prognostic and therapeutic biomarker in thyroid cancer.
Data Source
The terms ‘HGF’, ‘c-met’, and ‘Thyroid disease, Thyroid nodule(s), Thyroid carcinoma’ were used both separately and in reciprocal conjunction to search MEDLINE for articles published from January 1996 up to December 2016.
Current State of Knowledge: Identified Biology of HGF/c-met
Since HGF was isolated in 1983 as a serum factor appearing after partial hepatectomy, several effects on cellular growth, motility, and morphogenesis have been reported. 11 Mainly, HGF protein consists of a heterodimeric structure including 2 chains, 69-kDa α (heavy chain) and 34-kDa β (light chain), by which it can bind to the cognate receptor, namely, c-met.11,12 This is a proto-oncogene product belonging to the tyrosine-kinase receptor family that, upon binding of the specific ligand and activation of the tyrosine-kinase domain, provides multiple binding sites for SH2 molecules through autophosphorylation of Tyr 1349 and Tyr 1356. 13 These molecules act as intracellular transducers by recruiting and activating several effectors, including phosphatidylinositol 3-kinase (PI3-kinase), rat sarcoma (Ras), adaptors GRB2 and SHC, the docking protein Gab1, and STAT3.14-18 Through HGF binding to c-met, specific intracellular effectors are activated to promote different cellular responses, such as proliferation, cell scattering, and morphogenesis.19,20 For instance, the scattering effect of HGF/c-met is mediated by PI3-kinase effectors, whereas the proliferative action is mediated by GRB2 and Ras, 15 and the morphogenetic action is mediated by STAT3, a member of the signal transducers and activators of the transcription family18,21,22 (Figure 1).
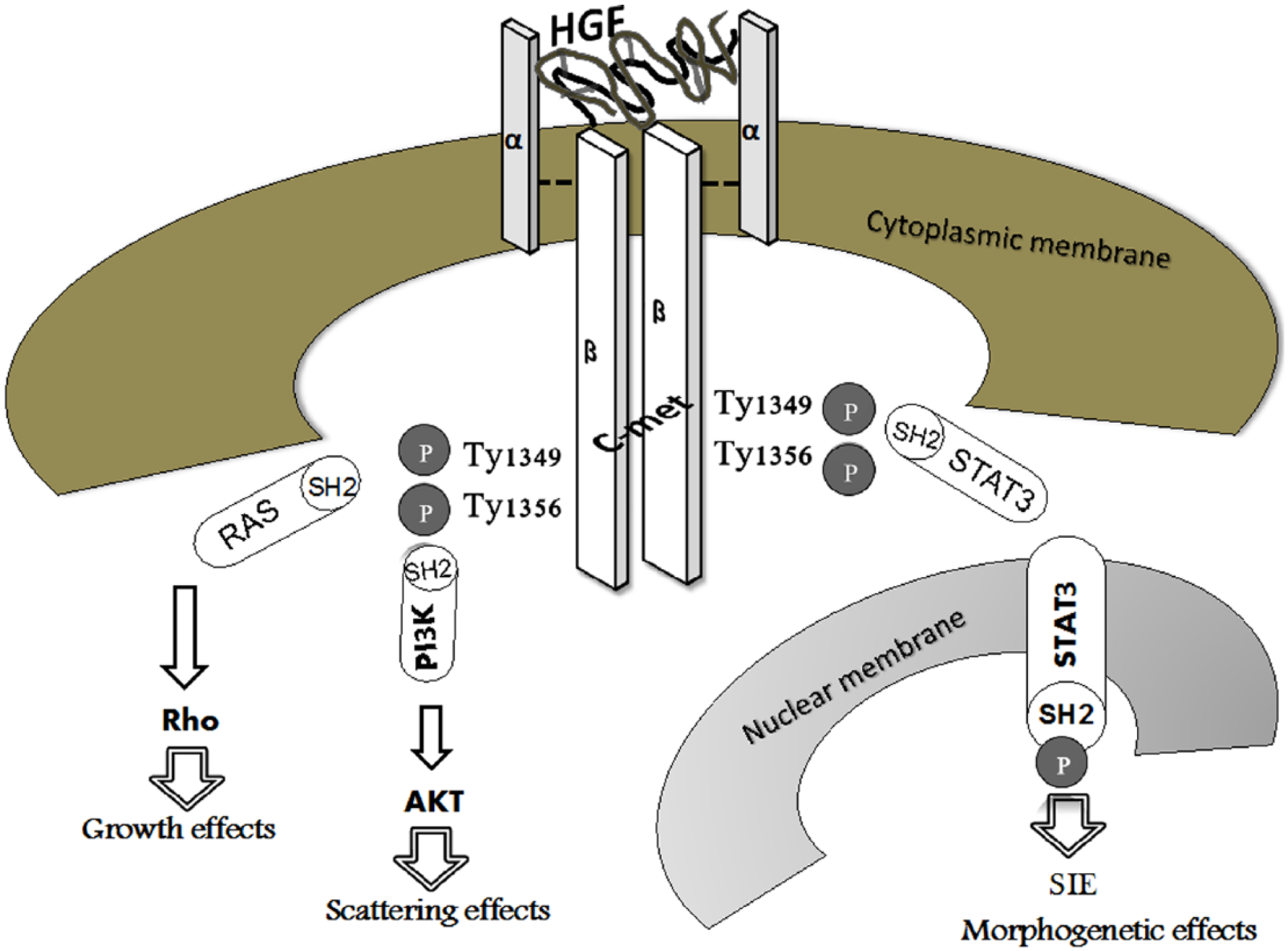
HGF/c-met axis. HGF/c-met interaction leads to c-met dimerization as well as phosphorylation of 2 tyrosine residues, Tyr 1349 and Tyr1356, both located in the C-terminal tail and responsible for docking sites for multiple substrates showing SH2 domain. Through SH2 domain, Ras, PI3K, and STAT3 effectors are triggered to induce cellular growth through Rho activation, scattering effect by AKT recruitment, and morphogenetic response by migration of STAT3 to nucleus and subsequent to its binding to SIE. AKT indicates protein kinase B; HGF, hepatocyte growth factor; met, mesenchymal epithelial transition factor; PI3K, phosphatidyl inositol-4,5-bisphosphate 3-kinase; Ras, rat sarcoma; Rho, rhodopsin; SH2, Src homology 2; SIE, sis-inducible element; STAT3, signal transducer and activator of transcription 3.
The ligand HGF and the receptor c-met share 2 distinctive characteristics. First, both genes lie on the long arm of chromosome 7, at loci q21.1 and q31.3, respectively. These genetic regions show high susceptibility to loss of heterozygosity (LOH) occurring in microsatellites flanking the HGF gene, namely, D7S660, D7S630, D7S492, D7S689, and D7S657, as well as in D7S486 and D7S465 microsatellites which are located near the c-met gene region. 23 Second, in normal development, ligand and receptor are expressed by cell lines of different embryological origins: HGF is secreted from mesenchymal cells, whereas c-met is expressed on the cellular membranes of epithelial cells.21,24,25 Interestingly, in malignant proliferation, HGF has been detected also in tumour epithelial cells along with its receptor, raising the possibility of c-met activation through an autocrine loop.21,24,25
Based on cellular distribution, tissue expression of members of HGF/c-met axis has been proposed to serve as diagnostic biomarkers of proliferations occurring in the thyroid, whereas serum HGF levels have been associated with benign growth of goitrous nodules also occurring in the context of Hashimoto’s thyroiditis (HT).21,25,26 It is also conceivable that inflammatory cells (ie, macrophages) infiltrating the thyroid gland may represent a mesenchymal source of HGF production, thus contributing to aberrant cell growth in HT.
Similar to other tumours,27-29 the cornerstone evidence supporting HGF and c-met utility in the diagnosis of thyroid malignant lesions is built on evidence of specific cellular localizations for each member of the HGF/c-met axis. When thyroid benign lesions shift towards malignancy, the HGF/c-met cellular disposition is dysregulated to the extent that abnormal epithelial cells can simultaneously express the ligand and the receptor.21,24 Among thyroid cancers, papillary thyroid carcinoma (PTC) is associated with marked overexpression (up to 100-fold) of HGF/c-met, which, instead, is rarely expressed in other histotypes, such as follicular, anaplastic, and/or medullary thyroid cancer (Figure 2). Overexpression of HGF/c-met has been found in 75% to 100% of PTC, regardless of the histological variants30-33 (Table 1). Our group has also correlated the expression of HGF/c-met with that of STAT3, which is known to mediate morphogenetic effects, suggesting that this autocrine pathway may be relevant for the establishment of the papillary phenotype. 34

Immunoreactions of hepatocyte growth factor (HGF) in cases of benign colloid goitre and papillary thyroid carcinoma (PTC). (A) Unstained HGF follicular thyroid cells (grey arrow) and HGF stain located on membrane and cytoplasm of stromal cells (black arrow) surrounding thyrocytes, respectively (original magnification ×400). (B) HGF cytoplasmic and membranous immunostaining in PTC follicular cells (black arrow) and unstained HGF stromal cells (grey arrow), respectively (original magnification ×400).
HGF/c-met expression in benign and malignant thyroid lesions, as well as in normal thyroid tissue.

Abbreviations: ATC, anaplastic thyroid cancer; FA, follicular adenoma; FC, follicular carcinoma; FTC, follicular thyroid cancer; GD, Graves’ disease; HGF, hepatocyte growth factor; HT, Hashimoto’s thyroiditis; OA, oncocytic adenoma; PTC, papillary thyroid carcinoma.
In addition, HGF/c-met expression by tumour cells has been related to metastatic spread and a worse prognosis. Besides its antiapoptotic and proliferative effects, HGF/c-met promotes cellular motility and invasion (hence the name scatter factor). Thus, its expression may distinguish more advanced and aggressive cancers at higher risk of metastatic dissemination.32,35,37,38
From these phenomena, 2 consequences of HGF/c-met mechanisms of action have been noted: first, in noncancerous growth, HGF appears as a mesenchymal cytokine adopting paracrine loops to bind c-met localized in adjacent epithelial cells. Conversely, in malignant nodules, gain of HGF expression on epithelial cells indicates an improvement in the ability of HGF to use autocrine mechanisms.21,24 Some data reporting LOH occurring in HGF and the c-met gene region have recorded HGF epithelial expressions in PTC which were never associated with loss of genetic material for microsatellite markers pertaining to HGF or c-met.22,23 Furthermore, LOH in HGF and c-met gene regions has been observed in follicular and anaplastic thyroid carcinomas only, both failing to express the HGF/c-met axis. 23 These features together strongly imply an HGF involvement in the aberrant epithelial growth of PTC.
Future Perspective: Ongoing Therapeutic Strategies
As a result of the cellular responses induced through HGF/c-met axis in cancer tissues, both HGF and c-met represent an attractive target for personalized antitumour strategies. Recently, there has been much interest in the prognostic and therapeutic implications of the expression of members of the HGF/c-met axis by several epithelial and nonepithelial malignancies, arising from both endocrine and nonendocrine tissues.27,28,39-43 The identification of biomarkers of response will lead to more effective targeting of this pathway for cancer therapy. Therefore, if the HGF/c-met axis is to be used as a target for treatment, it will be important to identify predictive biomarkers to select those patients likely to benefit. Potential predictive biomarkers for anti-HGF/c-met–targeted therapies should include the immunohistochemical expression of molecules such as c-met, phospho-c-met, HGF, and downstream signalling proteins (RAS-MAPK, PI3K-AKT, and STAT3, to name a few). These putative predictive biomarkers should be evaluated for therapeutic stratification of patients. 39 Immunohistochemical profiles of the HGF/c-met pathways depict several cancers, including PTC and colon carcinomas, as well as pituitary adenomas, which may benefit from these novel therapeutic agents.21,22,33,44
Several drugs targeting HGF/c-met have been developed and are now being tested in clinical trials with encouraging results (Table 2). These include anti-met and anti-HGF monoclonal antibodies and antagonists of biological HGF,40,41,42,43 to be used alone or in combination with tyrosine-kinase inhibitors. 45 Among c-met inhibitors, few agents have proved to be capable of cutting off the HGF signal. Mainly, the non–adenosine triphosphate competitive c-met inhibitor, tivantinib (ARQ 197), when combined with erlotinib, has shown an appreciable response rate in phase 3 study of patients with locally advanced or metastatic nonsquamous non–small cell lung cancer.42,46,47
Clinical trials adopting HGF antagonist or anti–c-met drug agents in cancer therapy.
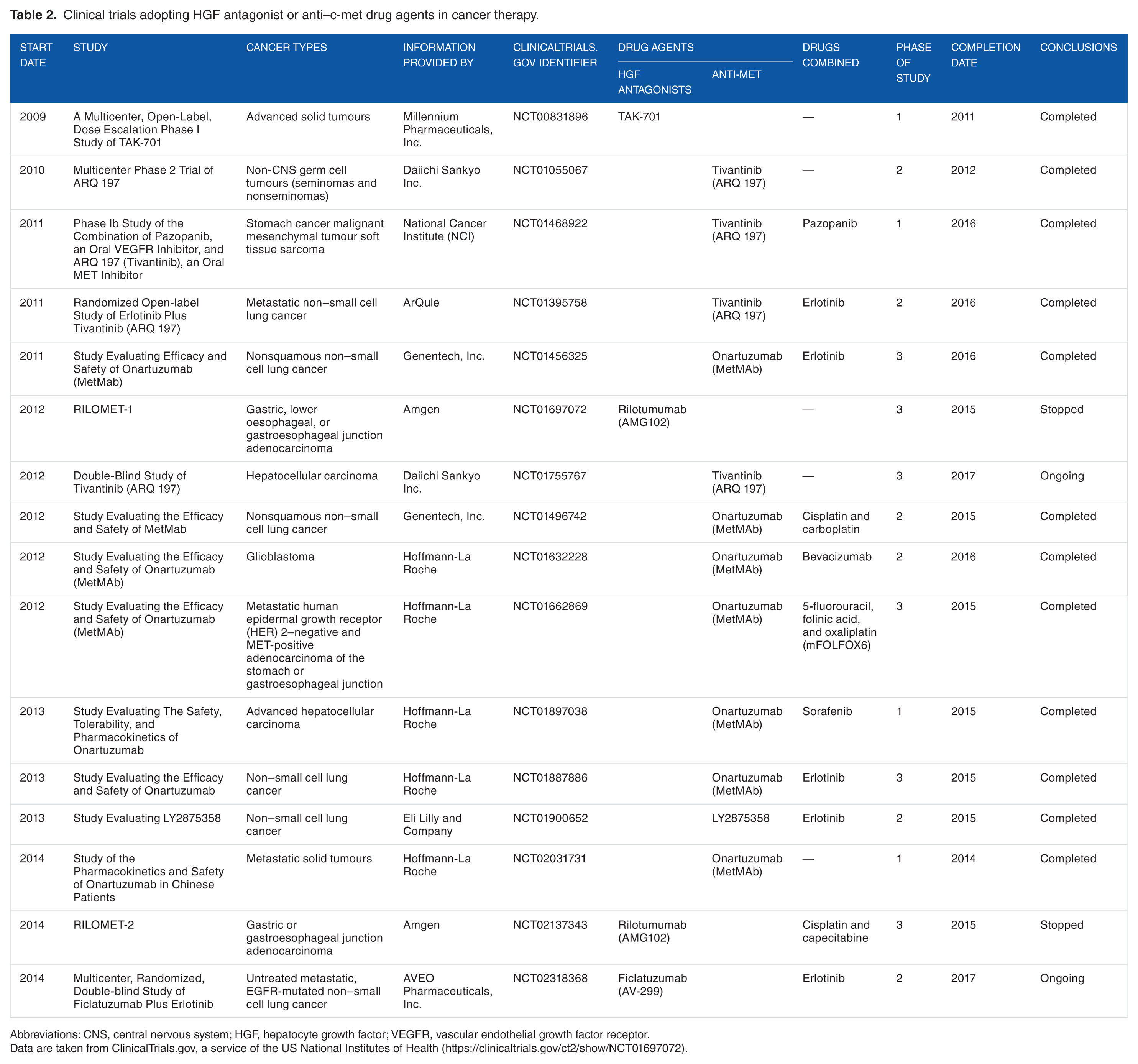
Abbreviations: CNS, central nervous system; HGF, hepatocyte growth factor; VEGFR, vascular endothelial growth factor receptor.
Data are taken from ClinicalTrials.gov, a service of the US National Institutes of Health (https://clinicaltrials.gov/ct2/show/NCT01697072).
Rilotumumab (AMG102) has been reported as the humanized anti-HGF monoclonal antibody capable of blocking HGF/c-met signals by preventing HGF binding to c-met. 41 In vitro, there was evidence demonstrating the inhibition of the HGF signal through rilotumumab-induced apoptosis in cell lines of gastric carcinomas expressing c-met. 28 On these promising data, 3 pivotal global phase 3 trials, namely, RILOMET-1, RILOMET-2, and MetGastric, were designed on the response of advanced/metastatic gastroesophageal cancer to rilotumumab and to onartuzumab, a humanized monovalent antibody to c-met (c-MetMAb) that neutralizes c-met by inhibiting HGF binding and receptor dimerization.41,42 However, data from RILOMET-1 showed no valid therapeutic efficiency of rilotumumab in terms of overall survival and progression-free survival (PFS), and no subgroups of patients seemed to benefit with rilotumumab, including those with higher percentages of cells expressing c-met.42,43 Both studies, RILOMET-1 and RILOMET-2, were stopped. Also, preliminary results of the MetGastric trial showed that the addition of onartuzumab did not improve PFS in the unselected population or in the MET-positive subgroup.42,43 Further study is necessary to determine the combination of targeted therapy that will translate into improved survival.
Finally, an emerging model of cancer plasticity resulting in a phenotype switch from an epithelial to a mesenchymal cellular state may further support the development of new drug targets. Either HGF is released from the surrounding stromal cells, resulting in a constitutive paracrine c-met activation, or coexpression of HGF and c-met leads to autocrine activation, targeting the HGF/c-met pathway may disrupt the interactions between tumour cells and their microenvironment, significantly increasing cancer treatment efficacy.
Conclusions
In this review, we have evaluated the present knowledge on HGF/c-met axis expression in thyroid disease with a special point of reference to the impelling demands to look for markers of thyroid cancer that would also serve as therapeutic targets.
Similar to other cancers, HGF/c-met signal expression and cellular distribution in thyroid tumours may help to identify patients eligible for potential targeted therapies with HGF/c-met inhibitors or antagonists. This may be relevant for iodine-refractory cancers, whose treatment is still a challenge.50,51 Moreover, HGF/c-met expression may be used for diagnostic and prognostic applications in the context of other well-accepted clinic-pathological prognostic parameters for DTC (age, gender, pTNM stage, histological subtype, and malignant nodule topography) because very few new markers have revealed prognostic value per se.52-56
Footnotes
Author Contributions
MT wrote the first draft of the manuscript. RMR contributed to the writing of the manuscript. MT, AC, SG, MS and RMR agree with manuscript results and conclusions. MT and RMR jointly developed the structure and arguments for the paper. SG revised English language. MS and AC made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totalled 1662 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures and Ethics
The authors have provided to the publisher with signed con-firmation of compliance with legal and ethical obligations including, but not limited to, the following: authorship and contributorship, conflicts of interest, privacy, and confidentiality. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. The authors have nothing to disclose.
