Abstract
Tunneled central venous access devices (T-CVADs) are used in selected neonatal cases, and their placement often requires a peel-away introducer. In premature infants and neonates, when the introducer is significantly larger than the catheter, the procedure may be technically challenging because of the small size and fragility of the veins. This study describes a modified technique for T-CVAD placement in neonates weighing less than 5 kg. The proposed approach combines a Seldinger over-the-wire insertion with retrograde tunneling and proximal trimming, with the aim of avoiding the use of a peel-away introducer and simplifying catheter placement. Eight neonates underwent T-CVAD placement using this technique. No procedure-related adverse events or early catheter migration were observed. Most catheters (7/8) were removed at the end of the prescribed therapies, while one was removed due to late tip migration. Overall, the Seldinger over-the-wire insertion appeared feasible, safe, and effective in this small case series. The combination of proximal trimming and retrograde tunneling allowed accurate catheter length adjustment, reduced the risk of dislocation, and preserved the catheter’s tapered tip, without observed procedure-related complications.
Keywords
Introduction
Central venous access devices (CVADs) have become essential tools for the management of critically ill patients, with tunneled non cuffed centrally inserted central catheter (Tnc-CICCs) being particularly indicated for patients at high risk of CVAD-related infections, such as those admitted to neonatal or pediatric intensive care units (NICUs or PICUs). While these devices are widely used in older patients, their application in neonates and infants poses significant challenges due to the fragility and small size of the available cannulation vessels. The introduction of ultrasound-guided cannulation has greatly enhanced the safety and precision of these procedures, especially in neonates.1 –3 Pre-procedural evaluation, using established protocols such as Rapid Central Venous Assessment (RaCeVA), 4 allows for the accurate selection of the appropriate vessel and optimal exit site, minimizing procedural complications and improving patient outcomes.5,6 To address the lack of dedicated devices for neonates and infants, it is common practice to adapt small adult peripherally inserted central catheters (PICCs) for central venous access, 7 provided that the minimal “vein-to-CVAD ratio” of 3 mm of vein for each millimeter of catheter diameter,8,9 or a CVAD size less than 45% of the vein diameter, 10 is respected. The use of PICCs necessitates the application of a peel-away introducer. However, we acknowledge that in several high-quality PICC systems the peel-away introducer is only minimally larger than the catheter itself, and in such cases the associated risk may be limited. Nevertheless, the technique still presents two potential disadvantages in neonates. Firstly, in clinical settings where introducers are relatively larger or less refined, their use may increase the risk of injury to the small and fragile vessels compared with the simpler method of dilating and advancing a 3–4 Fr catheter over a guidewire.
In addition, although the position of the microintroducer can be continuously assessed with ultrasound, removal of the guidewire during catheter advancement eliminates a stable intravascular reference that can be visualized up to the superior vena cava. Maintaining guidewire visualization may provide additional reassurance regarding the intravascular course and final direction of the catheter, potentially reducing the risk of unintended deviation. 11
Proper tunneling enhances catheter stability and reduces the risk of infection. To overcome the limitations previously mentioned, we modified the insertion technique by omitting the introducer and advancing the catheter directly using a Seldinger approach, followed by distal trimming and retrograde tunneling. This manuscript aims to describe our experience in neonates weighing less than 5 kg and to discuss the safety, feasibility, and potential benefits of this alternative technique.
Methods
A specific institutional informed consent for CVAD implantation is obtained from parents or legal guardians prior to the procedure. This consent explains the technique, potential adverse events, and available alternatives. At our institution, since 2022, we have utilized a web-based database prospectively collect data on all vascular implants referred to our team. The Research Electronic Data Capture (REDCap) tool, available at our institution, was used for this purpose. REDCap is a secure, web-based platform designed to support data capture for research studies. It offers: (1) an intuitive interface for validated data entry; (2) audit trails to track data manipulation and export procedures; (3) automated export features for seamless data downloads into standard statistical packages; and (4) procedures for data integration and interoperability with external sources.12,13 Data collected included demographics, primary medical condition, indications for catheter placement, details of the implant procedure, fixation methods, and perioperative complications (early and late). We conducted a registry search for all neonates requiring a Tnc-CICC implanted using an ultrasound-guided percutaneous technique with retrograde tunneling. Inclusion criteria included a weight below 5 kg, a target central vein diameter of 2 mm or greater, and tolerance for retrograde tunneling with an over-the-wire technique using tapered proximal trimming catheters. In our institution, both anterograde and retrograde tunneling, as well as CVAD insertion over the guide without a micro-introducer, have been standard practices in neonates for many years. The study was exempted from Institutional Review Board approval, as it was considered a retrospective analysis of a clinical practice.
Catheters and equipment
We exclusively used the Vygon Lifecath CT PICC Easy 3 and 4 Fr, 60 cm catheters for this study. As far as we know, this is the unique catheter with distal trimming available on the market. A 4 Fr blunt-tipped, disposable steel tunneler (Medcomp) was used for tunneling.
Catheter insertion technique
All procedures adhered to a standardized protocol. Before the venipuncture, the RaCeVA protocol was followed to systematically assess the neck and supra/infraclavicular veins via ultrasound and select the optimal vein and exit site. 2 The procedure was carried out under strict aseptic conditions, which included hand hygiene, skin antisepsis using 2% chlorhexidine in 70% alcohol, and the use of maximal barrier precautions.
Venipuncture: ultrasound-guided venipuncture was performed using a 21 G needle, with an in-plane approach and long-axis view of the vessel (Figure 1(a)). A straight soft-tip nitinol guidewire was inserted once the vein was accessed (Figure 1(b)).
Dilator insertion: a small incision (approximately 2 mm) was made at the venipuncture site. The dilator included in the catheter kit was carefully advanced to expand the subcutaneous tissues, creating a passage for the catheter (Figure 1(c)).
Catheter insertion: the catheter was introduced using the Seldinger over the wire technique (Figure 1(d)). The tapered tip of the catheter was particularly effective in this phase, as it allowed for easy advancement through the dilated subcutaneous tract with minimal resistance.
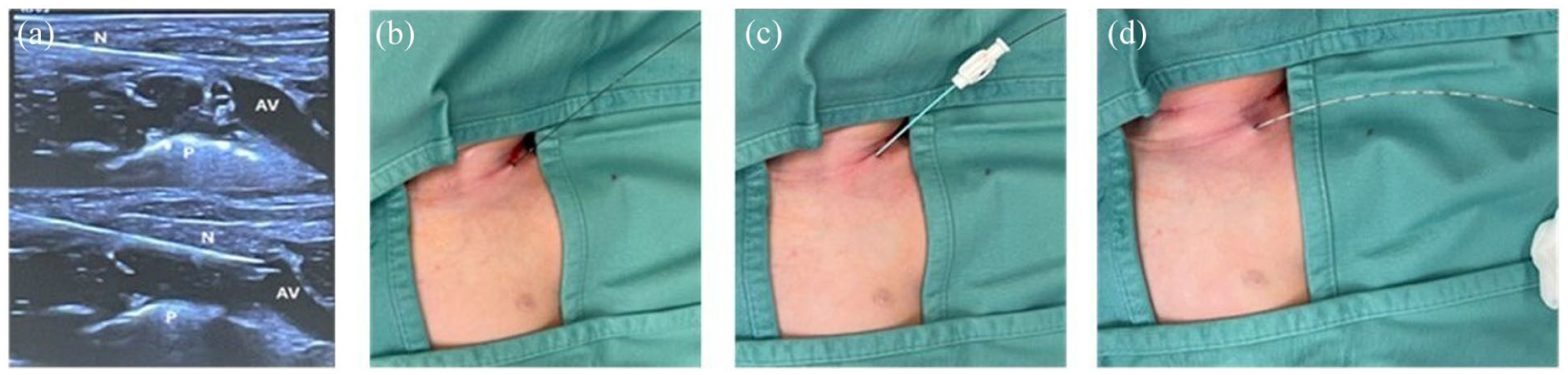
(a) Ultrasound guided venipuncture. N: needle; P: pleura; AV: anonymous vein. (b) Guidewire insertion. (c) Dilator insertion. This dilation is critical to minimize tissue resistance and prepare for the smooth insertion of the catheter. (d) Catheter insertion over the guide. The catheter smoothly tracks along the guidewire, the tapered design facilitates the advancement into the vessel without kinking or resistance.
The retrograde tunneling process is a straightforward and well-established technique. In our experience, this method allows for smooth and efficient subcutaneous tunneling from the venipuncture site to the exit point with minimal risk of complications.
Exit site selection: a small incision is made to prepare the site for tunneling (Figure 2(a)). The proximal catheter end is securely connected to the tunneler (Figure 2(b)).
Tunneling: the tunneler is advanced subcutaneously from the venipuncture site to the pre-determined exit site (Figure 2(c)).
Catheter positioning: The catheter is carefully pulled through the tunnel, ensuring proper positioning and alignment (Figure 2(d)).
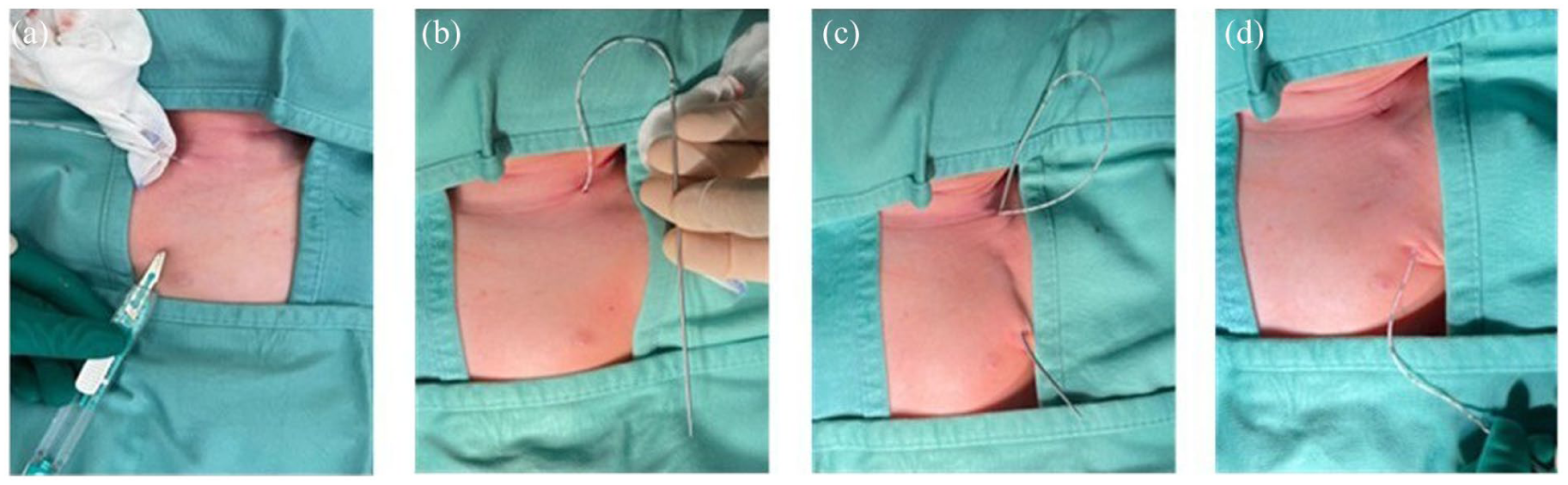
(a) Selection of the exit site. A small incision was made to prepare the site for tunneling. (b) The proximal end of the catheter is securely connected to the tunneler. (c) Tunneler is advanced from the venipuncture site to the pre-determined exit site. (d) The catheter is carefully pulled through the tunnel, ensuring positioning and alignment.
This technique ensures a correct subcutaneous pathway of the catheter, reducing the risk of kinking or migration. Once the catheter was tunneled, the power injectable connection was attached to the distal end of the catheter. Functionality tests were performed to confirm the catheter’s patency and readiness for use. The position of the catheter tip was verified with intracavitary ECG guidance to ensure correct placement at the atrio-caval junction. 14 The catheter was secured using a subcutaneous anchoring system (securAcath—Interrad Medical, Plymouth USA) to prevent dislodgement and ensure stability. 15 If the catheter length exceeded the optimal configuration for nursing management, it was shortened, and the connection system was reattached. This ensured an optimal setup for ongoing care and caregivers’ management of the exit site. The exit site was dressed with a semi-permeable transparent dressing, selected for its high breathability to maintain optimal site conditions.2,16
Results
At the time of data collection, eight neonates received a Tnc-CICC implanted using an ultrasound-guided percutaneous catheter and retrograde tunneling technique, meeting the inclusion criteria. All procedures were performed by a senior anesthetist, and data were collected by the Vascular Access Team—a multidisciplinary group of experts in venous catheter placement and care. Patient characteristics are detailed in Table 1. Catheter insertion using the Seldinger over-the-wire technique was successfully completed in all patients. Retrograde tunneling was performed without complications using a single-use tunneler. No catheter migrations or tunneling difficulties were encountered, and no immediate complications related to venipuncture or catheter placement occurred. Specifically, there were no cases of pneumothorax, arterial puncture, hemothorax, or failure to advance the guidewire or catheter. All catheters were inserted into the brachiocephalic vein. Four patients received a 3 Fr catheter, while the other four were implanted with a 4 Fr catheter. The only deviations from the protocol occurred in the three patients weighing less than 2.5 kg, where a micro-introduction kit with a 24 G needle, rather than the standard 21 G, was used to provide a gentler approach to the vessel. No other significant deviations from the standard protocol were made. In total, 379 catheter days were recorded. Seven (87.5%) catheters were removed at the end of therapy or because they were no longer needed, while one (12.5%) catheter was removed prematurely due to tip migration. No long-term adverse events were observed.
Descriptive variables. Data are expressed as n (%) or median (IQR).

VAD: vascular access device; dd: days; NICU: neonatal Intensive Care Unit; F: French; CLABSI: Catheter Associated blood Stream Infection; CR-DVT: catheter related deep venous thrombosis.
Discussion
This study describes our experience with the Seldinger over-the-wire technique combined with retrograde tunneling for long-term central venous catheter placement in neonates weighing less than 5 kg. The cannulation of the brachiocephalic vein in neonates and preterm infants has been recently well described 16 and is considered both feasible and safe. The proposed technique is simpler and faster than the traditional method, as it reduces the number of procedural steps. The key innovations in our approach are centered on two main aspects. The first is the use of proximal trimming catheters, which allow tunneling to be performed after the catheter tip has been advanced over the guidewire to its final position at the cavo-atrial junction. The second innovation is the omission of the catheter introducer. Typically, the catheter kit includes a peel-away introducer inserted over a dilator. In the classic approach, the dilator-introducer is advanced over the wire into the vessel. The wire and dilator are then removed, and the catheter is inserted through the introducer into its definitive position. However, the dilator has the same diameter as the catheter, and in some devices, the introducer can pose a significant risk to neonates due to its length (7 cm) and width. Specifically, for the Vygon Lifecath CT PICC Easy system, a 3 Fr catheter requires an introducer with an external diameter of approximately 1.72 mm and a 4 Fr catheter one of approximately 2.05 mm. In our technique, the catheter is advanced over the guidewire without the use of an introducer, after dilation. The peel-away technique is the most commonly adopted method for placing tunneled catheters, and when performed correctly—particularly under continuous ultrasound guidance—it remains an excellent and reliable approach. Nevertheless, it is not completely free of potential technical challenges and carries an increased risk of complications. In neonates with very small and delicate vessels, the use of a peel-away sheath needs specific steps and careful attention, to avoid kinking of the sheath, ensure proper opening during withdrawal, and maintain adequate depth control. Improper management of the peel-away sheath theoretically may introduce additional risks. For example, if the sheath–dilator unit is advanced too deeply, its distal tip could reach the atrial region, while insufficient advancement may increase the likelihood of losing venous access once the guidewire is removed, potentially leading to procedural failure. These considerations do not detract from the overall safety of the peel-away method, which, when executed appropriately under real-time ultrasound monitoring, rarely results in complications. Within this context, the main advantages of our over-the-wire approach lie in its procedural simplicity for selected neonatal cases. By avoiding the peel-away sheath, the technique eliminates the risk of air embolism and removes the need for continuous depth monitoring of the introducer. By using the Seldinger technique, the guidewire can remain in place throughout catheter advancement. Keeping the wire in position provides a stable and continuously visible landmark under ultrasound, which may assist in controlling the trajectory of the catheter—particularly in very small neonates, where the distance between the entry site and the final tip location is short. This step may contribute to a smoother progression of the catheter and can help reduce the likelihood of technical difficulties during placement.
The advantage of proximal trimming is that conical taper of the catheter’s tip is preserved. The specific conical form allows the catheter to advance smoothly and gently over the guidewire (Figure 3(a) and (b)) minimizing resistance and reducing the risk of tissue trauma during insertion.

(a) Original catheter tip with tapered form and (b) trimmed catheter with truncated tip.
Cutting the end of the catheter (proximal trimming) instead of the distal tip has been discussed, but few experimental studies have examined the potential clinical implications of distal trimming. These studies have suggested that distal trimming may expose irregular or non–factory-finished surfaces. However, these findings derive from heterogeneous models, including in vitro assessments and animal studies, and their relevance to neonates undergoing central venous access is uncertain.17,18 Importantly, there is no evidence to date that distal trimming increases thrombotic risk or clinical complications in neonatal CVADs, and in fact distal trimming remains the standard practice worldwide for PICCs and many small-caliber catheters.
This study has several limitations. It is a single-center case series, and larger studies should confirm reproducibility. The small sample size does not allow us to draw firm conclusions but only the feasibility and safety of the technique. It is a retrospective design despite the prospective data collection in our database and we did not have a control group of patients treated differently. As this study describes a modified technique in a small case series, we do not have statistical analysis to present.
Trimming the catheter at the end of the procedure allows for customization of an optimal length, improving the management of the exit site. This approach avoids an excessively long extravascular portion, which can be challenging to manage and uncomfortable for the patient.
In this small series, our results indicate that the method seems to be both safe and effective, with high success rate in tunneled long-term catheter insertion and no short- or long-term complications. The simplicity of the technique makes it easy to implement, even in neonates, while ensuring accurate catheter placement. The approach does not require any specific skills beyond those already required for standard CVAD insertion in neonates and is gentler on the vessel. Performing retrograde tunneling after catheter placement offers some potential advantages: it facilitates precise length adjustments, minimizes the risk of dislocation, and preserves the catheter’s original tapered tip. We speculate that by preserving the tapered tip of the catheter, the use of the dilator can be avoided, further simplifying the technique.
Footnotes
Author contributions
AD conceived the original idea for the article, enrolled the patients, drafted the first version, and managed the database. AW drafted the definitive version of the manuscript, performed the statistical analysis, ensured proper database maintenance, and selected the editorial channel. SB contributed to the concept of the paper. CM assisted in the revision of the manuscript. GC, SL, CPdP helped with patient enrollment. AM supervised the revision and facilitated the completion of the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Institutional Review Board waived the need of an informed consent due to the retrospective observational analysis of a standard practice.
