Abstract
Background:
Arteriovenous fistula (AVF) dysfunction commonly results from hemodynamically significant stenosis, which alters the local turbulence and generates characteristic vibrational signatures. A Hemodialysis Access Vibration and Sound Evaluation (HAVSE) device is a low-cost surface accelerometer designed to quantify these mechanical signals. This pilot study evaluated whether HAVSE could detect physiologically coherent changes in the AVF vibrational amplitude before and after angioplasty.
Methods:
We prospectively enrolled adults with dysfunctional arteriovenous fistulas who were scheduled to undergo angioplasty. HAVSE recordings were obtained immediately before and after intervention at predefined anatomical locations. Vibrational amplitude was derived from cardiac-cycle–specific accelerometry and expressed as a dimensionless postoperative-to-preoperative ratio to minimize inter-patient variability. Stenoses were categorized as perianastomotic, outflow, or central. Paired Wilcoxon signed-rank tests and Hodges–Lehmann estimates were used to assess relative postoperative changes. The study was prospectively registered with the Australian New Zealand Clinical Trials Registry (ANZCTR) under the identifier ACTRN12619000281190.
Results:
Nineteen patients were included in this analysis. In isolated perianastomotic stenosis, postoperative vibrational amplitude consistently decreased (median ratio 0.55; 95% confidence interval, 0.20–0.86; p = 0.03), consistent with reduced turbulence following angioplasty. In isolated outflow stenosis, the postoperative amplitude increased (median, 2.10), reflecting the restoration of physiological high-flow turbulence, although the difference did not reach statistical significance. Multifocal disease produces heterogeneous signals, and central venous stenosis does not generate reproducible surface vibrational changes. Neither absolute nor relative amplitude thresholds reliably distinguished stenotic from non-stenotic sites.
Conclusions:
Surface vibration analysis using HAVSE demonstrated physiologically coherent patterns in isolated perianastomotic and outflow stenoses but limited discriminatory performance in multifocal or central venous disease, supporting further device development and validation in larger studies that incorporate Doppler ultrasound correlations and standardized acquisition protocols.
Keywords
Introduction
Chronic kidney disease affects approximately 11% of Australian adults, with more than 27,000 individuals receiving renal replacement therapy in 2020.1,2 Hemodialysis remains the predominant modality and requires reliable vascular access capable of sustaining high extracorporeal blood flow. Among the available access types—arteriovenous fistulas (AVFs), arteriovenous grafts, and central venous catheters—AVFs are preferred because they offer superior patency, decreased infection risk, and improved long-term survival.3–7 Additionally, AVFs are more cost-effective than other access modalities throughout the dialysis lifespan. 8
Despite these advantages, AVFs remain vulnerable to progressive dysfunction driven by endothelial injury, neointimal hyperplasia, and hemodynamic stress,6,9–12 which result in juxta-anastomotic or outflow stenoses that disrupt laminar flow, increase turbulence, and reduce blood flow access. Notably, these impairments compromise dialysis adequacy and substantially increase thrombosis risk.13–16 Early identification and treatment of stenosis are essential to preserve AVF function and optimize dialysis delivery.3,4,6,17
Clinical examinations remain the cornerstone of AVF surveillance. Thrill propagation changes, bruit frequency alterations, and high-frequency vibratory component development often indicate underlying stenosis.15,18 Although duplex ultrasonography provides accurate anatomical and hemodynamic assessments, routine surveillance has not consistently demonstrated improved clinical outcomes and has significant cost and resource implications.3,19–21 Consequently, most centers rely primarily on physical examinations supplemented by selective ultrasound or angiography.
Recent studies have explored the objective quantification of vascular acoustic signatures using phonoangiography, demonstrating that stenotic AVFs exhibit characteristic sound frequency and intensity changes, particularly when combined with machine-learning algorithms.22–30 Nonetheless, these systems capture acoustic energy rather than the mechanical vibrations that clinicians traditionally assess during palpation.
A Hemodialysis Access Vibration and Sound Evaluation (HAVSE) device was developed as a simple, low-cost surface accelerometer to quantify mechanical vibrational signals generated by AVF blood flow. Foundational hemodynamic work by Soliveri et al. demonstrated that AVF stenosis-induced turbulence produces measurable oscillatory patterns propagating to the skin surface. 31 Similarly, surgical literature confirms that anastomotic geometry and venous outflow configuration strongly influence turbulent flow and stenosis behavior, most notably in the study by Franchin et al. 32 These data provide a strong physiological rationale for vibration-based diagnostic assessment.
This exploratory pilot study evaluated whether HAVSE can detect vibrational differences between dysfunctional and restored AVFs. Specifically, we examined whether the relative changes in surface vibrational amplitude differed before and after angioplasty, and whether these patterns varied across perianastomotic, outflow, and central venous stenoses.
Methods
Study design and oversight
This prospective single-center feasibility study was conducted to determine whether surface vibration analysis could identify hemodynamically significant stenosis in arteriovenous fistulas (AVFs). The study protocol was approved by the Royal Perth Hospital Human Research Ethics Committee (RGS0000396), and written informed consent was obtained from all participants. The trial was prospectively registered with the Australian New Zealand Clinical Trials Registry (ANZCTR) under the identifier ACTRN12619000281190. Because this was an early-stage evaluation of a novel device, operators were not blinded to clinical findings or procedural details; this absence of blinding is recognized as a methodological limitation.
Participants
Adults aged ⩾18 years with dysfunctional autogenous AVF who underwent clinically indicated endovascular interventions were eligible. The inclusion and exclusion criteria for patient recruitment are summarized in Table 1. AVF dysfunction was defined using standard clinical indicators, including palpable thrill or bruit changes, rising dialysis venous pressure, prolonged post-dialysis bleeding, or cannulation difficulties. Arteriovenous grafts were excluded because synthetic conduits substantially alter the vibrational transmission. Additional exclusion criteria included involuntary arm movements, inability to maintain limb stiffness, and lack of consent.
Inclusion and exclusion criteria.

AVG: arteriovenous graft; AVF: arteriovenous fistula; HAVSE: Hemodialysis Access Vibration and Sound Evaluation.
Summary of eligibility criteria for patient recruitment, including requirements for autogenous AVF, clinical indicators of dysfunction, and exclusion factors such as involuntary limb movement or arteriovenous grafts.
HAVSE device and signal acquisition
The HAVSE system consists of a low-cost surface accelerometer enclosed within a stabilized casing incorporating:
fixed protruding rims to prevent excessive compression,
a dampening sponge interface to minimize movement artifacts, and
stable contact platform to improve signal fidelity.
The patients were positioned with their forearms comfortably supported to minimize extraneous motion. Measurements were obtained at predefined anatomical locations: the anastomosis, outflow vein, and non-stenotic reference site. Although the casing design reduces the operator-dependent variability, the prototype does not incorporate pressure sensors, and full standardization of the applied force is not achievable. Inter-operator reproducibility was not assessed in the pilot phase.
Signal processing and normalization
The accelerometry signals were recorded at a high sampling frequency and stored for offline analysis. The vibrational amplitude was defined as the peak-to-trough magnitude of cardiac-cycle-synchronous oscillations and averaged across multiple consecutive beats to reduce physiological noise. Representative raw signals, amplitude extraction, and systolic–diastolic profiles are displayed in Figures S1 to S3.
Because the absolute amplitude varies substantially between patients owing to differences in soft-tissue damping, fistula depth, and intrinsic vibratory characteristics, amplitudes were expressed as dimensionless relative ratios, calculated as follows:
Relative vibrational amplitude = postoperative amplitude ÷ preoperative amplitude
Ratios <1 and ratios >1 indicate reduced vibrational energy after angioplasty and increased turbulent energy, respectively.
Stenosis classification
Stenoses were classified angiographically as:
perianastomotic (juxtaanastomotic region),
outflow (cephalic, basilic, or brachial segments), or
central venous (subclavian, innominate, or superior vena cava obstruction).
Because central venous lesions lack a superficial skin-level correlation, HAVSE cannot directly interrogate these areas. Therefore, measurements at the anastomosis and a distal non-stenotic site were descriptively analyzed as surrogates.
Statistical analysis
Statistical analyses were performed using Python (NumPy, SciPy, and StatsModels) and StataSE 17. Data were assessed for normality using the Shapiro–Wilk test; all continuous variables demonstrated significant non-normality (p < 0.05). Continuous variables are reported as medians, interquartile ranges (IQRs), and bias-corrected 95% confidence intervals (CIs), and categorical variables as counts and percentages.
This study primarily aimed to determine whether HAVSE detects physiologically meaningful postoperative changes in the surface vibrational amplitude. Within-patient and site-specific relative ratios (postoperative/preoperative) were used to minimize interpatient variability from soft-tissue damping, AVF depth, and intrinsic vibratory differences. Analyses used within-patient, site-specific relative ratios (postoperative ÷ preoperative). Paired comparisons were performed using the exact Wilcoxon signed-rank test, which is robust for small samples and non-normally distributed data.
Between-site comparisons (stenotic vs non-stenotic) and comparisons between stenosis types were performed using the Wilcoxon rank-sum test. Effect sizes were summarized using Hodges–Lehmann median differences with bias-corrected 95% CIs to provide precision and directionality estimates.
Given the exploratory intent and small sample size, all analyses were univariate, and no adjustment for multiple comparisons or multivariable modeling was performed. Missing data (<5%) were handled using complete-case analysis.
To explore the potential diagnostic performance of HAVSE, receiver operating characteristic (ROC) curves were generated for perianastomotic, outflow, and central venous stenoses. The areas under the curve (AUCs) were interpreted descriptively, acknowledging that the study was not suitable for a diagnostic accuracy assessment.
A two-sided p-value <0.05 was considered statistically significant, but all p-values should be interpreted cautiously due to the small sample size and the feasibility of the study design.
Results
Patient characteristics
Of the 26 screened patients, 19 were included in the final analysis. The reasons for exclusion included canceled procedures, incomplete recordings, and inadequate signal quality. The baseline characteristics are presented in Table 2. The median patient age was 49 years (range, 27–70 years), and 58% of the patients were men. Most AVFs were radiocephalic or brachiocephalic. Primary stenoses were perianastomotic, outflow, and central venous in 47%, 26%, and 26% of the patients, respectively. Secondary stenoses were present in 37% of patients.
Baseline demographic and clinical characteristics.
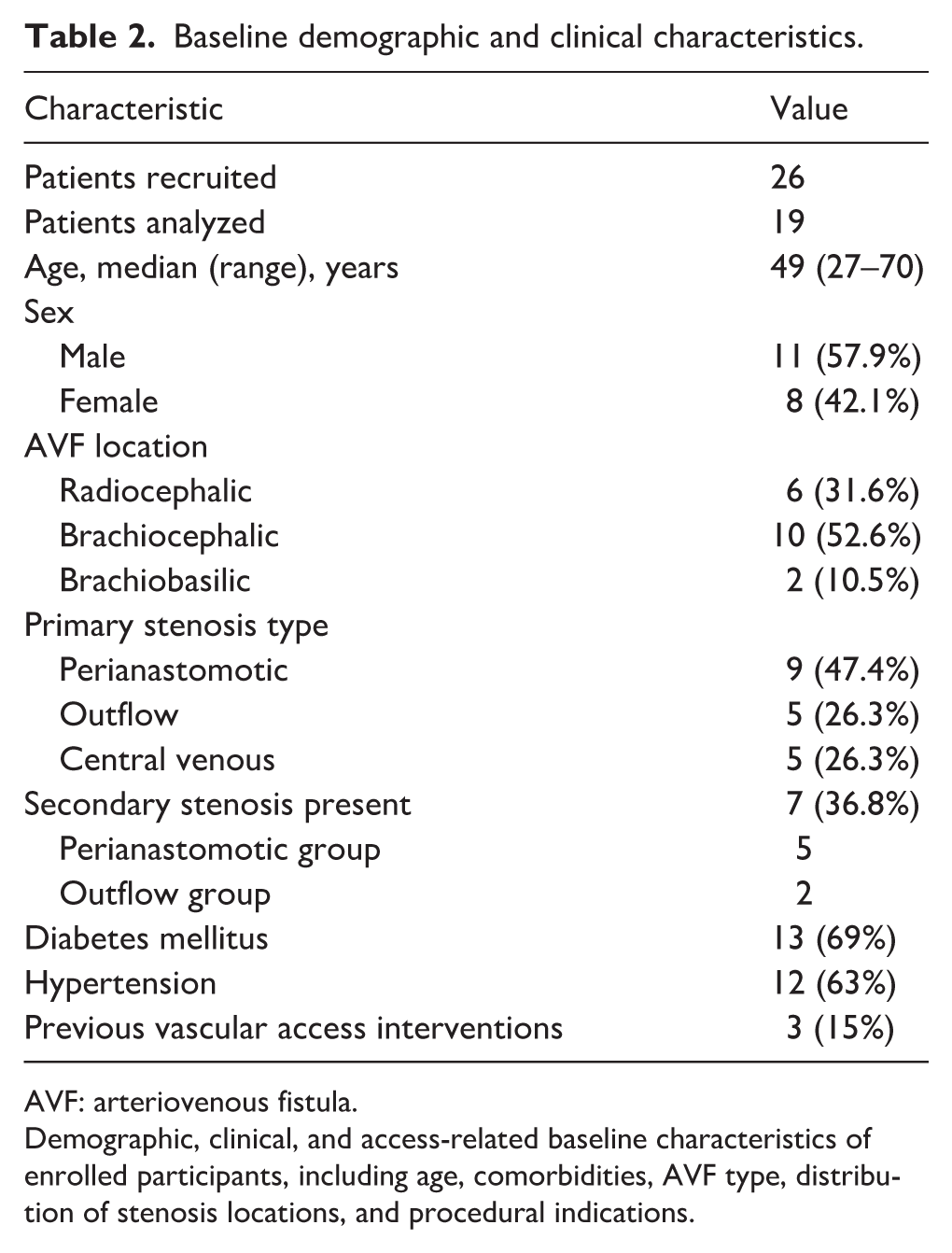
AVF: arteriovenous fistula.
Demographic, clinical, and access-related baseline characteristics of enrolled participants, including age, comorbidities, AVF type, distribution of stenosis locations, and procedural indications.
HAVSE signal characteristics
Representative device configurations and signal morphologies are displayed in Figures 1 to 4, with supplemental figures illustrating the unfiltered accelerometry traces and cardiac cycle amplitude extraction. Despite considerable interpatient variability in absolute amplitude due to differences in soft-tissue damping, fistula depth, and intrinsic vibratory characteristics, beat-to-beat waveform stability was preserved across participants, thereby supporting the use of dimensionless postoperative and preoperative ratios for within-patient comparisons.

HAVSE device components.
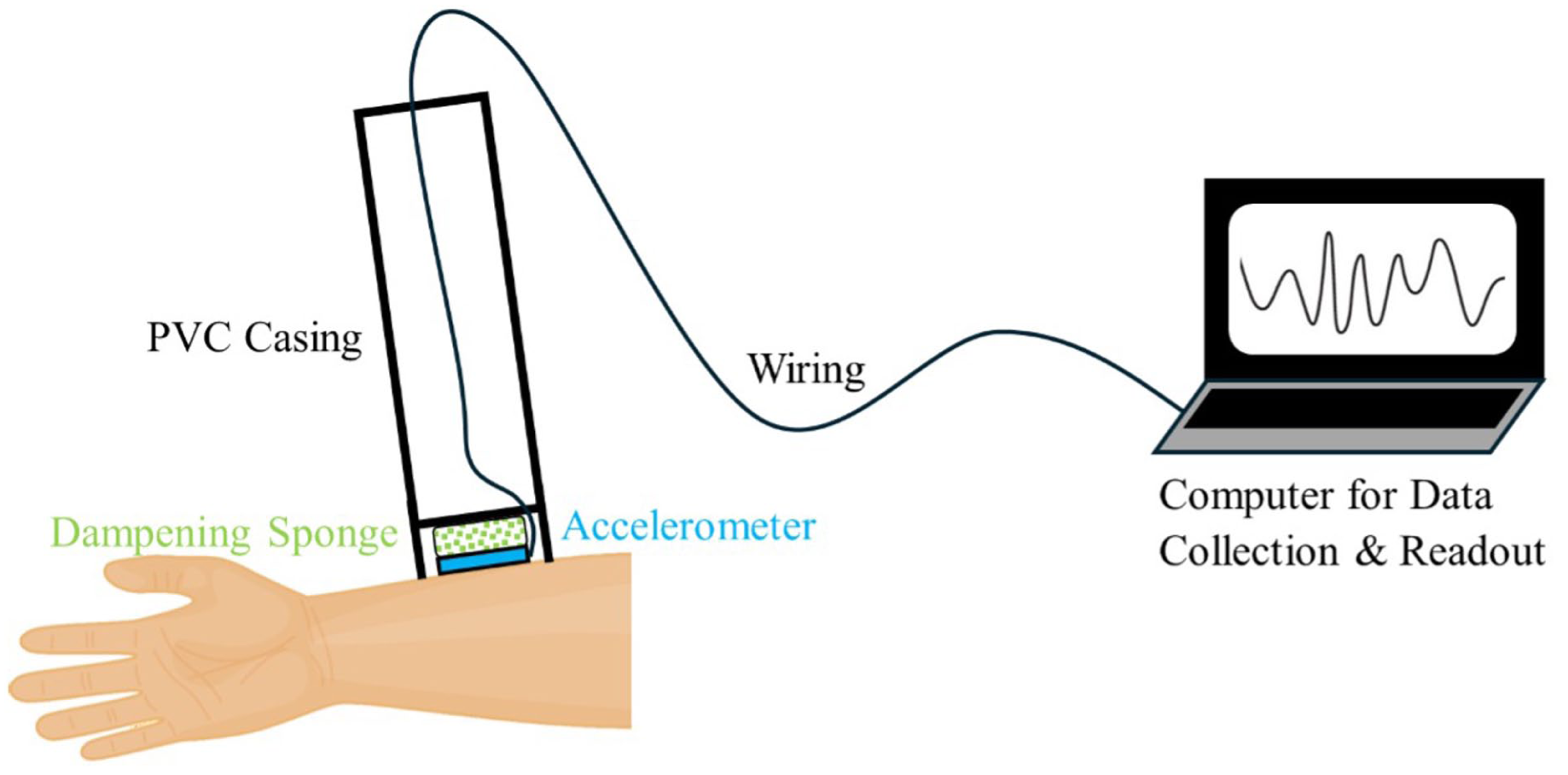
HAVSE sensor and signal pathway.
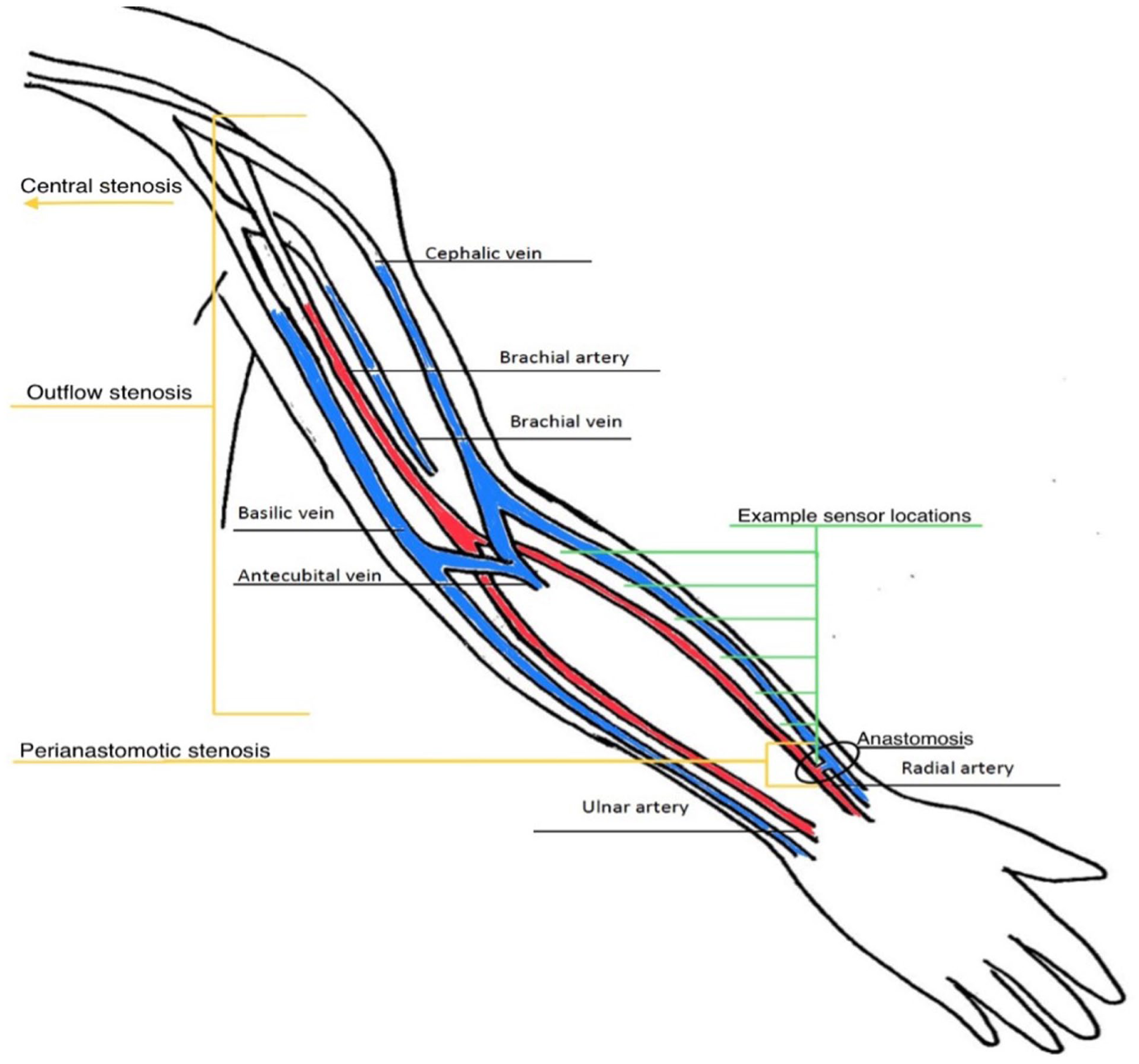
Anatomical regions of stenosis.
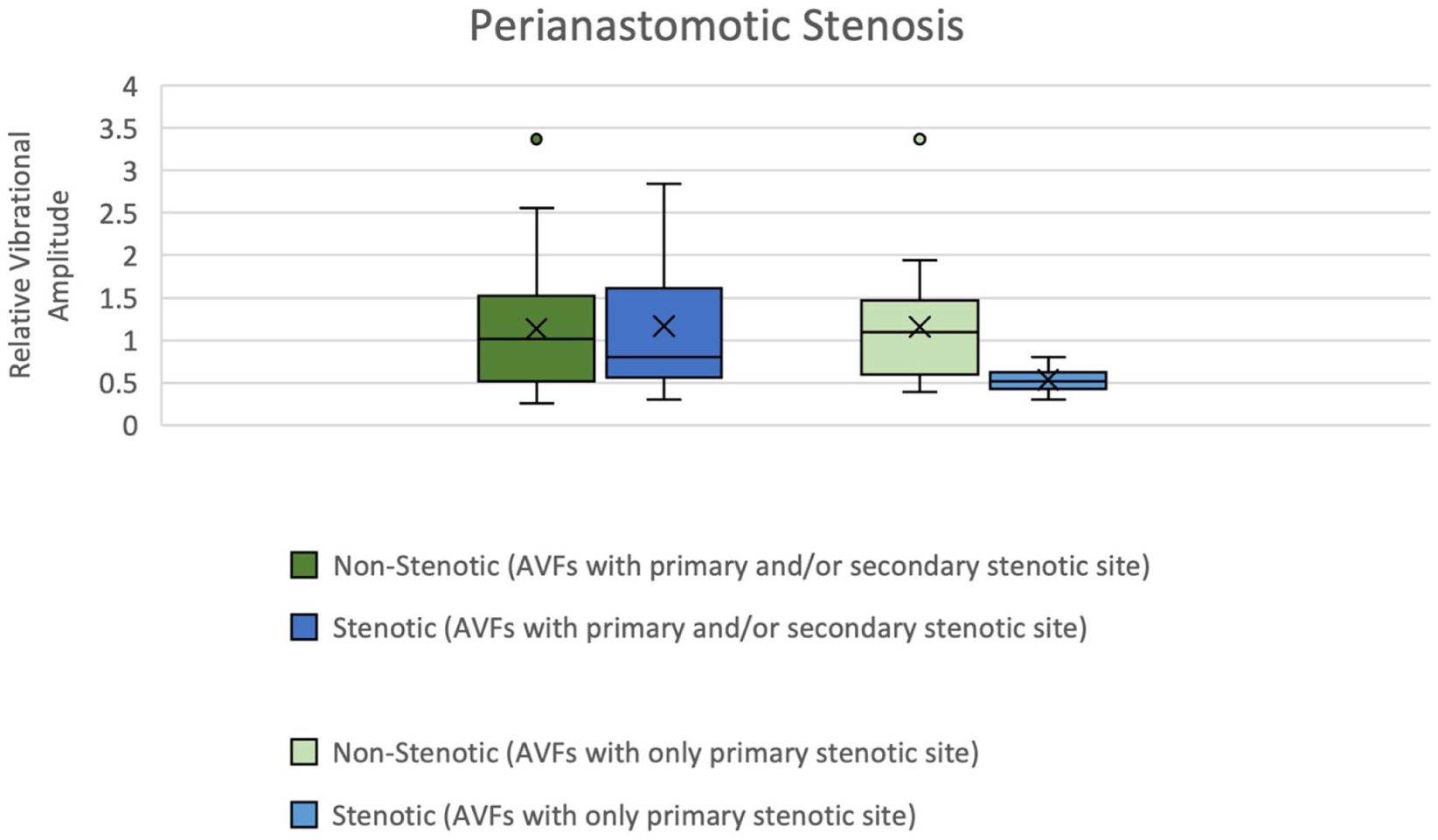
Relative vibrational amplitude in perianastomotic stenosis.
Perianastomotic stenosis
The relative postoperative-to-preoperative vibrational amplitude ratios by stenosis type and recording location are summarized in Table 3. Among patients with perianastomotic stenosis (n = 9), postoperative amplitude ratios varied widely (median, 1.10; IQR, 0.72–1.62; p = 0.41). Nevertheless, when restricted to those with isolated perianastomotic lesions (n = 4), responses were physiologically coherent: postoperative vibrational amplitude consistently decreased (median ratio, 0.55; IQR, 0.34–0.77; 95% CI, 0.20–0.86; p = 0.03; Figure 4), aligning with the expected reduction in turbulence in the juxta-anastomotic zone following a successful angioplasty.
Relative vibrational amplitude changes by stenosis type.
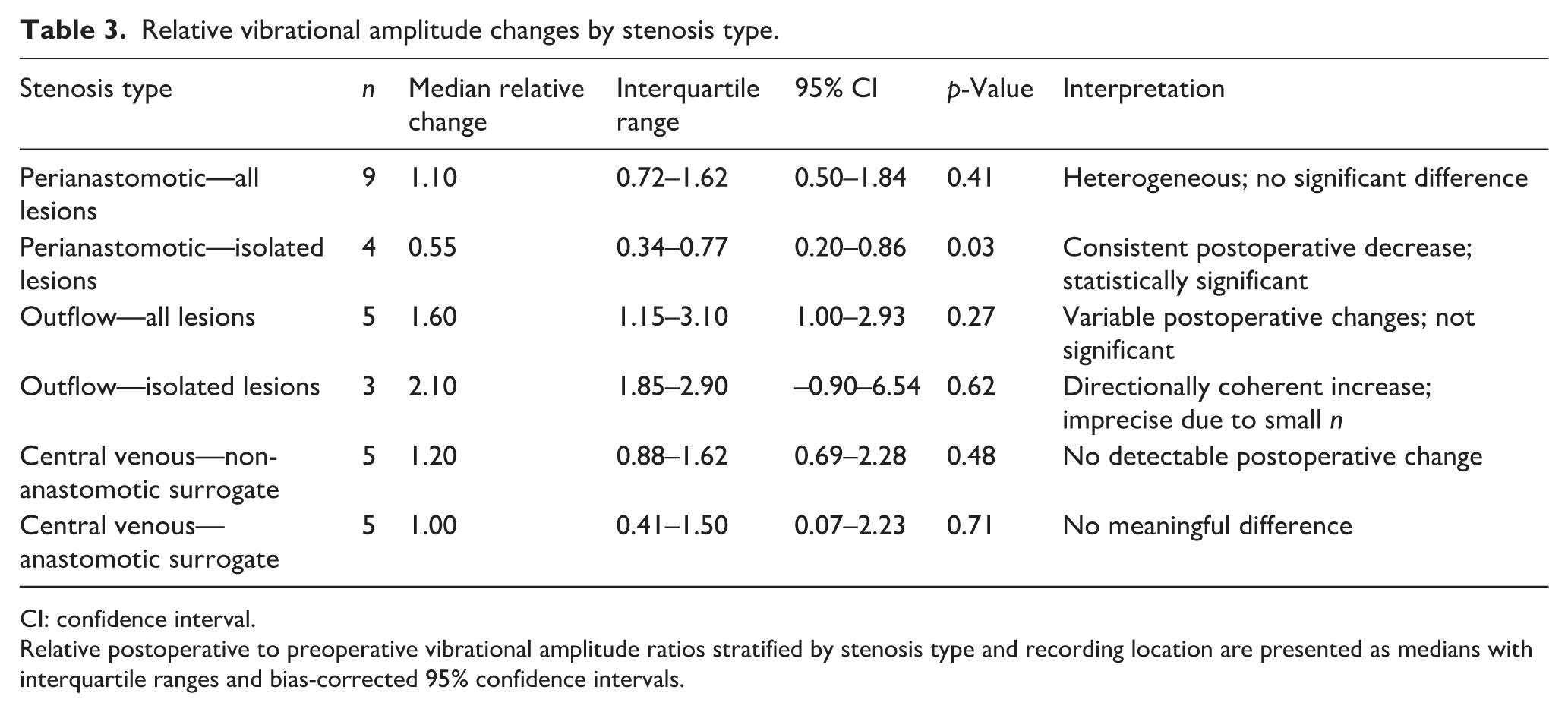
CI: confidence interval.
Relative postoperative to preoperative vibrational amplitude ratios stratified by stenosis type and recording location are presented as medians with interquartile ranges and bias-corrected 95% confidence intervals.
Outflow stenosis
In all outflow stenosis cases (n = 5), postoperative amplitude ratios demonstrated substantial dispersion (median, 1.60; IQR, 1.15–3.10; p = 0.27). The corresponding postoperative-to-preoperative vibrational amplitude ratios are shown in Figure 5. When analysis was limited to isolated outflow lesions (n = 3), postoperative amplitude increased in all patients (median ratio, 2.10; IQR, 1.85–2.90), consistent with the restoration of physiologic high-flow turbulence downstream of the anastomosis. Although directionally consistent, the effect did not reach statistical significance (p = 0.62) owing to the small sample size (Figure S1).

Relative vibrational amplitude in outflow stenosis.
Central venous stenosis (CVS)
CVS did not produce reproducible vibrational responses at superficial recording sites. Surrogate measurements at the anastomosis and adjacent non-stenotic locations demonstrated heterogeneous postoperative ratios (anastomosis: median 1.00; IQR, 0.41–1.50; p = 0.71; non-stenotic site: median 1.20; IQR, 0.88–1.62; p = 0.48). These findings indicate limited propagation of central venous turbulence to the skin surface (Figure S4).
Exploratory diagnostic performance
ROC curve analysis demonstrated modest perianastomotic stenosis discrimination (AUC 0.63), poor outflow stenosis discrimination (AUC 0.59), and no central lesion discrimination (AUC 0.51; Table S1). The diagnostic performance deteriorated in the presence of multifocal disease, where multiple turbulence sources confounded signal interpretation.
Overall, isolated stenoses exhibited physiologically coherent postoperative changes in HAVSE vibrational amplitude, whereas multifocal and central diseases produced heterogeneous, non-discriminatory signals. The absolute and relative vibration thresholds did not reliably differentiate between stenotic and non-stenotic sites across the entire cohort.
Discussion
AVF dysfunction manifests as progressive luminal narrowing, disturbed flow profiles, and increased turbulence, generating characteristic vibrational signatures along the vessel wall. Foundational work from the Istituto di Ricerche Farmacologiche Mario Negri IRCCS—most notably the study by Soliveri et al.—demonstrated that stenosis-induced turbulence produces measurable oscillatory patterns that can be detected non-invasively and that vascular remodeling modulates these flow instabilities over time. 31 Their hemodynamic modeling provides a strong mechanistic foundation for vibration-based diagnostics and directly supports the physiological principles underlying HAVSE.
Surgical literature further reinforces the role of turbulence in access failure. Franchin et al. 32 demonstrated that small-diameter anastomoses and technical variations in AVF construction substantially influence turbulent flow and complication rates. Together, these insights position HAVSE within a broader scientific continuum in which surface vibration assessment represents a quantitative extension of classical physical examination (thrill/bruit evaluation).
Because of the HAVSE accelerometer-based design, the device measures mechanical vibrations generated by blood flow. Therefore, high turbulence areas, particularly near stenotic lesions, are expected to produce greater vibrational amplitudes. Notably, stenosis relief should theoretically reduce turbulence and lower postoperative readings, consistent with the observed behavior of perianastomotic stenoses, which exhibit reduced vibrational amplitudes after angioplasty. These findings align with those of previous hemodynamic studies, which demonstrate maximal turbulence in the immediate vicinity of stenotic lesions.22,33
In contrast, outflow stenoses demonstrated a increased vibrational amplitude after correction. Prior research has found that physiologically functioning AVFs naturally exhibit high post-anastomotic turbulence owing to elevated flow rates,31,34 and outflow patency restoration likely reestablishes this high-flow state, yielding increased HAVSE readings.
The absence of distinct vibrational changes in the central stenosis was expected because HAVSE measurements were not performed directly over the lesion, underscoring the fundamental limitations of surface-based detection and indicating the need for future methodological strategies to evaluate centrally located stenoses.
Patients with multiple stenotic sites display broad variability, reflecting complex hemodynamic interactions across the access circuits, thereby suggesting that HAVSE is currently the most applicable treatment for AVFs with single isolated stenosis.
Furthermore, the vibrational amplitudes at the non-stenotic sites overlapped with those at the stenotic sites, implying potential false positives. Nonetheless, relative amplitude ratios near 1.0 may help reliably rule out isolated stenoses. Notably, no universal amplitude threshold emerged; instead, relative changes normalized within individual patients appeared more informative, underscoring the potential value of baseline HAVSE recordings during AVF creation.
Methodologically, HAVSE records amplitude in non-standardized sensor units; thus, all analyses employed dimensionless normalized ratios to enhance comparability across patients. Operator variability was reduced but not entirely eliminated through the prototype’s fixed protruding rim and dampening sponge. Nevertheless, formal reproducibility testing has not been performed, and comprehensive acquisition protocols are required.
The Doppler ultrasound correlation was not included because of feasibility constraints in the periprocedural pilot setting. Although a direct comparison of velocity ratios and anatomic stenosis severity will be essential in future validation studies, the present study was designed primarily to evaluate the device’s feasibility.
This study had some limitations: small sample size, unblinded design, AVG exclusion, manual sensor placement, non-calibrated measurement units, univariate analysis only, and the absence of duplex ultrasound or correlation with clinical outcomes. These limitations reflect the exploratory nature of early-phase feasibility testing.
Despite these constraints, HAVSE has significant potential as an inexpensive and portable AVF surveillance adjunct, particularly in resource-limited settings. Importantly, the device can support bedside or dialysis-unit assessment, at-home monitoring, and early detection of access dysfunction. Future refinement may incorporate machine-learning-based signal interpretation, as suggested by prior work.25–30 Ultimately, whether such surveillance improves morbidity or mortality remains an important question that has similarly constrained the routine screening role of duplex ultrasound.
Future developments should prioritize multicenter evaluation, standardized acquisition protocols, calibration to SI units, reproducibility analysis, integration with duplex ultrasound and clinical outcomes, assessment of arteriovenous grafts, advanced algorithmic interpretation, and longitudinal studies to evaluate the predictive value for access failure.
Conclusion
The HAVSE prototype shows promise as a low-cost mechanical vibration sensor for detecting hemodynamic changes associated with AVF stenosis. Clear postoperative reductions or normalized vibrational amplitude increases were observed at stenotic and non-stenotic sites, indicating that surface vibration analysis may complement established approaches to vascular access surveillance. Although preliminary and constrained by a small sample size and early-stage methodology, these findings support the continued development, refinement, and validation of vibration-based diagnostics validation. Further studies are warranted to determine whether this technology can significantly improve clinical outcomes, streamline surveillance practices, and enhance long-term AVF management.
Supplemental Material
sj-docx-1-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-docx-1-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-docx-2-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-docx-2-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-jpg-3-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-jpg-3-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-jpg-4-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-jpg-4-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-jpg-5-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-jpg-5-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-jpg-6-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-jpg-6-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Supplemental Material
sj-jpg-7-jva-10.1177_11297298261417721 – Supplemental material for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study
Supplemental material, sj-jpg-7-jva-10.1177_11297298261417721 for Preliminary evaluation of a novel surface accelerometer (HAVSE) for dialysis access surveillance: An exploratory pilot study by Justin K Liew, Bryan Tan, Bernadette Lee, Gabrielle Stratford, Monique Sandford, Patrik Tosenovsky, Mark Thomas and Bibombe Patrice Mwipatayi in The Journal of Vascular Access
Footnotes
Acknowledgements
We sincerely thank all patients who participated in this study, as well as the dialysis staff and vascular access teams at Royal Perth Hospital for their outstanding support during data collection and clinical coordination. We also appreciate Dr James E. Dodd for his invaluable contributions to the manuscript, whose insightful review greatly enhanced the quality of our work.coordination.
Author contributions
Justin K Liew supported the study through methodology development, software and data processing, validation, and critical manuscript review. Bryan Tan provided engineering support and early technical development, building on the original study concept proposed by Patrik Tosenovsky, and assisted with data acquisition and manuscript review. Bernadette Lee participated in manuscript review. Gabrielle Stratford contributed to the preparation of the final draft. Monique Sandford’s participant recruitment and data collection should be appropriately recognized. Patrik Tosenovsky contributed the original study concept, provided clinical oversight and supervision, assisted in interpreting findings, and critically revised the manuscript. Mark Thomas offered methodological guidance, supported the interpretation of results, and contributed to manuscript review and editing. Bibombe Patrice Mwipatayi served as the senior author, interpreted the findings, critical manuscript revision, and final approval of the submitted version.
Data availability
De-identified data may be obtained from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received no external funding. It was conducted as a voluntary pilot project to assess feasibility and data acquisition. Grant funding may be pursued for future, larger studies.
Ethical considerations
Ethical approval for this study was granted by the Royal Perth Hospital Human Research Ethics Committee (RGS0000396).
Consent to participate
All patients gave written informed consent after receiving verbal and written explanations of the study.
Consent for publication
Not applicable; no identifiable patient data are included in this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
