Abstract
Self-administered capillary blood collection devices are increasingly used in both clinical research and patient care. The Tasso+ device, a CE-certified system designed to collect 500 µL of whole blood, is generally regarded as safe and user-friendly. However, complications related to its use remain insufficiently documented. We describe the case of a female study participant who developed an unexpected wound complication after using the self-blood collection device. Although the blood collection itself was successful, the participant later required surgical treatment for a gaping lesion at the application site, which ultimately left a small permanent scar. To our knowledge, this represents a previously unreported adverse effect of the device. The case raises concerns about the interaction between device application and skin tension lines and underscores the need for further evaluation of safety aspects in self-administered blood collection systems.
Keywords
Introduction
The growing need for remote healthcare solutions and decentralized clinical trials has led to increased interest in self-administered capillary blood collection devices. These devices allow patients or study participants to collect blood samples at home or through non-medical staff, reducing the need for in-person clinic visits.1,2 In addition, recent studies suggest that capillary self-sampling can achieve good analytical agreement with venous blood, is associated with less pain than venous blood sampling 3 and shows high user satisfaction.4,5 Current self-blood collection devices are mostly CE-certified and Class II Lancet 510(k)-Cleared system designed for the self-collection of approximately 500 µL of whole blood. 6
These devices are generally considered safe, and user-friendly, and allow almost pain-free drawing of blood.3,4 Other than black-out/fading due to blood-drawing, adverse effects associated with their use have not been described yet. As these devices become more widely used, it is crucial to understand and report any unexpected adverse events to ensure patient safety and improve device design.
Case presentation
A female 43-year-old participant of the Blood-it-yourself study (DRKS00033172) successfully collected approximately 500 µL of whole blood using the Tasso+ device (Tasso Inc., Seattle WA, USA) at a general practice on May 8th, 2024. The study is an observational study aimed to evaluate the feasibility and accuracy of self-administered capillary blood collection using self-sampling devices. Feasibility results of the Blood-it-yourself study have been published, 4 whereas the manuscript reporting analytical accuracy is currently in preparation. The study compares laboratory results from self-collected capillary blood samples with those from standard venous blood draws. Conducted in German primary care and rheumatology practices, the study includes adults aged 18 or older who provided informed consent. Key outcomes include measurement concordance, sample quality, and user experience with the self-collection device.
The participant was healthy with no known chronic conditions, especially no known tendency for complications in wound healing such as keloid or hypertrophic scars. The incision site on the left upper arm was inspected by study personnel as part of the pre-screening prior to enrollment as wounds, scars, or ulcerations were exclusion criteria for participation. The self-collection procedure was observed by study personnel and was performed according to the manufacturer’s instructions. The participant applied a quick-healing bandage to the incision site as directed. The participant rated the perceived pain during self-blood collection as similar to the pain experienced during the venous blood draw, giving both blood draws a score of 2 on a Likert scale from 0 to 10, where 0 represents no pain and 10 represents severe pain.
Later the same day, upon removing the bandage, the participant noticed a gaping, horizontal wound approximately 3–4 mm in length at the incision site (Figure 1(a)). Although the wound appeared clean and showed no signs of inflammation, the participant sought medical attention from a surgeon. To prevent potential disfiguring scarring, the wound was closed with a single interrupted 6-0 non-absorbable monofilament suture (Figure 1(b)). Surface anesthesia with Mepivacaine 1% injection solution was used for anesthesia.
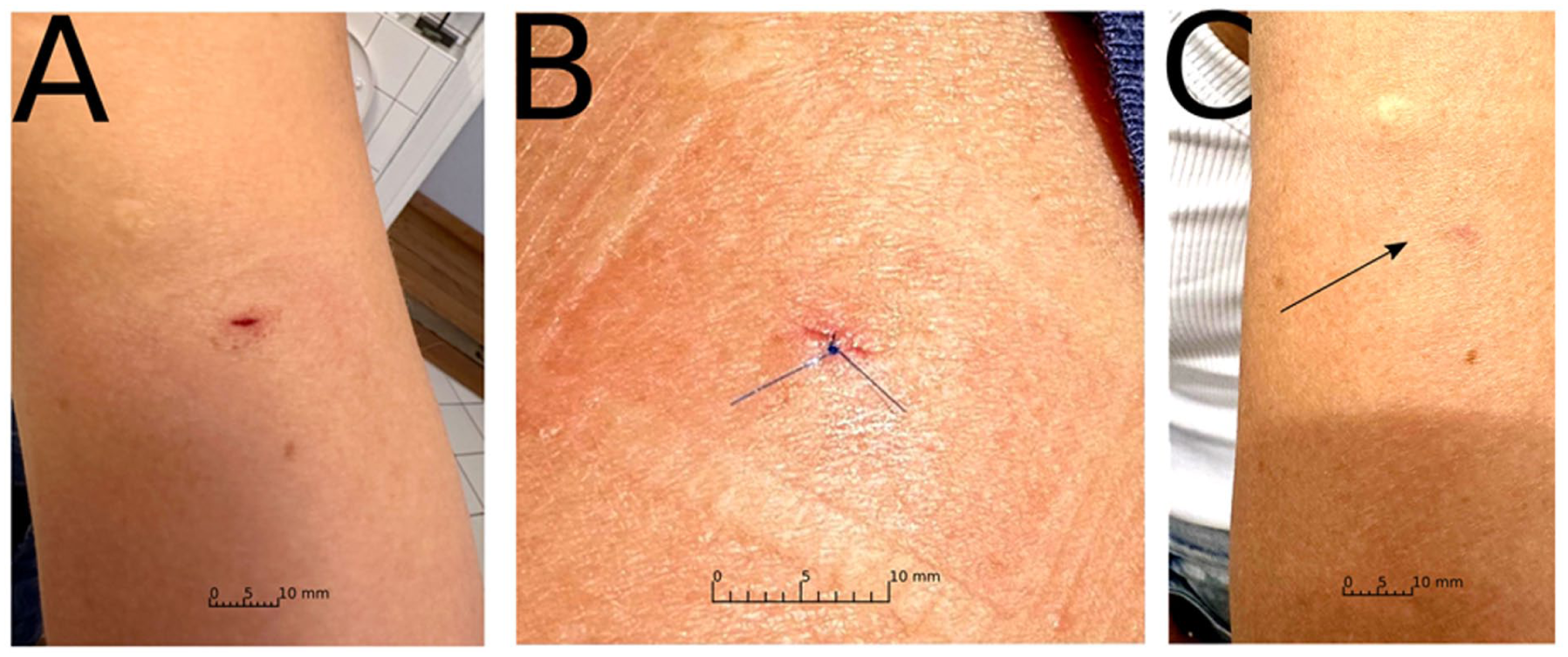
(a) Incision wound approx. 7 h after using Tasso+ device, (b) wound closing with suture immediately after 1(a), close-up image, and (c) scar after 48 days.
The participant subsequently contacted the study team, reporting that she had not been informed about the possibility of scarring as a potential adverse event. She provided photographs documenting the incision immediately after blood collection, after wound closure, and following complete healing after 48 days (Figure 1(c)). The images confirmed the presence of a small permanent scar at the incision site.
Discussion and conclusion
This case report describes an unexpected complication following the use of a self-blood collection device. To our knowledge, scarring has not been previously reported as an adverse event associated with this device, nor is it mentioned in the official product information. While the resulting scar was small, measuring only a few millimeters, it is important to note that even minor visible marks can be psychologically distressing to patients, particularly when they occur in visible areas such as the upper arm or as an unexpected outcome of a seemingly minor procedure. In contrast, venipuncture at the cubital fossa rarely leads to scarring, even with repeated blood draws from the same site. Needle punctures typically heal with minimal scarring compared to blade-incisions, highlighting how the method and site of blood collection can significantly impact cosmetic outcomes. Although capillary blood-taking devices have been marketed as pain-free, it should be noted that they still incise or puncture the skin.
As self-sampling devices become increasingly integrated into clinical and research workflows, an evaluation of their benefits and risks is essential. Recent studies have demonstrated good analytical agreement between capillary self-sampled and venous blood measurements for many laboratory parameters, and overall low levels of pain 3 and high user acceptability have been reported in primary care settings.4,5 However, these studies did not systematically assess device-related adverse events. In one study conducted in 2021, 6 out of 60 participants (10.0%) reported problems with the healing process, and five of them (8.3%) developed a scar as a secondary outcome. 7 Since then, device designs and materials could have changed compared to our study conducted in 2024. To better understand the current frequency and clinical relevance of such cutaneous complications, adequately powered studies specifically designed to assess these outcomes are needed. In this context, the Blood-it-yourself study contributes additional real-world evidence, with feasibility results already published 4 and analyses on analytical accuracy currently in preparation.
The occurrence of scarring in this case may be attributed to the incision approach of this device using a single blade (Figure 2(a)). The horizontal incision on the upper arm is perpendicular to both Langer’s lines (topological lines of the human skin that correspond to the natural orientation of collagen fibers in the dermis) and relaxed skin tension lines. 8 The underlying tension may lead to scar widening and deterioration. 9 When incisions are made across Langer’s lines, they are prone to dehiscence and may result in more pronounced scarring. This principle underscores the importance of considering skin tension patterns in both surgical planning and medical device design.

Mechanisms of incision for capillary blood drawing: (a) Tasso+ device with a single blade and (b) TAP II using a circular microneedle array.
It is worth noting that other self-blood collection devices, such as the “Touch Activated Phlebotomy” TAP® system (YourBio Health Inc., Medford MA, USA), utilize an array of microneedles (Figure 2(b)) resulting in a circular incision to draw capillary blood. 10 At present, it is unclear whether this approach leads to fewer or less noticeable scars compared to a single linear incision.
The incident was reported to the responsible health authority, the German Federal Institute for Drugs and Medical Devices (BfArM), under reference number AZ 20035/24. Following their assessment, the institute recommended that the manufacturer provide more detailed descriptions of potential complications in their patient information materials.
This case raises important questions about the design of self-blood collection devices and the need to consider skin tension lines in their development. It also highlights the importance of comprehensive adverse event reporting in the post-market surveillance of medical devices. While the individual burden of this small scar may seem minimal, it is crucial to consider both the potential cumulative impact on a population level and the variability at the individual level. For instance, with quarterly blood draws, the upper arm may be at risk to become significantly scarred. The individual burden can also vary greatly, making it essential that patients are informed in advance about the possibility of scarring. If these devices were introduced on a large scale, even a low incidence of scarring could translate into a significant overall burden. In conclusion, the report documents a previously undocumented complication of a self-blood collection device. The horizontal incision design, which runs perpendicular to Langer’s lines on the upper arm, may increase the risk of wound gaping and subsequent scarring. Although the resulting scars in such cases are typically small and tend to fade over time, they may still be distressing for some individuals, particularly when unexpected. Healthcare providers and researchers should therefore be aware of this potential complication and consider including it in patient information and consent processes. Further research is warranted to determine the frequency of this event and whether design modifications could mitigate this risk.
Footnotes
Acknowledgements
The authors would like to acknowledge the support of Christopher Schuchardt who helped with patient recruitment and sample analyses.
Author contributions
FM: Conceptualization, Funding acquisition, Methodology, Project administration, Writing—original draft, Writing—review & editing. AH: Data curation, Figures, Writing—review & editing. JS: Writing—review & editing. EH: Writing—review & editing. LK: Writing—review & editing. PB: Patient treatment, Writing—review & editing. DS: Methodology, Project administration, Writing—review & editing.
Data availability statement
The dataset presented in this article are not readily available due to ethical/privacy restrictions. Requests to access the datasets should be directed to the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Blood-it-yourself study as part of the Blut-Mobil project was funded through the European Social Funds (ESF), Grant No.: ZAM 5-87006761. The funder had no role in the design, data collection, data analysis, and reporting of this study.
Ethical approval
This study protocol was reviewed and approved by the Research Ethics Board of University Medical Center Göttingen, approval number 23/12/23.
Consent to publish
Written informed consent was obtained from the respective participant for publication of the details of their medical case and any accompanying images.
Clinical trial registration
The study was registered in the German Register for Clinical Trials (DRKS00033172).
