Abstract
Background and objectives:
Central venous catheters are recommended for chemotherapy administration, but peripheral intravenous catheters are frequently used. This study examined venous access practices (central vs peripheral) for administering antineoplastic agents to patients with solid or hematologic malignancies and assessed the associated educational aspects.
Design, setting, and participants:
A questionnaire developed according to the Enhancing the Quality and Transparency of Health Research Guidelines was distributed to Italian hematology/oncology nurses who performed intravenous chemotherapy administration and peripheral vein cannulation. Of 171 questionnaires returned, 141 were completed.
Results:
Statistics showed that 33% of respondents used peripheral veins for at least 25% of patients within 30-day period. Peripheral veins were chosen because of organizational challenges, urgency, and central catheter issues or malfunctions. Irritant and vesicant drugs were administered by 60% and 49.3% of respondents. The study found 81.7% of nurses received additional education on chemotherapy administration. While 95.8% of nurses informed patients about chemotherapy side effects and extravasation signs, 77.5% did not document this in patients’ charts. Informed consent forms often lacked specifications for peripheral vein administration (63.9%), essential for safe antineoplastic agent administration.
Conclusions:
The survey highlighted the need for standardized guidelines for intravenous chemotherapy, as peripheral veins serve multiple purposes.
Keywords
Introduction
Intravenous antineoplastic agent administration is essential for treating solid and hematological neoplasms. It can be infused into central or peripheral veins. Vascular devices for chemotherapy delivery are divided into central vascular access devices (CVADs), 1 including peripherally inserted central catheters (PICCs),2,3 Hickman-type tunneled catheters (e.g. Hickman), totally implanted ports (PORTs), 2 and short peripheral intravenous catheters or cannulas (PIVCs), inserted in forearm veins. 4 Short PIVCs are common among cancer patients. In Italy, peripheral catheterization is performed by various healthcare professionals, including registered nurses. The tools are readily obtainable and cost-effective, while catheter insertion is typically rapid. 5 Choosing between CVAD or PIVC depends on considerations, including infusion pharmacological properties and patient-specific factors like vascular health, geographical location, access to outpatient facilities for device maintenance, and expected treatment duration.6,7 For vesicant medications using a short PIVC, the device should be newly inserted and removed post-infusion. The infusion should be brief and supervised by medical personnel. 8 A study on vesicant and irritant chemotherapy extravasation at National Cancer Institute (NCI)—designated cancer centers found events occurred more frequently with PIVC than with CVAD. 9 This indicates the importance of proper antineoplastic agent infusion management. Italian registered nurses receive comprehensive training in short PIVC insertion and general infusion management during internship and work placement. Nonetheless, nursing staff must understand specific departmental drugs like antineoplastic agents. Nurses must be also trained to manage emergencies, such as irritant or vesicant drug extravasation, and patients must be trained by nursing staff to recognize extravasation signs and symptoms.10–12
Methods
Purpose
This study aimed to determine the current practice of selecting venous access (central vs peripheral) for administering antineoplastic agents in patients with hematologic and solid malignancies and to assess specific education on this topic. A questionnaire was developed, and a survey was conducted among hematology/oncology nurses in Italy to estimate the frequency of administering antineoplastic agents for hematologic and solid malignancies via short PIVC in routine clinical practice and to explore the reasons for this practice. The secondary objectives were to evaluate the additional education and training provided to nursing staff and to identify educational needs on this topic.
Study design and setting
This observational cross-sectional study followed the Enhancing the Quality and Transparency of Health Research (EQUATOR) guidelines and Checklist for Reporting of Survey Studies (CROSS). 13
In April 2021, a voluntary survey was created using Google Forms. The link was emailed to head nurses of 50 hematology/oncology divisions associated with the Nursing Group of the Italian Lymphoma Foundation (Fondazione Italiana Linfomi (FIL)). 14 A letter explaining the survey objectives invited registered nurses involved in short PIVC insertion and intravenous chemotherapy administration to participate. Head nurses forwarded the link to their staff via email, which was used solely for this purpose. Participants completed the questionnaire anonymously without providing personal information, ensuring that the researchers could not identify them. The questionnaire was developed by an expert committee comprising board-certified physicians and registered nurses from the Hematology Division of Fondazione IRCCS Istituto Nazionale Tumori (INT), Milano, Italy, and approved by the Heads of the Division of Hematology, Health Care Profession Management Division of the Fondazione IRCCS INT, and the Chief Medical Officer of FIL. Registered nurses need only the capability of PIVCs insertion and intravenous antineoplastic drug administration to participate, representing Italian oncology–hematology nurses. No method adjusted for sample non-representativeness, and there was no predefined sample size. The questionnaire included 30 multiple-choice questions in three sections: (1) demographic information, education and work experience, and work setting (questions 1–9), (2) specific education on chemotherapy administration to cancer patients (questions 16–30), and (3) reasons for selecting central or peripheral intravenous catheters for chemotherapy administration (questions 10–15). The survey took ~15 min to complete. Responses were collected between April 26 and May 17, 2021, with a 3-week response window. A copy of the survey is available in the Supplementary Materials.
Data collection
Survey data were summarized using descriptive statistics, showing the results as absolute frequencies (number of cases) and percentages (%) for categorical variables. Percentages were calculated based on the number of participants who answered each question (i.e. participants who did not visualize or answer a specific question were excluded from the denominator). Data were extracted and analyzed using Microsoft Excel Professional Plus 2019 sheets (Microsoft Corp.) Sensitivity analysis was not performed.
Ethical considerations
The survey received email approval on April 15, 2021, from the Head of the Division of Hematology (also the Scientific Vice-Director) and Head of the Health Care Profession Management Division at Fondazione IRCCS INT, Milano. Subsequently, the Chief Medical Officer of FIL approved the questionnaire via email on April 23, 2021. Participation was anonymous and voluntary, with individual participant data remaining inaccessible to the research team. By accessing the online link, the nurses automatically consented to participate in the survey.
Results
The survey included 171 registered nurses from 50 hospitals across Italy. Of these, 141 questionnaires were fully completed and 30 were partially filled. Table 1 details the participants’ sociodemographic, educational, and training characteristics. As the questionnaire was distributed internally by participating divisions to the nursing staff, calculating the response rate was not possible.
Main sociodemographic characteristics of the participants in the multicenter population survey.
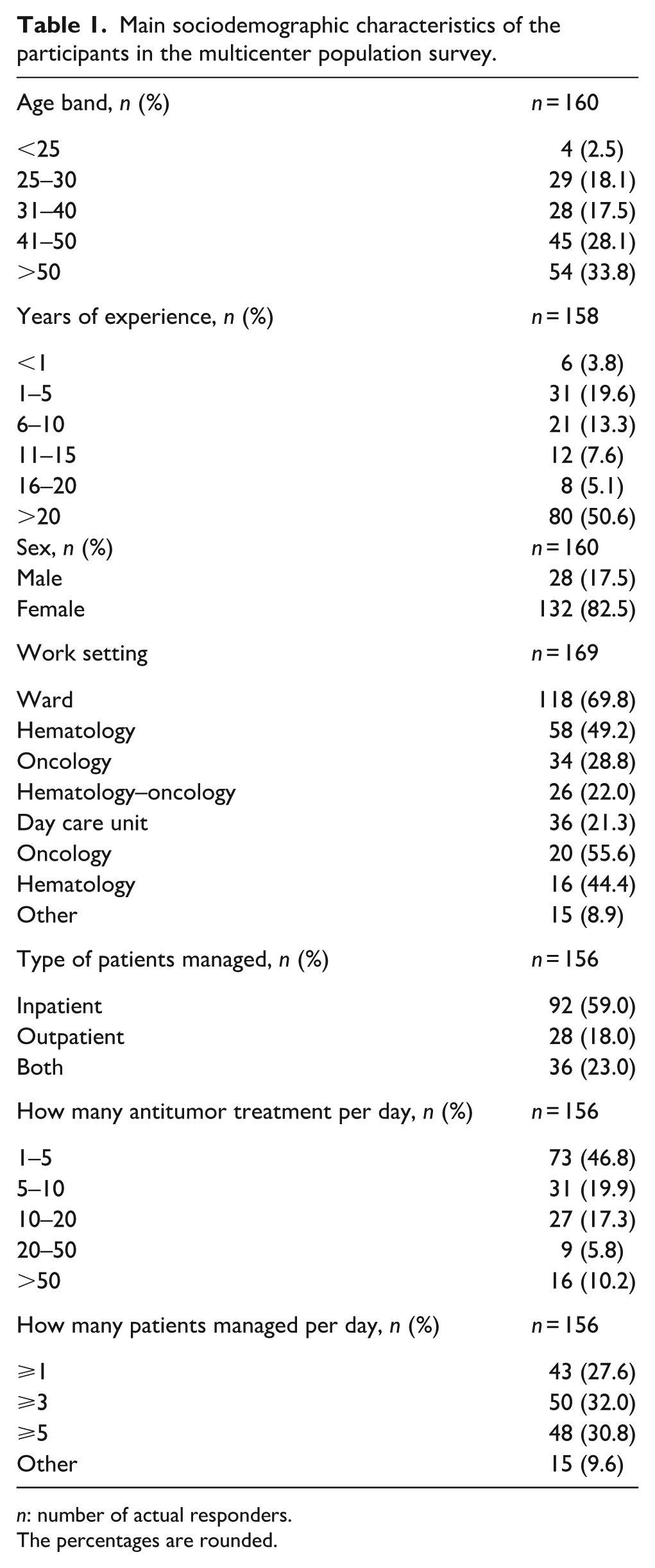
n: number of actual responders.
The percentages are rounded.
Most respondents were female (82.5%), over 30 years old (79.4%), with more than 20 years of experience in about half of the sample, and over a third had 10 or fewer years of experience.
Regarding nurses’ training for intravenous anticancer therapy, 116 out of 142 respondents (81.7%) received specific training on chemotherapy administration through hands-on and frontal teaching (77 out of 115 respondents, 67%). However, 47 of the 142 respondents (33.1%) lacked specific training in selecting the most appropriate vascular access device (VAD). Additionally, 26 of the 142 respondents (18.3%) lacked specific training in handling extravasation. Meanwhile, 138 out of 141 respondents (97.9%) considered training in chemotherapy administration a very high or high priority, especially among newly hired nurses.
Irritant and vesicant chemotherapeutic agents 15 were infused via PIVCs in 60% and 49.3% of cases, respectively.
The responses also highlighted a lack of standardization in managing extravasation; 88 out of 142 respondents (62.0%) reported that no multidisciplinary team was available to handle this adverse event in their hospital.
Regarding the choice of central versus peripheral venous access for anticancer therapy, although CVADs were more commonly used (Table 2), a large proportion of patients received chemotherapy through peripheral veins (>75% of patients treated in the last 30 days for 11.8% of respondents, >50% for 12.4%, and 25%–50% for 13.7%). However, it was not possible to determine the number of patients with hematological malignancies or solid tumors who received chemotherapy via the peripheral vein. The reasons for using a peripheral vein, in decreasing order of frequency, were as follows: (1) hospital organizational issues, (2) urgent initiation of antitumor treatment, (3) non-functioning CVAD, (4) disease-related obstacles to CVAD placement, (5) administration of mobilizing chemotherapy (cyclophosphamide), and (6) other reasons (Table 3).
Administration of chemotherapy through a central versus peripheral venous access over a 30-day period.
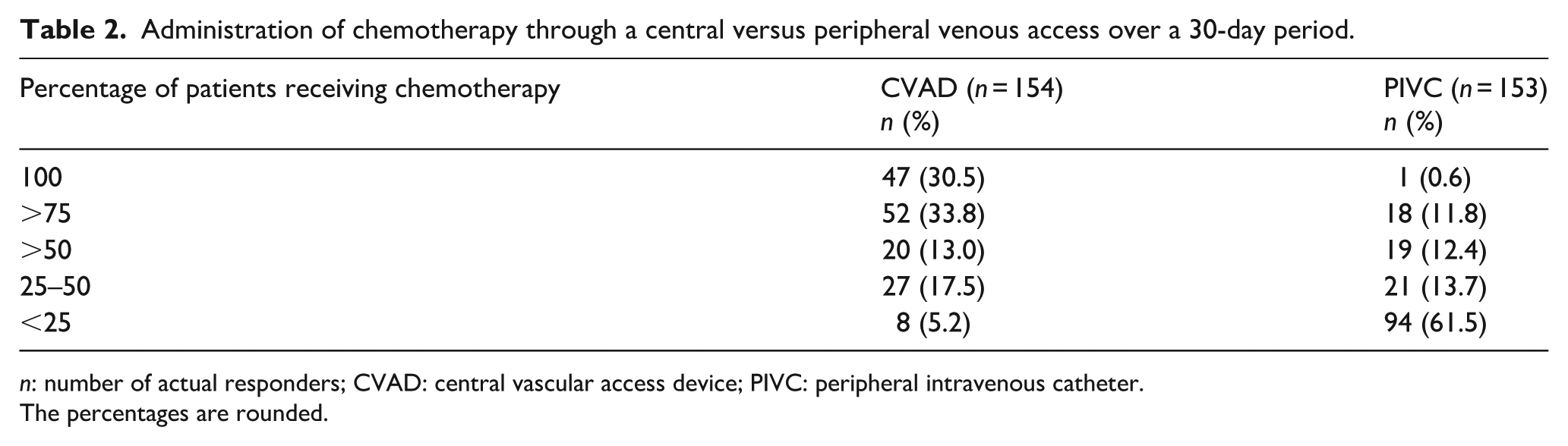
n: number of actual responders; CVAD: central vascular access device; PIVC: peripheral intravenous catheter.
The percentages are rounded.
Reasons for delivering chemotherapy through a peripheral venous access.
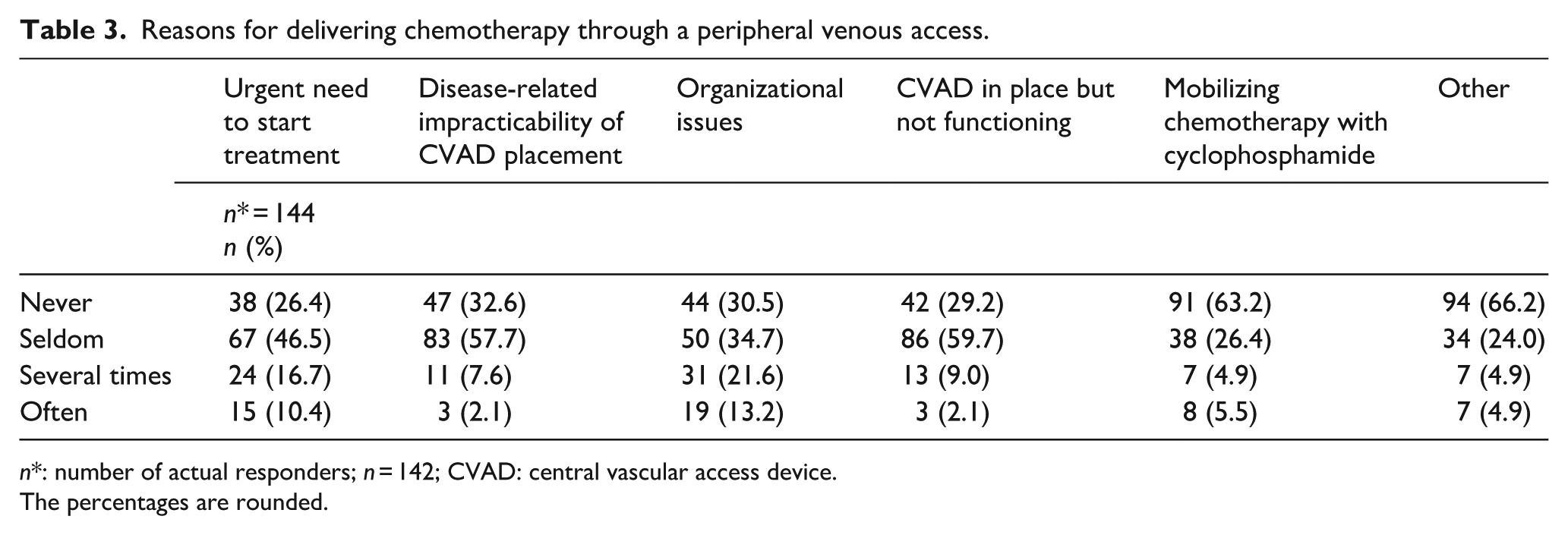
n*: number of actual responders; n = 142; CVAD: central vascular access device.
The percentages are rounded.
Regarding chemotherapy patient education on extravasation signs and symptoms, most respondents (135 out of 141, 95.7%) perceived this as a very high or high priority and performed it (136 out of 142, 95.8%). Only six respondents did not consider patient education to be important. However, only 32 out of 142 respondents (22.5%) documented patient education in the patient charts. Furthermore, 92 out of 144 respondents (63.9%) reported that the Informed Consent Form did not mention chemotherapy delivery through peripheral veins.
Discussion
Appropriate vascular access selection is essential for safe antineoplastic agent administration, with nurses playing a key role. Various societies have developed venous access catheter selection algorithms.16–18 Moreover, international guidelines from organizations like the Oncology Nurses Society (ONS), 19 Infusion Nurses Society (INS), 6 and World Conference on Vascular Access (WoCoVA), 8 establish CVADs as the standard of care for vesicant and irritant chemotherapy. Conversely, PIVCs are considered inappropriate except in rare circumstances, though patients with hematologic malignancies might require both CVAD and PIVC during treatment. 20 Therefore, nurses must assess PIVC appropriateness when starting chemotherapy according to international standards. Despite recommendations, our survey found that PIVCs are still used for vesicants and irritant agents, mainly due to organizational reasons or urgent treatment needs. This practice deviates from evidence-based standards and increases the risk of complications. Furthermore, a recent study by Faccini et al. 21 confirmed that non-central devices, including midlines, have higher failure rates and adverse outcomes, confirming peripheral access inadequacy for chemotherapy. Consequently, our findings highlight the need to revise the ongoing use of this practice in light of international standards and patient safety issues.
Sometimes, patients decline CVAD insertion due to concerns about invasiveness or complications and opt to receive antineoplastic agents through short PIVC at their request. This situation is documented in the literature, 22 showing that patient preferences can impact the choice of vascular access. However, this has not been thoroughly examined in the current study.
Aligned with the study’s secondary aims of evaluating standardized courses for Italian oncology nurses, given no certification requirements, questions were designed to determine whether nurses had received additional ward or theoretical training in the following areas: (1) short PIVC insertion, (2) management of antineoplastic agent infusions, (3) venous access device selection in oncology and hematology patients, and (4) extravasation management.
The survey showed that training on safe antitumor treatment was perceived as a very high or high priority by most respondents. However, 18% and 33% of respondents had not received training on intravenous chemotherapy administration and vascular access choice, respectively. Additionally, 18% reported no training in handling chemotherapy extravasation. Contrary to these findings, recent literature reaffirms the necessity of specialized and continuous training in administering antineoplastic and immunotherapy agents for patient safety. 23
Survey data also showed that 96% of respondents provided patient education, but only 22.5% recorded it in the patient charts. This suggests that the information was provided orally; however, recording is important from a legal and insurance perspective. Additionally, patient education is a vital nursing responsibility and ONS standard of care. 24 Expected chemotherapy side effects comprise major educational content for cancer patients. 25 Early recognition of extravasation signs and symptoms following chemotherapy via the peripheral vein should be included so that patients can seek timely intervention, if necessary. 26 Thus, our survey highlighted the lateral underestimation of patient education.
Finally, informed consent forms should state that patients were informed about the risks of antineoplastic agent infusion through peripheral veins; however, only 36% of respondents reported this. Nurses should note that omitting this information may lead to legal issues.
Limitations
This study had limitations, and results should be interpreted cautiously. Given limited research on antineoplastic drug administration via short PIVC in patients with hematologic and solid malignancies, this study represents a novel contribution. The sample size indicates the need for further international studies to confirm these findings. As the questionnaire was anonymous, responses from the same hospital could not be identified. Most respondents trained and worked in Italy, limiting the generalizability of the findings to other countries. Despite its limitations, this survey can help identify areas for improving patient safety.
Implications
This study revealed a gap between the guidelines for administering antineoplastic agents through intravenous access and actual practice. A difference also exists in chemotherapy administration training among nurses, indicating the need for structured training for all oncology nurses, as recommended.5,27 In fact, the reasons nurses cited for choosing peripheral access (organizational barriers, malfunctioning CVAD, urgency, and patient-related issues) are not clinically acceptable alternatives when vesicant or irritant agents are administered but reflect organizational failures that expose patients to risk and create medico-legal implications for healthcare professionals using PIVCs. Healthcare institutions should implement standardized vascular access algorithms aligned with guidelines, ensure multidisciplinary vascular access teams, and establish accountability frameworks for device selection and documentation. Informed consent forms should specify the type of venous access and associated risks. By addressing these gaps through education, protocols, and documentation, decision-making can become more transparent and safer, improving patient outcomes and efficiency.
Based on the survey results and other organizational factors, Fondazione IRCCS INT, Milano, assembled a multidisciplinary team in 2024 to revise the internal procedures for VAD selection, placement, and management. This ongoing initiative aims to develop a clear VAD selection process and ensure prompt CVAD placement for new treatments, particularly irritant or vesicant drugs. Additionally, Fondazione INT has revised and is promoting new educational courses for nurses on CVADs guidelines, antineoplastic agent administration, and patient education.
Conclusions
This cross-sectional study consisted of an online survey of registered nurses working in 50 hematology/oncology divisions affiliated with the Nursing Group of the FIL, involved in short PIVC insertion and intravenous administration of antineoplastic agents. This confirms discrepancies between the literature recommendations for using CVAD for chemotherapy administration and routine PIVC use. Additionally, the survey demonstrated a lack of specific training for nurses administering intravenous chemotherapy and insufficient documentation of patient education on extravasation sign and symptom detection. It is also evident that informed consent does not always report that chemotherapy will be provided through a peripheral intravenous catheter. These findings highlight the need for structured additional training for nurses on this issue.
Supplemental Material
sj-docx-1-jva-10.1177_11297298251398418 – Supplemental material for Selection of central versus peripheral vascular access in patients receiving chemotherapy: A survey among Italian nurses promoted by the Italian Lymphoma Foundation
Supplemental material, sj-docx-1-jva-10.1177_11297298251398418 for Selection of central versus peripheral vascular access in patients receiving chemotherapy: A survey among Italian nurses promoted by the Italian Lymphoma Foundation by Lucia Saracino, Serena Togni, Mariangela Cieri, Rosita Bianco, Federica Olivazzi, Daniela Manzo and Giuliana Nepoti in The Journal of Vascular Access
Footnotes
Acknowledgements
The authors thank all the nurses who participated in the survey. We also acknowledge Dr. Anna Guidetti, MD, from the Hematological and Medical Oncology Division at the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy, for preparing the questionnaire. We also acknowledge Ministry of Health for its support in partially financing the publication.
Author contributions
All authors have read and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research or authorship of this article. The authors received financial support from Bibliosan and Ministry of Health, Ricerca Corrente funds, solely for the publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
