Abstract
Background:
The impact of different percutaneous femoral access closure strategies on mortality and bleeding after transcatheter aortic valve replacement (TAVR) has been less studied. The aim of this study is to investigate whether different percutaneous femoral access closure strategies might affect systemic outcomes in patients undergoing TAVR.
Methods:
In this systematic review and meta-analysis, randomized trials comparing a suture-based closure (suture group) with a plug-based or suture/plug-based closure (control group) for hemostasis of the femoral access in patients undergoing TAVR were included. The primary endpoint of the study was all-cause death. Secondary endpoints included any bleeding (defined according Valve Academic Research Consortium (VARC) criteria) and unplanned endovascular or surgical repair at the access site. All endpoints were calculated at the longest available follow-up.
Results:
Four studies with a total of 1429 TAVR patients (suture-based group, n = 707; control group, n = 722) were available for quantitative synthesis. There was no statistically significant difference in all-cause death in patients who underwent TAVR with suture-based closure compared to the control group (risk ratio—RR = 1.04, 95% confidence interval—CI 0.60–1.82; p = 0.83). Similarly, there was no statistically significant difference between treatment groups for any bleeding and unplanned endovascular or surgical repair at the access site. However, there was a significant treatment effect for VARC bleeding type ⩾2 associated with the use of a plug-based or suture/plug-based closure in the control group (p for interaction = 0.01).
Conclusions:
In the context of contemporary TAVR procedures, percutaneous closure of the femoral access using any suture or plug-based strategy has no impact on hard clinical outcomes such as death from any cause.
Keywords
Introduction
Percutaneous transcatheter aortic valve replacement (TAVR), also referred to as transcatheter aortic valve implantation (TAVI), is a cornerstone of the treatment of patients with severe aortic valve stenosis, regardless of baseline risk profile. The common femoral artery access has been shown to be superior to alternative access sites and remains the percutaneous approach of choice for patients undergoing TAVR in routine clinical practice. 1 The hemostasis of the femoral access plays an important role in the TAVR procedure since it contributes to TAVR success across the entire risk spectrum of patients. In this regard, despite the considerable improvement in TAVR techniques and vascular access management observed in the last decade, vascular complications continue to occur in a non-negligible proportion of patients. 2 This is of utmost importance, as access-related vascular complications after TAVR are strongly associated with increased morbidity and mortality, thereby significantly impacting hard clinical outcomes.3 –5
Several strategies for the percutaneous closure of the femoral access after TAVR have been subject to clinical investigation in recent years.6 –8 In particular, the use of a suture-based closure (ProGlide™/ProStyle™, Abbott Vascular, Abbott Park, Illinois, USA), a plug-based closure (MANTA, Teleflex, Wayne, Pennsylvania, USA), or a hybrid suture/plug-based closure (ProGlide™/ProStyle™, Abbott Vascular, Abbott Park, Illinois, USA and Angio-Seal®, Terumo Interventional Systems, Somerset, New Jersey, USA) in patients undergoing transfemoral TAVR has been compared in randomized trials mostly powered for composite outcomes related to vascular access.9 –12 However, the impact on mortality and bleeding of different percutaneous strategies for the closure of femoral access after TAVR remains less studied or controversial. Therefore, this systematic review and meta-analysis was performed to investigate whether different percutaneous femoral access closure strategies might affect systemic outcomes of patients undergoing TAVR.
Methods
Search strategy, qualitative assessment, and outcomes of interest
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines (Supplemental Appendix A). Electronic scientific databases (MEDLINE, EMBASE, and Cochrane), scientific abstracts of major cardiovascular conferences, and clinical trial registration websites were searched from the inception of each database until March, 2025. The search was for randomized trials comparing vascular closure strategies for hemostasis of the femoral access in patients undergoing TAVR. All eligible studies were screened for further citations by reviewing the reference lists. Search terms included the keywords and corresponding Medical Subject Headings for: “femoral access,” “vascular closure device,” “TAVR,” “TAVI,” “plug,” “suture,” “trial,” and “randomized trial.” The full search term strings used for the database search are available in Supplemental Appendix B. The inclusion criteria for further assessment were: (1) TAVR via femoral access; (2) random allocation to either a suture-based or a mixed suture/plug-based vascular closure for hemostasis of the femoral access after TAVR; and (3) completion of the trial with ⩾30-day clinical follow-up. Patients receiving plug- and/or suture-based vascular closure devices for indications other than hemostasis of the femoral access after TAVR were ineligible for inclusion in the meta-analysis. Two investigators (FS and SC) independently assessed publications for eligibility at the title and/or abstract level. A third investigator (EX) helped resolve any discrepancies. Studies that met the inclusion criteria were subject to further analysis. The risk of bias was evaluated independently for each study, in accordance with the Cochrane risk-of-bias tool for randomized trials version 2 (RoB2) to assess the quality of included trials. 13 Composite quality scores were not assigned. 14
The primary endpoint of this study was all-cause death. Secondary endpoints included any bleeding (defined according Valve Academic Research Consortium (VARC) criteria) and unplanned endovascular or surgical repair at the access site. No protocol was prospectively registered for this study.
Quantitative synthesis
Trial-level data concerning the overall number of patients, mean age, and proportions according to male sex, type 2 diabetes, arterial hypertension, and prior myocardial infarction (MI) were extracted from each trial for descriptive purposes. Risk ratios (RRs) with a 95% confidence interval (95% CI) and p-value <0.05 were used to compare outcomes of interest between treatment groups. The study-level risk estimates were pooled using the Mantel-Haenszel random-effects model with Hartung-Knapp adjustment. The I2 statistic and a chi-square test quantified between-study heterogeneity, whilst between-study variance was measured using the Paule-Mandel estimator for tau 2 . Importantly, the use of the Paule-Mandel method or estimating tau 2 in combination with the Hartung-Knapp adjustment broadens the CIs for risk estimates, allowing for a better assessment of statistical uncertainty. 15 An influential analysis of the primary outcome was performed, calculating the pooled estimates by omitting one study at a time. A chi-square test for subgroup differences was used to evaluate treatment effect modification for the primary and secondary outcomes, conditional on the use of plug-based or suture/plug-based closure in the control group. The same analysis was also performed for bleeding VARC type ⩾2. For all outcomes, the 95% prediction interval of the pooled estimates was displayed. The impact of publication bias on the primary outcome was investigated by visual inspection of the funnel plot and by using Egger’s linear regression test, which is a statistical method used to detect and measure publication bias in scientific studies. All endpoints were calculated at the longest available follow-up according to definitions contained in the protocols of original trials. All analyses were performed using the packages meta in R (version 4.1.3; R Foundation for Statistical Computing, Vienna, Austria).
Results
The flow for the trial selection process is shown in the Figure 1 and the results of the risk of bias evaluation are reported in the Supplemental Appendix C. Four randomized controlled trials comparing a suture-based closure (suture-based group) versus a plug-based or a suture/plug-based closure (control group) for hemostasis of the femoral access in patients undergoing TAVR were included. Among the screened randomized controlled trials, one was excluded because it compared a surgical access technique for TAVR, 16 and another was excluded because it studied a suture-based vascular closure device that was different from the ones under study here. 17 The four randomized trial included in this analysis were: the Comparison of Strategies for Vascular ACCESS Closure after Transcatheter Aortic Valve Implantation 9 (ACCESS-TAVI; suture-based, n = 224 vs suture/plug-based, n = 230), the Randomized Comparison of Catheter-based Strategies for Interventional Access Site Closure during Transfemoral Transcatheter Aortic Valve Implantation 11 (CHOICE-CLOSURE, suture-based, n = 258 vs plug-based, n = 258), the MANTA versus Suture-based vascular closure after transcatHeter aortic valve replacement 12 (MASH, suture-based, n = 104 vs plug-based, n = 102), and the study from Yeh et al. 10 (suture-based, n = 121 vs suture/plug-based, n = 132). Table 1 presents an overview of the main features of the original trials included in the present analysis.
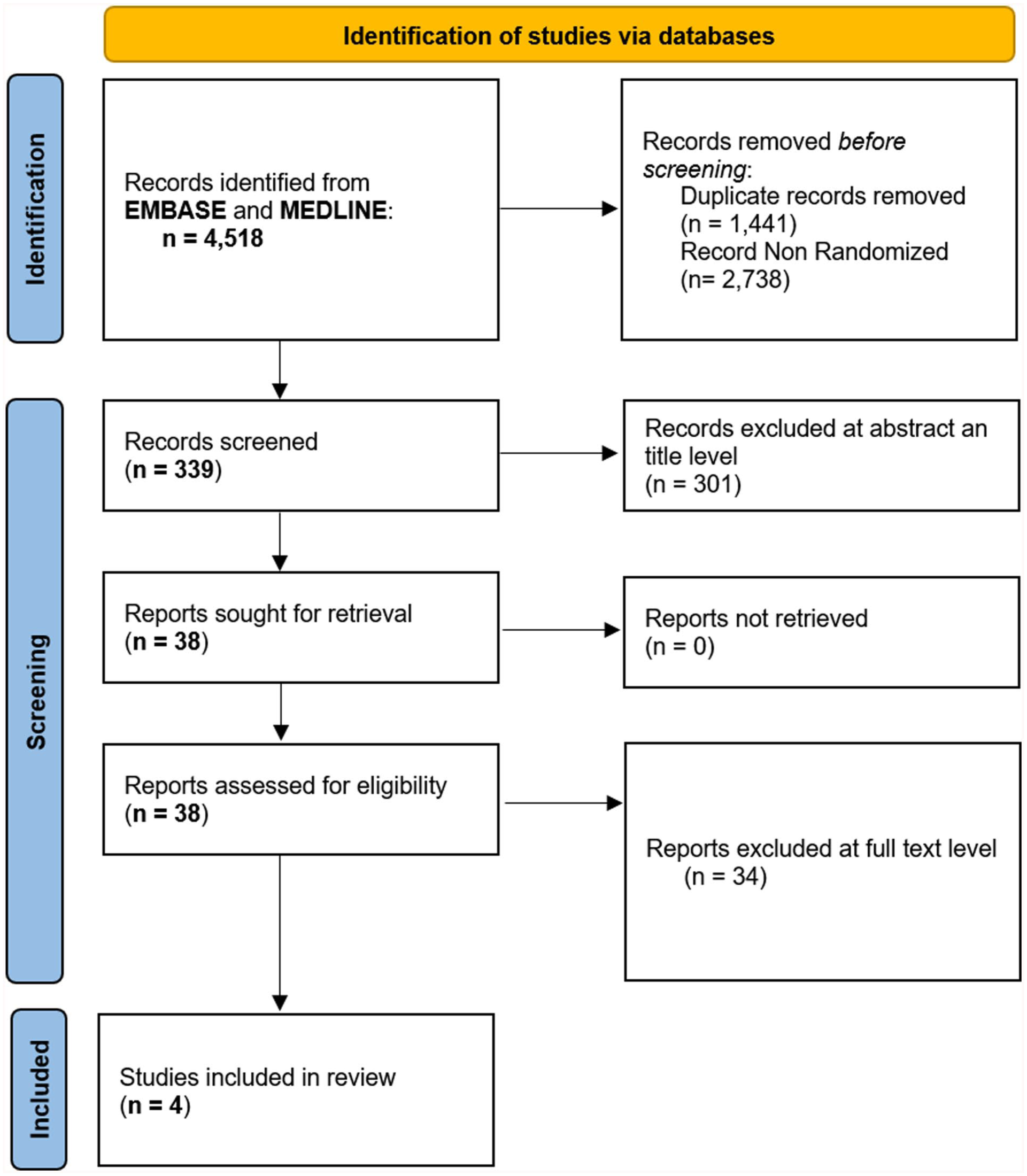
PRISMA 2020 flow chart for study selection.
Baseline features of patients included in each arm of the original trials.
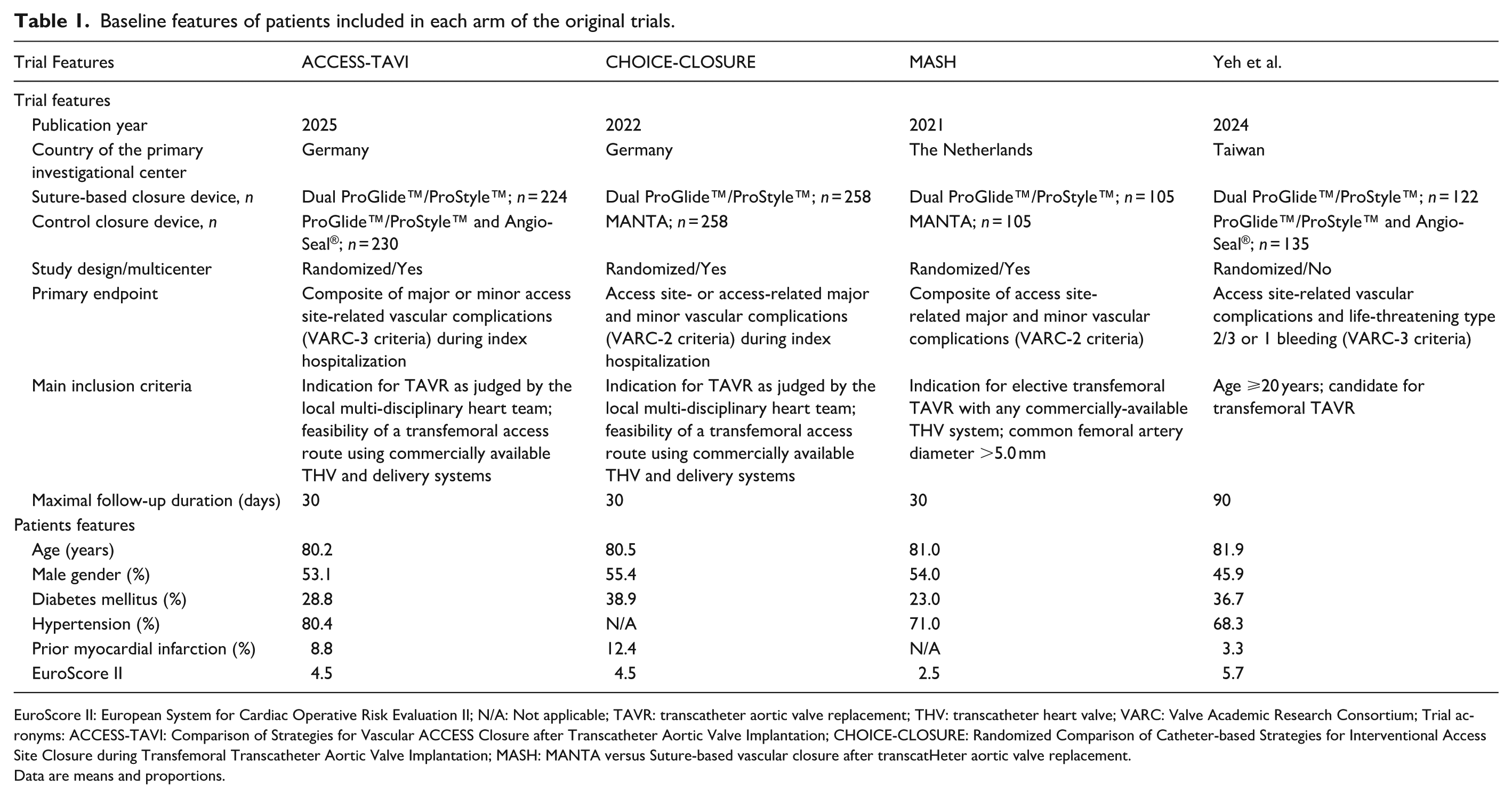
EuroScore II: European System for Cardiac Operative Risk Evaluation II; N/A: Not applicable; TAVR: transcatheter aortic valve replacement; THV: transcatheter heart valve; VARC: Valve Academic Research Consortium; Trial acronyms: ACCESS-TAVI: Comparison of Strategies for Vascular ACCESS Closure after Transcatheter Aortic Valve Implantation; CHOICE-CLOSURE: Randomized Comparison of Catheter-based Strategies for Interventional Access Site Closure during Transfemoral Transcatheter Aortic Valve Implantation; MASH: MANTA versus Suture-based vascular closure after transcatHeter aortic valve replacement.
Data are means and proportions.
Overall, a total of 1437 participants were enrolled in the original trials, and a number of 1429 (99.4%) TAVR patients were available for quantitative synthesis (suture-based group, n = 707; control group, n = 722). The baseline features of patients included in each arm of the original trials have been presented elsewhere9 –12 and are summarized in Table 1. The population aggregated for the present analysis had a median age of 80.7 years (80.3–81.4), more than half were males, one-third had diabetes mellitus, and nearly two-thirds were hypertensive. The mean European System for Cardiac Operative Risk Evaluation II score was 4.3 ± 1.3. The weighted median follow-up duration was 30 days.
Patients undergoing TAVR displayed no statistically significant difference in terms of all-cause death associated with a suture-based closure as compared to control (2.3% vs 2.3%; RR = 1.04, 95% CI 0.60–1.82; p = 0.83; Figure 2A). Similarly, there was no statistically significant difference in terms of any bleeding (18.0% vs 21.1%; RR = 0.86, 95% CI 0.63–1.17; p = 0.22; Figure 2B) and unplanned endovascular or surgical repair at the access site (8.9% vs 9.6%; RR = 0.91, 95% CI 0.40–2.04; p = 0.73; Figure 2C) between treatment groups. There was a moderate statistical heterogeneity for this latter outcome. Of interest, only one 12 out of four trials reported the surgical treatment of four access-site complications. Despite the limited value of the linear regression test due to the small number of trials available, no statistical evidence of publication bias for the primary outcome was found (p = 0.81). Consistently, the funnel plot displayed no asymmetrical distribution of included trials (Supplemental Appendix D). The influential analysis ruled out the possibility of imbalanced influence from any of the included trials on the pooled estimates for the primary outcome (Supplemental Appendix E).
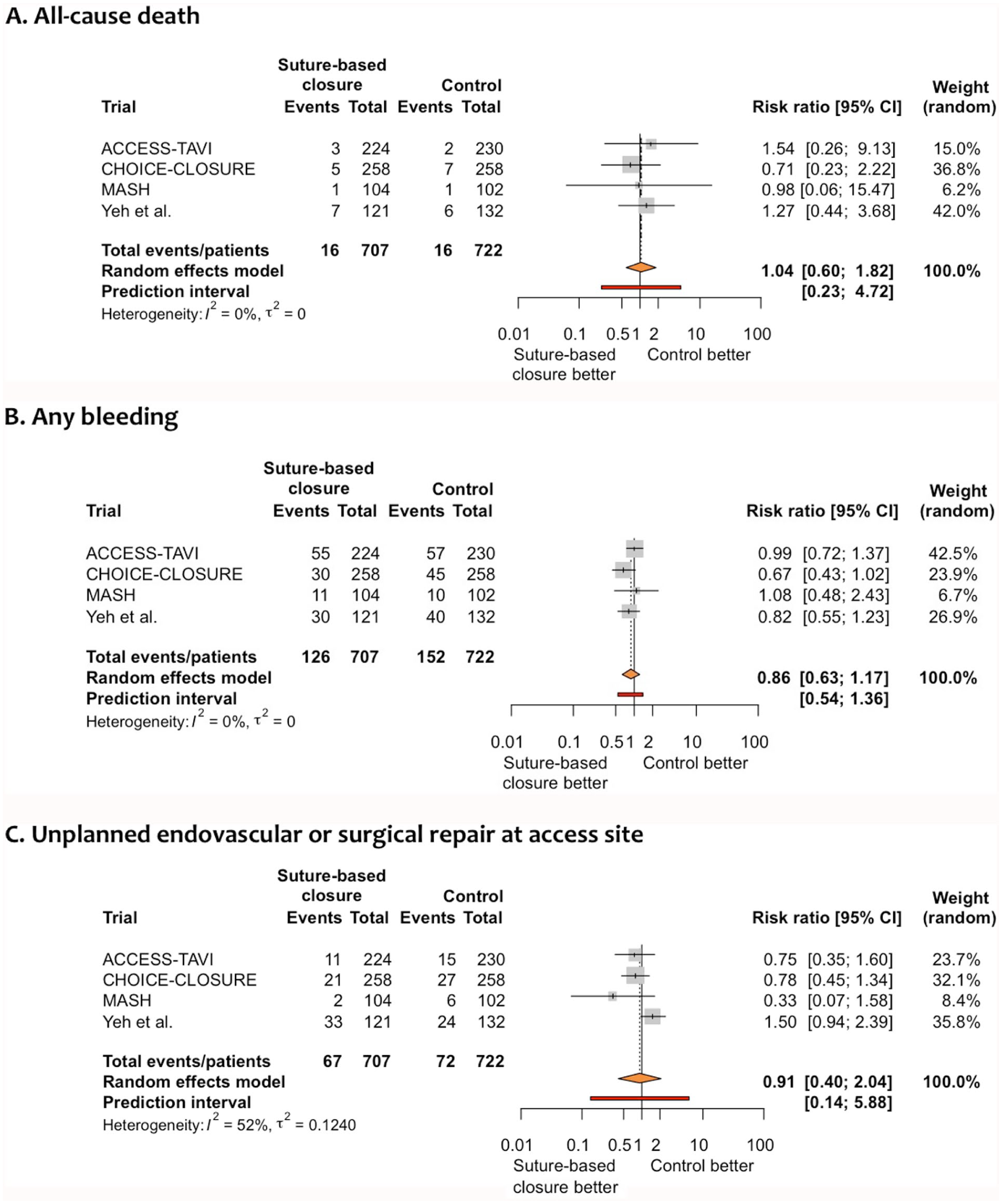
Forest plot of summary risk ratios for outcomes of interest associated with a suture-based closure versus control. The control group includes participants allocated to either a plug-based or a suture/plug-based closure. Trial acronyms are reported in the Table.
There was no significant change in the direction of the risk estimates for all outcomes of interest associated with a suture-based closure conditional to the use of a plug-based or a suture/plug-based closure in the control group (p ⩾ 0.27). However, there was a significant treatment effect for bleeding VARC type ⩾2 associated with the use of a plug-based (RR = 0.67, 95% CI 0.43–1.02) or a suture/plug-based closure (RR = 1.55, 95% CI 0.93–2.57) in the control group (p for interaction = 0.01).
Discussion
This meta-analysis showed that, in TAVR procedures, percutaneous femoral access closure, whether performed with suture- or plug-based devices, does not impact hard clinical outcomes such as all-cause mortality.
This analysis has several methodological strengths. These comprise the inclusion of the largest randomized population in studies addressing the research topic, the selection of clinical outcomes less susceptible to bias, and the adoption of a more conservative statistical method for calculating summary estimates. The results of this analysis merit careful consideration.
Meticulous pre-procedural assessment of access site calcification and vascular anatomy, as well as the continuous iteration of percutaneous TAVR systems, have considerably improved the safety of contemporary TAVR procedures. The present analysis found around 2.0% mortality after a median follow-up of 30 days in a cohort of TAVR patients with an intermediate preoperative risk as per the European System for Cardiac Operative Risk Evaluation II score. This aspect is of the utmost importance given that US regulatory agencies have recently approved TAVR treatment for asymptomatic patients with severe aortic valve stenosis, 18 a fact which is expected to lead to an expansion of the range of indications in the years ahead.
Despite having no impact on mortality, was found that using a suture/plug-based closure rather than a suture-based closure affected the risk of bleeding VARC type ⩾2. This finding has also been observed with the use of a suture/plug-based femoral access closure in several other scenarios requiring large-bore arteriotomies, such as with mechanical circulatory support systems and endovascular aortic repair procedures. 19 Although the risk of subsequent adverse outcomes associated with access-site complications in patients undergoing TAVR remains undisputed, access-related complications (including bleedings) observed in contemporary TAVR practice are rare, do not affect short-term survival, and require endovascular management in most cases, with only a few cases requiring surgery.
Further investigation is required into the long-term clinical impact of suture- or plug-based closure strategies for femoral access in TAVR patients. The present analysis cannot provide information on the long-term effects on the local vasculature associated with the use of a specific vascular closure strategy after transfemoral TAVR. In fact, previous observations have suggested that the strain applied to the vessel wall of the femoral artery due to the use of more than one suture-based closure device may lead to stricture or fracture at a later stage. 19 However, this mechanistic, cannot be investigate perspective in the current report.
Study limitations: Some limitations have to be considered in the interpretation of the present data. Importantly, this is a meta-analysis of aggregated data: an analysis based on individual patient data is required to definitely rule-out whether differences exist between femoral access closure strategies according to baseline risk profile, vessel diameter, calcium distribution, puncture guidance, type of plugs, and vessel characteristics at computerized tomography scan. Additionally, the study was not prospectively registered in a protocol registry; therefore, beyond the authors’ statement, the risk of selective reporting bias cannot be objectively excluded. Of note, despite collecting outcomes data of almost 1500 patients undergoing TAVR, the overall incidence of death from all causes after 30 days remained low in absolute terms. On the one hand, this finding argues in favor of the safety of contemporary TAVR procedures. On the other hand, the present analysis remains underpowered to reveal the prognostic impact of any local strategy for arterial access closure after TAVR. Additionally, this analysis has a follow-up limited to the very short period after TAVR. A longer follow-up duration would be desirable. Finally, it is important to note that, particularly for bleeding outcomes, all four trials used a validated VARC classification. However, an update to the VARC criteria occurred during the publication period of the included trials. As a result, the two earlier studies adopted VARC-2, while the two more recent ones used VARC-3. This discrepancy may introduce some heterogeneity in the reporting of bleeding events; therefore, the results for this outcome should be interpreted with caution.
Conclusions
In contemporary TAVR procedures, percutaneous closure of the femoral access using any suture- or plug-based strategy does not affect hard clinical outcomes such as all-cause mortality. However, the use of a suture/plug-based closure appears to affect the risk of clinically relevant bleeding compared to a suture-based closure. Further investigation is required to determine the long-term clinical impact of a suture- or plug-based closure strategy for femoral access in TAVR patients.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298251389645 – Supplemental material for Percutaneous femoral access closure and prognosis following transcatheter aortic valve replacement: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-jva-10.1177_11297298251389645 for Percutaneous femoral access closure and prognosis following transcatheter aortic valve replacement: A systematic review and meta-analysis by Fiorenzo Simonetti, Salvatore Cassese, Tobias Rheude, Hector Alvarez-Covarrubias, Costanza Pellegrini, Adnan Kastrati, Michael Joner and Erion Xhepa in The Journal of Vascular Access
Footnotes
Abbreviations and acronyms
CI: confidence interval
RR: risk ratio
TAVI: transcatheter aortic valve implantation
TAVR: transcatheter aortic valve replacement
VARC: Valve Academic Research Consortium
Author contributions
TR, MJ, EX, and SC were involved in study conception and design. SC performed the data analysis. MJ supervised the data analysis. SC together with FS, TR, MJ, and EX wrote the first draft of the manuscript. FS, TR, HA-C, CP, AK, MJ, EX, and SC were involved in data acquisition and revised the manuscript for important intellectual contents. All authors had full access to all the data, including statistical reports and tables and approved the manuscript for final submission.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Simonetti is supported by a research grant provided by the CardioPath PhD program. Dr Rheude has received speaker fees from AstraZeneca and SIS Medical AG. Dr Joner reports institutional grant support from Boston Scientific, Cardiac Dimensions, Edwards Lifesciences and Infraredx; consulting fees from Biotronik, TriCares, Veryan, and Shockwave; speaker fees from Abbott Vascular, AstraZeneca, Biotronik, Boston Scientific, Cardiac Dimensions, Edwards Lifesciences, Recor Medical and Shockwave; participation on a Steering Committee of Biotronik and Edwards Lifesciences; travel support from Boston Scientific, Cardiac Dimensions, Edwards Lifesciences, and SIS Medical AG. Dr Xhepa has received lecture fees/honoraria from AstraZeneca, Boston Scientific, and SIS Medical AG; and proctoring honoraria from Abbott Vascular; and institutional grant support from Abbott Vascular. Dr Cassese has received lectures/proctoring honoraria from Abbott Vascular, Shockwave Medical, SIS Medical AG, and Translumina GmbH; and institutional grant support from Abbott Vascular and SIS Medical AG. The other authors declare no potential conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki. Due to its retrospective and anonymized nature (Trial-level analysis), ethical approval and informed consent were not required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
