Abstract
Background:
Selecting the most appropriate vascular access is crucial to minimizing catheter-related complications and repeated-venipunctures. The objective of this study is to evaluate whether the use of a long peripheral catheter (LPC), compared to short peripheral catheter (SPC), results in a different number of patients requiring repeated venipunctures for blood withdrawal.
Method:
Single-center, parallel arm interventional randomized controlled trial. Patients were randomized to receive either a conventional SPC or LPC, placed by trained Emergency Department nurses without ultrasound guidance. The primary endpoint was the proportion of patients requiring additional venipunctures during hospitalization. Secondary outcomes included time to persistent withdrawal occlusion (PWO) and time to catheter failure. This trial was registered with ClinicalTrials.gov (NCT06290284).
Results:
In total, 294 patients were randomly assigned to either the intervention or the control group. The catheter was successfully placed at the first attempt in 133/147 (90.5%) and 103/138 (74.6%) patients in the SPC and LPC arms, respectively. No significant difference was found between the two study arms in the proportion of patients requiring a venipuncture after SPC or LPC insertion: 85 out of 147 patients (57.8%) in the SPC group and 77 out of 138 patients (55.8%) in the LPC group (p = 0.73), with a mean adjusted difference of 3.5% (95% CI: −7.4% to 14.4%; p = 0.529). Time to catheter failure was 3.07 (1.96–5.90) and 4.72 (1.97–8.82) days in the SPC and LPC groups, respectively. However, time to PWO was significantly longer in the LPC group (median 1.9 vs 1.1 days, p = 0.006).
Conclusion:
Although LPCs did not significantly reduce the need for repeated venipunctures, they demonstrated more favorable performance in routine Emergency Department practice.
Keywords
Introduction
Peripheral intravenous cannulation is one of the most frequently performed invasive procedures in Emergency Departments (ED) worldwide, with billions of short peripheral catheters (SPCs) inserted every year. 1 SPCs are essential for administering fluids, medications, blood products, and contrast agents throughout a patient’s hospital stay. 2 Given the frequency of this procedure and the fundamental importance of vascular access, it is crucial to select the most appropriate vascular access device to minimize complications. Issues such as insertion failure, phlebitis, infiltration, infections, obstruction, and thrombosis can significantly compromise patient safety and comfort. 3 In addition, repeated venipunctures, often necessary due to difficulties in drawing blood through the catheter, contribute to patients’ fear, pain, and discomfort. 4
Peripheral catheters can be classified by length into three categories: SPCs, which measure <6 cm; long peripheral catheter (LPCs) ranging from 6 to 15 cm and midline catheters, which are >15 cm in length.5,6 LPCs present a valuable alternative to traditional SPCs, particularly for patients requiring extended intravenous therapy or frequent blood sampling, as they may reduce the need for repeated venipunctures. 7 Unlike SPCs, LPCs can remain in place for up to 4 weeks, depending on factors such as catheter material and the insertion technique, which may involve either the direct Seldinger or the simplified Seldinger technique. 7
The introduction of ultrasound (US) technology has further influenced vascular access practices, making the placement of LPCs safer, faster, and more tolerable. 8 Research indicates that US-guided catheter insertion lowers complication rates and is more cost-effective than midline or central venous access devices. 9 Although the benefits of LPCs have been extensively examined in different hospital settings,10 –14 few studies have specifically examined their use as a first-choice option without US-guidance in the ED, particularly in patients without difficult intravenous access, classified by the Enhanced Adult Difficult IntraVenous Access (EA-DIVA) score, 15 who are subsequently admitted to inpatients wards. Considering the high turnover of patients in the ED and the need for reliable vascular access, 16 incorporating LPCs into routine clinical practice could significantly reduce the frequency of repeated venipunctures and improve overall patient outcomes. Therefore, the primary aim of this study is to evaluate whether the use of a LPC, compared to a SPC, results in a different number of patients requiring repeated venipunctures for blood withdrawal or the placement of new vascular access during their hospital stay. The secondary aim is to investigate whether LPCs are associated with a reduced occurrence of adverse events, such as phlebitis, catheter obstruction, or accidental removal, compared to SPCs.
Methods
Study type and setting
This study conducted a single-center, parallel-arm (1:1) interventional randomized controlled trial (RCT). The study was registered on ClinicalTrials.gov under identification number NCT06290284. This RCT was conducted in the ED of Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, an academic tertiary-level hospital in Milan (Italy). The ED for adults is one of the largest in the metropolitan area, serving over 55,000 patients annually. Between September 25 and November 17, 2023, adult patients presenting to the ED triage desk were evaluated by the triage nurse, who assigned a triage code and assessed the likelihood of hospital admission. Consolidated Standards of Reporting Trials (CONSORT) guidelines were followed. 17
Inclusion and exclusion criteria
Inclusion criteria were: age ⩾18 years, high probability of hospital admission based on the triage nurse’s clinical judgment, 18 and absence of difficult intravenous access placement (as indicated by the EA-DIVA score ⩽8). 15 Exclusion criteria included the presence of existing vascular access (i.e. another SPC, central venous catheter, or peripherally inserted central catheter), inability to provide informed consent, or situations where obtaining informed consent would unnecessarily delay clinical care. Due to the distribution of patient flow at the triage desk during the day-time and organizational constraints, 19 only patients presenting to the ED between 7:00 a.m. and 8:00 p.m. were screened for study eligibility by the triage nurse.
Sample size
We consulted with a team of experienced vascular access nurses at our institution and estimated a risk of 60% of repeated venipuncture during the hospital length of stay. To detect a 20% difference between groups with 95% confidence interval (CI), we determined a sample size of 230 patients. Since approximately one-third of ED patients requiring urgent care are ultimately admitted to the hospital, the total sample size was set at 294 patients, with 147 assigned to each study arm.
Randomization and masking
Sequentially numbered, opaque, sealed envelopes labeled with a patient identification code containing the randomization sequence were prepared by the administrative staff who were not involved in the study. These envelopes were then made available to the ED. When a patient was deemed eligible for participation in the study, the triage nurse opened the corresponding envelope. The randomization sequence was generated by the blockrand function in the R open source software to achieve an equal number of patients in each study arms. 20 Blocks of different size were permuted to ensure that assignment to a study arm could not be predicted. The triage nurse and the whole research team were blinded to the block size and number. Nurses and physicians responsible for placing and managing the catheters were aware of each patient’s group assignment, as the catheters differed visibly by design, making it impossible to blind patients and staff to the catheter type. However, the statistician received an anonymized dataset and remained blinded to the catheter type used for each patient.
Study intervention
Patients in the LPC group had catheters inserted by a trained ED nurse using the simplified Seldinger technique without US-guidance. The LPC used was a Midline PowerGlide Pro™ (Becton Dickinson Company, Franklin Lakes, New Jersey, USA), an 18 or 20-gauge, single-lumen, power-injectable catheter with a length of 8 cm. In the SPC group, patients received a SPC Venflon™ Pro Safety Needle Protected (Becton Dickinson Company, Franklin Lakes, New Jersey, USA), an 18 or 20-gauge catheter. To secure the LPC, Grip-Lok® (TIDI Products, Neenah, Wisconsin, USA) catheter securement device with wide silicone adhesive area was used to hold the catheter in place on the patient’s skin, thereby reducing the risk of movement and accidental dislodgment. A transparent, semi-permeable dressing was applied to both groups, allowing for daily visual inspection of the insertion site. Follow-up began immediately after catheter placement and continued even after patients were transferred to a hospital ward. During this period, the cannulation site and dressing were closely monitored. Catheter care was overseen by an expert nurse in vascular access, who evaluated the catheter’s status daily using and documented any complications (such as malfunction, phlebitis, pain, catheter occlusion, or accidental removal) in an observation chart. Additionally, the ability to withdraw and infuse through the catheter was assessed daily to determine the time to persistent withdrawal occlusion (PWO). To evaluate and manage complications such as phlebitis, the Visual Infusion Phlebitis (VIP) scale was used. This scale assigns a score from 0 to 5, in ascending order of severity of inflammation. 21 Patients were monitored until catheter removal, with all catheters used solely for peripherally compatible intravenous therapy.
Outcome measures
The primary endpoint was the difference between the two study arms in the proportion of patients requiring additional venipuncture for blood sampling or placement of new catheters after insertion of the SPC or LPC. This measure was selected over the number of venipunctures because the literature does not report a clinically significant difference in the number of venipunctures that may be relevant for a patient. Secondary endpoints included comparisons between the study arms regarding: the number of venipunctures required for catheter placement; time to catheter failure; occurrence of complications such as phlebitis, malfunction, and catheter-related thrombosis; proportion of patients with accidental catheter self-removal and the occurrence of PWO.
Data collection
All study data were collected using the REDCap database (Research Electronic Data Capture, version 11.0.3). 22 The electronic case report form collected demographic data (sex, age, and ethnicity) and clinical information, including Body Mass Index (BMI), the cause of ED admission, Charlson Comorbidity Index score, 23 other comorbidities, and hospital length of stay. Details on the vascular catheter were also recorded, such as device type and size, date of placement, anatomic site of insertion, and number of attempts. Complications related to vascular catheters were systematically documented and included malfunction, phlebitis with associated pain, accidental self-removal, occlusion, and catheter-related thrombosis. Catheter malfunction was defined as removal of the device due to inability to complete the prescribed infusion therapy, irrespective of the underlying cause (e.g. reduced flow or intermittent occlusion). Phlebitis and pain were recorded when inflammation of the vein occurred, likely due to the presence of the catheter, and were typically accompanied by local symptoms such as redness, swelling, and discomfort at the insertion site. 24 Self-removal denoted accidental displacement of the catheter by the patient. Occlusion was defined as the complete inability to infuse intravenous fluids through the catheter. Finally, catheter-related thrombosis was defined as a partial or total loss of full compressibility, with thrombus adherence to the vessel wall confirmed by Doppler US. 25 The use of Doppler US was not performed routinely but rather employed selectively to investigate uncertain or unexplained causes of catheter malfunction. The frequency of semi-permeable dressing changes, category of drug administration, reasons for catheter removal, number of venipunctures required, and time to the occurrence of PWO were also recorded. Patients were followed with daily assessments until catheter failure (defined as removal due to loss of catheter functionality from any complication while its continued use was still clinically indicated and required catheter replacement) or until discharge. 26
Ethical considerations
Eligible patients were invited to participate and received written information about the study’s objectives and procedures. After a comprehensive explanation, patients who agreed to participate provided written informed consent. Participation in the study was entirely voluntary. The study protocol adhered to the principles of the Declaration of Helsinki and received approval from the local Ethics Committee (Comitato Etico Milano Area 2) with approval number 934_2022.
Statistical analysis
Metrics of interests are summarized by descriptive statistics, mean ± 1 standard deviation (SD) or median with interquartile range (IQR), and presented in tables with baseline characteristics of patients at enrollment. Number of patients with repeated venipunctures in the two groups are reported as crude and adjusted proportions, by fitting a logistic regression model accounting for Charlson Comorbidity Index score, gauge size, and type of drugs administered. Time to PWO and time to catheter failure were summarized using the Kaplan-Meier estimator, and difference between groups evaluated with the log-rank test. All comparisons were done using Pearson’s chi-squared or Fisher’s exact test. A Poisson test was used to compare incidence rates between groups, calculated as the number of events per 1000 catheters days. Statistical significance was set at p < 0.05. Analyses were performed using R statistical software version 4.4.3. 20
Results
During the study period, 10,188 patients were screened for participation. Of these, 9894 were excluded, and 294 were randomly assigned to either the SPC or LPC group. In the final analysis, 285 patients were included, as 9 patients from the LPC arm were excluded due to more than three attempts (Figure 1).
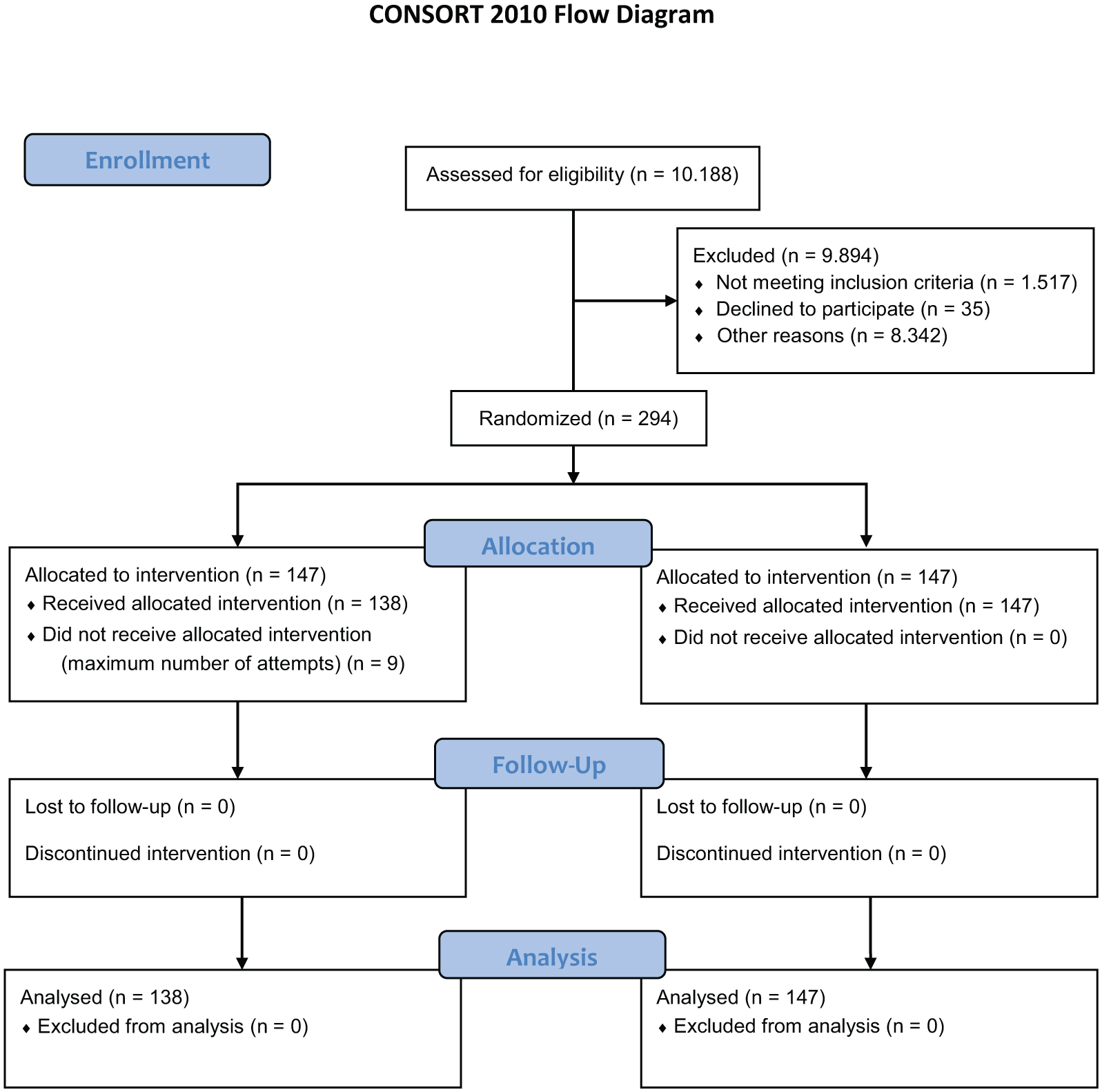
CONSORT 2010 flow diagram.
Table 1 summarizes the demographic and clinical characteristics of the study sample. The mean (SD) age was 70.4 (17.1) years, and 172 (60.4%) participants were male. The Charlson Comorbidity Index score and EA-DIVA score were similar between two groups. The median difference in hospitalization duration between the study arms was 2.5 (95% CI: 0–5) days. Among the 285 patients, 94 (32.9%) were discharged directly from the ED, whereas 191 required hospital admission. Internal medicine wards received the majority of admissions, accounting for 41.1% (117/285) of patients, while only 9.1% (26/285) were admitted to surgical wards.
Demographic and clinical characteristics.
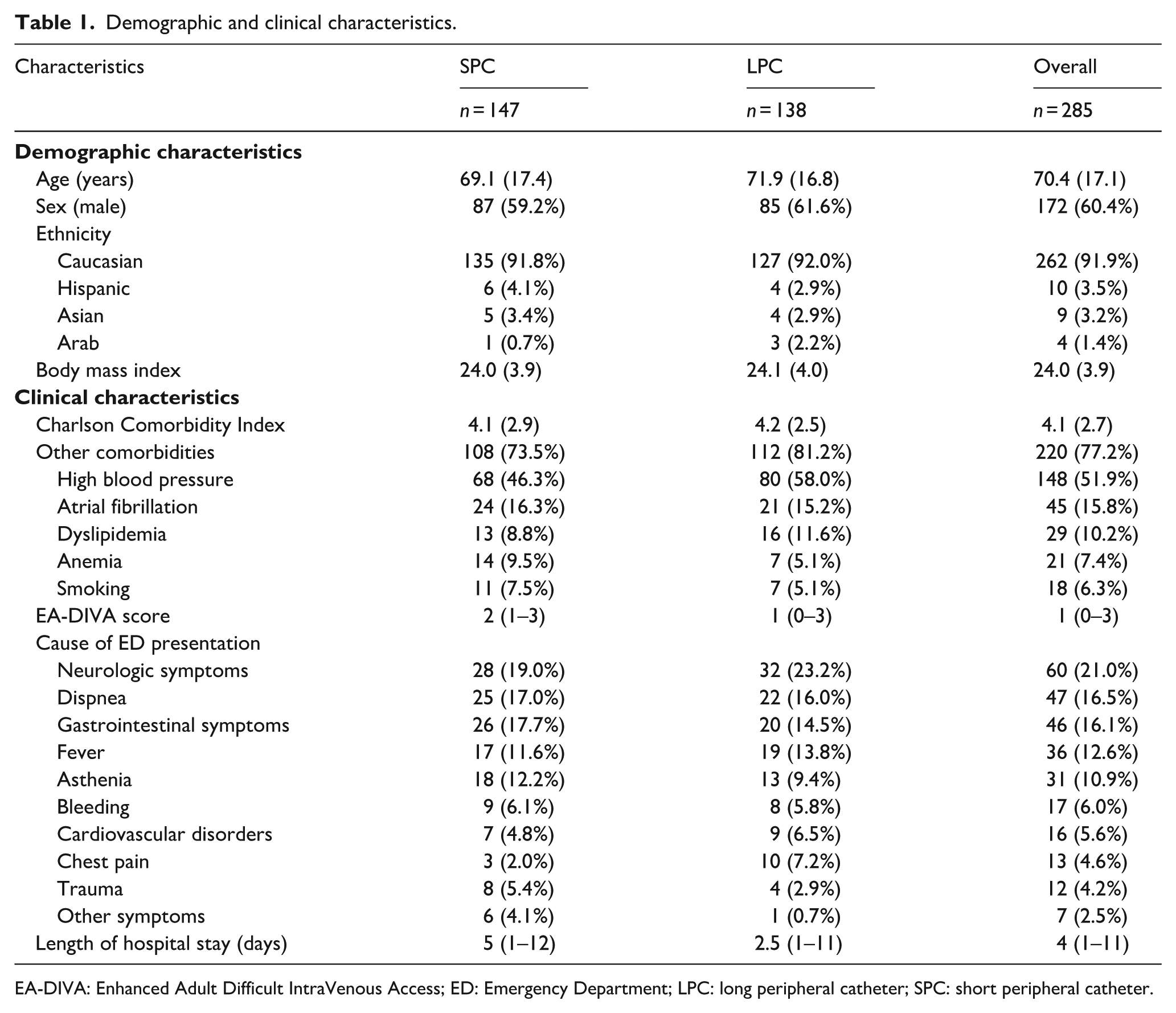
EA-DIVA: Enhanced Adult Difficult IntraVenous Access; ED: Emergency Department; LPC: long peripheral catheter; SPC: short peripheral catheter.
The catheter was successfully placed at the first attempt in 133/147 (90.5%) and 103/138 (74.6%) patients in the SPC and LPC arms, respectively. Other catheter-related characteristics are presented in Table 2. Regarding the primary study outcome, no significant difference was found between the two study arms in the proportion of patients requiring a venipuncture after SPC or LPC insertion: 85 out of 147 patients (57.8%) in the SPC group and 77 out of 138 patients (55.8%) in the LPC group (p = 0.730), with a mean adjusted difference of 3.5% (95% CI: −7.4% to 14.4%; p = 0.529). On day 2, the estimated catheter failure was 73.2% (95% CI: 64.1%–83.5%) in the LPC group and 72.9% (95% CI: 64.7%–82.1%) in the SPC group.
Vascular access characteristics.
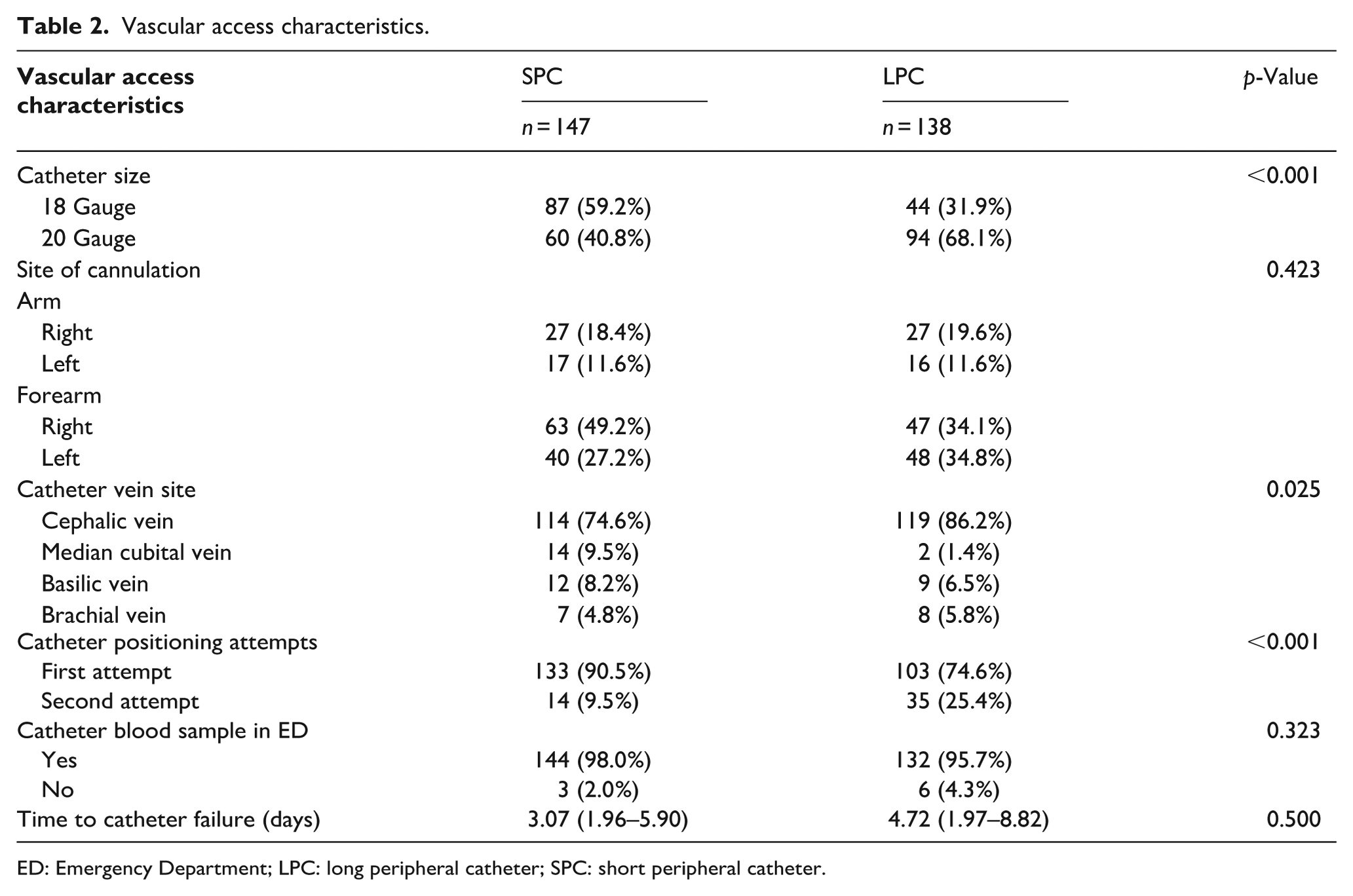
ED: Emergency Department; LPC: long peripheral catheter; SPC: short peripheral catheter.
As summarized in Table 3, additional venipuncture for blood tests or blood cultures were performed in both groups: 83 patients (56.5%) and 5 patients (3.4%) in the SPC group, and 75 patients (54.3%) and 6 patients (4.3%) in the LPC group. Specifically, 79 out of 147 patients (53.7%) in the SPC group and 74 out of 138 patients (53.6%) in the LPC group required an additional venipuncture due to the presence of PWO (p = 1.000). Radiological contrast agents were used more frequently for diagnostic procedures in the LPC group (p = 0.022). Overall, there was no statistically significant difference in catheter removal occurrence between the two groups (p = 0.074). The most common reasons for catheter failure were malfunction, phlebitis, and pain. Malfunction was the leading cause of catheter removal and occurred significantly more often in the SPC group (128.7 vs 71.0 per 1000 catheter-days, p = 0.013), while rates of catheter-related thrombosis were similar between groups. Figure 2 presents the patient-reported comfort levels (a) and the daily assessment of the catheter insertion site using the VIP scale (b). High comfort scores (NRS ⩾8) decline over the first 7 days post-placement. The VIP scale assessment shows that over 75% of patients with SPC remain in normal condition during this period. However, in the LPC group, the proportion of patients with catheters in normal condition drops to 60% after the seventh day post-placement. The Kaplan-Meier curves (Figure 3) show a statistically significant difference in time to PWO between the SPC and LPC groups (p = 0.006). In the SPC group, 50% of patients experienced PWO after a median of 1.1 (IQR: 0.9–2.0) days. In contrast, this occurred after 1.9 (IQR: 1.0–2.8) days in the LPC group. By the second day, the probability of the catheter remaining functional for both infusion and aspiration was 41.9% (95% CI: 32.8%–53.5%) in the LPC group, compared to 23.5% (95% CI: 16.5%–33.4%) in the SPC group.
Characteristics of vascular access management.
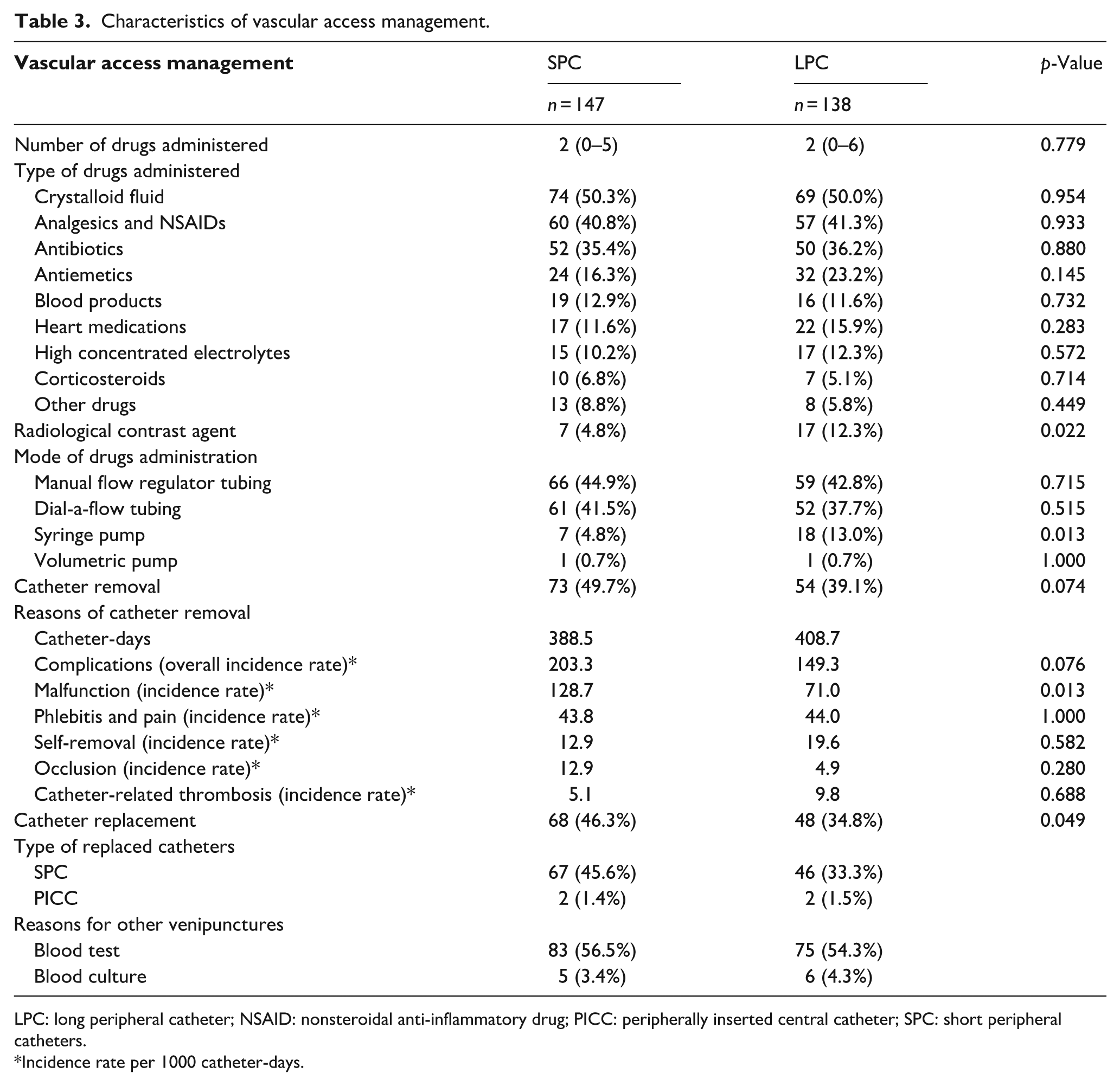
LPC: long peripheral catheter; NSAID: nonsteroidal anti-inflammatory drug; PICC: peripherally inserted central catheter; SPC: short peripheral catheters.
Incidence rate per 1000 catheter-days.
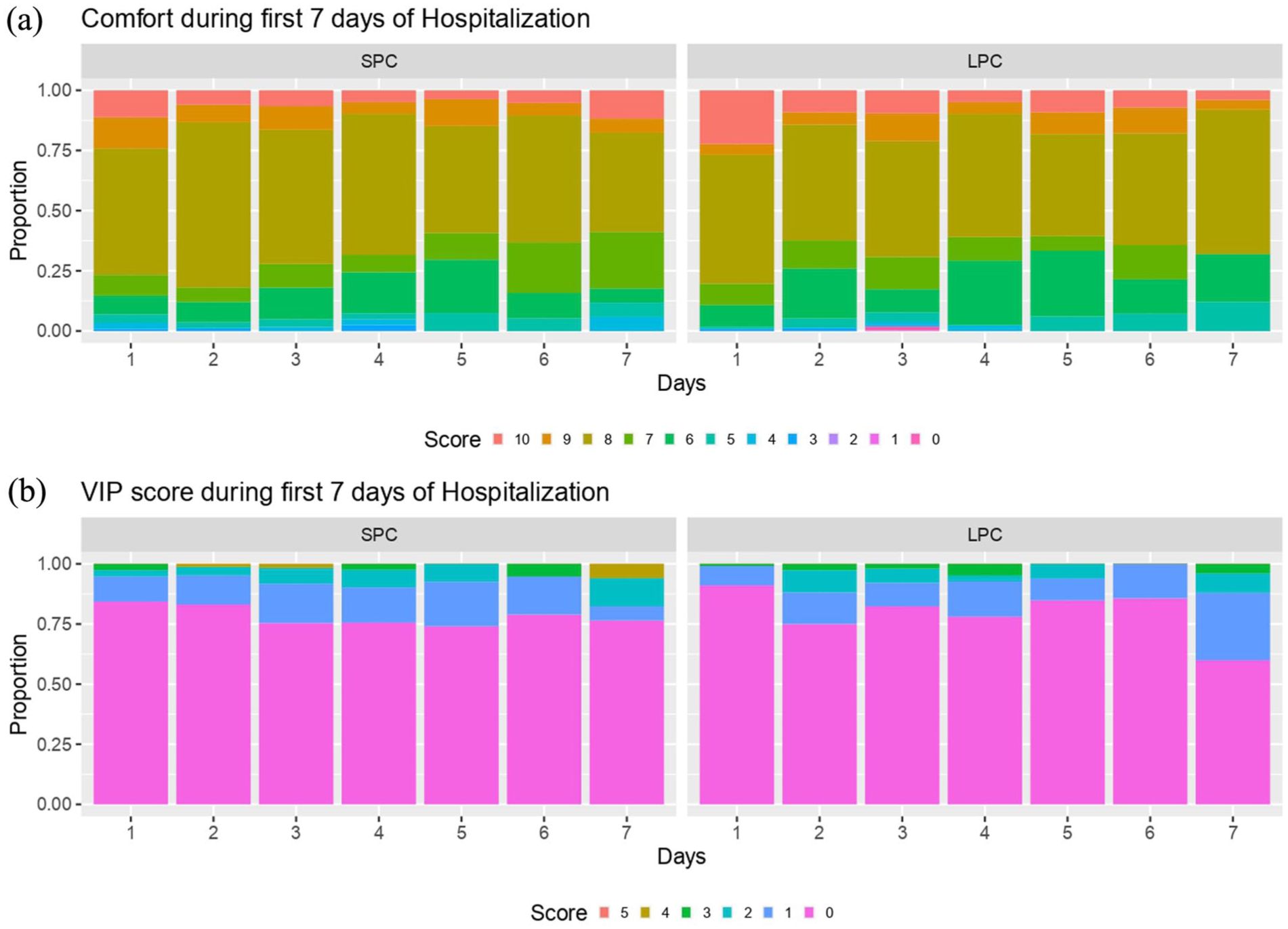
(a) Comfort during first 7 days of hospitalization and (b) VIP score during first 7 days of hospitalization.
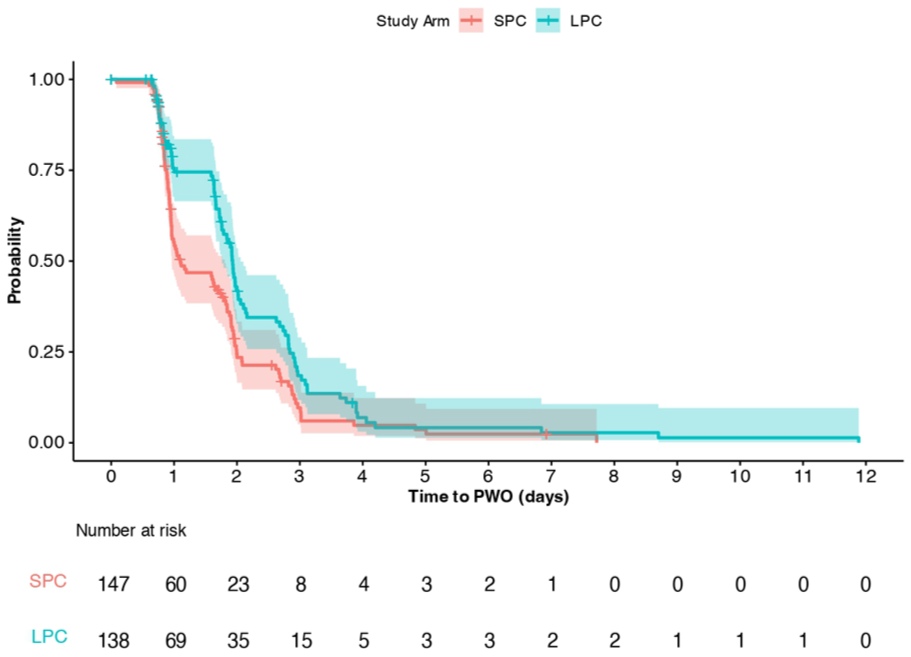
Time to Persistent Withdrawal Occlusion (PWO).
Discussion
This study represents the first RCT to evaluate whether LPCs, inserted by trained ED nurses using the simplified Seldinger technique, reduce the need for additional venipunctures during hospital stay compared to conventional SPC insertion. The results showed that the proportion of patients requiring repeated venipunctures did not differ significantly between the two groups, even when adjusted for factors such as catheter size, type of administered drugs, and patient comorbidities (as assessed by the Charlson Comorbidity Index score). Although these findings do not demonstrate a clear advantage of LPCs over SPCs in reducing the frequency of repeated venipunctures, several secondary outcomes provide valuable insights into catheter performance and the potential role of LPCs in routine ED practice.
Although the LPC group demonstrated a statistically significant longer time to PWO compared to the SPC group, this difference holds limited clinical relevance, given the typical length of stay in acute care hospital. However, this finding is clinically meaningful in another aspect: a prolonged time to PWO increases the likelihood of requiring additional venipunctures. When blood cannot be drawn directly from the catheter, alternative needle insertions become necessary, leading to increased patient discomfort and pain. The possibility of a single cannulation throughout the entire hospitalization could therefore reduce pain and improve patients’ quality of life and overall perception of intravenous therapy. 27 Patient satisfaction and comfort levels are important not only during cannulation but also regarding catheter securement devices use. In particular, the choice of this device can influence the comfort levels perceived by patients with either an LPC or a SPC. 28 An extended functionality may translate into fewer catheter replacements, especially for patients with hospital stays shorter than 36 h. These findings are consistent with previous research indicating that, when inserted and maintained appropriately, longer catheters such as LPC can remain functional for prolonged periods. 29
The incidence of malfunction was significantly higher in the SPC group, potentially attributable to a combination of factors such as the physical characteristics of the device, prolonged use, and less standardized management practices. 30 Furthermore, tip placement in superficial veins, such as those of the forearm, may predispose to mechanical obstruction and difficulties in blood aspiration, potentially leading to PWO. 30 Catheter tip location significantly influences the risk of catheter-related complications, as the placement of catheters in larger vessels with higher blood flow reduces the likelihood of thrombogenesis. This reduction is attributed to the accelerated blood flow, which facilitates faster dilution and transport of administered medications, and minimizes their contact time with the vessel wall. 31 Although the literature reports higher hemolysis rates for blood samples obtained from SPCs compared to venipunctures, 32 the advantages of drawing blood from an existing catheter include convenient access, reduced staff workload, lower costs, and less pain for the patient due to avoiding an additional venipuncture. 33 In contrast, LPCs did not show significantly higher incidence of phlebitis, pain, or catheter-related thrombosis compared to SPCs, even though LPCs were more frequently used for radiological examinations involving contrast agent. Unlike standard SPCs, the LPC evaluated in this study is specifically designed for power injection, enabling it to tolerate higher injection pressures safely. 34 This feature likely encouraged clinicians to use LPCs more frequently for diagnostic imaging requiring contrast agent, thereby reducing the need for additional venous access devices or dedicated power-injectable catheters. In settings where patients require urgent imaging diagnostics (such as in the ED) to identify the clinical problem, 35 having a power-injectable vascular device can streamline workflows, minimize delays, and enhance patient comfort.
These findings underscore the complexity of selecting the most appropriate peripheral catheter, as each device type offers distinct advantages and disadvantages depending on patient needs, the specific intravenous therapy, and local clinical protocols. In both study groups, the choice of catheter size was determined by the ED nurse. In the SPC group, an 18-gauge catheter was predominantly used, whereas in the LPC group, a smaller 20-gauge catheter was chosen more frequently. This approach may reflect the nursing staff’s lower familiarity with LPC placement; although nurses underwent specific training before the study, this limited preparation could not compensate for the extensive experience gained over years of placing traditional SPCs. The lower proportion of first-attempt success with LPCs may be attributable to limited proficiency with the simplified Seldinger technique. Notably, the use of US-guidance is essential for LPC insertion in DIVA patients, 36 while its application in non-DIVA patients is less common due to relatively easier vascular access and the lower complication rates associated with standard SPCs. 37 However, even in non-DIVA patients, conducting an in-deep US assessment of the venous system could be beneficial to guide the choice of the most appropriate catheter. 38
A key strength of this RCT lies in the comprehensive training provided to all ED nurses before study initiation, ensuring a consistent baseline of procedural knowledge about the simplified Seldinger technique among nursing staff. Another important strength is the strict follow-up protocol and detailed data collection, which allowed for thorough monitoring of catheter performance, complications, and patient-reported outcomes throughout the study period.
Nonetheless, this study has some limitations. First, this is a single-center RCT in which the decision to screen patients for potential hospitalization was based on the triage nurse’s clinical judgment, introducing an element of subjectivity in patient selection. Despite triage nurses exhibited an excellent ability to predict admission compared with other scores, 18 this approach may limit the generalizability of the findings to settings where other hospital admission procedures are used. Second, no cost-benefit analysis was conducted to evaluate the use of LPCs in relation to their time to failure and associated complications.
Conclusion
This RCT offers a new perspective on the utility of LPCs placed with the simplified Seldinger technique in an adult ED population without difficult intravenous access. While LPCs did not significantly reduce the need for repeated venipunctures, they demonstrated a longer functional lifespan and a favorable profile in terms of catheter malfunction. The potential to place LPC, particularly when multiple infusions or blood tests are performed, may be beneficial in busy ED settings. However, further multicenter trials are needed to fully delineate the cost-effectiveness, patient-reported outcomes, and optimal indications for choosing an LPC over a conventional SPC in ED practice.
Footnotes
Acknowledgements
The authors would like to express their deepest gratitude to all those who contributed to this study. Special thanks go to Maria Abbatangelo, Nadia Boasi, Paola Bosco, Cinzia Carugati, Cinzia Coldani, Elisabetta Corona, Luigi Giugno, Giovanni Greco, Chiara Marta, Grazia Molentino, Massimo Mudadu, Cosimo Nasuto, Myriam Neglia, Cristiana Piola, Elena Poliani, Lucia Ruzzo, Maria Tabini, Monica Tolentini, Yeral Aldo Vilchez Flores for their assistance in data collection and patient care.
Author contributions
Filippo Binda: Conceptualization, Investigation, Data curation, Project administration, Resources, Writing—original draft, Visualization. Alessandro Galazzi: Conceptualization, Resources, Writing—original draft. Federica Marelli: Investigation, Data curation, Writing—original draft, Visualization. Gianluca Salinaro: Conceptualization, Investigation. Matteo De Vecchi: Investigation, Data curation. Serena Sagna: Investigation, Data curation. Alessia Marcotullio: Investigation, Data curation. Jessica Calegari: Investigation, Data curation. Dario Laquintana: Conceptualization, Resources, Project administration, Supervision, Writing—review & editing. Simone Gambazza: Conceptualization, Data curation, Formal analysis, Methodology, Writing—original draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by Italian Ministry of Health—Current research IRCCS.
Ethical approval
Ethical approval for this study was obtained from the local Ethics Committee (Comitato Etico Milano Area 2) on 5 October 2022 (approval number 934_2022).
Informed consent
Informed consent was obtained from all patients in the study.
